Author: Dr. Michael Chase, 16th May 2019
SCOPE
This article describes the method and PRELIMINARY outcome of a relatively low cost “laboratory” (kitchen) experiment on the response of seawater pH to increases in atmospheric CO2.
When additional CO2 is added to the air above seawater the chemistry of the water is expected to change, and there is much focus on the pH measure of acidity. I find that the change in pH when equilibrium is established is readily detectable with a low cost portable pH meter with 0.01 resolution, but the measured size of the effect appears to be significantly smaller than reported in the recent scientific literature. This article deals mostly with the experimental method and results.
EXPERIMENTAL SET-UP
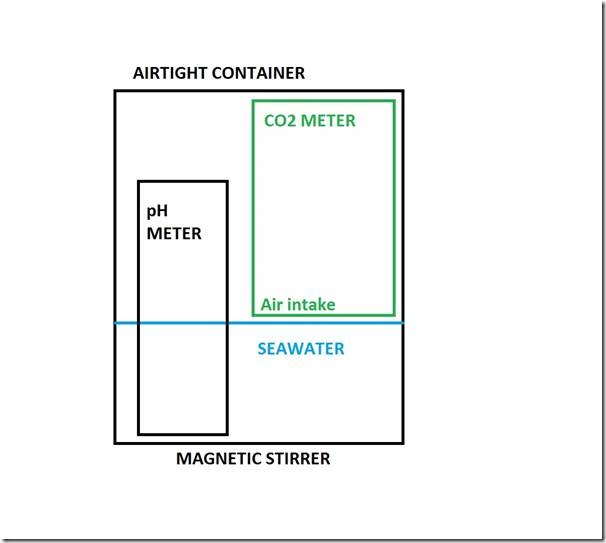
A schematic plan of the experimental apparatus is shown in the following figure. A 2-litre “airtight” food storage jar contains seawater and air, and portable (battery powered) instruments to measure seawater pH and atmospheric CO2 level, with alterations to the latter by injection of pulses of additional CO2. The water is mixed continuously by a magnetic stirrer.
The actual experimental system used is shown in the following photo, the pH meter is white, and the CO2 meter is grey, with its air intake visible just above the water surface. Food storage technology provided the 2-litre airtight container, the plastic “table” that holds the CO2 meter just above the water surface, and “Cling Film” to protect the CO2 meter from getting wet, with a hole cut for the air intake. Details of the meters used, and of the magnetic stirrer, are given at the end of the article.
The original plan was to use a small portable electric fan to mix-up the air, but it was found that the batteries ran-out after about an hour, insufficient time to achieve pH equilibrium, which is found to take around 10 hours.
CO2 GENERATION & INJECTION
CO2 was generated by adding vinegar to bicarbonate of soda in a beaker (with splash guard), injected via an unused ear syringe, a plastic syringe or eye drop dispenser could be substituted:
In a draft-free room the CO2 generated in the beaker can be thought of as a relatively dense fluid which remains there, and which can be drawn into a suitable device and injected into the 2-litre glass container. The aim is to get around 1000 ppm of CO2 in the 2-litre glass container, which is 1 part in a thousand, i.e. 2 ml of CO2 at atmospheric pressure, which can be easily generated and captured.
SEAWATER
Seawater samples of around 450 ml each time were obtained from the shoreline near Weymouth in Dorset, on the South coast of England. The shoreline had some seaweed, which may influence the pH. Each sample was stored in a sealed 500 ml plastic bottle in the “laboratory” overnight to warm it to the laboratory air temperature.
PRELIMINARY RESULTS
Each run of the experiment lasts around 12 hours, which is roughly the observed time required for pH equilibrium to be achieved, with data recording at 15-minute intervals. The runs start mid-morning and run to around 10 pm, a period in which the room temperature remains fairly constant.
