Guest post by Rud Istvan,
In the 3/19 Wuhan virus briefing with the FDA, team Trump made much of the possibilities for two therapeutic candidates, chloroquine and remdesivir. Having now done informed basic research on both, I found their stories intrinsically interesting, while enabling an early assessment of their chances of success. Hence this hopeful guest post.
Background
Wuhan coronavirus is an enveloped positive sense single strand RNA virus, meaning its core genetic RNA code is just one long chain coding directly for several proteins, surrounded first by a protective viral protein capsid coat, and then a lipid membrane ‘envelope’ from which project so called “E” (for envelope) and “S” (for spike) proteins. The S protein is what the virus uses to bind to and then invade the lung’s epithelial cells in order to hijack those cell’s reproductive machinery to make copies of itself using its RNA polymerase, itself encoded in about 2/3 of the core viral genetics. The newly assembled virions that then bud out to infect new cells also eventually kill the infected epithelial cell. Covid 19 disease is caused both by the death of those cells and the immune system’s eventual response to the infection.
The S spikes are also the reason this virus class is named corona, because the spikes make it look under SEM like the virus is wearing a crown.
Chloroquine
These are actually two closely related anti-malarials, hydroxychloroquine (the small French trial) and chloroquine phosphate (the larger Chinese trial). Both were developed in the 1950’s, and interestingly the main use now is to treat rheumatoid arthritis rather than malaria (which evolved resistance).
The discovery that certain classes of anti-malarials also affect rheumatoid arthritis (RA) was made quite by accident in 1951 by an asute doctor treating malaria in an RA patient. The problem then was the side effects of chronic RA use made them unacceptable for RA. The chloroquines were developed expressly as ‘milder’ side effect anti-malarials, and in the mid to late 1950’s there were a number of papers (I reviewed several for this post) reporting good RA safety and efficacy leading to global approvals for that indication.
The mechanism of chloroquine action on RA has long been well known. It increases a cell’s lysosomal pH. (Lysosomes are membrane bound cellular organelles [think tiny balloons inside the cell floating at a lower pH in the higher pH cytosol] containing about 50 enzymes, discovered and named in 1955.) This in turn changes their ‘leaked’ enzyme balance into the cytosol, which then inhibits the cell’s RA tissue antigen signaling, which in turn reduces the immune system’s attack on the RA tissue, slowing (but usually not stopping) progression of RA tissue damage.
The reason the Chinese and then the French thought to use chloroquine against Wuhan coronavirus is this same mechanism of action, albeit with different sequelae. The viral S protein binds to the epithelial cell wall’s angiotensin-converting enzyme 2 (ACE2) receptor. Raising lysosomal pH changes (via indirect enzymatic action) the ‘shape’ of ACE2 enough that the S protein cannot bind to it, thus preventing cell infection. Chloroquine changes the cell ‘lock’ so the viral ‘key’ doesn’t work. Does not undo damage from infected cells, nor prevent an infected person from shedding existing viable virus, but does stop the spread in an infected person’s body—a promising therapeutic for those testing positive.
Since safety is well known (the main side affect is retinopathy [vision problems] in 25% of patients over 50 that resolves [slowly] after discontinuation), the main FDA legal issue (FDCA Act of 1906 as amended) issue is to determine dosing and duration for this new indication. But for starters, the standard RA 250mg once a day generic cheap pill should suffice for emergency use authorization (EUA). As a ‘Big Pharma’ goodwill gesture, today (3/19) Bayer announced it donated 3 million 250mg chloroquine phosphate pills to the US to get started.
Remdesivir
This is a novel antiviral from Gilead that has a somewhat checkered past. It was originally developed for Ebola, where in African trials a few years ago it was shown reasonably safe but not very effective. It did, however, show efficacy against SARS and MERS in vitro. And, importantly, the NEJM reported a positive case outcome in Seattle patient zero under a compassionate use exception. The patient had visited Wuhan, returned to Seattle, began displaying symptoms, and was hospitalized on symptom day 3. By symptom day 8 X-ray showed clear lower respiratory tract viral pneumonia (diagnostic ‘ground glass’) and supplemental oxygen was started. Patient worsened, and intravenous antibiotics were started day 9. Patient worsened (proving viral pneumonia), so attending physicians consulted with FDA then had Gilead rush the experimental drug by air, with intravenous treatment starting day 10. Patient improved in 24 hours, was saved, and has since been discharged. For those interested, there is this NEJM case report providing a very hopeful proof of principle.
The reason Gilead tested it against SARS and MERS even though those two episodes died out naturally has to do with Remdesivir’s novel mechanism of action. The ‘drug’ is just an analog of the amino acid adenosine, one of the 20 amino acid (only, in all life on Earth, proving a common genetic ancestor) building blocks the viral polymerase uses to ‘assemble’ new copies of the viral RNA genetic code. The polymerase does not recognize the small difference between adenosine and the analog. Flood an infected cell with enough remdesivir molecules, and the polymerase will eventually grab one and add it to the ‘building’ RNA copy. Remdesivir is enough different that the polymerase is then blocked from adding any more amino acids to the RNA chain, so viral replication halts. Neat very basic molecular genetics provided at a basic science 101 level.
What Gilead scientists recognized was that the RNA code for Ebola RNA polymerase was very similar to SARS and MERS RNA polymerase, hence the in vitro testing. And when the Chinese first published the roughly 30,000 base RNA code for Wuhan coronavirus in January, it was evident immediately that it was another good RNA polymerase match, so they started immediate in vitro testing once viral samples were in hand.
Aside from price (Gilead is infamous for its Hep C cure that ‘only’ costs about $100,000 per treated patient), and scaled up availability (none yet, same issue that killed my 3 of 4 EUA for a persistent hand sanitizer in the 2009 swine flu pandemic), there are questions about dosing and treatment timing. There is some thought that remdesivir may not be useful past symptom day 10 or 11, typically when a patient worsens to need an ICU ventilator. The concern logic is simple. Remdesivir blocks virion replication in an infected cell, but not its spread to newly infected cells by virions from previously infected cells. So basically a quantity/quality argument saying eventually blocking further spread when you already need a ventilator for viral pneumonia is futile. Those clinical questions are why China is conducting a double blind (drug/placebo) trial on ~790 patients in Beijing and Gilead is conducting an unblinded smaller trial in the US, starting in Nebraska with Diamond Princess patients. The first results from both will be available sometime in April.
Further observations
Neither chloroquine nor remdesivir are just luck. The rapidity of their development against Wuhan virus reflects the enormously powerful insights that molecular genetics and molecular biology and their associated tools (sequencing, PCR, oligomer synthesis, protein structure) now bring to science and medicine. To echo the contrasts to climate science in my first post on Wuhan, this is as if we actually had now the computational power to avoid parameterization in climate models. Climatologists do not, but virologists do.
Chloroquine probably works, as AW previously posted. It would solve this pandemic’s key issue, progression to viral pneumonia requiring ICU ventilation. New York’s Governor Cuomo said yesterday that he has been told that without ‘bending the curve’ based on Italy, New York will require 27000 ventilators in a few weeks when the state only has 3000. Invoking the Defense Procurement Act cannot solve that mismatch in time without a ‘bent curve’ achieved via social distancing, frequent hand washing, and avoiding touching the mouth, nose, and eyes. All three are difficult but not impossible. Ambassador Dr. Birx is pretty clear about the dire consequences of Millennials ignoring these basic common sense recommendations during Spring Break this week in Florida. Here in ground zero Fort Lauderdale, our public beaches are closed, and the closure is policed.
But chloroquine still has the same Wuhan issue illustrated by its previous use for malaria–evolving resistance. RNA viruses like Wuhan coronavirus mutate rapidly (explained in my first post on this topic). The most conserved protein is necessarily the RNA polymerase. We know this from influenza, where it is the hemagglutinin and neuraminidase envelope proteins (equivalent to Wuhan S) that mutate so the annual vaccine is never ‘right’. Chloroquine may well be effective now, but if Wuhan coronavirus becomes endemic (now likely given its spread in Africa and Southeast Asia), then it is not a long-term solution like a vaccine. But it will probably buy the precious time to get a vaccine.
