By Thorstein Seim and Borgar T. Olsen
A review in WattsUpWithThat was performed on our paper by Kevin Kilty. It is found here. The reviewer´s criticism of the paper was summed in 3 points:
- The construction, calibration and use of the IR sensors.
- The energy balance calculation which searches for, but cannot locate leaks of energy to explain the null result.
- The many explicit and implicit uses of the Stefan-Boltzmann formula which are erroneous, and which cast doubt, not on the Greenhouse effect, but on the null result here.
We will try to answer his criticism and add more of the experimental details that he requests. But let us first make a summary of the experiment.
The experiment
The purpose of the simulated earth/atmosphere experiment was to a) be able to measure IR radiation, b) to reduce thermal energy losses to the surroundings and c) to measure temperature more accurately.
The experimental setup is shown in Figure 1: The front camber was added in order to separate the CO2 gas from the air in the rear chamber. This reduces heat loss from the rear chamber through the two windows due to heat conduction.

The one meter long, 50 cm wide and 30 cm high box, with a volume of 150 liters, is made of insulating 5 cm thick Styrofoam plates. The two chambers are separated by a 0.03 mm thin transparent plastic film. The window in the front of the box was also made from this film. The inner walls of the chambers (except the rear wall) are covered by thin, polished Al-foil. The Al-foil reflects most of the IR radiation and thereby reduces the heat loss through the walls. The length of the rear and front chamber is 30 and 70 cm, respectively. IR radiation was produced by heating a black-painted metal plate (or a thin, black painted Al-foil) to 100 oC by a 500W halogen lamp. A thermometer, measuring the gas temperature, was placed close to the roof in each chamber and screened from direct radiation from the heating plate.
An IR radiation detector is located in front of the window on the box (IR1). Another detector is placed behind the box (IR2) and measures IR backscatter radiation via a 6×6 cm window in the rear wall. To measure the heating of the inside of the rear Styrofoam wall with high accuracy, eight serial-connected and black-painted thermocouples was placed on the rear wall.
To avoid local convection and temperature gradients in the two chambers, a small fan with reduced speed is placed in each chamber. Energy input to the fans was small, only 0.6 watts. Since the gas expands during heating, each chamber has a small 5 mm aperture (covered with a piece of plastic) in the “roof” to avoid increasing the pressure. To check if infiltration from the surrounding air changes the amount of CO2 in the front, the CO2 level was inspected after the experiment. The chamber was still filled with CO2.
Construction, calibration and use of the IR sensors
To measure IR radiation and the backscatter generated by CO2, we constructed two IR detectors, using wide-band (3 to 24 μm) thermopile circuits with a nearly flat frequency response.
Calibration: To obtain a radiation spectrum close to that of a black radiator, we used a black iron pan, filled with water of temperature of 100 oC and allowed the temperature to fall to 15 oC. The measured relationship between the temperature of the radiating source and the output of the detector (in mV), is shown in Figure 2:
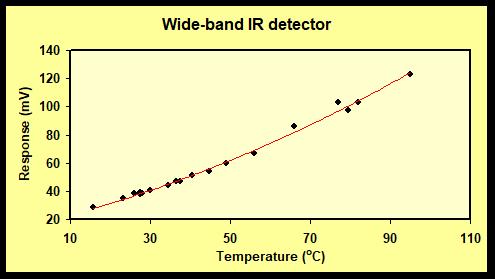
We see that the relationship is non-linear, not linear as expected from the Seebeck effect. Instead, we find that the voltage response depends linearly on the energy density of the radiation from the IR source. We computed the IR energy density output E (W/m2) from the pan, using the equation E = σT4 (the Stefan-Boltzmann’s law) where σ = 5.67 *10-8 W/(m2K4) and T is the temperature in Kelvin. The result is shown in Figure 3.
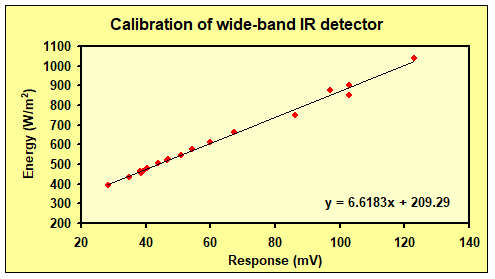
Getting a linear relationship supports the assumption that we can use the S-B equation to quantify IR radiation with the detector. But the reviewer pointed out that the IR source is not a perfect black body, and the emitted IR radiation might then be slightly lower than indicated by the S-B law (probably reduced by ca. 5%).
The reviewer also pointed out that there was a voltage offset of ca 20 – 30 mV in the detector circuit output. This offset is very small, compared to the operating range of the IR detector of more than ± 5 volts. The circuit used is a well-known one, f. inst. suggested by Hamamatsu.
The reviewer writes: “The sensor contains in addition to a thermopile, a highly accurate negative temperature coefficient (NTC) thermistor to aid in building a temperature compensation circuit”.
We were warned against combining the NTC and the thermopile circuit to compensate for variation in device temperature. It is much better to make separate circuits for the thermopile and then do the temperature correction in the computing procedure.
FOV (field of view)
The FOV of the detector is shown in Figure 4. A common way of defining the FOV is to use the half-angle, i.e. the value where the sensitivity is reduced to 50% of the maximum value. Figure 4 shows that the half-angle is close to ±5 degrees. At ±10 degrees ca 98% of the FOV is included.
With the length of one meter of the box the detector IR1 “sees” a circular area with a diameter of 35 cm, covering the heating plate and most of the rear wall. When the detector is placed in front of the window it then “sees” a 35 cm circular area of the 30 x 50 cm rear wall. The complete metal plate is “seen” within the half-angle FOV.
When the detector IR2 is measuring IR radiation from the interior of the box, it mainly “sees” the front chamber where the heated gas and windows radiates. The radiation is homogenously distributed across the two chambers, so the measurement situation is similar to that used during calibration.

As the reviewer points out the IR1 detector “sees” more than the heating plate, but the plate covers the main part of what the FOV measures.
Temperature measurements
The temperature increase in the two chambers during heating was equal (within the measuring accuracy) with air or CO2 in the front chamber. See figure 5. This is the most important result in our study.
