Guest Post by Willis Eschenbach
There is a more global restatement of Murphy’s Law which says “Nature always sides with the hidden flaw”. Parasitic losses are an example of that law at work.
In any heat engine, either natural or manmade, there are what are called “parasitic losses”. These are losses that tend to reduce the temperature differentials in the heat engine, and thus reduce the overall efficiency of the engine. In general, as a percentage parasitic losses increase rapidly with ∆T, the temperature differences in the engine. In the climate system, two main parasitic losses are the losses from the surface to the atmosphere by way of conduction and convection (sensible heat), and the losses from surface to atmosphere by way of evaporation and transpiration (latent heat). Both of these parasitic losses act to reduce the surface temperature with respect to the overlying atmosphere, by simultaneously cooling the surface and warming the atmosphere … nature siding with the hidden flaw to reduce the overall system efficiency. So I decided to see what the CERES data says about parasitic losses. Figure 1 shows the parasitic losses (the sum of sensible and latent heat losses), as a percentage of the total surface input (downwelling longwave plus shortwave).
 Figure 1. Parasitic losses (latent and sensible heat loss) from the surface to the atmosphere. Percentage of parasitic loss is calculated as the sum of sensible and latent loss, divided by the total surface input (downwelling shortwave plus downwelling longwave).
Figure 1. Parasitic losses (latent and sensible heat loss) from the surface to the atmosphere. Percentage of parasitic loss is calculated as the sum of sensible and latent loss, divided by the total surface input (downwelling shortwave plus downwelling longwave).
I was most interested in how much the parasitic loss changes when the total surface input increases. Figures 2 to 4 shows that situation:


 Figures 2-4. Scatterplots, parasitic loss in watts per square metre (W/m2) versus total surface input (W/m2). Parasitic loss is loss as sensible and latent heat. Gold line shows the loess smooth of the data. Red dots show land gridcells, which are one degree square (1°x1°) in size. Blue dots show ocean gridcells.
Figures 2-4. Scatterplots, parasitic loss in watts per square metre (W/m2) versus total surface input (W/m2). Parasitic loss is loss as sensible and latent heat. Gold line shows the loess smooth of the data. Red dots show land gridcells, which are one degree square (1°x1°) in size. Blue dots show ocean gridcells.
I was very encouraged by finding this result. I’ve written before about how at the warm end of the spectrum, parasitic losses would increase to the point where most of each new additional watt striking the surface would be lost as sensible and latent heat, and that little of it would remain to warm the surface. These graphs bear that out entirely. Here’s why.
The slope of the gold line above is the rate of increase in parasitic loss for each additional degree of warming. As you can see, the slope of the line increases from left to right, although the rate of increase goes up and down.
In order to understand the changes, I took the slope (change in parasitic loss divided by the corresponding change in surface input) at each point along the length of the gold line for both the land and the ocean separately. Figure 5 shows that result.
 Figure 5. Change in parasitic loss (in W/m2) for each additional W/m2 of surface input. “Wobbles”, the looped parts in the two graphed lines reflect subtle changes in the loess smooth, and can be ignored.
Figure 5. Change in parasitic loss (in W/m2) for each additional W/m2 of surface input. “Wobbles”, the looped parts in the two graphed lines reflect subtle changes in the loess smooth, and can be ignored.
Now, what are we looking at here? Well, this is how the parasitic loss changes as more and more energy is input to the surface. Where there is little surface input, the loss is low. In fact, at the South Pole the situation is reversed, and the net flow of energy is from the atmosphere to the surface. This is the result of huge amounts of energy being imported from the tropics.
The key point, however, is that as we add more and more energy to a given gridcell the amount of parasitic losses rises, in perfect accordance with nature siding with the hidden flaw. And at the right hand end of the scale, the warmest end, for every additional watt that is added, you lose a watt …
Is this relationship shown in Figure 5 entirely accurate? Of course not, the vagaries of the smoothing process guarantee that it isn’t a precise measure.
But it clearly establishes what I’ve been saying for a while, which is that parasitic loss is a function of temperature, and that at the top end of the scale, the marginal losses are quite large, close to 100%.
Now, as you can see, nowhere is the parasitic loss more than about 30% … but the important finding is that the marginal loss, the loss due to each additional watt of energy gain, is around 100% at the warm end of the planet. Here is the parasitic loss for the planet as a whole versus total surface input as shown in Figure 2:
 Figure 6. Change in parasitic loss (in W/m2) for each additional W/m2 of surface input, as in Figure 5, but for the planet as a whole.Change in parasitic loss (in W/m2) for each additional W/m2 of surface input. “Wobbles”, the looped parts in the two graphed lines reflect subtle changes in the loess smooth, and can be ignored.
Figure 6. Change in parasitic loss (in W/m2) for each additional W/m2 of surface input, as in Figure 5, but for the planet as a whole.Change in parasitic loss (in W/m2) for each additional W/m2 of surface input. “Wobbles”, the looped parts in the two graphed lines reflect subtle changes in the loess smooth, and can be ignored.
Note also that across the main part of the range, which is to say in most of the planet except the tropics and poles, about half of each additional watt of energy increase doesn’t warm the surface … it simply goes into parasitic loss that cools the surface and warms the atmosphere.
Best to all,
w.
PS—If you disagree with what I’ve said please quote my words. That lets all of us know just exactly what you disagree with …
Yes that field, the radiant cooling field. With 4/5ths of all energy leaving structures you’d need radiant cooling wouldn’t you?
If radiation was even important, somebody would make an industry of it even if it was niche or specialty clients or production.
Why doesn’t radiant cooling find itself ingeniously integrated into every one of man’s sophisticated,computer designed objects?
Because as soon as any thermally conductive atmosphere is present in the temperatures seen on earth, convection outstrips radiant loss.
