or Should the Montreal Protocol be terminated?
Mike Jonas,
For years, I had wondered about the Montreal Protocol and the ozone hole that started in 1979, and whether the science behind it all had been twisted to suit DuPont or others. But where would you look to find the answer? Then, last year, I saw a report that the ozone hole, which had been recovering as expected, was suddenly as large as ever. It could have been this report. So I started downloading some data. I knew that it would be a lot of work, I had no idea exactly what I would be looking for, and I suspected that I wouldn’t find anything. Lots of others must have looked and found nothing.
I was stunned by the first significant thing I found: There was an ozone hole long before 1979.
But first, why did I suspect twisting by DuPont? Well, I think ‘everyone’ did back then, but no-one could prove anything. It certainly looked like some sort of shenanigans were going on. For a start, the ozone hole was defined in the 1980s as the area of ozone less than 220 Dobson Units (DU). Why precisely 220? Well, it was clear that the number 220 was chosen so that the ozone hole started in 1979. A different number would have given a different start date, and it seems they really did want the ozone hole to start in 1979. It had nothing to do with 220 DU being some sort of safe level, the number was plucked out of the air just like today’s ‘need’ to restrict global warming to precisely 1.5C. DuPont’s lucrative patent for Freon (basically a chlorofluorocarbon (CFC)) expired in 1979. In 1974, with their Freon income to protect, the Chair of DuPont said that “ozone depletion theory is “a science fiction tale … a load of rubbish … utter nonsense”“. But in the 1980s DuPont changed its tune when it was clear that they would profit more from a replacement product. The prime function of the Montreal Protocol seems to be to sustain the manufacture of unnecessarily expensive refrigerants. Incidentally, Wikipedia joined in the shenanigans, no doubt for its own purposes, using the ozone hole to attack Fred Singer. Wikipedia (thank you, Wayback Machine), said: “Some atmospheric scientists (for instance Fred Singer, founder of SEPP and also a global warming skeptic) and industry-sponsored advocacy groups question or completely deny a link between CFCs and ozone depletion. It is fairly common to see completely nonsensical arguments …“. Today’s Wikipedia entry has no mention of Fred Singer or the “completely nonsensical arguments” which appear not to have been said by him anyway.
But I digress.
I downloaded a lot of data files for the southern hemisphere (Willis Eschenbach knows all about how many files you need to download sometimes), and loaded them into old-fashioned spreadsheets in my ancient desktop (sorry, w, I never got round to using R). When I looked at the ozone data for the south pole, there were ozone holes in 1964, 1966, 1969, 1974 and 1977. They may have been less pronounced than in more recent years, but they were there. I have written it up, it has been published, and my paper is open-access at WJARR. CFCs were oh so close to zero at the first of those ozone holes …….
There are obvious questions arising from this: If the ozone measurements at the South Pole showed column ozone repeatedly below 220 DU well before 1979, how could NASA’s Ozone Watch say ” total ozone values of less than 220 Dobson Units were not found in the historic observations over Antarctica prior to 1979“? Were NASA lying, or had they simply not seen the data? If they hadn’t seen the data then did they make their statement without checking the data? The South Pole would have to be the first place to look for pre-1979 ozone data relevant to the ozone hole. Or had the data been found to be wrong? – I could find no reference in the literature to the ozone data having been wrong.
If the gatekeepers ever take the trouble to refute my finding, maybe they will do it the way they refuted Qing-Bin Lu’s 2022 finding of a tropical ‘ozone hole’ – they refuted it by deflection (I refer to Qing-Bin Lu’s paper in my paper). Qing-Bin Lu was very careful to say that the standard definition of an ozone hole – ozone less than 220 DU – was not used, but the critics managed to leverage “looking at percentage changes in ozone, rather than absolute changes” into “the research was riddled with serious errors and unsubstantiated assertions“. The detail in the criticisms actually supports Qing-Bin Lu’s findings, but it seems the critics just didn’t want an ozone hole anywhere outside the Antarctic. Another curious thing about it all is that Qing-Bin Lu was giving them another opportunity for scare-mongering – why didn’t they take that up? Was it because this ‘ozone hole’, reportedly seven times larger than the Antarctic ozone hole and above where a lot of people actually lived, was natural?
The other thing of note that I found as I ploughed through the data was that the annual ozone minima not only didn’t correlate very well with CFC data, but also looked more like a phase change. Here’s my Figure 4, illustrating it:
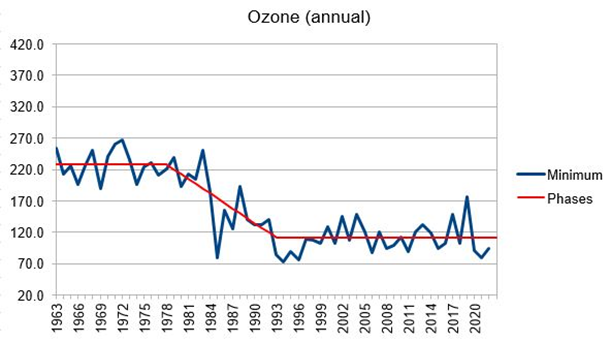
I was careful to derive the phases mathematically not manually, but it would obviously be credible to draw the phases differently. I did a check through the literature, and Bingo! an early 1980s phase change in south pole surface temperature had been detected (Lazzara et al 2012). Then I checked the south pole radiosonde temperature data, and there was a temperature phase change in the lower stratosphere and in the troposphere too. Everything tallied. Another thing I discovered after submitting the paper was that Qing-Bin Lu’s tropical ‘ozone hole’ is thought to have started in the 1980s – that tallies too.