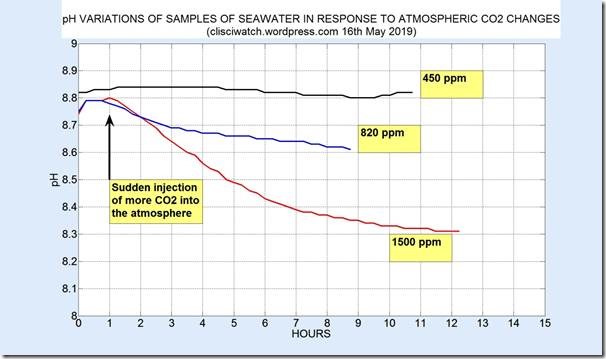
The following figure shows early results for 3 runs, one with no injected CO2 (uppermost data), one with injected CO2 such that around 820 ppm was measured (shown in blue with a constant +0.2 shift in all pH values), and one where the final CO2 level was around 1500 ppm:
DISCUSSION
If the recent growth rate of around 2.4 ppm per year of atmospheric CO2 is maintained then the level at the end of the 21st century will be around 600 ppm, with a change in seawater pH of around 0.15, which is considerably lower than figures quoted in the scientific literature. Note that the change of 0.15 in pH applies to seawater of 8.8 pH, which is at the high end of the observed range. Hydrogen ion concentration at a pH of 7.8 is ten times higher than for a pH of 8.8, so the change of pH in 7.8 pH seawater may be even lower than 0.15.
EQUIPMENT DETAILS
· pH meter: Hanna Instruments HI 98108
· CO2 meter: Kane-alert-CO2
· Magnetic Stirrer: Hanna Instruments HI 190M
Note: Water in a sealed container gives 100% humidity, and condensation may eventually damage the electrics of the meters, everything is thoroughly dried at the end of each run.
FURTHER INFORMATION
Readers can follow developments, and make comments, at the following website:
An Ocean Acidification Experiment


Where are the calcium carbonate minerals (a’la crushed sea shells) lying on the bottom of the “ocean”.
I would consider that a usefull “to do” addition to this experiment – repeat and compare results.
And, what about the photosynthesizing organisms. Photosynthesis is an alkalizing activity and can raise the pH of a bay or estuary above 10 on a sunny day.
The slight change in pH measured above shows that the seawater was a buffer system. But, without organisms doing their thing in the water, all bets are off.
How many people know that, when water moves through a coral reef, it comes out with a lower pH than when it went in, as general metabolism is a acidifying activity.
There is really no such thing as a constant pH in the real world, just as there is no such thing as a global temperature.
Plus 100, Charles and Ken!
Yes, I like it when my first thoughts appear right at the top of the comments.
are people really this stupid?…..of course they are
You can’t lower pH until you deplete the buffer
While I applaud the authors diligence in pursuing this experiment; I would prefer an experiment that more closely resembles the reality of the ocean environment.
So the first thing to eliminate is the magnetic stirrer. There’s no such thin in the oceans, and the effects of stirring totally distort what is actually going on. The author mentions ten hours for the system to reach pH stability. That is a surprising amount of time, but very important.
A recent article in the San Jose Mercury news paper, purported to describe a very scientific study by supposed scientists and chemists working at the Monterey Bay research facility.
The word pH was never mentioned in the article; just “ocean acidification”. So much for the “Scientific” claims. How could any chemist talk about “ocean acidification” and never mention pH, so I dismissed the entire article out of hand. The deep ocean which is in reasonable Henry’s law equilibrium with the well mixed atmosphere, is NEVER” acidic, and on line papers I quickly found described the pH as being in the 7.8 and higher range, up to 9.0.
The particular gist of this SJN article was the assertion that Silicon Valley CO2 emissions from automobiles was “pouring” through a coast range pass into Monterey Bay and acidifying it.
Well if they had measured their acidification in San Francisco Bay, which is much closer and shallower than Monterey Bay, they could have found even more massive ocean acidification; enough to eat holes in the bottom of ships in SF Bay.
I can think of few locations better for dumping automobile exhaust CO2, than Monterey Bay. It is very deep very close to shore. 10,000 ft depths are the order of the Monterey Canyon.
I did find two graphs showing the pH of the North Atlantic, and the North Pacific from the surface, going down to 3,000 meters.
At the surface, both oceans are very alkaline in the pH 8-9 region, and that pH drops rapidly down to 500 meters. Unfortunately there was no ocean Temperature profile to go along with that.
The north Pacific dropped down to the 7.8 or so range in 500 meters, and then slowly inched back up over the next 2500 meters, to respectably alkaline numbers.
So what is going on. Well for starters, you need to know that the downward long wave IR radiation from the atmosphere that is “warming” the planet, is all absorbed in the top 50 microns of ocean surface water which is thus warmed above the temperature of the top meter of ocean water. As a result of this temperature spike at the surface, the very surface layer has a lower equilibrium CO2 absorption.