But then those open roof radiators cool because when pointed to the sky they are receiving only minute amounts of energy down. Certainly not enough to warm them. In fact they cool to the point where they freeze sometimes,
these kinds of open radiant refrigerators are well known world wide. Open insulated container, fact sky at night away from direction of sun preferably, evaporation and radiation emit a lot of heat and things freeze even when the temp is above freezing.
That doesn’t help you though, the very core of my argument is the entire world uses something much, much more natural and easy to augment: convective cooling.
Willis Eschenbach says:
March 30, 2014 at 2:33 pm
Um … the field of radiant cooling?
Your lie filled rant doesn’t help your cause any either, where you simply melted down and started publishing bald-faced lies everyone on the thread knows aren’t true. I put like a dozen different references on this thread.
You’re just doing the “I’m not really in thermodynamics so I don’t have a reputation to blow: so I’ll just throw a low class invective filled meltdown into the thread.
I have one that went into moderation listing all my contributons Willis. It’s less than disingenuous to simply start ranting like someone off their meds just to tantrum and lie, when the evidence that you’re just tantruming is on the very page you’re doing it on.
Martin Lewitt says, March 30, 2014 at 3:26 pm:
You are trying to make the case that heat CAN pass from cold to hot. When it comes to radiation. Give it up. It can’t.
We agree on the result of reduced cooling. We do however NOT agree that you reduce a hot object’s cooling by letting the opposing cold object increase its heating. ‘Back radiation’ does not raise the temperature of the surface of the earth, Martin. Because that would make it an extra source of HEAT for the surface. A heat source heats (raises the temperature), a heat sink cools (lowers the temperature). And ‘back radiation’ to the surface comes from its heat sink. So you’re left in a check mate position. Because it can’t NOT be heat and then still heat the surface.
This is what I’m trying to tell you. You have to move away from the whole idea of the cooler atmosphere as a provider of energy (in effect a second heat source) to the warmer surface. It is ONLY a receiver of energy. A heat sink for the surface.
Reduced cooling comes from actually reducing the energy flow going OUT FROM the hot object to the cold object. By making the temp difference smaller (referring once again to the radiative heat transfer equation).
You see the difference? When you physically make the OUTGOING flux from the hot object smaller, THEN it is the energy supplied by a constant flux IN from the sun that piles up and make it even hotter. This is a situation that perfectly satisfies the laws of thermodynamics. You on the other hand are NOT making the outgoing flux smaller. You make it bigger by adding more INCOMING. And then it is NOT the energy from the sun that piles up and causes the extra warming. Then it is your extra addition of energy from the cooler atmosphere that does the trick.
And this is a direct violation of the laws of thermodynamics.
James (Rollins), I think I see the reason for the difficulty. We’re comparing net sensible and latent heat flows with individual radiation flows. Work with me here.

Let me take as an example the Kiehl-Trenberth global energy budget:
Now, set aside all of your considerations of whether it is right or wrong and look at what is being measured. The radiation flows are all the INDIVIDUAL flows, as is typical in multi-body radiation analyses.
But the sensible and latent heat flows are NET FLOWS. Let’s look at the sensible heat for example. It’s about 24 W/m2 as a global net average.
Now, during the day, the surface heats the atmosphere from the bottom, and the direction of the heat flow is from the surface to the atmosphere.
But during the night, the situation is reversed. The surface cools the atmosphere from the bottom, leading to thermal stratification, and the heat flow is from the atmosphere to the surface.
Note that the net heat flow, which is what is gained in the day minus what is lost in the night, is as we would expect, from the surface to the atmosphere. That’s the 24 W/m2 shown in the K/T diagram above as “Thermals”, a poorly chosen term meaning sensible heat loss.
And as a result, while as you point out the heat loss to the atmosphere during the day is quite significant, so is the nocturnal heat gain. it is the difference of these two, the net heat loss, that is shown above and that I have been discussing and referencing.
w.
Willis Eschenbach says: (to)
March 30, 2014 at 12:35 pm
david(swuk):
March 28, 2014 at 4:51 pm
So, ok bud, but your Bach is just a sharp as that Berger Bite I saw in your response to (Stephen was it – can`t reel back easy to check) so why the huff? Making you look “All Warmist” it is!
As for the term “magician” I of course was referring to the disappearing up your sleeve act you put on Solar N/IR Radiation when slipping from “Solar Radiation” to “Wavelenghts of Visible Light” in your claim that the Atmosphere does not absorb SR. There is no refuge for that switch and use of the words “denial” and “he knows….” are well justified in coming from a Scientist of your distinction on such fundamental fact.
But you don`t even stop there Sunshine in dishing me 5degC night sky temperature when it`s more like -40degF (pardon please my slip in writing deg,C) and the related and totally absurd 340W/M2 which is about all we are getting (minus the Real World wind chill effect) in full Sun here @ur momisugly ~50degN just now.
But tempus fugit and I must wish you an early to you bon nuit if not goodbye on this subject just yet.
James Rollins Jr says:
March 30, 2014 at 4:12 pm
James, this is a perfect example of why I insist, not ask but insist, that people quote what they are objecting to.
To start with, who are you addressing? Me? Kristian? Curt? Someone else?
Second, what are you claiming to be a “bald-faced lie”? Accusing a man of lying without quoting the lie is a sneaky underhanded tactic. IF it is me that you are accusing of lying, either quote me or leave me alone.
Third, don’t put quotes around words that are your own. Those are YOUR WORDS, not mine, and I said nothing remotely similar.
You are a piece of work, James. I’m trying to play nice with you, but your rage-filled invective makes it hard.
w.
Kristian says:
March 30, 2014 at 4:43 pm
No, no, that’s not what he said. Read it again. He did not say that HEAT can pass from cold to hot.