My paper covers both the non-correlation of ozone with CFCs and the matching phase changes in ozone and temperature. Those who don’t like non-emphatic words like ‘could’ and ‘suggests’ will be disappointed, but it really isn’t possible to prove much definitively from correlation or non-correlation with limited data. The analysis is complicated by the relationship between ozone and temperature being bi-directional (each affects the other), and of course, air moves. My paper does not argue against chemical reactions involving ozone and CFCs, but it does highlight that it really isn’t known just how much of the ozone hole the CFCs are responsible for.
Incidentally, the fact that air moves is very relevant. Say there’s an ozone hole near the south pole. Then it moves a little. One day, ozone at the south pole is over 220 DU, the next day it’s under. The ozone hole hasn’t changed at all, it just moved a little. There are many days in the south pole ozone record where there is a large change from the previous day.
My paper’s Abstract:
Abstract
Depletion of ozone over Antarctica was first observed in the late 1970s, and discovery of the Antarctic ozone hole was announced in the 1980s as having started in 1979. The ozone hole was defined as the area with total column ozone less than 220 Dobson units. Analysis of ozone, temperature, chlorofluorocarbon and nitrous oxide data from 1963 onwards suggests that the annual ozone minimum at the South Pole is related to lower stratospheric temperature independently of chlorofluorocarbons and nitrous oxide. There were ozone holes, ie. column ozone less than 220 Dobson Units, at the South Pole in several years before 1979 (the date that the ozone hole is reported to have first appeared) when chlorofluorocarbon concentrations were much lower than today and lower than in 1979. An early 1980s phase change in the lower stratospheric temperature at the South Pole at altitudes between 250 hPa and 100 hPa, and at some lower altitudes, coincides with a phase change in the annual South Pole ozone minimum. The phase change is not visible in chlorofluorocarbon or nitrous oxide data. This raises the possibility that, over a multi-annual or decadal timescale, lower stratospheric temperature has more effect than chlorofluorocarbons or nitrous oxide on atmospheric ozone concentration over the South Pole. Alternatively, temperature and ozone may both be reacting to some other influence.
The paper is open access and can be read in full at https://wjarr.com/sites/default/files/WJARR-2024-0531.pdf.
I mentioned above that this study would be a lot of work. A lot of the work is ploughing through the literature to see what has been discovered already, and checking that what I was finding hadn’t been found or refuted already. As I read the literature, I formed the opinion that a lot of scientists were sceptical of the CFC-depletion narrative, but they couldn’t quite put their finger on anything that definitively disproved it, so they were restricted in what they could say about it. Maybe my findings will help. Or, if NASA really did lie about ozone levels, or even if they simply ignored the data, then should the Montreal Protocol be ditched anyway?
There’s a really interesting statement in a phys.org article: “Lu’s observation of the ozone hole comes as a surprise to his peers in the scientific community, since it was not predicted by conventional photochemical models. His observed data agree well with the cosmic-ray-driven electron reaction (CRE) model and strongly indicate the identical physical mechanism working for both Antarctic and tropical ozone holes.“. Unfortunately, I didn’t see that until later.
I ask above whether the Montreal Protocol should be ditched. My study didn’t look at CFC chemistry, so it isn’t about that. The question is whether the Montreal Protocol is based on a lie. If CFCs are to be banned, they should only be banned for a valid reason. And remember, CFCs were very close to zero at the first of those ozone holes and there is no fuss about natural ozone depletion over where millions of people actually live. But in the paper I couldn’t say explicitly that anyone lied, so I said we would have to wait until ozone levels correlate with one of temperature and CFCs more than with the other. That could take a long time, especially if there’s another phase change. The reality is I suspect even worse – we will have to wait for the gatekeepers to get out of the way.
PS.
In the man-made CO2 “climate change” narrative, increasing CO2 supposedly cools the stratosphere. A global phase change in stratospheric temperature followed by decades of no temperature trend would pretty well knock that narrative on the head too. Qing-Bin Lu: “all the datasets show that significant O3[ozone] or LST [Lower Stratospheric Temperature] reductions only occurred in the 1980s and 1990s with no significant trends over the past ∼25 years. This is similar to the observation by Polvani et al. and that reported in the newest IPCC AR6. The latter states “most datasets show that lower stratospheric temperatures have stabilized since the mid-1990s with no significant change over the last 20 years” “. [my bold] Maybe that’s another reason why they were so keen to tear down Qing-Bin Lu’s paper.
Addendum from Charles
The article above leaves out the fact that satellite observations began just as the “Hole” was “Discovered” with the launch of The Nimbus 7 satellite on October 4th 1978. The Nimbus 7 contained the TOMS (Total Ozone Mapping Spectrometer).
Initially, the dearth of ozone was not noticed because the data was so far off from the expected range that it was discarded as invalid by the data processing algorithms. When the fact that something like 2/3rds of the instrument’s measurements were being discarded was finally noticed and analyzed, that’s when the panic set in about the “Hole in the Ozone”.
While the article above does cite a distinct global polar temperature regime change in the late 70’s that probably affected the ozone levels, it just seems too much of a coincidence that the panic set in just as a new measurement technique was deployed. Supposedly the TOMS measurements match ground based measurements to a reasonable degree; however, like the fact that “sea level acceleration” only shows up in Jason satellite measurements and not in tide gauges, there is a not insignificant chance that the “Ozone Hole” is an artifact of instrument changes.