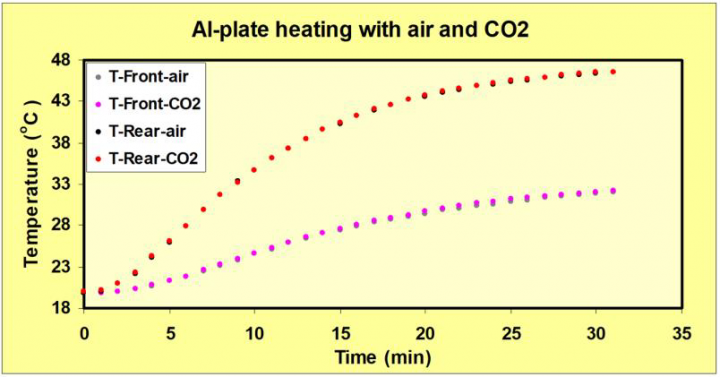
The reviewer seems to misinterpret Figure 5:
“This experiment is now repeated with the front compartment filled with 100% CO2. Now emitted IR radiation from the aluminum plate is partially absorbed by CO2 raising the temperature of the front compartment to around 33C. This causes radiation passing through the front window to drop temporarily”.
This is definitely not what we say. The back chamber heats up from 20 oC to 46 oC (upper two curves) and in the front chamber from 20 oC to 32 oC (lower two curves). The point is that the curves are identical for plain air and for 100% CO2 in the front chamber. This was surprising to us, since NASA (and Al Gore) claimed that we should have extra warming from 100% CO2. Also, the radiation passing through the front window does not drop temporarily but approaches a constant value. See figure 6.
“The missing IR is presumed to be redirected or reflected toward the rear compartment”.
We measure, not presume, that CO2 IR radiation is redirected or reflected toward the rear compartment.
IR measurements
The IR1 detector measured reduced IR output through the front window with CO2 in the front chamber.
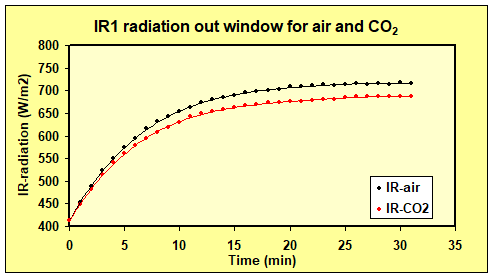
Figure 6. Absorption of IR radiation. Range: 2.5 – 20 µm. Heating is done with the Al-plate.
The detector is pointing at the center of the 100 oC heating plate. With CO2 in the front chamber the IR radiation decreased 29.8 W/m2 or ca 10%. This is close to what we find from the HITRAN data-base, i.e. 11.6% for a 70 cm long tube. The slightly lower measured IR walue might be due to the fact that the detector FOV is slightly larger than the heating plate. Anyway, an error of a few % will not influence our results and conclusions in any significant way.
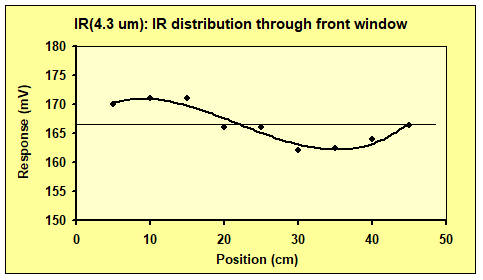
To find how much IR radiation is leaving trough the front window, we need to know how much it varies at different positions. For this we used a thermopile detector without a lens, giving it a wide FOV. The detector was used to measure IR output along the 50 cm wide front window. The spectral sensitivity is a narrow band in the 4 μm region where CO2 absorbs/emits IR radiation. The result is found in figure 7, showing that the output is close to constant, with roughly ±4 % variation.
The IR2 detector measured increased IR radiation hitting the rear wall with CO2 in the front chamber. See figure 8.
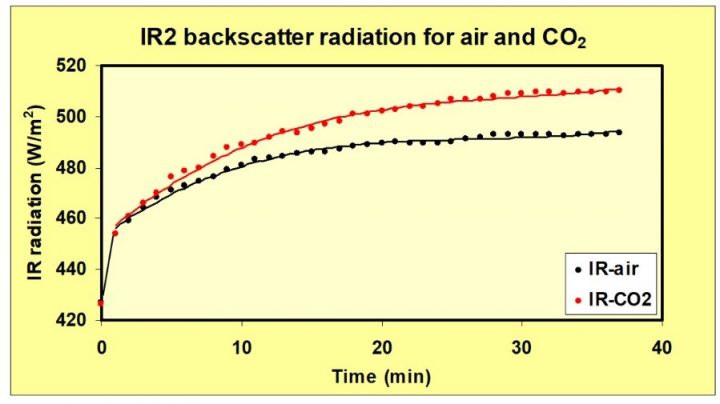
Figure 8. Backscatter (increased IR radiation measured by IR2), received by the rear wall of the box, increased 17 W/m2 with CO2 in the front box. Heating is done with the Al-foil.
The IR2 detector “sees” mainly the IR reflected from the two windows and the gas within the two chambers. The distribution of reflected IR is relatively homogenously distributed in the chambers, verified by figure 7. The measurement situation is therefore not very different from that used to calibrate the detector.
The reviewer have some objections:
“When the apparatus is in operation the aluminum plate at 100C radiates IR and causes the rear compartment to reach a steady temperature of about 46C. The authors “compute” the irradiance of the back surface using the Stefan-Boltzmann law. When the front compartment is filled with air all of this “computed” IR power is supposed to exit the front window”.
What we do: We use the calibrated IR detector IR1 to measure the amount of IR leaving through the front window, with air and then with CO2 in the front chamber. We find that more IR energy is absorbed in the box with CO2 in the front chamber. We use the calibrated IR detector IR2 to measure the amount of IR hitting the rear wall. We find that more IR energy is received by the rear wall with CO2 in the front chamber.
Misuse of the Stefan-Boltzmanns law?
Review comment:
“The apparatus here is not a cavity. It is transparent on one end and partially so on the other. Having a substantial fraction of its surface transparent means that placement of materials and their detailed radiation characteristics matter.[2] The first order of approximation to IR radiation from something that is not a cavity, and not isothermal, is to use the Stefan-Boltzmann law, but to assign appropriate emissivities less than 1.0 to different materials. The blackened aluminum radiator has an emissivity close to 1.0. It is perhaps 0.96, but the bare Styrofoam is far from black at IR wavelengths. An accepted estimate of emissivity of this material is 0.60; i.e. at any temperature it will radiate only 60% as strongly as the Stefan-Boltzmann law predicts”.
The reviewer states that we must include the emissivity ε in the S-B equation to get the correct relationship between temperature T and IR energy flow E:
E = εσT4
We are in an advantageous situation since we can measure the IR radiation and the temperature of Styrofoam when it is heated! This was done and we found that, in our experimental setup, the value of ε was measured to be 1.0 ± 0.025, not 0.6! So maybe the chambers behave a bit like a cavity after all… This result also negates the criticism of using the S-B equation in the calibration procedure.