Remdesivir may be a longer-term therapeutic solution, because it tricks the conserved RNA polymerase. But its cost and efficacy remain to be determined.
*****CORONAVIRUS: STOP GOING OUT – IT IS GOING TO KILL PEOPLE – WARNS ICU DOCTOR
video – 8 minutes 45 seconds
“Stop this virus” ROFLMAO… that to me sounds like stopping the wind. Dr. Chicken Little et al don’t seem to realize that there is no stopping influenza or cold viruses. These viruses were around long before humans travelled the world easily and increased transmission rates.
The sad fact is that everyone that lives long enough in modern societies today will probably get Covid-19 eventually, if not this year then next, if not next year then some year after that. Shutting down the global economy has the potential to be magnitudes worse than the worst case scenario with this novel cold virus. Millions of people died from economic hardship related causes in the USA alone during the Great Depression, we’d better hope a new cold is the worst thing that comes out of all of this.
I don’t know where you get your history from, but you need better sources. There were no deaths from economic hardship in the US during the depression.
Yes, people will probably get the virus someday. However they will be a lot better off if they get it at a time when hospital beds are available and the medical system isn’t in the process of breaking down from over use.
That’s why we try to isolate, in order to slow down the number of people getting the disease at any given time.
Well: I think the idea is that there is significant evidence, regardless of specific reasons, that wealth affects length of life. So picking whether or not people starve to death may not present the full picture. MIT has done studies, and there are lots of other studies.
In US there is less likelihood of people dying because they cannot afford to heat their homes, like in UK. But in general, wealth of a nation leads to decreased mortality, me thinks.
Mark W is correct life expectancy increased during the depression era.
My dad (1926-2017) told me that people took care of each other during the depression and WWII, which made all the difference from today. Now folks try to “watch out for #1” and “make sure nobody gets mine”.
That might have been a mitigating factor for the less fortunate of that previous era.
Penicillin discovered in 1928. If looking for a cause for life span increases during the depression, that was surely also a factor. Diet changes and reduction of smoking (did it?) May also have contributed.
Life spans apparently actually increased during the Great Depression of the 1930’s in the US. See below link for some interesting explanations.
https://www.history.com/news/great-depression-economy-life-expectancy
But the WWII came along, and well, we all know what happened then.
There were no deaths from economic hardship in the US during the depression.
and GOOGLE says:
Of six causes of death that compose about two-thirds of total mortality in the 1930s (Fig. 4), only suicides increased during the Great Depression. Suicide mortality peaked with unemployment, in the most recessionary years, 1921, 1932, and 1938.Oct 13, 2009
so, YOU ARE WRONG and GOOGLE is your friend
“Stop this virus” ROFLMAO…
===≠========
That isn’t the issue. If everyone gets sick on the same day, we don’t have the 5 million ICU beds we would need 10 days later, and the death rate would skyrocket.
But if we can spread this out over time, we can dramatically reduce the death rate. Same number of people get sick, but different numbers of sick get life saving medical treatment.
Good paper, thank you Rud. Lots of bog words. 😉
I’m sending a copy to my doctor.
big words
I liked “bog” better. They sure bogged me down, right clicking for a definition every other word!
“Taiwan Says It Warned WHO About Coronavirus In December, But Its Warnings Were Ignored”
https://dailycaller.com/2020/03/20/who-taiwan-coronavirus-warning/?utm_source=&utm_medium=email&utm_campaign=12322
More grumpy pre-coffee news:
I was scheduled to travel to SE Asia via China on 4Feb2020 and I cancelled because of cascading quarantines due to the corona virus. Now we learn that a coronavirus carrier off a cruise ship was allowed back into Canada on 21Feb2020 without being quarantined – MORE THAN THREE WEEKS AFTER I CANCELLED MY TRIP on 28Jan2020.
I don’t claim to be that well-informed on global health issues, rarely bother to take my vitamins, and am not interested with the popular obsessions of personal health that fascinate younger generations. I don’t watch TV news or subscribe to a newspaper. So how the heck did I know more than three weeks ahead of our slothful Canadian government that something was seriously amiss?
All our government imbeciles had to do was read the news – on January 31, 2020 the USA banned foreign nationals who had travelled to China from entering the USA, and any US citizen who has traveled in China had to undergo health screening upon entry into the country and was asked to self-quarantine for 14 days.
https://www.theverge.com/2020/1/31/21117403/trump-coronavirus-ban-travel-non-us-citizens-china
This is what I’ve come to expect from Canadian governments at all levels – at best, they are asleep at the switch. More likely the Libranos were busy plotting a way to scam billions of taxpayers’ money for personal use – the Justin Trudeau Liberals make the Jean Chretien crooks look like choir boys.
Well in Holland a majority of parliamentarians were refusing a debate on coronavirus als late as jan. 28 and jan.29. Now we’re shy of being a police state like most countries. Time for coffee I guess.
You answered your own question. You told what you don’t use as news sources, all the others are better.
Two of my relatives returned from Vietnam to Canada March 7rh. They had 2 observations. Eveything in Vietnam was closed and noone at YVR mentioned anything about 14 day self-isolation.
We returned to Canada from the US March 2nd by car, same thing. No health questions.
From my experience, it was only after Sophie Trudeau tested positive that the Canadian government began to get off its lazy ass.
Much too busy jet setting around the world giving out taxpayer money and snapping selfies. Karma is a bitch.
PRIME MINISTER JUSTIN TRUDEAU CONVENES THE INCIDENT RESPONSE GROUP FOR AN UPDATE ON THE CORONAVIRUS SITUATION
February 2, 2020
Ottawa, Ontario
https://pm.gc.ca/en/news/readouts/2020/02/02/prime-minister-justin-trudeau-convenes-incident-response-group-update
Today, Prime Minister Justin Trudeau convened the Incident Response Group to discuss the Government of Canada’s response to the novel coronavirus,
Minister of Foreign Affairs François-Philippe Champagne, lawyer
Minister of Health Patty Hajdu, Bachelor of Arts, Master of Public Administration
Minister of National Defence Harjit Sajjan, former detective with the Vancouver Police Department, a former Lieutenant Colonel with the British Columbia Regiment
and Minister of Public Safety and Emergency Preparedness Bill Blair, 39 years with the Toronto Police Service, the last decade as its Chief of Police.
JUSTIN TRUDEAU’S CORONAVIRUS TEAM: A LAWYER, TWO EX-COPS, AND A BACHELOR OF ARTS. ZERO QUALIFICATIONS FOR THIS TASK.
PLUS ÇA CHANGE, PLUS C’EST LA MÊME CHOSE.
CANADA INVOKES QUARANTINE ACT: Mandatory 14 Day isolation
March 25, 2020
https://torontosun.com/news/national/canada-invokes-quarantine-act-mandatory-14-day-isolation
Watch Justin Trudeau trying to look Prime-Ministerial as mandatory quarantine of those entering Canada from foreign countries is announced.
BUT TRUDEAU AND HIS FELLOW-IMBECILES ARE TWO MONTHS TOO LATE.
See my note at:
https://wattsupwiththat.com/2020/03/20/wuhan-coronavirus-therapies-scientific-background/#comment-2942967
I have no medical background, but I KNEW ENOUGH TO cancel my foreign trip on 28Jan2020 – THAT WAS THE TIME TO IMPOSE THIS QUARANTINE. NOW IS TOO LATE.