Here is a clip from a PBS documentary on the discovery of the “Hole” describing the discarding of out of expected range measurements and the ensuing panic when the data didn’t match preconceived expectations.
I have never understood the arguement about CFCs being the cause the Ozone Hole for the following reason. Most CFCs are produced/used/escape in the Northern Hemisphere and yet the Ozone hole is at the South Pole. Why is there not a bigger hole at the North Pole and how/why do all the CFCs migrate to the south pole? Do they prefer it there?
I have always suspected that there has always been an ozone hole so, thank you for showing that.
The other side of the depletion equation is creation of ozone. Ozone is beneficial because it absorbs UV. Ozone is created by the absortion of UV by molecular oxygen, of which there is plenty. There is a depletion of ozone at the poles, due to depletion of sunlight, not the abundance of CFC’s.
Ozone is metastable and has a relatively short half-life. Thus, it starts to decay during the dark Winter. However, the rate of decay usually accelerates in the Spring, when the sun comes back above the horizon. Therefore, there appears to be something to the claim about photo-catalytic destruction, when it is cold enough.
However, something that is rarely commented on is that during the southern Winter, when the ozone starts to deplete, there are typically high levels of ozone outside the perimeter of the circumpolar vortex. One can see that, despite the ozone being generated in the tropical stratosphere, the concentrations over the tropics are lower than near the Antarctic Circle. All the NASA ozone maps show these anomalous concentrations of ozone outside the circumpolar vortex. When the sun finally gets high enough, the circumpolar vortex breaks up, and both the very high and very low ozone levels disappear, apparently from mixing of air masses. There is still a lot we don’t know about what is going on. However, it appears that temperatures play a much more important role than CFCs.
Thanks Clyde, I’d suspected my understanding was too simplistic.
From: https://csl.noaa.gov/assessments/ozone/2010/twentyquestions/Q10.pdf
“Average daily minimum values over Antarctica are as low as −90°C in July and August in a typical year. Over the Arctic, average minimum values are near −80°C in late December and January. Polar stratospheric clouds (PSCs) are formed in the polar ozone layer when winter minimum temperatures fall below the formation temperature of about −78°C. This occurs on average for 1 to 2 months over the Arctic and 5 to 6 months over Antarctica (see heavy red and blue lines). Reactions on liquid and solid PSC particles cause the highly reactive chlorine gas ClO to be formed, which catalytically destroys ozone (see Q9). The range of winter minimum temperatures found in the Arctic is much greater than in the Antarctic. In some years, PSC formation temperatures are not reached in the Arctic, and significant ozone depletion does not occur. In contrast, PSC formation temperatures are always present for many months somewhere in the Antarctic, and severe ozone depletion now occurs in each winter season (see Q11). (Note that the dashed black lines denote the upper limits of the Antarctic temperature range where they overlap with the Arctic temperature range.)”
Also: https://uk-air.defra.gov.uk/research/ozone-uv/moreinfo?view=cfc-stratosphere
That’s all very nice and all but it doesn’t really explain the observed smaller ozone hole over the Arctic, does it? That the Arctic also has a hole is an established fact and a very quick search will turn up multiple articles discussing it.
Yes it does, only gets cold enough for shorter periods of time
But why is there an ozone hole at the Arctic at all? Surely, according the theory expounded at length, it should be inimical to noticeable ozone depletion in the Arctic?
“but it doesn’t really explain the observed smaller ozone hole over the Arctic, does it? “
It does.
The Stratospheric vortex over Antarctica is much more intense than the Arctic one, and as such is much harder for atmospheric waves to penetrate (SSWs), thus it gets much colder than the temperature required for the chemical process that attacks O3 and for longer.
Yes, it’s all about the chemistry. The British Antarctic Survey discovery of the ozone hole and their publication of their results in ’85 was the result of measurements they had carried out since ’56 which showed the decline which accelerated during the ’70s.
ozone_graph2.gif
I like this bit of honesty. Wish all climate “research” fessed up.
Greytide
The answer about the N v.s. S pole is that it is not nearly as cold and stable in the North, and there is no strong circumpolar wind herding the gases in the upper atmosphere into a distinct patch. If the Earth were to tilt 20 degrees, that pattern would be broken up and the ozone would be replenished from the tropics where most of it is created (due to sunlight). So it is only that isolation which allows the concentration to drop suddenly in spring and then open the cooling vent.
If the hole were closed it would warm the atmosphere detectably everywhere. It would be like turning off the air conditioner on a hot day.
That’s fine but it doesn’t explain why the CFCs migrate to the south when they are mostly released in the North.
Mixing takes some time generally, and heavier molecules move more slowly than others. Further, mixing between hemispheres might take months to years, but entropy makes uniform mixing the favored state regardless of entry point.
CFC concentrations are in fact slightly lower in the southern hemisphere because of mixing time lags.
https://www.ncei.noaa.gov/access/ocean-carbon-acidification-data-system/oceans/CFC_ATM_Hist2015.html
But there is an Arctic ozone hole, against all odds. I think the temperature and winds are a minor player, the main factor is probably the axial tilt.
O3 is diamagnetic and so moves away from the poles more quickly, relative to O2.
Not an answer or it would be the same at both poles.
Well, it’s part of the answer. Obviously it ‘s a small effect relative to atmospheric mixing. And the circular winds around the south pole mean the affected atmosphere is (a little) isolated form the rest of the world, which is not so at the North Pole.