He said that ENERGY can pass from cold to hot. If you shine a flashlight at the sun, the sun gets warmer … but not as much as the flashlight warms. In other words, there is a real physical flow of ENERGY from the flashlight to the sun. And there is a real physical flow of ENERGY from the sun to the flashlight. The energy is transferred in the form of photons of light.
But the HEAT, which is the NET ENERGY FLOW, only goes one direction—from hot to cold.
If you don’t understand what I just said, or if you disagree … buy a thermo textbook, I can’t help you.
w.
Kristian: Do you really not understand that electromagnetic radiation carries energy with it? Seriously?
Do you not understand that streams of electromagnetic radiation pass through each other unimpeded, like the example of two flashlight beams I recently brought up? And that this means that the energy these carry with them pass through each other as well?
Do you not understand that if a body absorbs electromagnetic radiation, it absorbs the energy carried by that radiation, and that this increases the internal energy of the object?
Do you not understand that if a body emits electromagnetic radiation, it emits the energy carried by that radiation, and that this reduces the internal energy of the object?
Do you not understand that we have known how this works down to the photon level since Einstein’s and Planck’s day, a hundred years ago? (And I will side with Einstein and Planck any day over Kristian!)
Do you not understand that a photon does not carry any information about its source, whether it was from thermal emission or not, and if from thermal emission, what the temperature of the emitting body was?
Do you not understand that for even for a thermally emitted photon of a given wavelength hitting a body of temperature T, the receiving body has no way of knowing whether the photon was from the “colder” (longer wavelength) part of the spectrum of a higher temperature body, or from the “hotter” (shorter wavelength) part of the spectrum of a lower temperature body?
Do you not understand that that the body’s probability of absorbing this photon and the energy it carries (= h * v) depends only on the body’s absorptivity at this wavelength, and not on any property of the emitting body?
Do you not understand that the metaphorical “heat flow” you keep talking about is simply a convenient way of talking about the difference between the countervailing gross energy flows carried in the electromagnetic radiation in the opposing directions – that it is not a physical entity at all?
Do you not understand that in any system but the absolute simplest of systems – those with more than two bodies, those with bodies of differing absorptivities/emissivities, bodies whose absorptivities/emissivities change with temperature, etc. – must be analyzed in terms of gross radiative flows, calculating heat transfer at the end?
Once you understand all of these things thoroughly, you can start providing a meaningful contribution to the discussion.
Curt says, March 30, 2014 at 5:23 pm:
I do understand one thing, Curt, and that is that you’re stuck in a naïvely simplistic 18th century concept world where the transfer of energy between two objects at different temperatures, where the hot object heats the cold object, is pictured as happening through distinct instances of heating and cooling in both ends by alternating absorption and emission of streams of ping pong ball-like particles flying along mutually independent and oppositely directed highway lanes that once accidentally put together (in what we today would call a ‘radiation field’) would still be readily distinguishable from one another and could also on a whim just as easily be disentangled again to stand as individual and working entities, take away the one and the other would still be there just as before, completely unaffected.
You STILL don’t understand even the slightest of what I’ve been saying on this thread, Curt. Still missing completely the EXCEEDINGLY simple point I’m making. You just don’t see it, do you?
The vigour of convection from a heated surface depends on the rate at which temperature declines with height as well as the absolute temperature of the surface.
The average temperature of a heated surface depends on both the amount of incoming radiation and the length of time that the radiation remains absorbed by the surface.
The rate of decline in temperature with height (the lapse rate) is set by the rate at which atmospheric density declines with height because the greater the density the more of any incoming radiation can be absorbed.
That all applies equally to atmospheres with and without radiative gases.
The slope of the lapse rate can potentially be affected by the presence of radiative gases in an atmosphere because the kinetic energy carried by such gases tends to spread the kinetic energy at the surface up through the vertical column.
That potentially changes the effective emission height for the atmosphere.
Note that changing the effective emission height need not involve a change in surface temperature if the lapse rate slope changes at the same time.
However, a reduction in the rate of decline in temperature with height also reduces the vigour of convection and less vigorous convection pushes less high up into such an atmosphere so the effective emission height drops back again.
Once more, the decline in temperature with height is determined by the decline in density with height and the original lapse rate is restored.
The reason for that being possible is the ability of the radiative gases to radiate directly out to space which means that the system can lose the same amount of energy to space as before radiative gases were introduced but without such a vigorous convective circulation.
The convective circulation in a non -radiative atmosphere must be more vigorous because it is then necessary to return all the energy flowing into convection back to the surface before it can be radiated out to space.
The thermal effect of radiative gases is, therefore, offset by an equal and opposite convective response.
The rules for condensing GHGs such as water vapour are different and need not concern us here.
Despite radiative capability within an atmosphere, the surface temperature, the effective emission height and the slope of the lapse rate are all kept stable on average over time.
If it were correct that radiative gases could contribute to a warmer surface than would otherwise be the case then the warmer surface would result in a faster decline in temperature with height and more vigorous convection.
The problem with that is that adiabatic convection returns as much energy to the surface on the descent as it takes away from the surface on the ascent so the more vigorous convective overturning would create a positive feedback loop which would result in a warmer surface and faster convective circulation with each successive convective cycle.
The atmosphere would soon be blown off into space by the heat building up at the surface but that hasn’t happened for the past 4 billion years.
Trick said:
“Only the sun using up its fuel can raise surface temperature mean from 255K to 288K enabled by increased atm. optical depth (opacity). Any sort “delay” in energy transmission uses up no other fuel adding 33K energy to earth system which can only come from radiative energy transfer of a nearby star – ”
Mass absorbs solar energy via conduction and any gases then engage in convection which is a slower method of energy transmission than radiation and so must heat the system if the rate of incoming energy remains the same.