There’s a horrible tale on https://going-postal.com/2020/03/fred-and-doris/ telling the story of an old couple subjected to Boris’s ‘self exclusion’. It basically tells the tale of a ‘don’t-care’ government attitude. Now, as an ex-Pommie, I’d like to think that it was merely carelessness on the part of the UK rule makers, but then, as a South African, I remember the stories of the Anglo-Boer war ‘concentration camps’ and the ‘don’t-care’ attitudes that led to so many deaths by disease. Having grown up in the post WW2 years, I have to say: Not My Country!
Yet, for whatever reasons, the UK has suffered far fewer deaths per million population than EU countries. As noted, Germany isn’t comparable because of different statistical and medical practices, eg not testing those who’ve died of pneumonia and other causes for Wuhan virus.
Italy has suffered 80 deaths per million, Spain 30, the Netherlands 8.0, but Britain only 3.5 so far. Being an island might help.
If you look at Willis’ latest coronavirus post, you will see that UK is only just below Italy for death rate against time since first case. If you subdivide the UK deaths into London and ‘the rest’, London has had about 10 deaths per million, and the rest about 2.5.
So far in the US, cases and deaths are also concentrated in big cities, and Italian cases in the North, with so many Chinese workers.
The U.K. is unlikely to track Italy in cases and deaths.
Willis’ graph starting all curves from time of first death shows the UK far below Italy.
“as been told that without ‘bending the curve’ based on Italy, New York will require 27000 ventilators in a few weeks when the state only has 3000”
Spare us.
Those stats are not based on infection increases but testing increases. For all we know real infection rates could be decreasing in reality.
So what about Italy? May be all there drugs are Mafia counterfeits or otherwise faulty and that’s killing Italians. Meanwhile the Koreans are using quinine and the Israeli govt. is sending millions of pills to the US.
Trump’s fear spreading newsconferences have made the country crazy, and certainly susceptible for misleading statistics. Sound familiar?
Quinine derivatives are also Zinc ionophores, they increase the concentration of intracellular zinc. The viral replicase enzyme of n-COV-19 is gummed up by Zinc slowing replication.
I have B-blood type (I naturally lack the antigen that is most effective against viruses)
I catch every virus that goes around taking weeks to months to get better. I’ve tried every product, by far the most effective is “zinc”
I’ve tried zinc throat sprays and nose sprays which are not effective as the zinc rapid melts. If you have a sore throat, break up a tablet and place small pieces between your cheek and gum and let it slowly melt. Most effective before bed absorbing all night, when you wake up the sore throat is gone. Do you feel a cough coming on? Crush a small amount between your fingers and breathe the dust, sucking air sharply. Same with the runny nose, crush a small amount into dust with your fingers (smaller than half a pea) and breathe it in. The powder will stick to the membranes delivering the medicine where it’s needed. The results are amazingly quick and will stop a cold before it starts.
Max:
I am B+ and grew up catching every dang cold, flu and sinus infection that went around. I was not aware of blood type vs virus until recently where I read O type is most resistant and A type least resistant. Is B type in the middle?
I have asthma when around cats and some other allergens, and get bronchitis every time I catch a cold. Running and cardio allowed me to gain control over it –which when I was 12 years old was against my doctors orders to be active. So I decided to not follow his advice.
At 55, and since my mid 40’s, I don’t catch so many, and when I do, they are extremely short lived.
It’s the Zn (CA/Mag/Zn) Vitamin D3, C, quercetin, juiced powders, that made the difference in my life. The thing is, I was most afraid of growing old and dying of suffocation. Now I am more robust than when I was in my youth.
One funny thing… when I get sick the point where I crave steak I know I’m just about over it. It’s a fluffy science barometer for me. Good health!
I’m A1, rh- and I nearly never suffer from any virus, only rhinovirrus. Rarely a influenzal infection, with low fiever, often without, so not knowing if it was a viral infection.
Born 52, smoking.
Please expand on the ‘juiced powders’, please…
Please expand on the ‘juiced powders’, please…
I vary the use of brands. Costco used to have a green green powder called Amazing Greens. They also still have a powder with lots of reds and greens, called Juce, Trader Joes used to have “Reds” and “Greens”
I like to make sure I get all the colors and a huge variety of well known organic juiced veggies so that all of the phyto’s are available to feed me.
Today I continue to use Juce and my wife is a naturopath and orders greens called Nourish Greens by Apex Energetics.
She can tell what I am in lack of through muscle testing, but I also eat way more of these juices than is called for.
I do this mostly because we will probably never stop finding specific vitamins and nutrients to supplement, but food has a nearly infinite ability to provide what we need, especially plants.
I swear by them, and have gotten rid of cancers that doctors promised would not go away.
People say it’s anti oxidants, or give a myriad other reasons. All I know if that my health has turned around over the past 15 years as I expected age would force it in the other direction!
Mario: like you, as a child I developed bronchitis after every cold. These attackes were sheer hell – I’d be fighting for every breath I took during the night, and vomiting as well. Our local doctors were wonderful, a home visit and antibiotics to follow brought relief. I had pneumonia twice as a child as well.
My doctor advised exercise – perhaps swimming, but the idea of being exposed to chlorinated water at the local pool didn’t appeal to me or my parents. Then I took it into my head to learn to ride a bicycle , at around age ten. I rapidly became a cycling fanatic, I rode hard, even trying a bit of racing in my teens. My bronchitis attacks feel dramatically within a year – the effect of exercise was unbelievable.
My eternal gratitude to my GP, my parents, and my older brother who built up my first bicycle for me. He found an old Raleigh frame which had been poorly brush painted. I stripped it to bare metal, my brother got it sprayed a nice cherry red at a local motor cycle dealership and then assembled it for me. All of those who helped me are gone – they gave me the precuous gift of health, and I remember them with fondness for their kindness.
Thank you for the comments! Exercise is magical…indeed!
I’m B- . Not heard of this virus problem before and I’m not going to test it. I have MGUS and diabetes both from unknown sources although I recently read a paper that suggested pylori ? bacteria, those associated with stomach ulcers can trigger both illnesses. The surgeon I had when treated for ulcers said that anglo Saxons lack a certain enzyme which can allow Pylori to thrive. It’s all conjector though
When I was child, when a sore throat was knocking at the door, I got a desinfecting tablet of Chinosol to gargle with after dissolving it in water. As I remberer it well because of effectiveness, I used it later too, it’s rare I have a sore throat.
Chinosol is a quinine product, chinolinsulphate-potassiumsulfhate for external use
I carry around with me a little refillable nip in which I use Crown Royal. I get about 30 sips per one once nip… I use it when I feel a cold may be trying to hit me.
Canadian rye whisky – breakfast o champions
good on ya…!
There is a suggestion that gargling with mouthwash reduces viral shedding in those already infected. No hard evidence.
I am B+ and almost never catch a cold and have never had the flu. The few colds I do catch is consistently caused from not getting sufficient sleep.
I may be an anomaly to your theory so I am not denying it.
Good for you!
That’s why a sample size of 1 is never enough data for true analysis… But whatever you’re doing, keep on doing it… and get sleep unless the pay off is worth it…
I saw what you did there ……
I thought I was being cute/clever? Please expound sir!
I saw that you were being cute/clever …..
Thank you, philincalornia, I got the error message on a full page warning, so I posted to another thread.
I am also B+, never have had the flu, and only get a cold every few years, then it is mild.
So now there is a sample size of 2.
I’m B+ and my immune system eliminates incipient colds, flu, infections in 36 hrs or less.
The whole blood type thing may be a total red herring, no-one has yet determined what the actual proportions of blood groups were in the are of Wuhan affected. They data is blood groups of those who died against WORLD AVERAGES and you get considerable deviation from that in many countries.
In the study was also a table with total proportions of the blood groups in Wuhan then compared with the infected. Though could of course have other reasons. Let’s say one type of blood live at one side of the city where the infection rate was higher. Will take another study to be sure.