The point being that it explains how gasses emitted north of the equator travel to the southern pole. They don’t. It’s not a chemical effect.
Based on your claim, ozone should show a gradient getting stronger away from the poles, especially in the Summer. That isn’t observed.
Diamagnetism will not overpower the wind. It’s not that powerful.
But within the polar vortex, it will cause an ozone donut.
Can you point to an ozone map that demonstrates your claim?
How about this?
ESA – Map of the ozone hole over the South Pole on 16 September 2021
It’s marked:
Interestingly, one can see a shift in the growth of the ozone hole to August.
Do you have similar data for the Arctic ozone hole?
Sorry.
https://www.cpc.ncep.noaa.gov/products/stratosphere/polar/polar.shtml#plot1
You can find it at https://ozonewatch.gsfc.nasa.gov/NH.html
No I’ve seen that, I was hoping that someone might be able to show annual variation rather than just the last 6 days.
Go the the site I linked to and click on meteorology , here’s the current data:
https://ozonewatch.gsfc.nasa.gov/meteorology/figures/ozone/to3capn_2023_toms+omi+omps.pdf
During periods of low solar activity, ozone production over the equator in the upper stratosphere decreases, as evidenced by a drop in temperature.
As a result of the breakdown of oxygen O2 in the upper stratosphere by photons shorter than 242 nm, excess energy is transferred to neighboring air molecules, causing an increase in their kinetic energy (temperature increase).
The molar mass and molecular weight of CCl2F2 is 120.914.
The basic CFC is a heavy molecule and getting it high in atmosphere is hard work. The ozone hole was known for decades.
https://gml.noaa.gov/ozwv/dobson/
PSC were blamed as the catalyst for the chlorine in CFC to destroy O3. By getting rid of CFC’s they also hurt fire fighting by getting rid of halon. The big red bottles were taken off flight lines and systems out of engine rooms on ships.
The sun effects the production of ozone.
Halon fire extinguishers were taken from airliners, too. The rain repellent sprays for windscreens also contains cfc’s, and was removed. After that, we would bring some Rain-X along to wipe the windscreens before flying.
(You’d think that a several hundred mph breeze would blow the rain off the windscreen, but it doesn’t. Go figure.)
There’s nothing more exciting when you run into a smoke filled computer room, watch your boss pull the Halon discharge handle … and nothing happens.
You’re lucky it didn’t go off. When we had a halon system we were told to get out as quickly as possible or end up dead. Apparently it condenses in your lungs and you basically drown.
Jeff I think you were lied to. One benefit of halon like DDT is it was safe around humans.
https://www.firetrace.com/fire-protection-blog/why-is-halon-banned#:~:text=Halon%20Gas%20Effect%20on%20Humans,%2C%20documents%2C%20and%20valuable%20assets.
The reason for getting out of the room as fast as possible is halon produces some very nasty compound when exposed to fire. If there is no flame, you are safe but linger when there is a flame and you don’t last long. I have got a whiff of freon 12 that was exposed to a flame and you know it’s something you shouldn’t be breathing. Fortunately it was just a little in one inhale so it didn’t cause any damage.
The second downside is it takes the oxygen out of the air by displacing it. Remain in the room and you will die from suffocation.
From the link provided:”Overall, Halon is safe around humans and can be used in occupied spaces. Halon suppression systems became widely properly because Halon is a low-toxicity, chemically stable compound that does not damage sensitive equipment, documents, and valuable assets.”
lack of halon extinguishers in a fire in an exchange that took our entire town off phone n net for 10days. no food no fuel no banking no nothing
It can be seen that the temperature of the stratosphere above the equator is falling again this year in the upper stratosphere.
The decrease in ozone can also be observed in the lower stratosphere, as ozone absorbs UVB radiation and is broken down by these photons into oxygen molecules.
Yes, the Montreal Protocol along with all the other government actions that aren’t having their planned for desired effect should be abandoned, and that would be most government actions.
I recall in the late 1990s being at a 3rd party political meeting and spending time talking with an older GE refrigeration engineer who said he and his colleagues were very upset with GE management when they caved in to the “CFSs are bad” crowd because the alternatives made inferior refrigerators. He had a small book he wrote on the subject. This was where I first heard that the whole CFS/ozone thing corresponded to the end of Dupont’s patent protection for freon. And who had the patent on the replacement? Why Dupont.
Worse than that was that in the protocol was a provision that paid countries to get rid of their CFC’s. So China manufactured then destroyed CFC’s and got paid by us.
I remember the era well. We had to go out and buy all kinds of recovery equipment, get our guys “ certified” to handle all refrigerants and needed a license to purchase the remaining ( and rapidly inflating) mixes that were being phased out under gov mandate. Ironically I just discovered my license to purchase R12 in an old wallet from the 1990s when packing up to move to the country.
As I understand it, ozone is created by the Sun’s radiation. Ozone has a limited life, mostly dependent on temperature. At minus 50 C, it’s half life is about 3 months and at 20C it’s a few days.
When the poles go into their respective winters, there is no significant sun for 6 months.
How is a hole not supposed to appear?
Exactly.
The hole develops in the spring because the return of sunlight allows the ClO cycle to start and break down the O3. Later in the season the UV accelerates the production of O3 exceeds the destruction by the CiO cycle.
In the winter when the sun is set the CiO cycle shuts down and no further reduction in O3 takes place.