Below that surface “film”, the temperature drops more or less linearly down to the thermocline, so the amount of CO2 the water can hold increases with depth, so the dissolved CO2 diffuses under a concentration gradient diffusion process, and literally pumps CO2 from the surface down to the colder ocean depths. Because of the constant depletion of that very thin surface layer, the water is soaking up CO2 and pumping it to deeper water.
So that 10 hours or so that our present author mentions for pH stability, is very important.
What is absorbed into the ocean surface is GASEOUS CO2, which doesn’t acidify anything. You just have to open a bottle of 7-up to see that what is dissolved in the pop is CO2 gas, which immediately bubbles out when you relieve the pressure.
So what happens now to that dissolved CO2 gas over apparently some hours, is that the CO2 reacts with the hydroxyl ions from the dissociated water, to form the weak acid: izzat (COOH)2. IANACH I think that is carbonic acid. The point being that is how dissolved CO2 eventually does change the ocean water pH, but because of the temperature gradient of the deep oceans, the “acidification” reduces at greater depth; not increases.
Anybody done any scuba diver on oceanic reefs at 500 meters depth to see how acidification is damaging the reef environment ??
No if you want a benign place to put some local excess CO2, Monterey Bay Canyon is a great place to put it.
GES
More on ocean acidification
https://tambonthongchai.com/2018/09/29/ocean-acidification-by-fossil-fuel-emissions/
This experiment is no good!
Don’t you know that only modelling predictions are acceptable nowadays?
Exactly! Haven’t we learned by now that playing with numbers on a computer is vastly superior to going out in the field and doing actual science?
Did they have Institutional Ethics Approval – can’t publish without that.
what’s the end result, how do fish react
I think you meant to ask “How do the fish feel?” Also, how do the polar bears feel about the fish?
Oh, and I’m afraid that Nature will not be able to print details of your interesting experiment.
Our peer review team found a number of fundamental flaws with your equipment, but they felt that there was no point in detailing them, since you might then point out that they were simply being put forward as excuses to drop your paper. The paper does not meet our strict standards, as it is obviously written to point out flaws in the current Climate Change hypothesis, which is settled science. And anyway, we have just discovered that there is no room to publish it in the next, or any future issues….
In other words, it is “flawed” and ‘thoroughly debunked.”
You are keeping me laughing!
b. cary’s was good, too.
You are on a roll, this morning Dodgy!
CO2 content on water is determined by Henry’s law and therefore the water temperature. What was the temperature of water?
That was my first thought.
” Each sample was stored in a sealed 500 ml plastic bottle in the “laboratory” overnight to warm it to the laboratory air temperature.”
So not the natural sea temperature, but laboratory air temperature. Caveat emptor.
Right. My first thought as well. This simple experiment should be run at different temperatures from ~0.5 C to around 40 C.
MAK
Actually, temperature, CO2 partial pressure (actually, fugacity), and salinity effect CO2 solubility in sea water.
Now we’re talking some real phase equilibrium thermodynamics when we get to use the term “fugacity”. The fugacity of CO2 in air at ambient temperature and pressure is very close to its partial pressure since the mixture is very close to ideal. The fugacity of CO2 in the liquid phase is extremely hard to model, especially for a biologically active sample. I do like the experimental approach of the measurements outlined above. The ratio of water to air will affect the resulting equilibrium, but the ratio in the experiment is probably more conservative than reality. This is not just a function of the depth of the water column, but depends on mass transport. Most of the water in the ocean is not at equilibrium with the CO2 in the air above it.
In the ocean one would have to correct for surface warming, which would outgas the CO2,thus raising Ph while turnover of the water would revert the Ph to the existing level.
Also the experiment does not allow for buffering from rocks in seawater which would presumably resist Ph change.
Given these caveats the results point to an even less of a Ph change to neutral than found in this excellent empiricle experiment.
Speaking recently on this site
https://wattsupwiththat.com/2019/04/23/podcast-fired-for-telling-the-truth-about-climate-alarmism-guest-peter-ridd/
Dr Peter Ridd commented that it was unclear what Ph change due to extra dissolved CO2 would do to coral reefs.
One wonders whether the intrinsic metabolism of the coral symbiots would simply use the extra CO2 to build themselves, thus naturally stabilising the Ph for the corals themselves.The extra CO2 would also drive the reaction to build CaCo3.H2O…, building coral skeletons.