Energy balance
Under Energy balance the reviewer claims that we have energy loss. Yes, of course. The radiation and thermal energy is flowing from the back wall through the front window (like IR from the earth surface to the space). After 30 minutes a close to steady state was established. IR energy from CO2 in the front chamber is partly lost through the front window, partly returned to the rear chamber. The point is that we do not observe any extra warming in the rear chamber despite of the increased level of IR radiation measured there. This is why we say that the back scatter heating theory might be wrong.
Energy content in gases
The reviewer presents an alternative theory:
“An atmosphere containing 70 cm of 100% CO2 at a pressure of 100kPa has effective emissivity of about 14%. Once this gas absorbs its limit of 14% of IR from the back compartment (i.e. 14% of 80 watts) and reaches an equilibrium temperature it does not reradiate this backward, but rather in all directions. It is reflected many times from the aluminum foil, with 4% being absorbed with each reflection, some passes out the front EDTA window, some passes the intermediate EDTA window and reaches the rear compartment. This could easily be only 10% of what had been absorbed in the front compartment”.
It seems that the reviewer believes that the energy just disappears through the walls and windows. He suggests that only 10 % of the energy flow that has been absorbed by CO2 in the front chamber reaches the rear compartment, which is ca 2W/m2 of 20W/m2. However, we measure an increased energy flow of 17 W/m2, not 2W/m2, returned to the rear chamber,
“The gas in the rear compartment contains so little CO2 that its emissivity (which equals its absorptivity) is probably in the neighborhood of only 1%. Thus, the null result of this experiment, rather than being a surprise, should be entirely expected”.
It is correct that the air (as well as the windows) absorbs (and emit) IR radiation, but the absorption in air is much larger than 1%. This was tested by us in a 30 cm long box Styrofoam box with a single window. The IR emission from the air in the box increased linearly with temperature in the 15 – 35 oC range. The increase of IR emitted by the air was significant, about 30% of the increased radiation from the Styrofoam walls. (The relative humidity of the air was ca 30 – 35%).
By measuring IR emission with/without the window we found that roughly 30% of the increase in the measured IR was emitted by the window. Since the temperature of the windows are the same with air and CO2, the IR contribution from them are also equal.
In Figure 8 the amount of IR radiation emitted back to the rear wall is shown, with air in both chambers (black circles). This is IR radiation received from the heated air and the heated windows. The measured IR increment is ca 65 W/m2, while the increase with CO2 in the front chamber is 17 W/m2, or an increase of about 25%. The radiation absorbed by the air is re-emitted in all directions and reflected by the Al-foil walls, with some absorption. By adding CO2 in the front chamber, less of the IR from the heating plate leaves the box through the front window. The IR2 detector shows that the IR level inside the rear chamber increases significantly, and remains high!
A final critical review comment:
“One additional error in applying the Stefan-Boltzmann law occurs in the translation of the calibration transfer function to an irradiance value. The calculation mentioned in the quotation about the calibration procedure implies a one-way transfer from the blackened pan to the sensor, when in fact the transfer is two way between the sensor and pan”.
Theoretically this could lead to an error, but it is too small to be measurable. The pan, filled with water, weighs 7 kilo, while the small detector weights ca 200 grams. It is placed only a few seconds in front of the pan and removed after the IR measurement is performed. The temperature of the detector is close to that of the surrounding room. The detector box is made of aluminum. When the measurements are done the box mainly reflects IR from the room, which is what the black pan “sees” between measurements!
Lessons learned
All of the reviewer´s statements about misuse of the S-B law has been rejected.
We were able to measure IR radiation with an accuracy of ±2.5 %.
IR backscatter from CO2 in the front chamber to the rear chamber does not increase the temperature of the rear wall and the air in the chamber, as assumed by the climate models.
0f course no rise in temperature. The intensity from CO2 emission is too cold compared to surroundings. The main CO2 atmospheric window around 10um equates to about minus 80degrees..hence only warms other objects at high altitudes. By then, so few molecules most is lost to space. Intensity at 4um is not much warmer. Never heard of anyone getting a moon- tan but the subs UV will burn skin in minutes.
The energy from CO2 is energy! It shares IR energy to other items whatever their temperature.
A warm plate emitting x amount of energy to space will lose energy at x/sec.
A cold plate emitting y amount of energy to the area of the warm plate will instantly (no change in warm plate temp) change the overall energy lost from plate from x/sec to x-y/sec.
At pressures used in this experiment there will be only a small amount of IR energy transfer. Most will be transferred from co2 to other substance (co2, walls, N2, o2, etc) by conduction and collision – the mean free time is very very much shorter than the time required to re-emit a photon.
Will it add energy from the surface that emitted in the first place if redirected back?
if a photon of energy hits a 100% reflective surface it will be reflected back to wherever normal mirror and light would.If it is a CO2 emitted photon it will be absorbed by the abundance of co2 in the air and the energy be passed on by collision to other stuff.
If it is not a reflective surface that the photon hits then the energy will be added to the material it hits
If a photon leaves a cold surface and eventually approaches a hotter surface there is no way it can decide to reverse direction if the hot surface is hotter than the cold. The energy the photon carried is simply added to the hot surface energy. It does not make the hot surface hotter it simply slows the cooling of the hot
This is true. However, since the energy of that photon is so weak it will not penetrate below the skin of that surface. Hence, it is almost always reradiated away within milliseconds.
Energy is energy – it does not remember from whence it came!
Energy absorbed by the surface will be conducted / emitted quickly but it slows conduction from below
The energy physics between conduction and radiation absorption are not the same. Much more complex than you let on.
In any event, this often balances out at night. It just takes a few milliseconds longer for the surface and air to reach the identical equilibrium point.
I’m betting dollars to donuts that the gas molecule “excess” energy is transferred into the material it contacts long before it is able to “re-radiate” that energy as a photon at LWIR or even lower wavelengths.
Here is a problem that needs to be addressed, the absorption of a lower energy EM wave. Even if both bodies emit and absorb equal spectra, albeit at different temperatures, the hot body already has higher excited molecules. That is why it is hotter.
Question: Can a lower energy EM wave with less energy from a cold body further excite an already excited molecule?
I am no expert here, but it is my thought from reading a lot, that the absorption levels are different and unique and not just linear, that is, any amount of energy is absorbed.
Jim, a blackbody simply absorbs all radiation, no matter the energy content of the photon in question or the temperature of the body that emitted that photon.
Thus if it hits a atom/molecule that is already high in energy, it simply adds to that energy, no matter the energy differences.
Unique absorption only is relevant for non-black body objects which only absorb at specific wavelengths.
ghalfrunt, temperature makes that a lot of different waves from any material is emitted, where the peak wavelength depends of temperature.
Opposite to that almost all energy that hits a material is simply absorbed, no matter the original temperature of the emitter or absorber, as far as that material acts as a black body.