I use the snuffing up of salt water into my nose and spitting it out my mouth at the first signs of a cold or flu. You can get a spray at the drugstore. About 1/3 to 1/2 tsp in a cup of warm water – it should not be enough to ‘burn’ your nasal passages. Pour in the palm and suff strongly up your nose. You will swallow a little bit. Add this to washing your hands. For Corona, I don’t know, but it wouldn’t hurt at worst.
It was noticed years ago that North Atlantic fisherman didn’t seem to get these diseases commonly while landlubber friends and family did. Presumably fisherman everywhere get some benefit but rougher seas obviously would be a factor.
There is a device called a neti pot, looks like a miniature tea kettle.
It is meant to be used with little packets of a salt crystals that is buffered and measured into an amount which will make an isotonic saline solution.
This is poured into one nostril while standing in the shower, and it pours all the way through the sinuses while you keep your head tilted to the side.
After pouring one pot through one nostril, you mix up another batch and pour it through the other side.
Washes out the sinuses but good.
Back when I was on chemo one time I had something going on where my sinuses smelled really awful.
The cure was every day a nasal decongestant, and a antihistamine, and neti pot as many times a day as I could get around to.
I kept it up afterwards.
I think it washes stuff out of the sinuses that accumulates over an entire lifetime.
If you look at a diagram of the sinuses in the face, it is obvious that stuff that gets in there has no easy way to get out…hence neti pot with hot saline.
http://medivisuals1.com/images/products/detail/R15359_01XG.jpg
Another view:
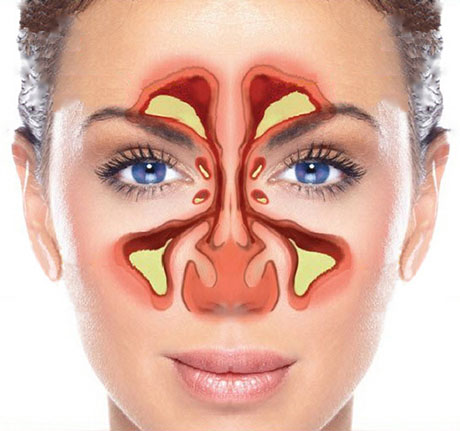
I do the same thing using neilmed sinus rinse. It comes with buffered salt mix.
I use spring water heated to about 98F. The right temperature, lack of chlorine and proper salt sooth and work like a charm. I lay back holding my sinuses full of the water for a minute to let it dissolve the dry stuff.
Importantly, DO NOT hold both nostrils when blowing your nose after this, as it will push the water with infection through Eustachian tubes. NOT good!
I do the same thing using neilmed sinus rinse. It comes with buffered salt mix.
I use spring water heated to about 98F. The right temperature, lack of chlorine and proper salt sooth and work like a charm. I lay back holding my sinuses full of the water for a minute to let it dissolve the dry stuff.
Importantly, DO NOT hold both nostrils when blowing your nose after this, as it will push the water with infection through Eustachian tubes. NOT good!
PS fantastic sinus descriptions! No wonder I used to get so many infections.
re: “I think it washes stuff out of the sinuses that accumulates over an entire lifetime.”
Hmmmm … Horseradish, hot yellow mustard? Seems to activate the sinus ‘flush’ mechanism too?
Nicholas,
So glad that the chemo worked! (and your sinus treatment, too)
And, very glad that you are still commenting at WUWT… 🙂
Take, over there (well, most people are sound asleep at 1:55 AM 🙂 ),
Your ally for science truth (that’s TRUTH, phone — heh, it tried to insert “fiction” there),
Janice
“Take CARE …”
Something for those considering Chloroquine to consider if they take ACE inhibitors. Those drugs deplete zinc, so you might need extra zinc to take advantage of chloroquine’s zinc ionophore property.
I’ve ordered some quinine supplement tablets as linked in the original WUWT post. Any idea what an appropriate dose might be?
I have seen 500mg.
In Australia a secret trial has been reported with good results in paptients from a cobination of Kalet, an HIV drug, and hydryxychloroquine. it has been released to 50 hospitals.
quinine won’t do anything read again the name of the drug … details matter.
Quinine is not good? I’ll have to switch back from G&T? Buggers!
It will help you with malaria but will do nothing for covid19 🙂
MikeP
I strongly advise against self-dosing! You really need to be under the care of a doctor while taking chloroquine. I suggest that you read the following article:
https://www.msn.com/en-us/health/healthtrending/virus-drug-touted-by-trump-musk-can-kill-in-just-two-grams/ar-BB11rIHa
For those strongly advocating for the immediate widespread use of hydroxychloroquine, note what the head of the FDA said during a presidential ‘presser’:
“However, Dr Hahn pointed out some concerns about the drug as a possible treatment for the virus. ‘We may have the right drug, but it might not be in the appropriate dosage form right now, and it might do more harm than good’, he warned.”
Note especially the remarks about side-effects in this article:
https://qz.com/africa/1822701/coronavirus-trump-says-malaria-drug-chloroquine-can-work/?utm_source=YPL&yptr=yahoo
And for those who are using Roy Spencer’s remarks about the anti-correlation between COVID-19 and malaria, note the remark that the use of chloroquine has declined in recent years, both because of a loss or efficacy and side-effects.
The 500mg is for Rhumatoid Arthritis. It was mentioned to show that it is comparatively safe at such high doses. Chinese paper indicated from tests that 6.9mg per dose was 90% of optimum. You may have to check to see what other studies for this indicate, but 500mg would seem excessive.
I think it is 6.9 milligrams per kilogram.
beware side effects – including death! (with 2x safe dose)
https://bnf.nice.org.uk/drug/chloroquine.html
Medcram on YouTube have an interesting video (Coronavirus update 34) explaining the way zinc works to protect the infected cell and why certain factors inhibit its usefulness.
How about increasing temperature and, especially, humidity in the offices, homes, schools, cars etc.? It is a well known measure to kill viruses outside of the human body, and to slow down transmission.
https://papers.ssrn.com/sol3/Papers.cfm?abstract_id=3551767
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3583861/
https://markets.businessinsider.com/news/stocks/humidity-helps-in-the-fight-against-covid-19-virologists-report-1028995035
UV light and high intensity. Instead of walking around cleaning desks with rags and alcohol sterilize them with UV. I’m not sure what intensity and duration would be needed but some water filters use them to kill off virus contamination. You don’t want to look at it without protective goggles.
Leo that is good idea. I sent those recommendations to CDC and never heard anything back.
After testing in hospitals, on March 17 Italy also listed hydroxychloroquine as a drug with preliminary positive results against Wuhan virus:
https://www.aifa.gov.it/-/azioni-intraprese-per-favorire-la-ricerca-e-l-accesso-ai-nuovi-farmaci-per-il-trattamento-del-covid-19
The Italians should be testing all the medications they been using for counterfeits and defects.
Teva to Donate Potential COVID-19 Treatment, Hydroxychloroquine Sulfate Tablets to Hospitals Nationwide
https://www.heraldsun.com.au/news/victoria/melbourne-scientists-twoyear-coronavirus-preparation-set-to-save-thousands/news-story/c957ea882c0fe579683d16faf63c83ee
HOSPITALS GIVEN HIV, MALARIA MEDS TO FIGHT CORONAVIRUS
A handful of Australian COVID-19 patients – some of the first to be infected with the deadly virus – were successfully medicated with malaria and HIV drugs in a secret trial which will now be rolled out around Australia within days.
Such was the success of the secret treatments fifty Australian hospitals will be given HIV medication Kaletra and malaria treatment hydroxychloroquine in a clinical trial which promises to “cure” the virus.
Don’t grow old (eventually this is a problem). Stay healthy (mostly in your own control).
“Don’t grow old”? Then you’re dead……
Yes, eventually it’s a problem.
In Mel Brook’s German accent, “Ja, Herr Schwartz is a heavy sleeper.”
Hey Rud ….. what have you read about the combination of HCQ and Azithromycin?