Got this from the net:
OZONE HALF-LIFEThe half-life of ozone refers to the amount of time required for half of a given quantity of ozone to decay into oxygen. Ozone is an unstable gas and readily decomposes into oxygen. Therefore, the ozone halflife is an important factor to consider when determining its effectiveness in various applications.
Typical Gaseous Ozone Halflife time as a Function of Temperature Half-life time Temp ∼ 3 months -50 ºC ∼ 18 days -35 ºC ∼ 8 days -25 ºC ∼ 3 days 20 ºC ∼ 1.5 hours days 120 ºC ∼ 1.5 seconds 250 ºC
As I read it; ozone is very unstable, readily decomposes to oxygen, and will not shut down its own destruction due to the sun setting; and is only slowed down by low temperatures. Would be interested in where you got your information.
Best,
That data is for atmospheric pressure, we’re talking about the atmosphere above 15km. The O3 concentration below ~12km and above ~22km doesn’t decrease (T -80ºC and -60ºC respectively).
The problem is accentuated by the fact that the Brewer-Dobson circulation is interrupted by the Antarctic circumpolar vortex during the Winter months. Thus, the decaying ozone can’t be replaces by new tropical ozone during the Winter months, unlike during the Summer. There is some loss of O3 during the Winter, just not as rapidly as during the Spring.
Wouldn’t the slowing of ozone loss be due to colder temperatures during the winter? And in the spring isn’t the increased rate of loss of ozone due to warmer temperatures then offset by the sun’s UV rays producing even more ozone? And on top of that, would any decrease in O3 through CFC depletion be offset by O3’s natural decay? Or, said another way, if O3 is going to revert to O2 anyway, what function can CFC’s have in the depletion of the ozone in our atmosphere? The physics of natural ozone depletion is in the paragraph I took from the net is a function solely of time and temperature.
As mentioned above that data is for O3 at the surface not at altitude.
While the circumpolar vortex is active, there is little to no warming. The most rapid losses of ozone occur when it is coldest, which supports the photocatalytic reaction hypothesis.
So, I went to Wikipedia for the information you are citing. Not the best source, but it parrots what you are telling me. But I don’t believe it. Why? Because the hole in the ozone layer is not acting as predicted by models. There is something being missed. But I accept your explanation for the bulk of what is happening within the ozone layer. Thanks.
Best,
SMS
“How is a hole not supposed to appear?”
Ozone continues to be transported to the South Pole during winter. As do the so called Ozone depleting gases.
Your reasoning for the appearance for the hole is not appropriate, as the hole is created by other mechanisms.
If your logic is true, then the hole is not a hole. The degradation of the ozone layer is not at the poles, but evenly throughout the stratosphere.
It is a canary in a coal mine. If it works, let’s try it with CO2, and then with COVID…
Don’t you know that when we humans discover a phenomenon it must have catastrophic implications and we humans absolutely must be the culprits. When did they stop teaching logic and examples of fallacious reasoning in University. Nice essay, thanks.
‘Humans are to blame’ is confirmation bias for so many of the leftist eco-loons that have zero knowledge but a lot of ‘feelings’ on the subject. It resonates with their feelings that modern society must be a bad thing because it has separated humanity from nature, without the understanding that this is what enables their lives to be longer, easier and more comfortable.
This is a most relevant article and comment. Prof Qing-Bin Lu (University of Waterloo, Ontario) has written papers on the topic for +10 years and has not been credited enough with his proposed and demonstrated chemistry.
Not mentioned above, is that the ozone hole is a massive heat vent for the planet which opens and closes. One of his comments is that he can explain all global warming and cooling from ~1960 based on the ozone forming and destroying processes alone.
There are observed chemical processes that destroy ozone, a major one being bromine, whether from human emissions or the oceans (oceans are the major source, and obviously natural).
Prof Lu first proposed a chemical path mechanism in a paper, a follow-on detailing it further using satellite data. Then he managed to replicate the chemistry in his atmospheric test lab in Waterloo proving the chemical path was viable.
This provoked an almost ridiculous response from the climate agitators because there were two huge groups endangered by his findings: the CO2-regulates-all-temperatures group and the CFC-destroys-the-ozone group. Both have massive industrial consequences. There is a blog site run by “Eli Rabbet” which took on Prof Lu who politely participated. It was truly humorous because Eli claimed to have replicated his study and found no such effect or correlation. Prof Lu replied that he (Eli) had deliberately used data from a satellite that didn’t pass over Antarctica. Duh.
In support of Dr W Soon’s claim that, “It’s the sun, stupid,” the GCR’s, which play a major part in ozone destroying chemistry activation, are modulated by solar activity. As I understand it, when the ozone concentration is low, the Antarctic region vents heat to space more effectively than when it is high, because ozone is a very effective GHG due to where it is.
Papers on the basics:
Lu, Q-B 2009 Correlation between cosmic rays and ozone depletion, Phys. Rev. Lett. 102, 118501 (1-4)
Lu, Q-B. 2010a, Cosmic-ray-driven electron-induced reactions of halogenated molecules adsorbed in ice surfaces: implications for atmospheric ozone depletion and global climate change, Phys. Rep. 487, 141-167 (published online in 2009).
Lu, Q-B 2010b What is the major culprit for global warming: CFC’s or CO2? J. Cosmology 8, 1846-1862.
Paper that provoked the usual suspects:
Lu, Q-B 2013. Cosmic ray driven reaction and greenhouse effect of halogenated molecules: culprits for atmospheric ozone depletion and global climate change. Int J. Phys. B27, 1350073 (1-38).