Of course, no physical laws are violated, as the total amount of energy per area emitted by a hotter body is higher than from a colder body, so the net energy transfer is from the hotter to the colder body, but still all energy that hits the hotter body from the colder body is absorbed.
A well known example is the use of CO2 lasers: maximum temperature of the laser 100ºC, single around 10 μm beam, which is the “peak” wavelength for a body at a temperature of -80ºC, which can melt steel at 1200ºC…
https://en.wikipedia.org/wiki/Carbon_dioxide_laser
0.04% is an “abundance”?
This is correct. The use of temperature to describe the amount of energy in an IR photon is more for comparative purposes. Low temperature equates to less energy. However, the energy itself is no different. You just need more low temperature IR photons to produce the same warming as a high temperature IR photon.
Where the amount of energy makes a difference is in absorption. High energy photons can penetrate a surface to greater depths than low energy photons. This is why the biggest result of increasing CO2 is to enhance evaporation.
photons hitting water will not significantly penetrate but they will conduct to air and warm that slowing cooling of the water i.e. similar to the water being warmed.
Yes, you will get some reduction in upward conduction but you also enhance latent heat release which I suspect is much greater. Also, the surface skin is often warmer than the water below because it is being warmed by constant IR. Hence, there would be no upward conduction. You could get some almost instantaneous reradiation.
The net effect would be more energy released which is called cooling.
no! the photon excites the surface molecule transferring energy, The surface molecule is pushed into “gaseous” form (steam or water vapour) removing the energy imparted by the photon from the water mass. How does this also cool the mass – its energy hasn’t changed?
Energy of photons is not determined by temperature, it is determined by wavelength. Power in the EM wave generated when a particle radiates is partly determined by the temperature and is in essence how many photons per area the EM wave has available. Look up power to photon calculator.
P/A = εσT^4
Seems like you are not an engineer who understands Thermodynamics.
Have a search for Wien’s Displavement Law which relates wavelength to absolute temperature (discovered before Planck came up with his general relationship)
Maybe you should do a little more research before you disparage a person’s knowledge. Ad hominem attacks only end up making you look smaller in the eyes of everyone. The least you could do is to refute my remark with the math that you use to show that the energy of a photon is not temperature dependent. Good luck dude!
I gave the equation of the power/area for the radiation of an EM wave from an atom/molecule. It is:
P/A = εσT^4
ε – emissivity
σ – SB constant
T – Temperature
The energy of a photon is determined by:
E = h * c / λ = h * f
E – the energy of a photon
h – the Planck constant,
c – the speed of light,
λ – the wavelength of a photon,
f – the frequency of a photon.
This link will explain the Wien’s Displacement Law is used to determine either the wavelength of maximum intensity or the temperature of a body whose maximum temperature occurs at a given wavelength.
http://hosting.astro.cornell.edu/academics/courses/astro201/wiens_law.htm
The equation is:
λmax = 0.29/T
Not all energy is manifest as temperature ie heat.
From the post:”The spectral sensitivity is a narrow band in the 4 μm region where CO2 absorbs/emits IR radiation.”
I thought we were talking about 15 micro?
Congratulations you seem to have proven Hoyt Hottel wrong in saying that CO2 has emissivity of practicality zero below 33 C. The charts Peta posted a link to showing emissivity of .0035 or so are wrong also.
There are additional weak CO2 absorption bands at 1.05, 1.23, 1.5, 2.14, 2.5, and 4.0 um
The NIST data sheet doesn’t show those. The 15 yes.
https://webbook.nist.gov/cgi/cbook.cgi?ID=C124389&Type=IR-SPEC&Index=1#IR-SPEC
They are in the MODTRAN absorber tables.
mkelly:
Your graph refers to Hottel 1954
So did my link
Your graph goes up to pressures of 100 bar.
Look at your graph, at a pressure of circa 0.5 Bar (is that the average pressure of the atmosphere) and it shows a CO2 emissivity of about 0.025
Almost exactly that of Oxygen/Nitrogen (Emissivity of 0.02 and that is why the lowest part of the atmsophere, where we live and hang out, is warm)
If that 0.0025 figure doesn’t kill the radiative green house effect stone cold dead, wtf does?
Never mind the right or wrong of it, it tells us that CO2 does nothing more than Oxygen and Nitrogen already do, even at the totally unrealistic temperature of 400 Kelvin.
But this graph only starts at 400 Kelvin (circa 127 Celsius) – barely relevant to Earth’s atmosphere and presumably using 100& pure CO2, not 400ppm
This graph, taken from Hottel 1954, was run at 340 ppm and 300 Kelvin, tells us a CO2 emissivity of 0.00092
is that really what all this fuss is about
Sorry mkelly, but Your Own Authority shoots you down
Hottel’s equation and charts uses a path length, partial pressure as well as emissivity factors and overlap with other gases (eg water vapor) to determine radiation absorption. The data in the table with units of atm-m are calculated values. I do not know if they are correct. Some people assume an atmospheric height for CO2 as 5 km others use 8km. Emissivity, which is a ratio to a black body is dimensionless. Albedo is a stupid term as it is related to a wavelength or temperature range.
For Albedo, I meant not related. Clouds have a range of absorptivity depending on the composition (water droplets or ice), varying thickness and varying density (of liquid and solids)
Thanks for pointing out that remark. This mention of a “lens” is new to me, and I didn’t find it in the original paper. The sensor their detector was based on has a thin silicon filter over it to prevent sensivity to near IR and visible light. Frankly I am not sure what idea they are trying to convey. The Eckert charts I refer to are organized differently than yours. I made a copy of yours ’cause it looks handy.
Emissivity within a temperature range is a comparison to the emission of a black body. Hottel’s work is correct. Have a look at the higher temperatures. CO2 will emit radiation in flames at a wavelength of 2.7 micron (or about 1073K) and at 4.3 micron (or about 674K). It will absorb some radiation from the sun but the sun emits practically no radiation in these wavelengths. The peak wavelength from the sun is about 0.6 micron (or 5700K). CO2 will absorb and emit radiation at 14.8 micron ( a temperature of about 200K) The Earth surface temperature is about 288K which corresponds to a wavelength of about 10 micron. The absorptivity/emissivity of CO2 in the range of temperature 200-400K is indeed very small as it absorbs in only a very small range around 14.8 micron and your figure of 0.0035 is certainly close the the actual figure. Further, the Earth surface emits very little radiation at a wavelength of 14.8 and on top of that there is practically no CO2 present -the partial pressure is only about 40 pascals. So basically using Hoyt Hottel’s formula the radiation absorbed by CO2 (before it emits to space at around 200K) is close to zero.
