This simple visual analogy that Ron House has designed can help readers not familiar with a contentious atmospheric modeling issue get a primer on the it. While not a perfect analogy (and by definition analogies often aren’t) it does help convey an important point: the predicted red spot has not appeared. For the more technically inclined, or for those wanting more, Steve McIntyre posted an interesting discussion at Climate Audit. – Anthony
Guest Post by Ron House July 29, 2009
Let’s say it’s a cold night and Fred climbs into bed:
 (A) Fred in bed.
(A) Fred in bed.
Will Fred use a blanket to keep warm? If so, the air will heat up close to Fred because his body warms the air and the blanket prevents it from moving away. On the other hand, as the night progresses, the air beyond the blanket will cool:
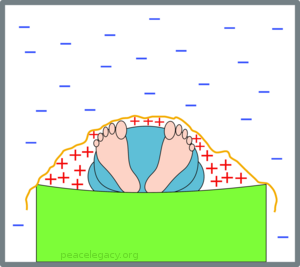 (B) With a blanket, the warm air collecting around Fred warms him up.
(B) With a blanket, the warm air collecting around Fred warms him up.
In the picture, the “+” signs show air that becomes warmer, and the “-” signs air that becomes cooler.
Now what if Fred (forgetful Fred) didn’t use a blanket? The warm air escapes and tends to rise (warm air being less dense than cold air):
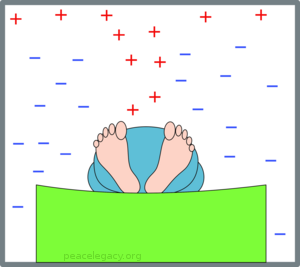 (C) With no blanket, warm air escapes and Fred shivers.
(C) With no blanket, warm air escapes and Fred shivers.
Poor Fred gets colder as the night wears on. But now we come to the point of the exercise: How do we know whether Fred used, or did not use, a blanket?
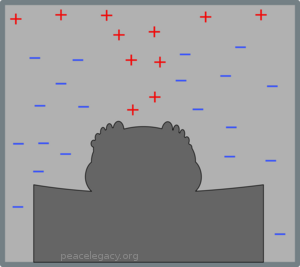
“Easy,” you say: “Take a look!” But let’s suppose that Fred is a very light sleeper, we dare not put on the light, so there’s no way we can see if there’s a blanket. But—surprise!—we just happen to have an infra-red scanner that can tell us the temperature of the air at various spots throughout the room. Depending on whether Fred uses a blanket, the temperature change in the room follows one of the two characteristic patterns we saw above; so if we check where the air gets colder and where it gets warmer as the night wears on, we know, for a fact, whether or not Fred used a blanket, even without being able to see it. If Fred did use a blanket, our scanner should show results like this (note how we can’t see the blanket, but we can be sure that it is there):
 (D) Warm air collects in a contained region, so there must be a blanket.
(D) Warm air collects in a contained region, so there must be a blanket.
On the other hand, if he does not use a blanket, we will see the temperature change in a pattern something like this:
 (E) Warm air escapes upwards, so we are sure there is no blanket.
(E) Warm air escapes upwards, so we are sure there is no blanket.
Once again, there is no doubt at all what is going on. In science, nothing is absolutely certain, but depending on which temperature pattern develops, we can be very, very sure indeed of the answer to the question: Did Fred use a blanket?
Now we can turn to the global warming question, whether the Earth is surrounded by a ‘blanket’ of anthropogenic (human-generated) greenhouse gas stoking up the temperature of the planet. The physics of a real blanket (as with Fred in the fable above) and a gaseous ‘blanket’ around the Earth differ, but just the same, different heat dissipation (or retention) processes will result in different characteristic patterns of temperature change. Just as Fred will be surrounded by something roughly resembling one of two quite different patterns of air temperatures, so likewise will temperature changes around the Earth have a quite definite pattern, depending on which climate theory is right. Scientists whose paycheck does not depend on agreeing with global warming alarmism will all agree with this simple statement. It’s part of the basic skill of having a ‘nose’ for physics.