Hopefully if he or those who follow him are allowed, such experiments could be planned, run and replicated.
The buffering capacity of the oceans is obviously quite high. That’s where all the world’s limestone came from, which holds most of the CO2 in our earlier atmosphere .. a process that operates over geologic timescales, not the mere 10 minutes to reach equilibrium in this tabletop experiment.
There is no such thing as “ocean acidification”. There is “limestone making”.
We also saw here an article recently that examined the mechanism by which simbionts are driven out of host coral. The bottom line was: a lack of CO2.
Higher temperatures in the water around the coral drives up the absorption rate of CO2, which is taken out of the water, leaving the pH higher than before.
This is a basic mechanism of coral growth. Anything that absorbs CO2 has the by-product of raising the pH in the water. This explains why the pH in a large enough reef is higher than the surrounding ocean water – it is short of CO2.
As with all terrestrial plants that have enough water, the limiting factor on growth of plants in the ocean is the availability of CO2 (food).
Not just CO2, but the trace Iron necessary to facilitate production of chlorophyll (Chlorosis) and photosynthesis..
I have a pet theory (hypothesis?) that suggests that maybe some of the bleaching is due to a lack of dissolved iron in the tropical waters making the algal symbiots too weak to reproduce.. and thus the coral dies.
Would love to see some experimentation on the idea.
Though chlorophyll, itself, has magnesium in the heme ring, not iron. And, if I am not mistaken, intracellular calcium would poison it, as does oxygen when the carbon dioxide concentration gets too low.
It is even easier to show that the ocean acidification thing is a load of garbage via basic facts:-
There is 48 times more CO2 in the oceans than in the atmosphere.
Doubling the concentration of CO2 in the oceans lowers pH by .4 points
The oceans are around pH of 8- alkaline.
For this argument, I am not even going to use the the huge effect of buffering from rocks on the ocean floor, which are what make the oceans alkaline.
If all the extra CO2 added to the atmosphere by evil humans recycling fossil fuels/plant food dissolves in the oceans, the atmospheric concentration needs to increase 48 fold to double the CO2 concentration in the oceans. To lower pH by .4 to pH 7.6 (still basic) requires the equivalent atmospheric concentration of 408 x 48=19,584ppm !!IE .04% to 2%.
It will need to double again and still not hit acidity. Still no factoring the buffering capacity of rocks.
Anyone still believe the fairy tales?
Sea water contains a lot of living creatures, so I would be concerned with photosynthesis & possible decay.
I think it would be sensible to use a coarse filter on the sea water to avoid anything big getting in (but you may want an active population of smaller creatures).
If possible, would be to maintain the original temperature and exposure to sunlight – that avoids any effects purely related to changes in temperature on the creatures (like mass die off)
I would also do a “control” – by boiling the sea water and then letting it cool – if that were giving much the same results then the effect of living things is probably small (over a short time).
Earth’s oceans hold over 50x the CO2 of our atmosphere. A warming earth also means a warmer ocean. Warmer water can hold less CO2. Ergo: less carbonic acid and higher PH. The slight increase in atmospheric CO2 would balance this effect but I’m not convinced we should get ocean acidfication.
Nice experimental work! I’ve done plenty of tests like that in labs over the years. About the only thing different I’d’ve done is use a CO2 cylinder, regulator and a rotameter. But that would just be to make CO2 supply easier.
As to your data, seawater pH changes considerably more than that due to temperature. Willis Eschenbach had a nice graphic of the pH versus latitude and depth. Ranges from 7.2 to 8.1, which is nearly a 10 times change in [H3O+].
The other aspect is the activity of HCO3- ions. Calcification is dependent upon pH and upon availability of bicarbonate. So as you increase pCO2 you are reducing pH but also increasing [HCO3-] availability. And as corals annually have massive numbers of offspring the effects of natural selection would rapidly kick in to favour the progeny most able to cope with lower pH/higher [HCO3-].
Unfortunately that isn’t easy to test in a 3 year PhD project, so we don’t have any decent data on coral adaptability.
Here is Willis’s pH article:
A Neutral View of Oceanic pH (2015)
Full marks for effort but, as other posters have pointed out, there are so many other factors to take into account (not to mention the question of scale) that it is not actually very useful.