No other ‘fuel’ is needed to raise the surface temperature beneath an atmosphere. All the necessary fuel was consumed in the sun.
To clarify, I should have said:
” the greater the density the more of any incoming radiation can be absorbed either directly or from the heated surface.”
It is important to realise that the bulk gases which have negligible radiative absorption capability are well able to absorb conductively from the surface especially when recycled by convection.
James:
Since you tried to pull rank on me by calling me an “amateur”, I will mention my background. I have worked professionally as an engineer for over thirty years now, dealing with thermal issues on an almost daily basis in my work. I have graduate and undergraduate degrees in mechanical engineering, with extensive coursework in thermodynamics, heat transfer, and fluid dynamics.
Some of the interesting thermal issues I’ve dealt with lately in my professional capacity – I mentioned the first two of these above:
A customer of ours is using a power electronic amplifier with a metal heat sink that has been black anodized to enhance its radiative output for cooling. The electrician wiring up the cabinet did not realize that the black anodized surface is not a good electrical conductor and tried to ground the amplifiers through the surface of the heat sink. All sorts of flaky electrical issues resulted.
On a new power electronic design, the electrical and thermal designers want very high-flow fans on the heat sink to keep the electronics as cool as possible, which would extend their lifetime. The marketing people want lower-flow fans so they will be quieter. We are in the midst of testing to validate the thermal model predictions. I am mediating this.
A customer of ours who was using motors in a vacuum chamber at a synchrotron did not adequately account for the added temperature that would result from the loss of conductive/convective cooling, leaving only radiative cooling. I had to work with them to reduce electrical current in the motor to the absolute minimum to keep the motor within temperature specs.
A lot of the world’s large telescopes use our electronics. Most of them are at such high elevations (e.g. 4500m) that they are beyond the standard range of conditions for listed cooling requirements due to the less dense atmosphere. I have had to work with them to qualify specialized cooling systems in these environments.
In one of these big telescopes, the scientists wanted to minimize the power dissipated by our electronics because the convection currents inside the dome were distorting their optics (like the air over a fire). We came up with a scheme of selective “underclocking” of the key digital circuits, which significantly reduced their dissipation at times when high performance was not required.
In each of our heat sink designs, we have to decide whether it is worth the cost of black anodizing the outer surface to enhance the radiative cooling. When there is forced convection as well, we usually conclude that going to a higher-flow fan is more cost effective.
In a new heat sink design, we have to decide whether we should go with an extruded design, where all of the fins must be parallel, or with a cast design, which provides for more flexibility in managing the convective flow, but has more up-front tooling costs.
Since I used to design opto-electronic components, I stay active on those engineering forums. I recently helped an LED lighting designer understand how light from an LED generated in the semiconductor “junction” at ~35C could melt his lens coatings, which meant they were getting close to 100C.
So no, I am not an amateur in this field in any sense of the word.
Now, on to the more substantive points. I said earlier that you read, but you don’t understand. With each post you make, you just confirm my assessment. In your post of March 29, 2014 at 11:43 pm, you object to Willis’ and my use of the basic SB radiative output equation to calculate the radiative output. You say we are ignoring other heat transfer modes.
This is a very odd claim, for several reasons. First, the whole point of Willis’ post was to try to calculate the magnitude of these other modes of heat transfer by subtracting out the radiative transfer. How can anybody claim he is ignoring these modes in an attempt to calculate them? (I also talked about other heat transfer modes in my posts, so I was not ignoring them either.)
Then you link to an Engineering Toolbox page that completely backs up our use of the SB equation. It even emphasizes the difference between gross radiative output and net radiative exchange that we keep talking about, and you still don’t seem to understand. But to repeat one more time: Willis is talking about GROSS output, you are talking about NET exchange.
By the way, the page YOU cite has a very nice link to a table of radiative emissivities of different materials that completely backs up Willis’ claim that metals have very low emissivities and most natural materials have very high emissivities. You claimed that this would require different laws of physics — do you really think Engineering Toolbox believes in different laws of physics for metals?
Now, you seem to be upset that we are not explicitly calculating convective losses in the same style that we are calculating radiative losses. (Any claim that we have ignored them is just factually false.) In Willis’ case – to repeat – he is using a database of radiative measurements to try to calculate the other losses by subtraction. Because in the natural world, it is very hard to compute or measure convective transfer directly given all of the variables in play.) I was able to show that even just gross radiative output from the human body led to a large power imbalance over the metabolic production; computing convective losses would just have made the imbalance worse.
I carefully explained how in typical earth environments, most of this imbalance is compensated for by the ambient radiation. This makes the net radiative balance small, such that the resulting radiative loss, plus convective loss and evaporative loss, often with slight insulation (i.e. clothing), balance the metabolic power gain of the body nicely.
I’m sure that in many of the thermal systems you deal with, convective transfer dominates over NET radiative transfer. In my professional experience, when forced convection is used (i.e. pumps or fans over heat sinks), the convective transfer so dominates over the NET radiative transfer that you can safely get away with ignoring the radiative transfer. I suspect that you often do this.
You keep talking about systems “on earth” having convective transfer as the primary transfer means. But you don’t understand the deeper reason why that is true. Willis and I keep talking about GROSS radiative outputs. Why? Because focusing on this underlying mechanism allows us to look at a much wider set of systems. The reason NET radiative transfer is small for a lot of earthly systems is that we are engulfed in a significant sea of background ambient thermal radiation.
By the way, an incandescent light bulb operating at its rated voltage transfers well over 80% of its power to ambient through NET radiative transfer. Only a small minority of the power is transferred through conductive/convective means. Many high temperature furnaces have multiple layers of reflective radiative insulation to minimize the input power required and to permit higher temperatures to be reached.