It was the combination used to clear one cohort of the French patients in 6 days. IIRC, chloroquine alone cleared 60 percent while the combination cleared ‘all’ in 6 days. Why azithromycin antibiotic helps dunno unless some of the pneumonia was in fact secondary bacterial pneumonia, which quite common especially in elderly patients—the reason there are pneumonia vaccines for the two most common causative bacteria.
Maybe an artifact. The study was small (40 patients) and the endpoint a bit murky (no detectable virus in the nose and throat is NOT the lower respiratory tract). There is a youtube of a US doctor analyzing the study rather critically.
Rud: Great article and very well written!
That is my experience wrt to the use of azithromycin. Secondary infection is treated with antibiotics. In my case, I was reasonably sure I had no bacterial infection and waved the antibiotics. In no way did I want to kill off my hopefully healthy gut flora with antibiotics.
At 55, my immune system is evidently still working well enough to enjoy (/sarc) short duration viral infections… feed the body with loads of mixed nutrients… carefully. Be in touch with how you feel… which is tough since we are relatively good at feeling change, but not good at feeling stasis.
One needs to invest in keeping the old body ahead of the curve, where medical does not really chime in much in that respect. So it’s a lot of anecdotal research and accumulation of knowledge –that requires conscious change in what works and does not. Fluffy science sometimes.
no detectable virus in the nose and throat is NOT the lower respiratory tract)
Don’t forget, the corona tests are based on curtailment exactly there, thoroat and nose, so what.
Nose and throat are primarily where the virus is spread from?
Nose and throat are good places to swab for the virus. It will be there.
However, the virus attacks the lungs… not really easy place to collect a sample. But the mucus from the lungs will make its way to sinus and throat through cilia action.
Sneezing and choughing is the way of infection, using the way as Mario wrotee it.
Yup! Basic stuff now. And I am NOT a doctor.
The French treatment results with combined HCQ and Azithromycin are encouraging, and can be seen in this video presented by prof. Didier Roualt (in French)
https://www.mediterranee-infection.com/coronavirus-diagnostiquons-et-traitons-premiers-resultats-pour-la-chloroquine/
The table at 15:24 shows a difference between No treatment vs. HCQ alone vs. HCQ + Azithromycin, although the patient number is rather small. Concerning Azithromycin, there are several studies in the PubMed showing it having antiviral activity, too. Personally, if I would catch the COVID-19 with pulmonary symptoms, I would prescribe myself HCQ + Azithromycin + zinc.
15% of the patients “dropped out” because of death or transfer to the ICU
I was just wanting to see if you had any new info. Azithromycin also has some antiviral properties, particularly against rhinovirus. It is a protein synthesis inhibitor and may have some secondary impacts on the ability of the virus to replicate its proteins, though I don’t know if SARScoV2 produces its own enzymes for protein synthesis or if it uses the host cell ribosomes.
Anyway, I know it is cheap, as is HCQ, …. seems worth it to give it a try, cause as the French study noted, the CQ has an effect on its own. The Redemisvir is probably going to be expensive unless the gov makes them sell it in the cheap.
And price is your main concern when you are fighting for your life with viral pneumonia?
Also saw a report that chloroquine can also reduce or eliminate cytokine storm which for many COVID patients is the fatal factor causing lung fluid buildup. Any comments on how this works and effectiveness of chloroquine in preventing?
There are better drugs for that.
“Azithromycin inhibits constitutive airway epithelial sodium channels in vitro and modulates downstream pathogenesis in vitro” by Fujikawa’s team in Japan is available in English on line as free full text.
I am not certain the French chose Azithromycin merely on the basis of antimicrobial action. There are some indications the drug contributes disproportionally to anti-biotic spawned bacterial drug resistance &, as such, employed with clinical discretion.
By the way R. Istvan – I’ve enjoyed reading your WUWT comments over the years & find them well written.
Thanks Rud for the article. Much appreciated. I am now a lot less dumb than I was!
15% of the French Patients were removed from the trial because of death and transfer to ICU
26 started in the trial, 1 died, 3 went to ICU,
20 stayed in the trial, and the results are from those who stayed in
beware the survivor bias
I’m guessing skeptics forget the dedication to statistical significance.
Based upon reported cases, US death rate has fallen to 1.35%, but many cases must be going unreported.
https://www.worldometers.info/coronavirus/
Our current case rate of 50 per million population is about on a par with the UK (48).
Not to be picky, but it’s really not accurate to say “the United States”, as this pandemic is primarily isolated to NY. WA seems to plateaued. CA seems to be containing it fairly well too. All the other states are in just double digits or in the 100s.
I’d like to see the FDA actively evaluate these treatments immediately. In particular, HCQ + Azith, both cheap generics.
True, the disease, if not the virus, is indeed concentrated in a few big, mainly coastal cities, although every state has reported at least one case, with WV the last to be hit.
I have been wondering why the U.S. experts are so resistant to using Chloroquine, which is cheap and readily available. Growing up in Pakistan, I was prescribed Chloroquine twice for malaria. Subsequently, I was advised by my company to take Chloroquine for two weeks before I traveled to malaria infested area. I never suffered from adverse side effects. Is it because they prefer the expensive, U.S. developed Remdesivir?
FDA will test it too.
Who said they were resistant to using it?
Dr. Anthony Fauci.
You are mis-interpreting his remarks.
Unless you think that finding the correct dosage isn’t important.
Exactly, before my trip to brazil I was given a mandatory prescription to start weeks b4. This drug MAY have some prophalaxis ?? properties or at least lessen the impact of the infection. Reports seem to indicate that quantities of HCO are being gathered. Why? My local drugstore had some weeks ago, but is now trying to get more.!!! Why would it not be immediately available to any who can tolerate it? Worst case, we don’t get malaria.(joke) If we don’t get back to normal, the cure may be much worse than the scourge. If this drug knocks down the curve dramatically we all win; if not, we ‘ve lost nothing. The cost of HCO is nothing compared to the cost of continuing in place.
Doug
theres still a lackof testing done so actual numbers would be higher
John Tilman: I get the same calculation as of this morning. As I have always believed, testing will bring the denominator up by at least an order of magnitude (my hunch) because there are no tests available. The measurement has been, and still is to a large extent, skewed to people who are sick enough to seek medical help.
This site is very good. https://www.worldometers.info/coronavirus/country/us/
I hope that the mild, misdiagnosed or unnoticed and unreported cases are indeed an order of magnitude greater than those reported.
If fatality be one percent, and 60 million Americans were infected, as during the 2009 swine flu pandemic, then 600,000 would die. If the mortality rate however actually be 1/10 of one percent, then “only” 60,000.
My hope however is that Wuhan won’t infect 60 million. If we can hold the spread to, say, six million, then 1/10 of one percent would mean just 6000 lethal cases.
At this point, who knows? We can only hope and take whatever actions promise to limit infection and severity.
Yes sir… and once in a while I need to ground and remind myself that this virus is a new and probably soon-to-be just part of the common cold for which humans have dealt with forever.
With all of our efforts to stop the influenza virus and with vaccinations prevalent in society, we have many millions infected annually in the US.
I don’t get vaccinated for flu because so often the best guess as to the prevalent strain most likely in each winter is wrong.
But I guess the older I get, the better the odds a flu vaccine confers.
I am in the same camp and same wrt flu shot. It’s always last year’s strain and this year what goes around is something new. I have not gotten flu in a decade by the way.
These antivirals taken seasonally in low doses might be better than vaccines or maybe would be complimentary.
Scissor, the keyword complimentary!
1) The human body is a wonder of self defense especially when fortified with good ingredients.
2) Antivirals can make life for the virus very difficult.
I would resort to number 2) after number 1) screams for help, and only when I know that’s the case. 🙂
” John Tillman March 20, 2020 at 1:24 pm
I don’t get vaccinated for flu because so often the best guess as to the prevalent strain most likely in each winter is wrong. ”
And twice they had it totally wrong to what was actually in circulation yet there was no increase in deaths. So nobody got vaccinated to what was in circulation and no increase in deaths shows it is in placebo category (IMHO).