Replies came from Rolf Muller, Jens-Uwe Grooß, Dana Nuccitelli et al, and then Prof Lu replied (2014) in Int. J. Mod. Phys. B28, 1475003.
You can see he was on the right track in 1999. Find papers by Lu, Q-B and Madey, T.E. There are at least 5.
Mike,
I gather you are relying on radiosonde data pre-1070, and I presume you are integrating the measured O3 in the observed column. But the Dobson unit is the O3 between surface and space, and radiosondes are seeing only part of that. Is that why you are getting sub-220 values pre-1979?
Do the post 1979 calculated by Mike Jonas agree with the official numbers? If they do then where’s the problem?
Missed some history classes in school, Nick?
🙂
Obviously a typo. Hardly worth joshing about.
Well, nobody was getting medieval with him, were they?
I wonder if Duke William brought the radiosonde technology with him when he invaded 4 years before? Were they mentioned in the Domesday book?
Nick and others jumped on me pretty good a few days ago when I made a mistake in a comment.
Fair cop.
When I give some out, I always expect to get some back at their earliest opportunity.
The reverse applies too.
I enjoy a bit of back & forth with Nick.
He’s clearly very knowledgeable and with expert numeracy skills and intelligence.
It does confound me however how anyone as clever as Nick can get smoked by the agw conjecture.
The whole construct sounded, looked and smelled like bullshit right from the get-go.
Many other ideologues are similarly smoked I guess.
Like they said in the X-Files –
“I WANT to believe!”
The paper lists the data sources:
Radiosonde daily temperature data is used for the South Pole at a range of altitudes and times of day (data is available from 1961 for temperature) [4].
the USA’s National Oceanic and Atmospheric Administration (NOAA) Amundsen-Scott (South Pole) station daily ozone data (from 1963) [5].
NOAA Southern Hemisphere annual CFC and N 2 O data (from 1765, although all CFC data is zero until 1936) [6].
So the ozone data is from the South Pole station, radiosondes were used only for temperature data.
Mike,
OK, I looked at that South Pole data, which has quite a lot of missing days. Here is a plot of the number of days in each year when the ozone reading was less than 220
You can see that before 1979, there were just a few years with a few days in that category. They weren’t much below, and the effect might very well have been removed by a reasonable noise filter. Something happened after 1979.
In that data set there are quite large chunks of data missing. I don’t think this affected the conclusion about years before 1979, but probably adds to the irregularity of later data.
Yes, Nimbus 7 was launched. Prior to the launch of the TOMS in late-1978, the only measurements available were after the sun came up over the horizon, and what was measured was the slant-range path between the ground station and the sun. What was measured was a different geometry than the satellite measurements, and was much more limited in coverage.
But these are results from the same ground based spectrophotometer that Mike used for his article. Despite what he says, it shows essentially no ozone hole activity before 1979.
Here is the difference between 1974, counted here as an ozone hole year, and 1994. Plot is of ozone measure vs day of year. Note that 1974 had a few values just dipping below, around mid year. 1994 had a deep dive in Oct/Nov, the months of the actual ozone hole.
Nice summary. I haven’t looked at any detail into ozone hole worries for a long time, but I seem to recall that much destruction of ozone occurs at heterogeneous reactions on ice crystals. A cooling stratosphere might lead to more such ice crystals. Moreover, I have thought at times about the form of ice crystals being a function of temperature — prismatic needles vs. plates etc. and as such might provide very different surface environments for these reactions, a basis for phase changes, in other words.
Looks like a big research project to me.
I am always suspicious of panics and hysteria related to enviromental concerns because they so often turn out to be wonderful rent-seeking opportunities.
Kevin K
You are correct. See the papers by Prof Lu listed above. The chemistry of the crystals and gases are activated by free electrons. It also explains why the ozone disappears with the rising sun at the end of winter, not all the time
No one ever considers that ice crystals have electrostatic charge.
I have some friends who are total magick woo-woo crystal types. They have a business where they cast their magick crystals in some polymer and make all manner of ‘healing’ appliances. One of which is a drinks coaster. Put a plastic tumbler of water atop the coaster in the freezer, and it creates spectacular images in the ice.
This leads me to ponder … charged particles bobbing along in the equatorial current, with a velocity perpendicular to the magnetic field …
What I meant from this, is that a needle shaped crystal would have a very strong charge at the needle point, just as a lightening rod shape enhances the charge (attractance) of the rod.
Free energy?
Just add hugely expensive collectors?
As the alternatives to “Ozone Depleting” CFC’s are less efficient, I wonder how much more CO2 is being produced? Yes, newer equipment is more efficient than older CFC units due to design improvements, but if those and other modernizations were made using CFC’s as the refrigerant, they would be even more efficient.
Make windmills, PV solar arrays, and related backup/transmission lines: More Net CO2
Make systems convert to less efficient refrigerants: More Net CO2
Why are we doing this again?
First — thanks for doing the work and writing the paper.
Back when this issue was presented on WUWT (~12 years ago), I informally looked at the compounds involved and found reports that an active ocean surface ejected plenty of the chemicals being blamed as “bad” by the activist community. I concluded they were p—ing up the wrong tree.
2nd: Wikipedia, at that time, was a cesspool of misinformation about AGW, and likely still is.