What, then, are our main competing climate theories? The IPCC’s reports are based on results from a collection of climate computer models; they have nothing else. These are simply computer programs that, in essence, contain a computerised version of the assumptions and beliefs of the climate modeller as to how the climate of the planet works. Whether these assumptions are well-founded is another question, but the key point is that whatever these assumptions may be, when the climate model is run, it generates its ‘predictions’ by calculation of hypothetical futures for the behaviour of the atmosphere. These ‘futures’ contain, as an essential element, predictions of the changes of atmospheric temperatures at various heights above the planet and the various latitudes all the way from south pole to north pole.
The indisputable fact about these atmospheric temperature predictions is that if the pattern doesn’t happen, the model is wrong. Just as Fred won’t warm up if he isn’t surrounded by warm air, likewise the effects on the Earth of global warming cannot happen if the cause of the warming —the warm air—isn’t there.
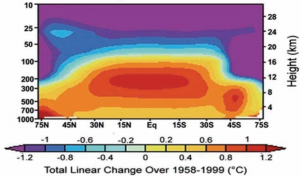
So now we come to the graphs that clinch the matter. All global warming models predict some sort of developing ‘hotspot’ in the atmosphere above the tropics. Here is the graph for one of the models, but they all look roughly similar:
 (F) Model predicts air above the tropics heats up. from the NIPCC Report p. 107
(F) Model predicts air above the tropics heats up. from the NIPCC Report p. 107
This picture shows the air from 75 degrees north to 75 degrees south (the equator in the middle) and up to 30 km above the Earth. We can think of this air pattern as corresponding to the pattern in Fred’s bedroom when Fred used a blanket: although the actual mechanism is different, something is ‘keeping the heat in’, so to speak. Just as we did with Fred in bed, we can compare reality with this picture. Is the heat in the real atmosphere doing what the model predicts? Here is the temperature trend in the real world:
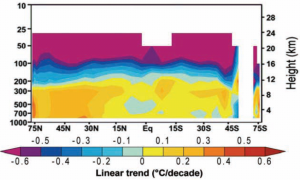 (G) Real world trend develops no hotspot. from the NIPCC Report p. 106
(G) Real world trend develops no hotspot. from the NIPCC Report p. 106
What have we actually proved here? Well, proved, without possibility of error, nothing, of course: no question at all about the real world ever has a complete perfect proof as an answer, so don’t be misled if someone says the world still might be heating due to CO2 despite the absence of the warm spot that is supposed to do the warming. Of course anything might be happening; but how likely is it? Well how likely is it that Fred has a blanket, but the air around him is getting colder just as if he had no blanket, and yet Fred is warming up despite that? The two questions have the same answer: not very.
Yet surprisingly, some proponents of global warming alarmism actually resort to this very strategy. “True,” they say, “the hot spot isn’t developing. But that is because the heat is being stored up elsewhere—it’s “in the pipeline”—and one day it will burst forth with even greater severity and vengeance.”
What can we make of that claim? Well, thinking back to Fred again, it amounts to this: We use our temperature probe in Fred’s darkened bedroom and we see a pattern like that in (E) above, corresponding to no blanket: Fred should be freezing! But actually, the heat has all gone into Fred’s body, despite the complete absence of the hot air which is the mechanism for making it do so. In other words, Fred got warmer by disobeying the second law of thermodynamics—in other words, by magic. Likewise, if someone says heat is being secretly stored somewhere by global warming, despite the absence of the very mechanism that does the warming, they are saying global warming is happening by magic. That is the harsh truth of the matter.
One thing I have learned whilst studying the global warming question is that, like many other physical systems, the climate is constrained by limits that can be understood by any intelligent person willing to learn some simple physics. The ‘hotspot’ is one of them. Anyone talking down to you and telling you you have to take the word of some mythical ‘consensus’ of ‘experts’ is trying to hoodwink you.
Let’s say it’s a cold night and Fred climbs into bed:
 (A) Fred in bed.
(A) Fred in bed.
Will Fred use a blanket to keep warm? If so, the air will heat up close to Fred because his body warms the air and the blanket prevents it from moving away. On the other hand, as the night progresses, the air beyond the blanket will cool:
 (B) With a blanket, the warm air collecting around Fred warms him up.
(B) With a blanket, the warm air collecting around Fred warms him up.
In the picture, the “+” signs show air that becomes warmer, and the “-” signs air that becomes cooler.
Now what if Fred (forgetful Fred) didn’t use a blanket? The warm air escapes and tends to rise (warm air being less dense than cold air):
 (C) With no blanket, warm air escapes and Fred shivers.