But if you don’t understand that NET radiative transfer on earthly systems is relatively minor because the temperature differences are relatively small, so the received radiation is almost as great as the emitted radiation, you aren’t prepared to analyze systems where this is not true.
Just as an incandescent bulb filament at 3000K transfers the large majority of its power to a 300K ambient through NET radiative transfer, a body at 300K can have dominant radiative transfer as well. That’s what the T^4 dependency dictates.
Getting “off earth” now, let’s look at space. We all agree that the mass density is incredibly low, so when we are considering space, conductive and convective transfers are completely negligible. When it comes to radiative transfer, how do we consider it? Well, it’s obvious that space is virtually transparent to electromagnetic radiation – we can detect it from billions of light years away.
Do we treat it as a very cold (~3K) radiating body, or as “nothing”? In any practical thermal analysis, it really doesn’t matter. If you account for the cosmic microwave background radiation that was discovered 50 years ago, you use an ambient radiation level of a few microwatts per square meter. Ignoring this, you change the results by about one part in a hundred million.
If you are in low earth orbit, you can get a few hundred W/m^2 of reflected solar and/or thermal emitted power on one side of you. If you are in direct sunlight, you get ~1360 W/m^2 on one side of you. But there are cases where you get neither of these, and you must be able to survive without any significant GROSS radiative flow coming to you, even as your GROSS radiative output remains high.
Just as with high-temperature furnaces on earth, one of the solutions is multiple layers of reflective insulation. This has the benefit of also working to keep out high levels of GROSS radiative flow coming at you, as with direct sunlight on you.
I brought up the well-known phenomenon of the virtually instantaneous flash freezing of waste water expelled from space ships into space. Since everyone agrees that there is no conductive/convective heat transfer in the virtual vacuum of space, how would you explain it?
No those are your
“frustrated, resentment fueled invective rant streams we all read Willis.
That was you who melted down and went into a paragraphs long invective filled rant that had absolutely zero scientific relevancy about any part of the arguments at hand.
————-
Willis Eschenbach says:
March 30, 2014 at 12:55 pm
your endless
voluble
ranting without making the slightest attempt to provide
citations,
observations,
facts, or
mathematics to support your
manifold claims
is pathetic,
unpleasant,
distractive, and
totally meaningless.
NOBODY CARES WHAT YOU THINK, JAMES.
This is a science site. Look up the meaning of “Nullius In Verba”.
We care about science here, not your ceaseless raving about your incorrect thought processes.
James Rollins Jr says:
March 30, 2014 at 9:11 pm
Great, James, I got it. You’re in a serious snit, plus you have a rogue quotation mark that has inserted itself into your text. I hate it when that happens.
Look, I’m sorry to hear that you have your knickers in such a twist, but so what? Given your past behavior, why should any of us care if you are happy or angry?
Now, how about you respond to this scientific comment of mine above, which is more to the point than your internal state of upset?
w.
You’ve got a cartoon drawn by a clown who thought CERES graphs spelled unstoppable hellfire in the sky, which you apparently believe in. You’re the one with the graph showing more energy leaving an object than hitting it, you respond to how you explain it.
People in the real scientific fields don’t make up charts with more thermal energy leaving objects than arriving. It breaks the laws of physics.
Willis Eschenbach says:
March 30, 2014 at 9:52 pm
Now, how about you respond to this scientific comment of mine above?
Also your graph shows the earth absorbing 324 watts of it’s own previously expended energy; coming down from the atmosphere as backwelling; however the very link you gave,
shows clearly: in figure 3:
“Figure 3 Time series of downwelling infrared irradiance
for May 21, 2007. A clear sky was present from
midnight-midnight local time.
353 watts – not the 400 you claimed it represented,
occurs, in daylight.
That’s
In the presence of the sun. That’s infrared downwelling, total, with the sun.
The amount of atmospheric infrared is much,smaller at night, and there’s most certainly no 400 w/sq/meter infrared back radiation seen.
http://vortex.nsstc.uah.edu/atmchem/docs/DEPSCOR_progreport_9_13_07.pdf
Everybody go look at Willis’ Figure 3. Look at the “353 watts downwelling infrared.”
Look at what time of day it is.
That’s not “back radiation” that’s infrared radiation primarily, from direct solar downwelling.
If you understood what you are talking about you’d have realized space suits aren’t made with heaters.
Pretense you could even calculate the temperature of an object washed with fluid
utterly ignoring the fluid, is exactly what you both did, in front of everyone here.
Repeatedly. You were doing it
while I was pointing out to everyone you were repeatedly doing it.
Nobody told you calculate the surface temperature utterly ignoring the portion removed by convection and conduction. Ever. In your lives.
When radiant transfer from an object is calculated, energy removed by conduction and convection are accounted for first.
Not second.
Then the radiant fraction is calculated.
Don’t pretend the people catching you in these fundamental errors need to have it explained to us.
Don’t pretend that if you are just allowed to say you didn’t do what you just did, it would be like you didn’t do it.
Don’t pretend because you gave up and used wrong calculations
because you can’t begin to dream how to do it with the right ones,
or because no one can, it makes it
like I’m the one who did it wrong, not you.
You did it wrong, multiple times. I caught you doing it wrong.
Regarding water changing phase in space, It’s the change of pressure that makes it turn to ice.
just as the change of pressure that makes it turn to ice in the vapor > ice phase change process in storm cells.
Water doesn’t work in refrigeration systems because when it goes from high pressure to a lower one, it turns to ice.