Well you were apparently misinformed, the Chlorine compounds contained in the ocean are water soluble and at a result are washed out of the atmosphere and don’t make it up high enough to cause problems. By contrast the Freons etc are not soluble or reactive and so do reach high enough in the atmosphere to be broken down by UV and react with the O3.
The problem with calling low ozone a “hole” is that most people think hole and think missing when in fact is simply “lower than X”, with X being an arbitrary number … just like saying the Oceans are getting “more acidic” when in fact they “may” be getting less basic … both Ozone Hole and Ocean Acidity are propaganda scare tactics …
Yes, it should more properly be called a ‘thinning’ or a ‘lessening’ rather than a ‘hole’ but that is what everyone understands as the situation now.
Would that hole be between the ears?
A hole between the ears is generally called a nostril – that’s a free anatomy lesson for you!
But a hole is more scary sounding than a thinning.
Actually it’s called a ‘hole’ because all the O3 between 15km and 20km disappeared when normally that is where the maximum O3 concentration is (typically ~15 mPa).
I read it as being a depletion of O3 between 14 and 21 km, not a complete absence. But that’s still only about half the thickness of the ozone layer (depending on the source it goes up to anywhere between 35 and 50km) so still a lessening or thinning, technically not a whole ‘hole’.
Well if you’re thinking of it as a vertical ‘hole’ then it is a ‘thinning’, if you think of it as a horizontal ‘hole’ then it looks as below. When we had the stronger holes it was pretty much a complete absence between 15 and 20km.
SouthPoleMin_profile_1994_2001.png
That is only important for certain geometries of incoming light rays. The sun never comes in vertically.
What does that have to do with it? The O3 between 15km and 20km is destroyed.
The importance of ozone is that it absorbs SW UV, which is potentially dangerous to living organisms. The absorption is a function of both the concentration of ozone and the path length of the incoming light, integrated over the entire ray path. Just as much UV can be absorbed by a long path (oblique) through low ozone concentration as would occur with a short path (vertical) through high ozone concentration. Actually, with the sun on the horizon, the path of rays may take them through the anomalously high concentrations outside the circumpolar vortex. Unfortunately, little has been published on the actual surface UV flux.
If the light arriving on the ground comes in mostly above or below the depleted altitudes then the ozone depletion has no practical consequence. What is needed is a thorough light ray tracing taking into account the topography of Antarctica and the variation of ozone with altitude.
You are aware that the Earth is a sphere I take it?
And in the absence of ozone, more UV with the capability to create ozone will make it to lower levels, with the major difference of ozone having a shorter half-life in the denser atmosphere.
Also, the sun never gets directly above the so-called ‘hole’ and what light does make it to the surface has a long slant-range through air that is enriched in ozone from the Brewer-Dobson circulation.
Story Tip
Very interesting case history in the NY Times on the Dryas Comet theory. With some striking quotes that are reminiscent of the way global warming has taken off. There is little or no evidence for the Comet hypothesis, and yet its become widely believed. In this case not so much by academics, but by a smallish group of advocates and, more surprisingly, by the general public. Despite widespread skepticism by most serious academics, little or no evidence for it, and much against. So why?
https://www.nytimes.com/2024/03/05/magazine/younger-dryas-comet-civilization.html
I have yet to see any actual, real evidence that the ozone hole did not exist prior to 1979, nor have I ever seen any actual, real proof that human activity has anything whatsoever to do with it. All the actions forced on us to date have had ZERO effect on the ozone hole. It grows and shrinks in a quite cyclical manner totally divorced from human activities.
It was published in Joe Farman, Brian Gardiner and Jonathan Shanklin’s Nature paper, I posted a version of it above.
Er no that’s evidence that it existed before 1979. 2hotel9 asked for evidence that there was no ozone depletion before then.
I believe Dobson (of Dobson Units fame) detected ozone depletion over the Antarctic in the 1930’s, long before the introduction of CFC’s.
Correction: Gordon Dobson discovered Antarctic ozone depletion in 1956, still well before the adoption of CFC’s.
In short, the Ozone Hole is natural and has little or nothing to do with CFC’s.
Gordon M. Dobson, Forty Years’ Research on Atmospheric Ozone at Oxford: A History, 7 APPLIED OPTICS 387, 403 (1968) (“[The values for September and October 1956 were about 150 units lower than was expected”).
Freons were discovered in the 1920s and were being widely used in refrigerators by the 30s (8 million refrigerators using Freon in the US by 1935). Carrier introduced the home air conditioner using Freon in 1930.
Versus how many refrigerators in 1979? My home didn’t have a refrigerator until about 1953, and similarly we used a ‘swamp cooler’ in Phoenix in the early-1950s.
I was addressing the comment that “1956, still well before the adoption of CFC’s”, which is not the case.
Unless you are counting coups, it is only germane if there was a significant penetration of adoptions. Eight-million out of maybe 30-million homes in 1935 probably didn’t create a measurable amount of fugitive releases from new appliances. The rest of the world also hadn’t caught up with the early adopters in the US.
Thank you for the link. I had read that it had been detected previously by Dobson, but had never seen the source.
I agree the evidence is there but I think what 2hotel9 was asking for was what was used to back up the claim by NASA’s Ozonewatch that ozone depletion started in 1978 or 79?
In 1928, Thomas Midgley, Jr. aided by Charles Franklin Kettering invented a “miracle compound” called Freon.
https://www.thoughtco.com/history-of-freon-4072212
Really, the graph shows a fairly steady value of about 300 in the 50s and 60s then shows a drop down to 200 in the 70s and 80s.