(C) With no blanket, warm air escapes and Fred shivers.
Poor Fred gets colder as the night wears on. But now we come to the point of the exercise: How do we know whether Fred used, or did not use, a blanket?
“Easy,” you say: “Take a look!” But let’s suppose that Fred is a very light sleeper, we dare not put on the light, so there’s no way we can see if there’s a blanket. But—surprise!—we just happen to have an infra-red scanner that can tell us the temperature of the air at various spots throughout the room. Depending on whether Fred uses a blanket, the temperature change in the room follows one of the two characteristic patterns we saw above; so if we check where the air gets colder and where it gets warmer as the night wears on, we know, for a fact, whether or not Fred used a blanket, even without being able to see it. If Fred did use a blanket, our scanner should show results like this (note how we can’t see the blanket, but we can be sure that it is there):
 (D) Warm air collects in a contained region, so there must be a blanket.
(D) Warm air collects in a contained region, so there must be a blanket.
On the other hand, if he does not use a blanket, we will see the temperature change in a pattern something like this:
 (E) Warm air escapes upwards, so we are sure there is no blanket.
(E) Warm air escapes upwards, so we are sure there is no blanket.
Once again, there is no doubt at all what is going on. In science, nothing is absolutely certain, but depending on which temperature pattern develops, we can be very, very sure indeed of the answer to the question: Did Fred use a blanket?
Now we can turn to the global warming question, whether the Earth is surrounded by a ‘blanket’ of anthropogenic (human-generated) greenhouse gas stoking up the temperature of the planet. The physics of a real blanket (as with Fred in the fable above) and a gaseous ‘blanket’ around the Earth differ, but just the same, different heat dissipation (or retention) processes will result in different characteristic patterns of temperature change. Just as Fred will be surrounded by something roughly resembling one of two quite different patterns of air temperatures, so likewise will temperature changes around the Earth have a quite definite pattern, depending on which climate theory is right. Scientists whose paycheck does not depend on agreeing with global warming alarmism will all agree with this simple statement. It’s part of the basic skill of having a ‘nose’ for physics.
What, then, are our main competing climate theories? The IPCC’s reports are based on results from a collection of climate computer models; they have nothing else. These are simply computer programs that, in essence, contain a computerised version of the assumptions and beliefs of the climate modeller as to how the climate of the planet works. Whether these assumptions are well-founded is another question, but the key point is that whatever these assumptions may be, when the climate model is run, it generates its ‘predictions’ by calculation of hypothetical futures for the behaviour of the atmosphere. These ‘futures’ contain, as an essential element, predictions of the changes of atmospheric temperatures at various heights above the planet and the various latitudes all the way from south pole to north pole.
The indisputable fact about these atmospheric temperature predictions is that if the pattern doesn’t happen, the model is wrong. Just as Fred won’t warm up if he isn’t surrounded by warm air, likewise the effects on the Earth of global warming cannot happen if the cause of the warming —the warm air—isn’t there.
So now we come to the graphs that clinch the matter. All global warming models predict some sort of developing ‘hotspot’ in the atmosphere above the tropics. Here is the graph for one of the models, but they all look roughly similar:
 (F) Model predicts air above the tropics heats up. from the NIPCC Report p. 107
(F) Model predicts air above the tropics heats up. from the NIPCC Report p. 107
This picture shows the air from 75 degrees north to 75 degrees south (the equator in the middle) and up to 30 km above the Earth. We can think of this air pattern as corresponding to the pattern in Fred’s bedroom when Fred used a blanket: although the actual mechanism is different, something is ‘keeping the heat in’, so to speak. Just as we did with Fred in bed, we can compare reality with this picture. Is the heat in the real atmosphere doing what the model predicts? Here is the temperature trend in the real world:
 (G) Real world trend develops no hotspot. from the NIPCC Report p. 106
(G) Real world trend develops no hotspot. from the NIPCC Report p. 106
What have we actually proved here? Well, proved, without possibility of error, nothing, of course: no question at all about the real world ever has a complete perfect proof as an answer, so don’t be misled if someone says the world still might be heating due to CO2 despite the absence of the warm spot that is supposed to do the warming. Of course anything might be happening; but how likely is it? Well how likely is it that Fred has a blanket, but the air around him is getting colder just as if he had no blanket, and yet Fred is warming up despite that? The two questions have the same answer: not very.