Moisture In A Refrigerant System
“A single drop of water may look harmless, but to a refrigerant system, it is a monster,
First, it creates “freeze-ups.” Moisture will be picked up by the refrigerant and be transported through refrigerant lines in a fine mist which forms ice crystals at the point of expansion (expansion valve).” Ice crystals retard or stop the flow of the refrigerant, causing loss of cooling.
http://www.robinair.com/acsolutions/acvacuum/acvacuum.php
There’s as much, or more sun-side infrared stream where the space shuttle orbits. If you were in the realm of understanding much of this you’d know why water turns to ice when it’s suddenly depressurized.
You’d also realize the amount of infrared flux where the shuttle orbits is quite high and the concept of there being less infrared where the sunlight arrives full strength is more evidence of you truly, amateurish stature regarding this entire subject Curt.
Curt says:
March 30, 2014 at 8:26 pm
You’re right I’m not formally trained in thermodynamics. I’ve worked around it a lot though.
I’m a mechanical engineer.
I answered you Curt but when I pushed Post twice the thread didn’t appear.
It’s the change in pressure that makes the water outside the shuttle change phase.
It’s what makes it change phase identically in the troposphere in storm cells when it rises as vapor and contracts into ice.
It’s what makes it change phase similarly when water gets into a refrigeration unit, and it’s the reason water can’t be used in a modern compressor type refrigeration unit.
For someone telling everyone we need to study you sure don’t know the answers to a lot of extremely basic questions, Curt.
Good. Then we seem to agree: You let your flux down from the atmosphere to the surface heat it directly, that is, not by reducing the outgoing flux (decreased cooling), but by adding to the incoming flux (increased heating). Can’t do that.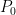 , the system will be in dynamic equilibrium at a constant temperature when the outgoing flux equals this. If we assume that the reservoir is a perfect blackbody that can only radiate energy out through the single transparent surface with surface area
, the system will be in dynamic equilibrium at a constant temperature when the outgoing flux equals this. If we assume that the reservoir is a perfect blackbody that can only radiate energy out through the single transparent surface with surface area 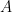 , we get:
, we get:
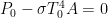


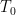 is the dynamic equilibrium temperature of the system in this case.
is the dynamic equilibrium temperature of the system in this case.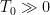 , its entropy change is zero. The entropy gain of the ~0 K “reservoir” of space (per unit time) is thus enormous, the entropy change of the reservoir is zero, and it is left as an exercise for the audience to realize that the entropy decrease of the star is much, much less than the entropy gain of anything else in the system or the surroundings at their much lower temperatures (per unit time). The second law is very happy, and of course the equations above are the first law for this system.
, its entropy change is zero. The entropy gain of the ~0 K “reservoir” of space (per unit time) is thus enormous, the entropy change of the reservoir is zero, and it is left as an exercise for the audience to realize that the entropy decrease of the star is much, much less than the entropy gain of anything else in the system or the surroundings at their much lower temperatures (per unit time). The second law is very happy, and of course the equations above are the first law for this system.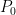 above. Because I am lazy and I only am proving a point, we will make this material perfectly transparent to the incoming SW radiation
above. Because I am lazy and I only am proving a point, we will make this material perfectly transparent to the incoming SW radiation 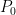 and perfectly absorbing at the LW radiation wavelengths emitted by the reservoir at the range of temperatures it might reasonable reach. This change is not necessary — Grant Petty, for example, provides a more detailed single layer model (still easily solvable) where one can make the absorptivity of the SW and LW radiation independent parameters from 0 to 1. One can make the absorptivity of the layer a full function of the wavelength instead of splitting it into only two pieces. The cost of making these changes is one has to work harder and integrate over the spectra in the end, but it changes nothing of the qualitative demonstration that is my purpose.
and perfectly absorbing at the LW radiation wavelengths emitted by the reservoir at the range of temperatures it might reasonable reach. This change is not necessary — Grant Petty, for example, provides a more detailed single layer model (still easily solvable) where one can make the absorptivity of the SW and LW radiation independent parameters from 0 to 1. One can make the absorptivity of the layer a full function of the wavelength instead of splitting it into only two pieces. The cost of making these changes is one has to work harder and integrate over the spectra in the end, but it changes nothing of the qualitative demonstration that is my purpose.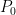 source and emits blackbody radiation as before, but the radiation it emits is all absorbed by the perfectly absorptive intermediate layer. This layer increases in temperature until it emits away all of the radiation it receives as blackbody radiation. The system still has to be in detailed balance — it has to lose energy to space at the rate it receives it,
source and emits blackbody radiation as before, but the radiation it emits is all absorbed by the perfectly absorptive intermediate layer. This layer increases in temperature until it emits away all of the radiation it receives as blackbody radiation. The system still has to be in detailed balance — it has to lose energy to space at the rate it receives it, 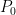 . The side of the thin surface layer of area
. The side of the thin surface layer of area 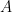 just outside of the transparent surface bounding the reservoir that is facing space therefore has to radiate at temperature
just outside of the transparent surface bounding the reservoir that is facing space therefore has to radiate at temperature 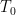 . Outer space is the only actual sink for the incoming power
. Outer space is the only actual sink for the incoming power 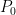 .
. —
— 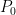 as SW radiation from the external energy source (which could be a laser, a star, anything hot enough to radiate with a peak in the SW/visible range where the thin layer is approximately transparent), and
as SW radiation from the external energy source (which could be a laser, a star, anything hot enough to radiate with a peak in the SW/visible range where the thin layer is approximately transparent), and 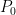 as LW back radiation from the thin layer interpolated between the reservoir and outer space at
as LW back radiation from the thin layer interpolated between the reservoir and outer space at 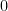 K.
K.


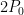 of long wavelength light from the reservoir and absorbs it all. It is at temperature
of long wavelength light from the reservoir and absorbs it all. It is at temperature 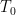 and radiates it symmetrically from its two (inner and outer) surfaces —
and radiates it symmetrically from its two (inner and outer) surfaces — 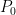 back to the reservoir and
back to the reservoir and 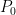 out to space. Space is cold, dark, and far away and acts as a perfect
out to space. Space is cold, dark, and far away and acts as a perfect  K reservoir. The first law is explicitly satisfied for both the reservoir and the interpolated absorber layer.