Hottel’s experimental work was for furnace optical depths at 1bar on order of 10 meters so of course the gas emissivity was so close to zero. For earth’s atmospheric gas optical depth, the global emissivity is ~0.8 looking up.
“Emissivity” as commonly understood is nonsensical for a gas. It is basically a short cut method for design work for fired furnaces and boilers. For those applications it works. For scientific research, the radiance/absorption bands need to be taken into consideration, otherwise you are assuming CO2 acts like a black body, which is an error.
JamesD,
You provided an excellent, spot-on comment . . . one that is often overlooked by those that should know better.
Thanks, and +42,000 intergalactic credits!
Could someone explain if I understand this correctly. (or not…which is more likely)
Back radiation from CO2 has a certain frequency and from it an associated energy. The surface will absorb this radiation provided that the molecule has an open vibrational level of that same frequency as the incoming radiation. If the door is open, as it were, the molecule absorbs the energy and jiggles around more increasing the temp.
But what happens if the molecule already has all it’s vibrational levels occupied through absorbing energy through conduction of other molecules next to it? Does the incoming radiation then get scattered without imparting any energy as the frequency door is shut?
I always thought that as the water at the surface of Earth is a much higher temp than the place where the CO2 is radiating in the atmosphere, it would be more likely that the vibrational modes at the surface would be all filled up by the energy imparted by conduction at a higher temp? So although the molecule is exposed to the radiation, it doesn’t absorb it as it’s already full up from energy elsewhere. Does it work like that?
thanks wattsupers….
That is the way it works. That is why long wavelength IR detectors need to be cooled.
Wrong, – semiconductor sensors need cooling but room temperature micro bolometers are at, surprisingly, room temp. Long wavelength IR adds energy to low thermal mass bolometers and raise their temperature (initially energy lost / gained from bolometer is dependant on its temperature and its surroundings temp (lens, camera body, metalwork plastic, electronics etc.) Add a bit more energy from focussed above absolute zero objects and it will warm. with the less energy state it was in balance energy in = energy out. Add more energy and its temp must rise to maintain energy in = energy out.
With a focussed object at -40°C the amount of energy transferred to the bolometer is very small and uncooled thermal imaging cameras usually have -40°C as the lowest detected temp. Remember there are NO cold photons to make a rt thermal camera work.
Solids and liquids absorb energy as grey bodies. They accept and emit a very wide band of frequencies gases on the other hand only accept and emit at certain frequencies
A ghg photon can hit a thing (cloud/earth/water etc.) and transfer its energy to that thing no matter what the things temperature is. Most things also lose radiation as a grey body. if energy in gt energy out it will warm. even if a photon of CO2 emmisions is very small, hit something with more than the energy lost and you can cut plastic/wood/metal with a co2 laser.
HOWEVER
The smallest amount of energy (i.e. one quantum) that an object can absorb from IR light with a wavelength of 8µm
Energy quantum = hν
h is known as “Planck’s constant”, and has a value of 6.63 x 10^-34 Joule seconds (Js)
so we need to know the frequency ν
νλ= c
ν = c/λ
ν = (3.00 x 108 m/s)/(8*10-6 m) s-1
plugging into Planck’s equation:
E = (6.63 x 10^-34 Js)*( (3.00 x 10^8 m/s)/(8*10^-6 m) s-1)
E (1 quanta) =2.49×10^-20 J at 8μm IR
I small calorie raises the temp of water at 19.5C by 1°C
4.182 joules=1cal
I quanta of IR at 8μm will RAISE 1 gm of water by (1 / 4.18)x2.49×10^-20 °C
=5.95×10^-21 °C!!
S 1 photon has little energy!
The surface will always absorb IR photons which induces lattice vibrations known as phonons.
Gas molecules will only absorb IR and increase in temperature if the photon frequency matches the frequency of one of the molecule’s normal modes and the two have vector directions of the same sign. In other words, if their vector directions have opposite signs, the incident IR photons and gas molecules still interact, but the molecule emits two photons of the same energy level in the same direction as the original photon in order to conserve momentum and energy – a process known as stimulated emission. That second part is typically ignored by those that subscribe to the backradiation GHG hypothesis.
The normal modes of the gas molecules are always active and that is why they are opaque to certain frequencies of IR in the first place – there is no “filled up” molecular modes. If a gas molecule absorbs an incident IR photon, the corresponding normal mode vibration/bend/stretch/scissor amplitude, as well as the velocity of the molecule, is increased and thus the total kinetic energy of the molecule increases. The temperature of a gas results from the average total kinetic energy (KE) of the molecules which is KE = 3/2kT + 3/2(r-1)kT where r is the number of atoms in the molecule, k is the constant of proportionality, and T is temperature in Kelvin. 3/2kT is the translational kinetic energy and 3/2(r-1)kT is the total energy of the internal molecular modes.
So you see for any monoatomic gas the temperature is derived entirely from the translational kinetic energy, the temperature of diatomic gases is equal parts translational kinetic energy and internal molecular energy, and for any gas with more than two atoms there is more energy within the internal molecular modes than in the translational kinetic energy. The energy of the translational kinetic and internal molecular modes are theoretically proportional given the relationship above – changing one will change the other for any gas molecule with more than one atom.
https://www.feynmanlectures.caltech.edu/I_39.html
https://www.colby.edu/chemistry/PChem/notes/NormalModesText.pdf
Maybe I have misunderstood how phonons are created but it seems like the surface of a solid/liquid must first absorb the EM wave at a certain frequency and the increased energy can then create phonons via vibrational/translational movement. This would seem to continue the frequency dependent absorption requirement.
Lattice absorption – University of Reading
Probably this is complex enough that a reply can’t be made effectively through comments, and I will write something more formal soon. But first, let’s see where I agree with Seim and Olsen, S&O.
First, S&O say the absorption in the front chamber when filled with CO2 is 11.8%. My reading of a graph by Eckert in the text Heat Transfer by Alan Chapman suggests 14%. Engineering correlations in heat transfer problems are rarely as accurate as plus or minus 10% and often in error by 20%. We agree, then.
There is also the mention of the terrible experiment described first on an NOAA web page, then done poorly by Al Gore, Bill Nye, and the BBC. I had physics students perform this experiment in three successive physics lab sessions per the recipe by NOAA and the results were summarized on WUWT long ago.
The conclusion was that the experiment produced a null result because the design was poor and the metrology was uncertain — among other things thermometers (thermocouples) can measure the same temperature differently by a couple of degrees C — temperature displays in factories can store an offset per thermocouple for just this reason. Of the Gore/Nye work I concluded:
While I applaud the effort of S&O to attempt this analog simulation of “greenhouse” backscatter, their effort seems to have some of these same issues of trying to measure and interpret small signals with uncertain metrology.