Are those average values or minimum values? How could they have obtained average values or minimum values for all of Antarctica prior to the launch of TOMS?
The made the measurements at the South Pole base, try reading their paper.
If you can’t provide me with a quick answer can I assume that you haven’t read the paper you are relying on?
No you can not, I nave read it several times, as I said if you’re so interested you should read it. I did provide you with a quick answer, I told you that they made it at a single base, they made daily measurements. The stats in their paper from which that figure is derived were the monthly averages for October.
They also showed the measurements of F11 and F12 in the SH from 72 to 80 which increased from about 50ppt to 200ppt. Annual daily plots of O3 were also provided, the daily average O3 in October averaged about 300 over the 16 years from 1957-73, ranging from extremes of 270 to 330. The comparable average over the 4 years from 1980-84 was ~230 with value for 1984 being ~200.
So, no justification for destroying Human Industrial production, Energy production or Agricultural production. All. A. Lie. Totally natural occurrence completely unaffected by human activity.
So, in spite of serious research showing this to be an entirely natural mechanism of Global Climate the Big Lie was created and perpetuated at a vast cost to the Human Race.
Regardless of the repugnance of the idea, the Human Race as a group entity clearly has a death wish.
Only part of the Human Race does.
“Some atmospheric scientists (for instance Fred Singer, founder of SEPP and also a global warming skeptic) and industry-sponsored advocacy groups question or completely deny a link between CFCs and ozone depletion. It is fairly common to see completely nonsensical arguments …“.
Years ago I read a paper by Fred, where he wrote “wihtout CR nothing will happen to ozone” (paraphrased). What I don’t know is, if it was his own finding or if he was referring to a paper from Lu. I couldn’t find that today, not in goolag, not via Duckduckgo.
Fred Singer was correct: nearly forty years after the Montreal Accord the Ozone Hole is still here, and as big as ever.
In the mean time maybe we should work out some numbers on the cost of the Montreal protocol. A time may come when those numbers are useful for civil litigation against those who propagated this probable scam:
Some days it seems there is no end to the destruction some people will inflict on the defenseless to earn one more dollar, and no limit to the lengths politicians will go to to jump on the bandwagon so they can hold on to power. But we as a voting populace also need to examine our compliance with “save the world” propaganda schemes and our lack of critical thought and questioning every time a new “threat” is leveraged to do harm to human society.
Here’s an excellent summary of the historical observations and development of the ozone hole narrative https://digitalcommons.law.buffalo.edu/cgi/viewcontent.cgi?article=1024&context=belj
Wow! A humdinger. Thanks for posting it.
Hmmm … which politician championed the dangers of “The Ozone Hole”?
Didn’t he then make millions championing the dangers of “CAGW”?
By the way, the attack on Fred Singer was ignorant and unwarranted. He was the one to first propose measuring the vertical profile of stratospheric ozone from future satellites by using backscattered ultraviolet solar radiation, using the Dobson method from above the stratosphere. He was a top astrophysicist who was the first director of the U.S. weather satellite program.
https://www.annualreviews.org/doi/pdf/10.1146/annurev.pc.43.100192.000245
Fred Singer’s proposal in 1957 https://agupubs.onlinelibrary.wiley.com/doi/abs/10.1029/TR038i004p00469
Very nice. I can answer the question should the Montreal Protocol be terminated. Yes it should. Government and inter government outfits have been shown to be untrustworthy. There is damn little evidence to show that we should take anything they say seriously.
Thanks, everyone, for measured and helpful comments. Charles in his addendum (which we discussed beforehand) says “it just seems too much of a coincidence that the panic set in just as a new measurement technique was deployed“. Yes it does, but then I have thought for a long time that it was a tragedy that satellites started in 1979, almost exactly when the climate switched from cooling to warming. The data I used was non-satellite and spanned before and after 1979, and the ozone and temperature data were from different sources, so I think the phase shift is real, and the coincidence with satellite instrumentation was just that, a coincidence. Such coincidences are extremely rare, so the issue is well worth airing. However, I do wonder whether the ‘ozone hole’ being defined to start in 1979 was designed to be at the end of the Freon patent, and if so, why? I doubt we will ever know.
It might be interesting to discover how long DuPont and NASA had been working together, whether there were vested interests and long-term contracts that influenced both parties. I can find references of DuPont working closely with NASA from 1969 and Apollo 11 right up to the Mars probes and it might go back further than that. Interesting that it was NASA’s Ozonewatch that established the 1979 date, to DuPonts benefit.
PS. At the end of the video posted by Charles, they say “What had we done?”. No consideration whatsoever that what they were looking at could be natural.
The detection of the ozone hole was not done by satellites it was done by ground measurements dating from 1956, so yes it is a coincidence in timing.
As I pointed out above, the measurement geometry and coverage are different for ground-based measurements and satellites.
So what?
Ground-based measurements with a spectrophotometer provide a single point on the ground where it can be expected there will be variations with cloud cover and elevation. That is why there are many missing data points. As I recollect, it was time-consuming to make the measurements. Whereas, the TOMS and subsequent satellite measurements provide a map of the distribution showing where the high and low concentrations are. That is, the spatial variation from a constant altitude. The lows move around over time. So what? The difference is a unique sample of one versus samples of thousands.
The first measurements which showed the depletion of the Antarctic Ozone were reported in the paper by the BAS in Nature in ’85. It was later confirmed by NASA satellite measurements.