Vaccines work best against genetically stable targets. Smallpox was the best target you could ever ask for. Influenza is the other end of the chart.
I tend to disagree, call me a sometimes anti vaxer. I take the big vaccinations of course… the bad ones that are known to stay in circulation until wiped out largely.
John Tillman said:
“I hope that the mild, misdiagnosed or unnoticed and unreported cases are indeed an order of magnitude greater than those reported.”
Presumably you also hope that they somehow or other don’t infect others.
No, that is not what was meant by John Tillman. Think about what he meant. We have not been counting people who have symptom or mild symptoms and have no idea how many are actually infected. The solid assumption is that there are many more. No one wants that to be true. But we want to know how many there are that have not been counted. The common sense realization of this is that we could rest assured that this dilutes the given mortality rate. If it’s an order of magnitude greater than we have counted this far, that means the mortality rate is likely equivalent to the Flu… and panic will subside. It’s basic math or pre-algebraic.
PS – that is where most of the spread is coming from… undetected cases mostly
mario says:
“PS – that is where most of the spread is coming from… undetected cases mostly”.
Exactly. And given the inability to test the whole population of the world (or the USA) then you need to go with the data available.
Otherwise, it’s just wish-thinking. And, another useless academic navel gaze.
What Mario said.
I was referring to the magnitude of unreported cases. However many there are are already contributing to the spread. Or not.
The hopeful part is that fatality rate is ten times lower than derived from reported cases.
Exactly John
I’ve been looking at the data and it seems that this virus spread seems to top out at about 2 mo. Provided that China is telling the truth, their spread covered Jan-Feb. Washington State has plateaued (Jan 20-Mar 20) and is now reporting just a few cases per day. This could be good news, as maybe the NY outbreak will ease up in April, hopefully sooner with the social distancing. Italy (mid Feb) should plateau off around the middle of April if the two month window holds true.
That’s with draconian measures. Something you will not see here until it gets really nasty.
Most of the dropping china cases was due to the government shutting down companies, banning gatherings, and forcing people to stay home. The number of new cases started to drop after the quarantine.
In washington most of the original cases were in one assisted housing complex for the elderly. They were all quarantined and many of the stafff and other secondary contacts were traced down and also quarantined. South korea also did this and added extensive testing of the population allowing them to track down many unknown cases and implemented targeted quarantines.
So the 2 month drop you are seeing is mainly due to people and governments increasing sanitation and implementing quarantines. Also note it is now believe that some of the first cases occurred in China way back in October of last year. So it continued to spread for 4 months before emergency actions were taken.
Increasing temperature and humidity indoors reduces transmission of flu-like viruses. Warmer and more humid air kills viruses outside the human body faster.
This is a well known poor man’s measure. People used it in Ukraine against the Swine Flu in 2009.
Now a study confirmed that it works for the COVID-19 virus.
“Increasing temperature and humidity indoors reduces transmission of flu-like viruses.”
So why aren’t the authorities recommending the use of humidifiers?
Fascinating stuff, Rud. Thanks for your work.
Like warfare, one needs defense in depth when dealing with complex issues; there is no one magic pill. Hopefully these drugs will give us time.
Also as in strategic bombing, you have to go back and hit the target again.
Adenosine isn’t an amino acid, but a nucleotide, ie the nucleobase adenine attached to a ribose sugar and a phosphate group. It’s one of the five such nucleotides in DNA and RNA, arguably the most important.
Remdesivir is an adenosine nucleotide analog.
Also amino acids are the building blocks of proteins, not nucleic acids.
Yep, I was going to make the same comment.
Yes, me too, but putting on my Mr Pedant hat, adenosine is a nucleoside and becomes a nucleotide when 5′-kinased to AMP.
There’s an international trial in progress to test the various drugs. 10 countries are joining in. Guess which major country isn’t taking part?
Canada?
I have not heard a word on the news in spite of contacting them.
He said “major” country … 🙂
LOL. Except for size Canada ain’t major (and hockey but that season on hold).
G7!! Canada (used to) punch well above our weight and still is considered a ‘major’. Trudeau progressives are trying to drag us back to the stone age!
Basketball, insulin and Oscar Peterson! Great contributions to the human mosaic. A sport enjoyed (playing and watching) by hundreds of millions, a medical treatment that has saved the lives of tens of millions if not hundreds and one of the all time great jazz piano masters.
Not to shabby for a small population. Not bad at all. Now, “What have you done for us lately?”….
I’ve read the Chinese have also tested high dose IV vitamin C and found it to be helpful in treating COVID-19. However, I have not seen any mention of this in the US, almost as if it is being intentionally ignored. If the vitamin C efficacy is true, it would be an easy to use treatment and should be widely available and it would be a shame if it was not put to use.
Add lime to your Gin and Tonic.
(I’m serious)
“add lime to your…” And do what me and my wife do. We use half a lemon and a touch of pure maple syrup added to a tall glass of water every day. After washing the lemon or lime, we grate the skin to let the zest call into the drink. Huge amount of bioflavonoids and extra C
“a lemon and a touch of pure maple syrup added to a tall glass of water every day.”
I’ve just discovered that adding vanilla extract can also make water more palatable.
I’ll try it… huh!
I’ve found that adding scotch to water also makes it more palatable. Especially if you skip the water..
Will Lemon also work?
For the last time tonic has quinine .. not the right thing other than it makes up part of name of the active drug being discussed. Quinine as a single ingredient has no know effect for covid19 so go an read again the name of the active ingredients.
10 g per day for moderate cases; 20 g per day for severe cases. No deaths, and 3-5 day reduced hospital stays. One patient near death received 50 g over 4 hrs and survived. No bad side effects for any of the patients.
http://orthomolecular.activehosted.com/index.php?action=social&chash=0a09c8844ba8f0936c20bd791130d6b6.148&ref=facebook&sh=eb7027deb126917dbc7dd4cf5defc54d
The vitamin C was administered by IV. Oral vitamin C is rapidly eliminated in the urine so plasma concentrations remain relatively low. As an aside, IV vitamin C was recommended by Linus Pauling (1970’s) for some cancer and other treatments. However the NIH refused to test by IV and “rigged” the studies with oral C with negative results. FDA/NIH has historically preferred denigrating or ignoring any cheap therapies for major medical issues and instead pursues approval of expensive drugs/fixes to support the pharma industry. Major distrust of the FDA by a significant portion of the public is one reason alternative medicines and approaches receive so much support.
So just because they (FDA) say there will be “approval” of vitamin C or chloroquine for COVID treatment doesn’t necessarily mean much, as they can delay by insisting on more “studies” (which I’ve have heard Dr. Fauci refer to). Of course rapid approval of an expensive anti-viral med would be right up their alley. Possibly Trump could go around them with an executive order. I guess we will see how this plays out.
The doses would cause problems for your kidneys so they give it via IV.
High doses can cause diarrhea if taken orally, that’s why IVs are given. There is controversy over whether they cause kidney stones.
http://orthomolecular.org/resources/omns/v09n05.shtml
Yes, and here’s the biology of vitamin C that justifies its use as a vasopressor in septic shock, and, along with it’s well-known antioxidant effects, may be why the Shanghai medical authorities have recommended vitamins C for COVID-19 treatment. https://www.youtube.com/watch?v=RA7obbfGg_o
Yet, not a peep about this in the mainstream media.
The FDA seems not setup properly to deal with a novel virus that becomes a pandemic. In a perfect world we would do months or years of testing before allowing the drug. Unfortunately we do not have the time. If an older person has this virus and has underlying condition there is significant chance they will die. We have to weigh this against what we know about these drugs at the moment.
Suppose for instance we know based on past cases an 80 year old man with a pre existing lung condition has a 70 percent chance of dying under current best case practices. Do we try the drug on him ?