K reservoir. The first law is explicitly satisfied for both the reservoir and the interpolated absorber layer.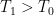 in detailed balance. Its net entropy change as it absorbs energy from the external source
in detailed balance. Its net entropy change as it absorbs energy from the external source 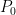 and as back radiation from the shell and and re-emits it all, neither heating nor cooling, back out through the transparent surface that bounds it as blackbody radiation is zero. The net entropy change of the thin absorber layer at the constant temperature
and as back radiation from the shell and and re-emits it all, neither heating nor cooling, back out through the transparent surface that bounds it as blackbody radiation is zero. The net entropy change of the thin absorber layer at the constant temperature 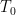 as it absorbs energy emitted from the reservoir at rate
as it absorbs energy emitted from the reservoir at rate 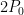 and re-emits it all, neither heating nor cooling, is zero. The entropy change of space is strongly positive as it temperature is very, very low. The entropy loss of the original source of the incoming SW power
and re-emits it all, neither heating nor cooling, is zero. The entropy change of space is strongly positive as it temperature is very, very low. The entropy loss of the original source of the incoming SW power 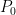 is much smaller than this if it is, as might reasonably be assumed, at either a much higher temperature or the result of operating a laser that overall produces entropy.
is much smaller than this if it is, as might reasonably be assumed, at either a much higher temperature or the result of operating a laser that overall produces entropy.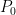 being SW radiation being produced by a very hot star with a temperature vastly greater than either
being SW radiation being produced by a very hot star with a temperature vastly greater than either 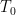 or
or 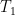 associated with its SW blackbody peak, for example). The only actual source of incoming power is the star. The only final sink in the system is outer space. Net energy transfer occurs from the star to the reservoir, from the reservoir to the thin layer, and from the thin layer to outer space but in dynamically steady state the entropy change of both reservoir and layer are zero anyway. Net heat never spontaneously flows from colder things to hotter things or whatever related claptrap you might wish to assert if you are a disciple of Postma.
associated with its SW blackbody peak, for example). The only actual source of incoming power is the star. The only final sink in the system is outer space. Net energy transfer occurs from the star to the reservoir, from the reservoir to the thin layer, and from the thin layer to outer space but in dynamically steady state the entropy change of both reservoir and layer are zero anyway. Net heat never spontaneously flows from colder things to hotter things or whatever related claptrap you might wish to assert if you are a disciple of Postma. , the reservoir with the LW absorptive layer is strictly warmer with the layer present (heated by SW that bypasses the layer altogether) than it would be without it. No laws of thermodynamics are harmed by this demonstration — the equations are the first law, and if one determines the entropy change of the constant temperature objects in the two systems, the overall entropy change of the Universe is strictly greater than zero. There is nothing any more surprising in this than there is when a block of passive perfect absorber material inside a shell maintained at temperature
, the reservoir with the LW absorptive layer is strictly warmer with the layer present (heated by SW that bypasses the layer altogether) than it would be without it. No laws of thermodynamics are harmed by this demonstration — the equations are the first law, and if one determines the entropy change of the constant temperature objects in the two systems, the overall entropy change of the Universe is strictly greater than zero. There is nothing any more surprising in this than there is when a block of passive perfect absorber material inside a shell maintained at temperature 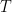 is itself, in dynamic equilibrium where the radiation it receives from the shell is balanced by the radiation it loses to the shell, at the same temperature
is itself, in dynamic equilibrium where the radiation it receives from the shell is balanced by the radiation it loses to the shell, at the same temperature 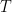 .
.
Of course I can. For one thing, I don’t tell the flux what to do. It doesn’t “heat” or “cool” the surface. The surface itself can be at a fixed temperature and emit radiative flux continuously as long as there is net energy delivery to the surface through all channels to balance it. Is it “heating” flux? No. “Cooling” flux? No. The temperature isn’t necessarily changing. It is outgoing flux. It carries energy away from the surface. It is, in fact, the flux of the Poynting vector through a surface with an outward directed normal, period.
Flux coming down from the atmosphere is nothing special. It has no memory of how it was created. It is electromagnetic radiation, the flux of the Poynting vector through a surface with a downward directed normal, period.
When I put that surface at the boundary of some (otherwise adiabatically isolated) heat reservoir with fixed volume and no internal source of free energy (so it does no work), we have three possibilities. If incoming flux exceeds outgoing, the reservoir will increase its temperature. If outgoing flux exceeds the incoming flux, it will decrease its temperature. If they balance, the temperature will remain constant. If one knows the heat capacity of the reservoir, one can make this entirely quantitative.
Nothing in this system knows about the temperature of anything else. The radiation incident on the surface of the reservoir does not have to come from a single source, be “black body” radiation (that is, have any particular spectral character) or come from a body or fluid that has any particular temperature or a single temperature at all.
The simplest description of a single layer greenhouse effect comes from just such a reservoir. Consider two systems. In the first case, incoming short wavelength flux enters the otherwise adiabatic reservoir through its one transparent surface, where it is completely absorbed by the unit emissivity medium within. It heats until the outgoing long wavelength flux from the reservoir through the transparent surface precisely balances the incoming radiation. If we assign the incident power (the integrated flux through the surface in the incoming direction) the symbol
or
or
Note well that the only source of incoming flux is the SW source indicated — otherwise we are assuming that the temperature of the surroundings of the system is 0 K, so there is no incoming thermal LW radiation. This works find as a model of a planet in a vacuum, as space surrounding the planet in all directions except the SW source (the star) is basically at 3 K.