Yet surprisingly, some proponents of global warming alarmism actually resort to this very strategy. “True,” they say, “the hot spot isn’t developing. But that is because the heat is being stored up elsewhere—it’s “in the pipeline”—and one day it will burst forth with even greater severity and vengeance.”
What can we make of that claim? Well, thinking back to Fred again, it amounts to this: We use our temperature probe in Fred’s darkened bedroom and we see a pattern like that in (E) above, corresponding to no blanket: Fred should be freezing! But actually, the heat has all gone into Fred’s body, despite the complete absence of the hot air which is the mechanism for making it do so. In other words, Fred got warmer by disobeying the second law of thermodynamics—in other words, by magic. Likewise, if someone says heat is being secretly stored somewhere by global warming, despite the absence of the very mechanism that does the warming, they are saying global warming is happening by magic. That is the harsh truth of the matter.
One thing I have learned whilst studying the global warming question is that, like many other physical systems, the climate is constrained by limits that can be understood by any intelligent person willing to learn some simple physics. The ‘hotspot’ is one of them. Anyone talking down to you and telling you you have to take the word of some mythical ‘consensus’ of ‘experts’ is trying to hoodwink you.
**********
If you look at the ideal gas law, PV = nRT, an increase in temperature will result in an increase in the product of pressure and temperature.
**************
That should read “in an increase in the product of pressure and volume”
Bob D,
“Here’s a question for you though regarding pressure (I’ve thought about this for a while and it confuses me) – If there was an atmosphere but no GHGs, wouldn’t the average emission level be at the surface, since nothing is preventing the emission of IR to space from the surface? Wouldn’t that then imply a surface temp of about 255K (ie: Ts=Te assuming e=0 by Stefan-Boltzmann), to ensure radiative balance?”
Yes.
“And wouldn’t that then determine the pressure (and volume) by PV=nR(255) because the atmosphere is free to expand or contract, in principle?”
No. That formula is valid for a volume of gas all at the same uniform temperature and pressure. We would still get pressure dropping with altitude. And if there was convection, you could still get the temperature of the air above the surface falling even further below the Stefan-Boltzman temperature, while the surface remained at that temperature.
As I said above, it’s how the top of the troposphere is cooled to -54 C.
jim,
“I can see where adding some CO2 would raise the “zero point” where radiation in = radiation out (at least I think that is what you mean.) But I don’t see how that raises pressure.”
It doesn’t raise the pressure. It moves the “zero point” to a different place, where the pressure is different.
It’s the pressure difference between surface and average emission altitude that controls the temperature difference between surface and the “zero point”.
“When you move the pipe up in the dammed lake, you actually add mass to the vertical dimension. This accounts for the increase in pressure in the lake when the level gets higher.”
The height of the water, equivalent to mass, is analogous to heat, not pressure. It’s an analogy, not an identical piece of physics, and water pressure does not correspond to atmospheric pressure. Moving the pipe is analogous to increasing the total heat in the atmosphere.
*********************
Stevo (10:46:40) :
It doesn’t raise the pressure. It moves the “zero point” to a different place, where the pressure is different.
It’s the pressure difference between surface and average emission altitude that controls the temperature difference between surface and the “zero point”.
*************************
So, are you making the “environmental lapse rate” argument?
Jim,
I suppose you could call it that. I just think of it as “the greenhouse effect”.
No, I get that, I think we’re in agreement. I suppose I was confused by your statement above: “Pressure is the reason the surface is warmer than -24 C, which is the average temperature it would be if we had the same sun, but no atmosphere.”
It seemed you were saying that the atmosphere’s pressure alone would raise the temperature above the S-B temperature, and I couldn’t see how it would do that. Under the scenario you mentioned, the maximum temperature would still be at the surface, and would be 255K. In other words, we would have an atmosphere, but it would start at 255K at the surface and decrease vertically from there.
The way I read your comment, I thought you were sort of implying that if there were no GHGs we would still have temperatures higher than S-B simply due to pressure. I’ve seen this argument advanced before you see, and it’s never made sense to me.
I think I’ve misinterpreted your statement, and you meant “Pressure in the presence of GHGs is the reason…”
**************
Jim,
I suppose you could call it that. I just think of it as “the greenhouse effect”.