Doctors can try these drugs under “compassionate use’ and obtain FDA approval. I uderstand that doctors are using these without FDA approval for this particular use.
Actually, we use drugs “off label” every day all the time.
No need to wait for an FDA approved indication. HCQ is already FDA approved. Has a known and reasonable safety profile. We have small randomized placebo controlled trials.
That’s being said this is NOT the first time HCQ has been studied as an antiviral. Every previous time it has nor panned out.
Stevek
If that little old man was you, I bet your answer would be “hell yes”.
I’d say,
“In a[n] [im]perfect world [such as we live in] we would do months or years of testing before allowing the drug.
Definitely hell yes !
What is it going to do?Kill him? Fire in the hole! Light the candle!”Etc.
A drug can be used “off label”. None of the drugs being tried on my chronic cough are for cough, but research shows they can sometimes work. After 25 years of coughing, off-label doesn’t look so bad.
I generally agree with you. However playing devil’s advocate here, aspirin was the new drug they used in similar untested fashion to fight the spanish flu in 1918. Today it is widely believed that this considerably increased the death rate. Aspirin lowers fever and reduces inflammation which is why they used it. They were fighting the symptoms. However fever has a reason and a purpose. They were interfering with the body’s efforts to fight off the infection.
I have always thought to “sweat it out” When I get a fever, I get as warm as I can tolerate. And interestingly, your body feels cold, so it encourages you to wrap up. If you can get into a sauna (that’s not too dry) or steam room and make sure to hydrate, your cold could be quelled quite fast.
However if you’re in bad health, you may not be able to take it… that’s where things go wrong fast.
PS – today I would not go to a public steam room with sickness…
same thing works for me. If the bed isn’t soaking wet from sweat, you need to add another layer.
Hah! Absolutely…
The sweat did a job on me (through evaporative cooling) when I get up to empty my bladder… Dang the shivers were intense as I crawled back into my damp sheets… Unfortunately for me it took 5 days for fever to break with this WuFlu.
It wasn’t just that… The dosages of aspirin that were given were absolutely huge, sometimes several grams per day. At those dosages, continued over several days, one of the major side effects is pulmonary edema, which doesn’t help at all when you are trying to fight a lower respiratory infection.
Excellent summary. BTW, adenosine is not an amino acid, it’s a nucleoside.
Anyway, it would seem that a cocktail might have good effect, and at least one is being evaluated.
Nucleotide.
My mistake. It is a nucleoside, ie the nucleobase and ribose sugar combo, without the phosphate group.
This kind of error does not instill confidence – just kidding. I used to know some of these things off the top of my head. Now if I want to be sure, I look it up.
My first mistake was trusting my memory.
“My mistake.”
Really?
Fancy that.
Nucleotides are phosphate esters, nucleosides are not. Adenosine is a nucleoside, which is being confused with the nucleotide ATP.
” The ‘drug’ is just an analog of the amino acid adenosine, one of the 20 amino acid…”
NO — it is a nucleoside, AKA nitrogenous base, which when phosphorylated form the bases that make up the genetic code of DNA or RNA. As far as I ever heard, there are 5 of them. There are, however 20-odd amino acids that form proteins. This kind of basic error does not induce confidence in the rest of the post.
For stuff like this, Wiki is reliable: https://en.wikipedia.org/wiki/Nucleotide
Nucleotide, not nucleoside, which is the nucleobase and sugar combo, without the phospate group.
Sorry, it is a nucleoside, ie lacking the phosphate group.
Albeit with a minor correction that Remdesivir is a nucleotide analog of one of the four nucleotides found in RNA, this is a solid summary of its drug effects (and potential limits) on this coronavirus as well as chloroquine and hydroxychloroquine.
I’m learning a lot about RNA, cytosol and molecular biology. 😉 More that I think I really want to know, but this is orders of magnitude better than listening to the various talking heads on TV/radio/social media.
Hopefully this will be a light at the end of the tunnel.
The higher the number of unreported cases, the lower the actual death rate.
Interesting, it’s been reported that virtually all of the deaths (>99%) in Italy involved patients with other serious health issues.
“…virtually all of the deaths (>99%) in Italy involved patients with other serious health issues.”
Sounds like heatwave statistics.
What would we do without nanny-governments and scary news media?
I found a post that said 13 healthcare workers in Italy have died from the coronavirus, so this is concerning and somewhat contradictory to the above.
Apparently, this is ongoing research. It appears that as death reports are sent from hospitals to the National Institute of Health, they are examined. A day or two ago the number was 105, this last one was 300+. So the numbers will likely adjust over time until all reports are examined.
“I found a post that said 13 healthcare workers in Italy have died from the coronavirus, so this is concerning and somewhat contradictory to the above.”
Probably they got a heavier viral load. (But why should that make such a big difference?)
Immune system overwhelmed?
Like the hero doctor in Wuhan, dead at only 36 and presumably healthy.
https://www.dailymail.co.uk/news/article-8130479/99-patients-killed-coronavirus-Italy-existing-illnesses-study-finds.html
Quote below is from link in prior post. It went into moderation because the word “k!lled” is in the URL
I’m wondering if the high mortalities in diabetes and hypertension patients are due to their pre-existing conditions or due to the ACE inhibiting drugs they take, which upregulate ACE2 expression possibly leading to nastier infections.
Here’s the link in case the first post never comes out of moderation. You’ll have to change the “!” to “i” in the word “k!lled” in the URL.
https://www.dailymail.co.uk/news/article-8130479/99-patients-k!lled-coronavirus-Italy-existing-illnesses-study-finds.html
Nearly every morbidity listed in that article – diabetes, hypertension, atrial fibrillation and ischemic heart disease are treated with ACE inhibitors and angiotensin receptor blockers (ARB), which upregulate ACE2 expression in the lungs (don’t know how prevalent for the last two, but they do appear to be treatments for those conditions).
Could this imply that not the hypertension is the risk factor, but the ACE inhibitor is the culprit?
That’s what I’m wondering, and researchers in that link are as well. A letter in the Lancet said that there are other options for hypertension patients, but I can’t remember what they were. Something with the word “channel” in it.
icisil, that’s probably the calcium channel blockers – I’m gonna switch my medication back to those tomorrow, they work well for me. Thanks for the link.
Yep that’s it.
https://www.bostonglobe.com/2020/03/19/metro/is-it-safe-take-ibuprofen-amid-coronavirus-pandemic/
As an aside, my doc has me on amlodipine for hypertension (high blood pressure). This is a calcium channel blocker, not an ACE inhibitor.
Yep, that’s the one – Amlodipine 5mg is what this Golden Age traveller is prescribed.
Calcium Channel blocker for r-e-l-a-x-e-d heart muscle & blood vessels – Ahhh. ;>)
That and some diuretic to keep me running! Way back in ’67 I was a DanD!
Hey, Covid19, we can’t dance together – we go nuthin’ in common!
Steely Dan – Hey Nineteen = https://www.youtube.com/watch?v=eAHQ-9Fniac
I’m also blood type A+ = the only test I ever got an A+ on!!
I know I’ve posted this link to the Information is Beautiful website, so pardon the duplicate. They’re updating their COVID-19 graphics every 2-3 days with the latest figures, and they’ve added a section showing how “multiple conditions” increase the risk of death.
https://informationisbeautiful.net/visualizations/covid-19-coronavirus-infographic-datapack/
Now, these are serious conditions like active cancer, COPD, diabetes, stroke, etc., so contracting the Wuhan virus on top of these would be like being hit by a speeding truck. Of the deaths in this study (Italy), 25% had one condition, 26% had 2 conditions and 48% had 3 or more conditions. Of the deaths, 1% had no serious condition. (Keep in mind the usual caveats: data in flux, small sample size, etc. Check the graphic in a few days for updates.)
Thanks Rud, here’s a way we can help with research.