I am being called to cook Saturday morning breakfast for my children, but I will return occasionally today with additional comments.
Kevin Kilty; How would you design an experiment to prove your point?
As I have been trying to outline in my comments here, the first step is to get absolute control of all the various means of heat transfer that act to confuse the outcome of this experiment. The signal is small and it takes very little to confound the measurements. Then, one might reorganize the experiment to get larger signal to noise (a longer section of CO2 filled section might be one way), but there is also something that this experiment cannot duplicate and that is the temperature gradient that exists in the atmosphere, but does not here.
This temperature gradient is important because, among other things, the apparent emissivity of a layer of CO2 laden air is a function of temperature. It is larger at cooler temperatures simply because of the distribution of absorption lines in the CO2 spectrum.
I have to say that S&O went to a great deal of trouble and effort in this experiment, but the amount of design required to prove that “backscatter” does not work as supposed is greater still. Mechanical engineers design boilers and furnaces and ovens all the time, and if IR radiation didn’t behave as theory supposes, we would have noted the discrepancies long ago. So I wouldn’t have bothered with this effort to begin with, or at least until I had reason to believe something was amiss with theory first.
The purpose of the simulated earth/atmosphere experiment was to a) be able to measure IR radiation, b) to reduce thermal energy losses to the surroundings and c) to measure temperature more accurately.
And yet this box is nothing like the [real] Earth. And that is the problem with this.
There is a good reason for the consideration of the Occam’s razor in the scientific analysis and scientific endeavors.
The box experiment in question is not meant as a replication of “real” Earth system, but more like an experimental method in testing a main part of the Radiative “theory” of climate.
Doesn’t stand as an explanation of how climate
ticks, but more like showing the merit or the lack of it, in the consideration of the IR radiation hypothesis towards climate, as an explanation of it.
Most of times is easier and feasible to figure out the ways how something doesn’t work, by continually testing and experimenting,
and therefore getting closer and closer to the chance of figuring out the actual way of that working…
…instead of just simply jumping from tree to tree with no any particular method of feedback assessment,
and just like that declare from the top of the higher tree there;
the indisputable
most exotic appealing find coming across, regardless of any merits or not, there.
well, just saying.
cheers
Ah, an interpreter.
Duly noted
Cheers.
The main transfer of energy at normal atmospheric pressure is contact The mfp is so short secondary photon emission dose not have time to occur. This experiment is measuring CO2 capture of photons (near 100% if normal atmosphere is used) and near 100% if CO2 rich atmosphere is used The rest is conduction
AKA saturation. So, no added absorption in the lower atmosphere where it could make a difference.
No in the lower atmosphere collisions are very frequent and transfer of energy to another molecule is rapid (probably n2 or o2 “For the nitrogen molecule already considered at a temperature of 293 K and at a pressure of 1 bar, a collision frequency of 7·10^9 1/s” 140 pico secs between collisions) freeing up the co2 molecule to absorb more radiation at its resonant wavelength.
The transfer of energy especially to non ghg of course warms the atmosphere
What was the spectral irradiance of the halogen lamp, and did you convolve with the spectral response of the photodiode?
It is not a photodiode they used, but rather a thermopile, which is to say a series connection of many junctions of some sort of materials. Some thermopiles actually use a series connection of ansi standard thermocouples, some probably use semiconductor materials, Copper and Copper +1 oxide has a very large seebeck coefficient. The manufacturer’s spec does not list the materials for this device as I recall.
At any rate the spectral response is set by the window covering the thermopile and this is, as the authors say, very flat response from 3 to 24 micrometers.
I missed this, thanks. Thermopiles usually need microvoltmeters to get decent resolution.
“The two chambers are separated by a 0.03 mm thin transparent plastic film. The window in the front of the box was also made from this film.”
What plastic was used?
Indeed, this is an important observation.
Plastic should reflect all IR wave lengths. However, I’m not used what a minimum thickness needs to be in order to do so.
Note that Seim and Olsen confirm my own empirical findings.
CO2 is a red herring.
https://breadonthewater.co.za/2021/04/05/unexpected-ice/
https://breadonthewater.co.za/2021/03/04/the-1000-year-eddy-cycle/
https://breadonthewater.co.za/2021/01/26/am-i-a-climate-denier-denialist/
polystyrene sheet is an insulator but has a thermal mass so will absorb energy adding thicker insulator does not stop it absorbing energy. the time for thermal stability will be increased by greater thickness.
Reflecting IR from imperfect aluminium sheet will improve matters but will still conduct to the insulation to which it was glued. Again thermal stability will be slow.
Perhaps a box constructed of thin silver coated Mylar sheets separated by very small gap (no contact!!) preferably “filled” with vacuum as James Webb telescope. Without the vacuum you still have conduction to slow stability.
There is a problem with this physics approved and proven insulation method in that many consider this to be a source of infinite power (see Iron Greenhouse on WUWT)
With aluminium lining you will have additional IR reflecting into the chambers which does not occur on earth.
Thermocouples work on the difference in temperature between junction of dissimilar metals commercial thermocouple measurements rely on one measuring junction and 2 other junctions at a accurately KNOWN temperature. The 2 other junctions are placed in good thermal contact to each other and to an accurate semiconductor temperature measurer. The 2 other junctions form 2 or more other sources of thermal emf (one for each change of temperature) but these can be kept minimal keeping all at the same known temperature.
This is usually done well in dedicated thermometers but less so in multimeters. measure the temp of boiling water after the multimeter has thermally equalised to the room. Then use a hair dryer to warm the meter and see the temperature reading change.
What was the exact construction of the temperature sensors?
How were temperature changes in the room accounted for?
You have identified a number of issues that are poorly resolved by the explanation of the apparatus. My response will try to clarify this to the degree possible.
But the apparatus remains the same in the two runs, only the gas filling the front box is changed.
The main effect measured here is the different thermal conduction of air and co2 and perhaps the major flaw the thermal conduction of the chamber separation window. By stirring the two chambers you will be ensuring that it is the window that is important not the gasses
Actually you will notice that temperatures of the front and back portions of the apparatus are different. This is why getting control of all heat transfer factors is important.
A vacuum . . . good luck with that! But I agree materials and structure are poor.
Kilty missed your main flaw. The application of equation 1a on a 1 meter thick atmosphere.
I am not sure what you are refering to, but I didn’t use 1a (in the original paper I presume) but rather used an engineering correlation presented as a graph from my old college heat transfer text. We can read my graph on pressure times path length down to about 1 pound per square-inch foot. In a nearby comment I stated that their estimate and mine agree with the capacity of the methods.