****************
No problem. I have never thought nor said that CO2 doesn’t raise the temperature a bit. I just am not convinced that it will lead to any kind of catastrophic warming. I’m certainly not convinced it will raise the global average over about 1.5 degrees per century. And even if it did raise temps 3-5 degrees per century, I would probably be for just letting society adjust as necessary with no restrictions on burning oil and coal. However, I would be for a Moon-shot effort to build all the nuclear power stations we could and to put in place the nuclear infrastructure that would entail, to include the completion of Yucca mountain. Cheap power can make many things possible. I don’t have a problem with solar and wind energy either as long as they can compete with oil, coal, and nuclear without subsidies. (And yes, I would be for the government subsidizing the insurance for nuke plants – a lot of people wouldn’t accept them without it and the odds of ever needing it are slim to none.)
“”” Spector (20:08:18) :
>>>
BTW, William F. Ruddiman has presented a theory that greenhouse gasses generated at the onset of human farming, 8000 years ago, may have prevented a next scheduled ice age. Scientific American, March 2005. “””
I believe that in the very same article, Ruddiman presents the results of two ice cores which during the last 1000 years or so, had records going in exactly the opposite direction at the same time. It was either the CO2 or the temperature; but the two cores said the exact opposite was happening.
So much for ice core proxies. (and results inferred from them)
“”” Jim Masterson (12:34:46) :
>> Kevin Kilty (06:23:09) :
. . .
I like Kiehl and Trenberth (Earth’s Annual Global Mean Energy Budget) 1997, not because the numbers are particularly accurate, but because it represents the quintessential model of the Earth’s average energy flows. So according to Kiehl et al., the atmosphere receives 67 W/m^2 directly from the Sun, 24 W/m^2 from sensible heat flux, 78 W/m^2 from latent heat flux, and 350 W/m^2 from the surface. (The surface radiates 390 W/m^2 which is what a blackbody radiates at a temperature of 288K, and the other 40 W/m^2 escapes through the IR window.) This adds up to 519 W/m^2 that the atmosphere absorbs on average. The atmosphere doesn’t radiate one-half to space and one-half towards the surface. It’s more like two-fifths (195 W/m^2) upward to three-fifths (324 W/m^2) downward. “””
Jim, I’m interested in your last assertion that the atmosphere radiates 40% upwards, and 60% downwards. That seems counter-intuitive to me. The emission of a photon from the atmosphere would seem to be required to be isotropic; unless somehow these molecules can sense gravity.
That would argue for the probabilities being equal for each direction. Then when you look at the temperature and density gradients in the atmosphere, and the resultant CO2 spectrum broadening; that would seem to favor the upward path rather than the downward path; since the downward path to increased air density and temperature, and CO2 concentration, would imply ever more frequent re-absorptions followed by presumably isitropic emission splits between the up/down directions.
Are your numbers the result of measurements or modelling ?
George
“”” Stevo (12:02:17) :
“So just what is it about CO2; and how about CH4, that is assymmetrical. Explain how this assymetry is any different from the assymetry of O2 and N2.”
Sorry. I was trying to keep it simple.
Molecules can contain thermal energy in a number of ways. Translation, rotation, stretching/contracting, bending, or by different parts of the molecule rotating with respect to other parts. Each sort of motion has a set of characteristic frequencies, and all the motions contribute to its absorption/emission spectrum.
If you take an N2 molecule, it can translate, stretch, or rotate about two axes. But rotating about the long axis doesn’t change anything, and you can’t bend it.
CO2 has three atoms in a straight chain, which can also bend. The carbon atom can be off-centre from the straight line between the oxygen atoms. And the two bonds can stretch independently.
H2O has three atoms in a bent chain, which means that rotating it about the third axis is also meaningful.
A molecule interacts with an electromagnetic wave via the charge distribution around it. For example, in H2O the oxygen has a negative charge compared to the hydrogens, so an electric field will pull the oxygen one way while pushing the hydrogens the other. Similarly, in CO2 the oxygens tend to be more negative than the carbon. Essentially, the asymmetry in this case is that the molecule contains atoms of different elements instead of all the same element. Figuring out exactly what sort of motion corresponds to what absorption line is a bit technical. But as a general rule, the more “messy” – big, complicated, asymmetrical – a molecule is, the more ways it can move and therefore the more likely it is to absorb/emit. There are greater and lesser degrees of symmetry. “””
Well Stevo you’ve just explained how the resonance absorption lines/bands of molecules come about, in particular with H2O and CO2, and I’m very familiar with that. But that energy is typically not re-emitted at the same photon freuency because the lifetimes of those excited states exceeds the mean time between collisions with the ordinary atmospheric gases; name ly N2, O2 and Ar. So the long wave Ir radiation absorbed by GHG molecules such as CO2 is rapidly transferred to the ordinary atmospheric gases as thermal energy, thereby raising the atmospheric temperature.