Right now there is a distributed computing effort to model the proteins and provide data to researchers going on at U of WA called Rosetta@Home. It is administrated by Berkeley U’s BOINC, which was born out of the now discontinued SETI@Home program.
You must download and install the BOINC manager software, which allows you to choose when and how clients use your CPU or GPU. It’s here==> https://boinc.berkeley.edu/download_all.php
Choose Rosetta from the client list during setup/first use. Some of the models you run will be part of the COVID research.
Rosetta will only use CPU, so if you have a GTX series or other powerful NVidia GPU, you can volunteer it at GPUGRID, where cancer, AIDS, virus (and soon COVID-19) research is always ongoing.
Earn badges, get intrinsic satisfaction, even signal virtue if you must, but the power of distributed computing is awesome to me and I enjoy participating in it. 😁👍
My computers run work units 24/7 and I use them normally, with the help of a great free utility called Process Lasso ===> https://bitsum.com/ which manages how apps use the CPU.
In the UK there are two malaria drugs Chloroquine (available with prescription) and Qualaquin (available without prescription to treat leg muscle cramps), both have side effects, the most serious are:
Chloroquine – Anxiety (attempts at killing oneself)
Qualaquin – Anxiety (behaviour change, similar to drunkenness)
If need be, I’ll rather be drunk than suicidal.
🙂
Chloroquine Side Effects
https://www.drugs.com/sfx/chloroquine-side-effects.html
Qualaquin Side Effects
https://www.drugs.com/sfx/qualaquin-side-effects.html
I miscalculated in a comment the other day, by taking the dose for prevention as being the same as for treatment of active malaria (same dose, but the prophylactic dose is weekly, not daily). Sigh…
Anyway, having recalculated, two to three cups daily of tonic water may do the job of prevention (looking at the different uptakes). So a few daily G&Ts ARE a reasonable precaution to take. Number depending on how strong you like them.
Writing Observer, I have a big bottle of tonic handy and recommend squeezing a lemon into into your glass. I admit to putting a tsp of sugar in the lemon juice. Very tasty.
Other serious side effects are, Retinopathy (long term use or high dose) and QT interval prolongation; so should never be taken with many anti-arrhythmic / antidepressants / psychotropic / anticonvulsants.
They also don’t work at all on the critical ill, they help those with mild symptoms. Many cases go straight from almost no symptoms to very severe and it offers no protection at all to that group. How many it stops from going from mild to severe needs proper clinical numbers under identical situations …. so two like for like hospitals with similar patients demographics. You see wide variation in all the statistics from within a single country in different regions, so getting exactly what the efficiency is hard to calculate.
Has anyone noticed that most of the links in Anthony’s original item on covid-19 and chloroquine have been barred by Google as ‘breach of terms of service’?
Is this because he has shifted some to point elsewhere, or might it be an attack by someone trying to close the site down?
I would be interested in a Mod or WebMaster reply….
Dodgy Geezer
I read the same thing on twitter today, somebody stated that the chloroquine research done in 2005 which shows it works against corona was not showing up on google anymore…
That is interesting.
There is an argument that, if chloroquine were advertised as fully proven, completely effective and readily available (which is not yet true), people would then cease self-isolating and avoiding crowds. If this happened, the medical services of major cities would be completely overwhelmed with new infections. Even if there was a simple treatment, the peak of people requiring attention would be too great for the existing infrastructure, and avoidable deaths would result.
So it makes sense to ‘flatten the curve’, even if there is a simple cure. It will be a delicate balance, because the techniques used to flatten the curve will result in disruption, and that will probably mean avoidable deaths….
So suppression of this news MIGHT be officially ordered. Equally, persons who see WUWT as a political enemy promoting evil ideas which will damage humanity might also be tempted to try to interfere with the free flow of information. And it would be easy for those persons to inform Google that WUWT is distributing dangerous fake news, and should be closed down – which Google would probably do as a matter of urgency. And, of course, it might just be an error….
The administration of the web site ought to be able to determine which, if any, of the above is true.
Sad that Big Government thinks censorship is preferable to facts!
The public aren’t listening to facts they are acting as a dumb mob.
LDB,
Yup:
Groups working in Salt Lake City and Hong Kong in 2004:
“Compounds approved for therapeutic use and in vitro inhibitors of severe acute respiratory syndrome coronavirus (SARS-CoV) were evaluated for inhibition in the mouse SARS-CoV replication model. A hybrid interferon, interferon alpha (IFN-α) B/D, and a mismatched double-stranded (ds) RNA interferon (IFN) inducer, Ampligen® (poly I:poly C124), were the only compounds that potently inhibited virus titres in the lungs of infected mice as assessed by CPE titration assays. When mice were dosed intraperitoneally (i.p.) with IFN-α B/D once daily for 3 days beginning 4 h after virus exposure, SARS-CoV replication in the lungs of infected mice was reduced by 1 log10 at 10,000 and 32,000 IU; at the highest dose of 100,000 IU, virus lung titres were below detectable limits. Ampligen® used i.p. at 10 mg/kg 4 h prior to virus exposure also reduced virus lung titres to below detectable limits. Nelfinavir, β-D-N4-hydroxycytidine, calpain inhibitor VI, 3-deazaneplanocin A and Alferon® (human leukocyte IFN-α-n3) did not significantly reduce lung virus titres in mice. Anti-inflammatory agents, chloroquine, amodiaquin and pentoxifylline, were also inactive in vivo, suggesting that although they may be useful in ameliorating the hyperinflammatory response induced by the virus infection, they will not significantly reduce the replication of the virus, the inducer of inflammatory response. Thus, anti-inflammatory agents may only be useful in treating virus lung infections if used in combination with agents that inhibit virus replication. In summary, the data suggest that induction of IFN by mismatched dsRNA or actual treatment with exogenous IFN-α can inhibit SARS-CoV replication in the lungs of mice.”
Note the lack of ambiguous language, focus on quantification of results, and the fact that this was not a study of pouring some chemicals on cells in a glass dish…which is very well known to not give the same results as what happens when live subjects have an infection and are given that same drug.
https://journals.sagepub.com/doi/abs/10.1177/095632020601700505
It’s the algorithms Sunny 😉
https://www.consumerreports.org/consumer-protection/fight-against-coronavirus-misinformation-shows-what-big-tech-can-do/
We need an internet without google & co
What kind of search results do the other search engines return?
_Jim, DuckDuckGo is fine I use it. There are also others that I’ve tried but didn’t like Startpage.
re: “DuckDuckGo is fine I use it. There are also others that I’ve tried but didn’t like Startpage.”
Here’s “the dope” on Startpage:
Minor point: adenosine is a nucleic acid, not one of the 20 amino acids that forms poly-peptides (proteins). It’s the “A” base precursor in the RNA and DNA coding 4 nucleic acids letters AGCU/AGCU as Rud mentioned.
errata: AGCU/AGCT (RNA/DNA)
I miss edit.
Sorry, adenosine is NOT an amino acid. It’s a purine ribonucleoside (nucleotide base = purine) attached to a sugar (ribose). Remdesivir is an analog of adenosine which is one of the natural substrates of the viral polymerase. The analog binds to the active site of the enzyme and cannot be processed further thus blocking this key enzyme’s activity (and viral replication).
Rud Istvan, thank you for the detailed and informative posting. I am on house quarentine here in Mendoza, Argentina, and enjoying good weather and practicing golf in my backyard with my dogs as caddies. The reality of this China Virus seems to be that following guidelines to flatten the infection rate curve and give the medical community a chance to get ahead of the virus is the best practice. For sure, those of us with money have an ability to guard ourselves and families that the others simply don’t have, and no amount of government intervention will save them all. Good on A. Watts for this involved and informative website, and good luck to all.
Hmm. Keep research records and you could probably publish (not here) on the “Effects of Canine Saliva and Dentition on Golf Ball Putting.” I have some old work acquaintances that would probably be far more interested in that than the latest CoViD paper…