I’m referring to what they write about (1a) and (1b). For instance:
“From the Equation (1a) and Equation (1b), we therefore should expect a measurable temperature increase in the rear chamber.”
Pure nonsense. According to the IPCC “The constant in the simplified expression for CO2 for the first row [the 5.35 in (1a)] is based on radiative transfer calculations with three-dimensional climatological meteorological input data (Myhre et al., 1998b).”
And that’s of course for the full air column. This experiment has nothing to do with eq. (1a).
> The two chambers are separated by a 0.03 mm thin transparent plastic film.
Transparent in the visible spectrum or…?
> …covered by thin, polished Al-foil. The Al-foil reflects most of the IR radiation…
Simple experiment. using an IR heater place a piece of polished Al-foil underneath and touch the underside. Surprise!
Kudos to the authors for engaging here at WUWT. I look forward to this discussion, and the opportunity to learn (many) new things. In primus, let’s all be polite to our guests. No advances are made via belittling, name-calling, and general hectoring of the correspondents. Save that for the politicians.
Indeed. Good thing their pictures aren’t posted, the children in the audience would immediately make fun of their looks, anonymously from the shadows.
when I tried measuring IR I tried to minimise the conduction of the gas from the window (now warmed by conduction between gas molecules) by blowing room temperature air between (no on) sensor and window but trouble is the air temperature fluctuates considerably makin these measurements poor.You seen to have no means of isolating conduction and photons.
Perhaps if you separated the 2 chambers and put double glazed window in each then blew fixed temperature air between the conduction confusion would lessen.
Also a hot metal plate in contact with the inner gas of the small chamber will transfer more heat by conduction than radiation. You need to isolate these conductions
You do not say how you maintain the fixed temp of “cold junctions ” – This is essential. Also how do you maintain a fixed room temperature during the course of the experiment. 5mm of polystyrene tile offers little isolation when measuring small differences. Did you accurately record the ambient temperature?
If the purpose of this experiment is to demonstrate with better apparatus that the Gore / Nye CO2 in a bottle with a heat lamp greenhouse “proof” is a pile of crap, it’s fine. As a means towards discrediting the GCMs, however, it’s awful. Try these instead:
https://wattsupwiththat.com/2021/03/14/the-problem-with-climate-models-2/#comment-3206701
A couple of things that I find questionable.
First: “To measure the heating of the inside of the rear Styrofoam wall with high accuracy, eight serial-connected and black-painted thermocouples was placed on the rear wall.”
Serial connection of thermocouples describes a thermopile, but that requires alternating junctions to be on opposite sides of the thermal barrier and results in measurement of the temperature difference between surfaces. Thermocouples connected in series placed on one surface will result in alternate junctions producing cancelling positive/negative voltages. The result will be something close to the temperature if the reference junction. Multiple TCs connected in parallel will provide an output of the average temperature as long as lead resistance is equal (using equal lead lengths, or swamping resistors).
Second: Visual appearance is not a good indicator of surface emissivity. 0.96 is the highest emissivity that is commonly achieved, but black paint may be as low as 0.85 and visually white surfaces can be 0.95. Polystyrene foam is typically about 0.90. When in doubt, measure with a calibrated emissometer.
Third: Why build your own radiometers instead of buying commercially made ones? The accurate calibration or radiometers is quite complicated and involves tight control and measurements of target temperature, emissivity, field of view, etc. I’ve purchased a number of them and paid for required regular recalibrations – not cheap.
I’m not saying that the results of the S&O paper are wrong, just somewhat skeptical that the experiment design is sufficient to accurately measure the subject phenomenon.
Good observations. To your first point about this…
“To measure the heating of the inside of the rear Styrofoam wall with high accuracy, eight serial-connected and black-painted thermocouples was placed on the rear wall.”
I didn’t like the black-painted description. This might make the thermocouples respond to radiation differently than the wall itself. As an analogous example, the reason we enclose weather instruments in a Stevenson screen is to keep radiation temperature from disturbing attempts to measure air temperature — the two can be very different.
The effect of photons only becomes significant above ~10km where the mean free path of co2 molecules and the time between collisions allows CO2 to re-release a photon before a collision transfers the energy
Which means any downward emission is likely to be reabsorbed before it reaches the surface by water vapor or clouds. Only emissions in the atmospheric window have much of a chance of reaching the surface.
Unless it reaches the surface it has no warming effect. It is just energy transferred from one part of the atmosphere to another.
So you are saying that the overturning/mixing of the atmosphere is not a feature – perhaps people advocating that it is this movement (Stephen Wilde) that gives the warming to 17°C should be informed.
I have informed Stephen of his errors more than once. Your attempted deflection does not change my point. In general mixing moves warm air upward where it radiates to space more easily. Doesn’t help your case.
I was wondering about the EDTA. I think what was meant is a poly ethylene terephtalate. Do we have an IR spectrum for that plastic film that was used?
The authors in their response have provided some information to help clear up my criticism about the energy balance. First, they supply the size of the pressure relief hole of 5 mm diameter. This is probably too small to be an issue, but we didn’t know until they supplied this value which was not in their original paper.
Second, we now know the EDTA film is only 30 micrometers thick. This too was not in the original paper. A film this thin cannot supply any resistance to the conductive flow of heat. So, to avoid a non-radiative flow of heat across the thin films, which are separating gases at different temperatures by 12-26 degrees C, we are depending mainly on a couple of thin films of gas. In the interior of the box the air is stirred by a fan which increases its film coefficient. Thus the largest impediment to non-radiation heat flow is the film coefficient on the exterior of the box, which could be 2W per meter squared per Kelvin — and at 26K Delta T over 0.15 square meters becomes 8W of heat flow. I think people should see why I had doubts about there being an accurate energy balance.
Kevin,
Issues re. heat loss / energy balance duly noted, but aren’t the following much more significant issues:
Staying on the topic of energy balance, let’s now consider heat flow, transient and steady state, through the styrofoam walls of the box. The steady value is related to temperature difference and thermal conductance across the material. The maximum temperature difference is 26C, and for styrofoam of thickness 5cm the conductance is 0.5W per meter squared of surface per kelvin of temperature difference. This is, itself, large compared to the 17Wm-2 backscatter they hope to see with a temperature measurment.
The transient signal comes from having to heat the styrofoam walls from ambient to a steady operating temperature. The Biot number for the walls is about 4 which is so large that we have to treat the problem as 1-D temperature diffusion across the foam. The problem of getting the walls to a steady temperature profile is one of diffusing energy into the styrofoam to raise its te,mperature, and this process is frustrated by the very low thermal diffusivity of styrofoam (the very reason it is a good insulator). To get the transient heat absorption down to 1W per meter squared requires about half a day. None of the graphs go beyond 30 minutes or so. How long did the authors allow any run to proceed? I don’t know is the answer.