The question that I raised was by what rule are those ordinary atmospheric molecules/atoms prohibited from radiation due to their non-zero Kelvin temperature.
Because of the thermal agitation of those molecules, whehter in the earth’s atmosphere or the sun’s they must radiate since they contain accelerated electric charges due to the thermal vibration. If the solar atmosphere can radiate essentially black body thermal radiation , so can the earth’s atmosphere, so the radiation from the atmosphere is a function of the temperature of the ordinary gases of the atmosphere and is unrelated in any way to the nature or species of the GHG molecule, that capture the energy to warm the atmosphere.
It’s myth that gases do not emit black body (like) radiation and only solids do.
George
>> George E. Smith (17:24:41) :
Jim, I’m interested in your last assertion that the atmosphere radiates 40% upwards, and 60% downwards. That seems counter-intuitive to me. The emission of a photon from the atmosphere would seem to be required to be isotropic; unless somehow these molecules can sense gravity. <<
We know that photons can sense gravity (thanks to General Relativity and the equivalence principle), but the Earth doesn’t have enough mass to make this property of photons important. We also know that the density of the atmosphere changes with altitude. There should be more absorbers/emitters closer to the surface. So, yes, these molecules can sense gravity.
>> That would argue for the probabilities being equal for each direction. Then when you look at the temperature and density gradients in the atmosphere, and the resultant CO2 spectrum broadening; that would seem to favor the upward path rather than the downward path; since the downward path to increased air density and temperature, and CO2 concentration, would imply ever more frequent re-absorptions followed by presumably isitropic emission splits between the up/down directions. <<
I’ve tried modeling this for multiple layers, and it isn’t isotropic. You get more radiation going downward than upward. It’s another property of the model.
>> Are your numbers the result of measurements or modelling ? <<
They aren’t MY numbers, but Kiehl and Trenberth’s numbers. The answer is both. If you read their 1997 paper, the top-of-atmosphere (TOA) outgoing value as measured by the satellite observations from Nimbus-7 and the Earth Radiation Budget Experiment (ERBE) is 235 W/m^2. If 40 W/m^2 comes directly from the surface, then that leaves 235 W/m^2 – 40 W/m^2 = 195 W/m^2 for the atmosphere. The other 519 W/m^2 – 195 W/m^2 = 324 W/m^2 must be going somewhere else (down’s a good direction).
These numbers change slightly with their new 2009 paper, but the general relationships remain about the same. (I still don’t know where they get the 40 W/m^2 value for the IR window. The 1997 paper calculates it wrong, and the 2009 paper gets it from the 1997 paper.)
Jim
ok well perhaps another analogy with c02 and its absorbtion bandwidth (12-18 microns) is sunblock. If you go to the Sahara with factor 5, then even putting the entire tube won’t stop the more harmful solar exposure. Yet put even a small amount of factor 60 and you’re better protected than any amount of facto 5. I’d estimate that c02 has a weak factor 5 in climate factoring. No matter how much more is added, no increase in effect is discernible
**************************
Jim Masterson (00:46:03) :
I’ve tried modeling this for multiple layers, and it isn’t isotropic. You get more radiation going downward than upward. It’s another property of the model.
>> Are your numbers the result of measurements or modelling ? <<
They aren’t MY numbers, but Kiehl and Trenberth’s numbers. The answer is both. If you read their 1997 paper, the top-of-atmosphere (TOA) outgoing value as measured by the satellite observations from Nimbus-7 and the Earth Radiation Budget Experiment (ERBE) is 235 W/m^2. If 40 W/m^2 comes directly from the surface, then that leaves 235 W/m^2 – 40 W/m^2 = 195 W/m^2 for the atmosphere. The other 519 W/m^2 – 195 W/m^2 = 324 W/m^2 must be going somewhere else (down’s a good direction).