We can easily compute the entropy changes associated with the energy movement through this system, because the system absorbs and radiates equal amounts of energy at a constant temperature
We now change the system in only one way. We add a thin layer of material that is a very good absorber in the LWIR band emitted by the reservoir, but is nearly transparent to the SW radiation delivered as
The reservoir absorbs energy from the
This surface radiates symmetrically on both sides, of course, as it is a thin layer of material that is a good conductor and its LW emissivity equals its absorptivity (Kirchoff’s Law). This means that it also radiates power $P_0$ back at the reservoir. The reservoir receives total power
The temperature of the reservoir in steady state is thus determined by:
or
The thin layer is in perfect balance. It receives
How about the second law? Well, as before the reservoir is at a constant temperature
In other words, the second law is entirely happy. Net energy always flows from hotter things to colder things (if we imagine
But in the end,
rgb
rgbatduke says:
March 31, 2014 at 12:34 pm
Dang, Dr. Robert, I do so enjoy your comments. They are detailed and solid, without getting sidetracked in the endless minutiae.
That is the clearest mathematical physics-based explanation I’ve read of why the Second Law is not harmed in building a greenhouse.
Live long and prosper, and continue to comment while you do, would be my wish …
w.
PS—As Dr. Robert commented on another thread, my own descriptions of how the “greenhouse effect” works break no new ground … but they are my own explanations that are a result of how I ended up understanding the poorly named “greenhouse effect”. Remember that I am totally self-taught, so I had to figure out for myself exactly what was going on.
I can’t express my surprise when I realized that the greenhouse effect works because a shell has two sides while a planet only has one side, and that the effect itself has nothing to do with gases, CO2, or even an atmosphere, you can build one out of steel … anyhow, my take on the same subject:
The Steel Greenhouse
People Living In Glass Planets
The R. W. Wood Experiment
So rgbatduke above proposes a perfect Black Body with a fixed window that is transparent to Solar Spectrum plus IR & LW EMR and which contains a reservoir of material that can reach a temperature at which it achieves radiative equilibrium with a Star by use of that fixed perfect window ALONE – AND – then go on to achieve an even higher temperature of equilibrium by the addition of a veil that is opaque only to the wavelength of its initially outward LWR.
Have I got that right rgb?
rgbatduke says, March 31, 2014 at 12:34 pm:
I’m sorry, Robert, that you had to spend all that time and effort to write such an extended post to try and distract from the real issue at hand here with words on top of words on top of piles of red herrings. For you see, all the words and red herrings in the world won’t help you cover up your central flaw, the blatant and embarrassingly obvious violation of physical laws that you commit.
Your base premise is wrong, Robert. And this renders your entire exercise thereafter in trying to drown my argument and displaying to the world your proficiency in physics utterly hamstrung.
I have to ask you the same thing I asked Curt: You simply don’t understand even the slightest of what I’ve been saying on this thread? Still missing completely the EXCEEDINGLY simple point I’m making? You just don’t see it?
I’ll try once more, then, to make it as clear as possible to everyone here what you’re doing.
Robert, what we’re trying to analyse here is a ‘heat transfer situation’. And trying to analyse such a situation, that is, interpret and describe what we think physically happens in a specific process giving a specific result, the first thing we need to make sure is that our explanation satisfies the Laws of Thermodynamics. If it doesn’t, our explanation, our interpretation of what’s going on, no matter how plausible and scientifically sound it appears to be, is WRONG. Start over. Try again. Then we have evidently misunderstood something essential.
I have explained to you and everyone else reading this thread now maybe ten times what exactly it is that you do that violates the Laws of Thermodynamics. But I guess I have to explain it one more time.
Energy comes in from the sun. It is absorbed by the earth’s surface, a cold reservoir (a heat sink) for the sun, warming it to a certain temperature, though far below the temperature of its hot reservoir, the sun. We will now follow this energy and see what happens to it according to Robert Brown.
First it is, after having warmed the surface, freely emitted back out as a loss, cooling the surface, making way for the next round of energy in from the sun. The energy ejected goes to the next cold reservoir (heat sink) in line, this time for the surface: the atmosphere.
The ejected energy is now absorbed by the atmosphere, warming it to a certain temperature, though one below the temperature of its hot reservoir, the surface.
So, here’s what Robert Brown does next. He takes the very same energy (well, not all of it, but some of it; quite a lot of it, in fact), originally come in from the sun, and throws it back to the surface, back into the hot reservoir, warming it one more time (the first time was when coming in from the sun, remember?), raising its temperature ABOVE where it ended up after the first time.
To sum up, Robert Brown want us to follow him in letting the ‘back radiation’ heat again some more its hot reservoir, where it just before came from as thermally emitted loss, warming the cooler atmosphere.
Remember, the energy coming in from the sun is all the time free to escape the surface again as loss. The extra surface warming is allegedly accomplished only upon the return of the ejected energy. The very same energy warming it on two separate occasions, raising its temperature first one time and then some more the next time.
This postulated back-radiated flux is apparently completely undiminished in power and ability to heat, no matter what it strikes, even after having been absorbed by the atmosphere, warming it. It is treated by Robert Brown just as an independent HEAT flux which is able to go and work as just that, heat, anywhere, even towards hotter.
I will end by once again repeating myself:
“(…) in nature, heat does not pass from cold to hot.
You can’t have the cake and eat it at the same time. But that’s what they expect. They want us to believe them when they say that the ‘back radiation’ is NOT heat, but at the same time they want the ‘back radiation’ to directly give a result as if it were. It can’t not be heat but still heat the receiving system (unless they want to argue that it’s in fact ‘work’).”
You do not reduce the cooling of something by supplying it with MORE energy, by ADDING energy to it. Then what you do is increase its heating.