Continuing with the topic of energy balance, let us now think about the problem of radiation transfer and measurement through the EDTA films. These film will radiate emitted power by virtue of their temeprature, but they are very transparent and they are probably reflective. What a person measures with a sensor looking through such a film is termed “radiosity” which is more than just emitted power from behind the film. In fact, some of the measured power could be coming from warm obects outside the apparatus which are reflected in the film.
I won’t even speculate about how large the effect might be. But keep in mind we are trying to reduce all these potential heat transfer magnitudes down to an uncertainty very small with respect to the 17W per meter squared that is our signal.
Firstly, and most critically, the above article does not cite how tightly the front window was temperature controlled to the nominal “100 °C” temperature. A mere difference of 5.5 °C in that temperature between running the experiment with air in the front chamber and then running it with CO2 in the front chamber would fully account for the ~10% difference in IR-radiation (W/m^2) noted in Figure 6.
Next, the above article has these statements “IR radiation was produced by heating a black-painted metal plate (or a thin, black painted Al-foil) to 100 °C by a 500W halogen lamp” and “To avoid local convection and temperature gradients in the two chambers, a small fan with reduced speed is placed in each chamber. Energy input to the fans was small, only 0.6 watts.”
It is not clear if the inward-facing surface of the aluminum foil front window (or the alternative “metal plate” inward-facing surface) was painted to enhance IR radiation emissivity values on this side. For example, the emissivity of standard, uncoated aluminum foil in the IR spectral region can be as low as 0.036 (https://www.researchgate.net/figure/Visible-light-absorptivity-infrared-emissivity-and-equilibrium-temperature-of-various_tbl1_328994550 ).
Using this value in the S-B equation with a 100 °C front window temperature and the experimental limit-low 20 °C internal front cavity gas temperature and radiation area of 0.5 *0.3 m^2 gives a total of 3.7 watts being radiated into the experimental box.
Similarly, in the experimental limit-high 32 °C internal front cavity gas temperate case, with all other facts being the same, a total of 3.3 watts would be radiated into the experimental box if the inward-facing surface was not painted.
In such cases, the 0.6 watts total of fan power input would then comprise 16% and 18% of the total power to be accounted for.
It is therefore incorrect to state, as the above article does, that “. . . so the measurement situation is similar to that used during calibration.”
Also, since Figure 6 and 8 graphs present experimental measurements of IR radiation in units of watt/m^2, one can surely question the source of heat for that radiation flux if the inward-facing surface of the front window material did not have very high emissivity in the IR spectral range.
There may be the case that the noted difference between pure CO2 and air being in the front cavity (the “money shot” Figure 6 and 8 graphs) is generally unaffected by this issue . . . it requires more specific details on the experiment setup than are given in the above article and more thought (on my part) about the net impact of the potentially not-insignificant input energy from the fans.
Do not all these factors, that you rightly call up, cancel out when the experiment is actually measuring the different heating effect when the chambers are filled with different gases. The losses and input are the same in all scenarios of the experiment so should have the same effect, cancelling out.
Importantly there was no difference in the heating effect in the presence of 100% CO2 to a standard air atmosphere. This puts doubt as to the heating effect of the back-scattered IR thus the existence of the GHE.
More importantly – there has never been published an experiment that proves the heating effect of CO2 that could lead to proving the GHE. Why is this so? Surely the experiment has been undertaken but the results have not received publicity.
OK, upon re-reading the above article 3 or 4 more times, here are the fundamental issues/flaws with what is being presented:
1) There is no accounting for the energy being lost to the mass of aluminum foil that is lining the inside of the box in the experiment, nor in turn to energy being conducted from the aluminum foil to the outer Styrofoam mass.
2) In this matter, one cannot simply argue that “shiny” aluminum foil will reflect most IR-radiation back into the interior of the box and thus provide additional “opportunity” to be absorbed by the gas inside. With the very low emissivity of aluminum foil in the relevant range of EM spectrum (0.036 per previous post) PLUS the fact that gas thermal convection undoubtedly overwhelms thermal radiation transfer from a 100 °C plate (or foil) to the aluminum-lined walls. The IR-photon-energized gas molecules will very rapidly lose their radiation-absorbed energy to other lower-energy molecules (be they air composition or just mostly CO2) or to the surrounding walls at time of collision, such time being on the order of micro-seconds to pico-seconds between collisions.
3) Figure 6 and its associated text say that with CO2 in the front chamber the IR radiation through the experimental setup DECREASED by 29.8 W/m2 from what it was with just air in the both chambers. Yet Figure 8 and its associated text say that with CO2 in the front chamber the IR radiation backscatter (supposedly just from the CO2) INCREASED by 17 W/m^2 from what it is with just air in both chambers. These claims present the question—raised by others—as to what accounts for the difference in power fluxes if the heating source is always maintained at 100 °C? That is, at near-equilibrium conditions (as asserted) where is that additional (29.8-17) =12.8 W/m^2 of IR flux going. It cannot be going out the front window since the flux there is being measured by sensor IR1 and it cannot be going out the rear since the flux there is being measured by sensor IR2. And it cannot be continuously absorbed by the CO2 under “steady state” conditions. And it cannot be continuously absorbed by the aluminum foil and Styrofoam enclosure under “steady state” conditions. Is the unaccounted-for radiation flux perhaps being the result of Figure 6 data associated with heating being via an “aluminum plate” at the rear, whereas Figure 8 data is associated with heating via an “aluminum foil” at the rear, as noted in the respective Figure titles?
Something is very wrong in the data as presented in the above article.
The interesting facts about CO2 being a Greenhouse gas are that it isn’t, mathematically and chemically it’s not possible. The biggest CO2 producers are living things, especially herbivores.
The biggest atmospheric driver of temperatures is water vapor.
There have been many good comments here. I am going to shut down for now. I had planned to tackle the issue of the calibration today, but I think I will defer that topic to a more complete posting I hope to present within a couple of weeks regarding measuring LWIR. Infrared measurements are more complex than most people realize if for no reason other than everything is a source of IR radiation.
Trivial observation – in figure 3 and associated text you refer to “energy density” but you are showing units of watts per square metre. I think you intended to say “power density.”
Why don’t we use a system where pure CO2 is circulated through transparent trays and exposed to direct sunlight? Theoretically it should trap and amplify tremendous amounts of heat which could be harvested and used to boil water which could then drive steam turbines.
You should apply for funding for this concept.
That will then determine whether the paymasters of ACGW really do consider Co2 to be the devil incarnate.
If you don’t want to pursue let me know – I would love to pitch this 👍
Such experiments have been attempted, including using CO2 in double glazing. but all have failed.