***************************
This is one of those ideas that does not pass the smell test. Either there is an inadequate understanding of what is happening or it's just wrong. It's kind of hard to "see" what's happening from the written description. Is there a chart that depicts their numbers somewhere? Also, if it is modeled, what mechanism does the model delineate to explain this counter-intuitive behavior? The obvious problem here is that the molecules will radiate in random directions. Also from morphological considerations, due to the curvature of the Earth and subsequently the atmosphere, actually there is a higher probability that a given photon will be radiated into space rather than hit the Earth. So, what is the mechanism responsible for this alleged behavior?
>> Jim (06:49:18) :
This is one of those ideas that does not pass the smell test. Either there is an inadequate understanding of what is happening or it’s just wrong. It’s kind of hard to “see” what’s happening from the written description. Is there a chart that depicts their numbers somewhere? Also, if it is modeled, what mechanism does the model delineate to explain this counter-intuitive behavior? The obvious problem here is that the molecules will radiate in random directions. Also from morphological considerations, due to the curvature of the Earth and subsequently the atmosphere, actually there is a higher probability that a given photon will be radiated into space rather than hit the Earth. So, what is the mechanism responsible for this alleged behavior? <<
The paper is online. Read it yourself (see figure 7). By all means don’t believe me. Model it yourself. Lots of things in physics are counter-intuitive.
Jim
Jim Masterson (07:21:35) : Thanks for the paper. I don’t have time to read it today, but I will get to it next week. It looks interesting.
***************
Jim Masterson (00:46:03) :
I’ve tried modeling this for multiple layers, and it isn’t isotropic. You get more radiation going downward than upward. It’s another property of the model.
*****************
OK, naive question here. I have an AMD Phenom 9850 Quad-core processor with 3.6 gig of RAM and 400 Gig hard drive.
1. Would your model run on my box?
2. Are you willing to share it?
George E. Smith,
“So the long wave Ir radiation absorbed by GHG molecules such as CO2 is rapidly transferred to the ordinary atmospheric gases as thermal energy, thereby raising the atmospheric temperature.”
The absorption of IR by greenhouse gases is irrelevant to the greenhouse effect. The only radiative physics that really matters with regard to GHGs is the final emission of IR to outer space.
You could think of it this way, if you like. The sun warms the surface, which warms the N2 and O2 touching it, which rises by convection to a high altitude, which transfers the energy by collision to H2O, which radiates to space.
The H2O radiating to space must do so at -24 C on average which is the amount required by Stefan-Boltzmann for heat balance, the N2/O2 at altitude is in thermal contact with it so must come to equilibrium at the same temperature, and once it’s done cooling and starts to descend to the ground again as part of the convective cycle, it’s temperature rises above -24 C as it gets compressed at lower altitudes. The temperature at the surface will therefore be considerably higher than -24 C, but not because of GHG absorption of IR.
“The question that I raised was by what rule are those ordinary atmospheric molecules/atoms prohibited from radiation due to their non-zero Kelvin temperature.”
Since I thought I already explained that, I suspect you probably don’t mean what I think you mean. Can you clarify or expand on why you’re asking?
“they must radiate since they contain accelerated electric charges due to the thermal vibration.”
Not necessarily. With a rotating N2 or O2 molecule, the acceleration on one end is equal in magnitude and opposite in direction to the acceleration on the other, so the waves they would emit will cancel.
“and is unrelated in any way to the nature or species of the GHG molecule, that capture the energy to warm the atmosphere.”
The atmosphere is not warm because GHGs capture the energy. This is the “trapped radiation” argument that I explained right at the start was wrong. Heat flows through the atmosphere by all sorts of mechanisms, including radiation, but the physics that decides what temperature it will be is just the bit I said. Convection combined with the change of pressure with altitude requires the temperature profile be a straight line with a given gradient, and the average altitude of emission to space fixes the straight-line intercept, because the temperature at the average emission altitude must be the Stefan-Boltzmann heat-balance temperature. The other heat flows will increase or decrease to push things back towards this state of affairs.
“It’s myth that gases do not emit black body (like) radiation and only solids do.”
Solids don’t emit perfect black body radiation, either. If solids don’t have energy levels in the right places, they’re completely transparent too. Take a close look at a Sodium Chloride crystal. The only difference between solids and gases in this regard is density.
I’ve heard this claim before as well about the “hidden heat”. A high school science student should be able to point out that this violates the law of conservation of energy. It does not simply “disappear” from detection. If the energy goes into some vessel, it should be possible to detect a heat anomaly from that vessel, because heat does not just stay somewhere. An energy system constantly seeks equilibrium and so heat travels to cooler areas. This movement can be detected by looking for a temperature anomaly from a heat sink.
Further, “temperature” (heat energy actually, of course) cannot “flow” from a cold source to a hot source.
Therefore, the AGW extremists’ mysterious pipeline” must (eventually) be hotter than what it will end up transferring heat to. So, if the “globe” is sucking in all of today’s heat energy, plus all the heat energy from 1998 through 2008, but world air temperatures have been decreasing over this same frame, where is the heat going? Hot air could heat up colder ocean waters anywhere they touch, but then how would the still-colder-on-average ocean waters begin heating up the average air masses above them? Why did ocean waters begin assuming this heat energy beginning in 1998 – but stop doing that absorption in 2025?
If the heat is going into the “oceans” (claiming the very true 1000x larger heat capacity of water compared to air), then how will the water suddenly and mysteriously change properties/methods/behaivor and suddenly begin heating up the air after 2025? There will not be enough of a temperature difference to move thirty years accumulated heat energy from the water “up” to the (suddenly assumed much colder for some reason?) air.
If the AGW extremists are going to claim that there is a thirty year “cycle” when the two masses (oceans and air) trade heat flow directions, where are the measured differences in the past thirty year cycles since 1890?
>> Jim (10:30:08) :
***************
Jim Masterson (00:46:03) :
I’ve tried modeling this for multiple layers, and it isn’t isotropic. You get more radiation going downward than upward. It’s another property of the model.
*****************
OK, naive question here. I have an AMD Phenom 9850 Quad-core processor with 3.6 gig of RAM and 400 Gig hard drive.
1. Would your model run on my box?
2. Are you willing to share it? <<
I have put the Kiehl-Trenberth model in an Excel spreadsheet (it’s that simple). Does your computer run MS Excel? I have no problem with sharing the spreadsheet model.
The atmosphere-layer model is mathematical. It runs on a piece of paper. I’ve generated similar atmosphere-layer models (which is why I know these models do not radiate 50-50 upward-downward). The layer model doesn’t match Kiehl and Trenberth’s numbers, so I don’t include it in the spreadsheet.
Jim
Just for reference: Black-body radiation is only emitted by black or partially black bodies. Transparent layers in the atmosphere only transmit or pass black-body radiation, whether that comes from the sun at 5780 deg K or is retransmitted by the earth at 300 deg K.
I believe that each “greenhouse” gas blocks transmission by converting the photons to heat (as phonons). This occurs at specific frequencies, determined by the molecular of structure and vibration modes of each gas. Generally, these absorption frequencies are in the infra-red range. I believe the heat generated by this process is primarily conducted to the surrounding atmosphere.
The “greenhouse” effect is caused by the fact that a higher temperature is required to force the same amount of energy through the remaining open bands. The sun is so hot that most solar energy arrives in the visible or optical frequency range where the atmosphere is transparent.
Please note that these comments do not apply to dust particles or clouds in the atmosphere. Carbon dioxide, the most notorious “greenhouse” gas, is optically transparent.
Dear bloggers,
For what it’s worth, my simplistic understanding as follows:
Ignoring reflective clouds, the atmosphere is essentially transparent to the high frequency, incoming radiation from the Sun and the Sun’s radiation is overwhelmingly the only significant source of energy to the Earth. The land and the oceans absorb this radiation and are warmed. However, the land and the oceans also absorb and emit low frequency radiation (IR) and only that portion of the emitted radiation that escapes directly to space can cool the Earth. Some of the Land and oceans’ emitted radiation is absorbed by the atmosphere. The atmosphere also emits radiation and similarly only that portion that escapes directly to space can cool the Earth.
The basis of AGW theory is that increasing atmospheric CO2 allows the atmosphere to absorb more radiation from the land and oceans that would otherwise have escaped directly to space. Therefore, it follows that the atmosphere must warm due to the proposed increase in its radiation absorption. If it hasn’t warmed then it hasn’t absorbed additional radiation.
Mark A.
PS Excellent site Anthony, I am learning heaps.