If anything, it shows the maximum pH change possible on 25 degree seawater with aCO2.
Susan,
Same thought with me. Sort of a neat first effort.
I wonder if they say how many thousands of dollars this cost.
What amount are they asking for in the next grant?
If I take a couple of Tums and then check my urine pH, think I would be able to publish?
No, they would probably piss on your results.
Publish, no. But you might be able to sell it the democrats as a salacious dossier, if you can allege Trump collusion.
Perhaps you should inform David Attenborough and the BBC, who clearly understand nothing about acid-base equilibria. https://www.youtube.com/watch?v=Lh1qdpnSrBs
Link Not available
Try this: https://www.youtube.com/watch?v=7t6fRN9gGlA
Great! Now all the baking soda is going to be restricted by stupid laws.
You’ll have to sign a Statement of Intended Usage and swear on a copy of Michael Mann’s hokey hockey stick book that you will only use it for making biscuits or cookies or to counteract your hyperactive stomach acid levels, which are what are really responsible for ocean acidification. And you’ll have to have the recipes in your hands when you show up at the checkout counter to pay for it.
Stock up on baking soda now, people. We’re doomed!
Too soon for conclusions maybe, but it deserves more attention. It would be interesting to scale up the experiment, perhaps to bathtub size, and verify the results. I wonder what supports the figures from the scientific literature.
And, add fish. In fact, scale up to a fish tank.
Make sure to lime the bathtub water for strict comparability.
A study of seawater pH near active volcanic CO2 vents in the Mediterranean (Kerrison et al., 2011) found that the pH immediately adjacent to the vent was still alkaline, despite being subjected to the equivalent of nearly 5,600 ppm CO2.
Partial pressure and fugacity (μatm) are a little lower than what the mixing ratio (ppm) would be, depending on temperature and humidity. However, they are close. A partial pressure (pCO2) of 350 μatm generally equates to about 350 ppm in the atmosphere. At nearly 5,600 ppm CO2 the seawater was still alkaline, not acidic.
David
Konrad Krauskopf remarks in his textbook on geochemistry that it is very unusual for sea water to even get to a pH of 7. The few cases observed were unmixed, anoxic zones rich in hydrogen sulfide. I think that I have read that the water immediately emanating from Black Smokers is acidic, but quickly become neutralized by mixing with the surrounding alkaline water. Yet, neither the temperature or acidity are barriers to abundant life around the Black Smokers.
“At nearly 5,600 ppm CO2 the seawater was still alkaline, not acidic.”
pH 7, the neutral point of pure water, is irrelevant in a buffered solution. As I often point out, if your blood reaches pH7, you will be dead (from acidosis). The question is always what will be the consequences of a shift in buffer equilibrium.
In sea water, the consequence is a conversion of the alkaline species carbonate to bicarbonate. This increases the tendency of calcium carbonate (shells etc) to dissolve – simple equilibrium chemistry. pH 7 has no role here. The main reduction in carbonate concentration takes place at much higher pH levels.
Stokes
If you are really interested in the “… consequences of a shift in buffer equilibrium,” then you should acknowledge that living calcifiers usually (if not universally) have chitin and mucous covering the surface of the shells and the reaction you are referring to takes place preferentially with the shells of dead organisms, which have lost their protection. Calcifiers are able to repair damage so the primary ‘cost’ is the expenditure of more energy to live in an environment where carbonate can dissolve.
Examination of a typical Bjerrum Plot shows that calcifiers tend to live most happily at a pH that corresponds to a minimum concentration of carbonate and near the plateau of bicarbonate.
https://chemistry.stackexchange.com/questions/15939/is-the-ocean-acidification-inconsistent-with-the-bjerrum-plot
It is interesting that sea creatures learned to adapt to pH changes by the use of metabolic pathways as they evolved, thrived and multiplied.
If they had not, we would not exist.
Our own blood pH of 7.4 mimics that of seawater, as does our reliance on sodium and chloride ions in our makeup.
This points to our origins in an alkaline salty environment
Lewis P Buckingham
Calcifiers have different ranges of optimal pH. I suspect that the particular range is related to what the pH was when the specie first evolved.
Nick,
Your point is valid… but I was just pointing out 1) that the process is essentially instantaneous, 2) it’s a diminishing returns function and 3) it’s pretty well insignificant below about 1,200 ppm… and even then, it’s not a “crisis”.
Nick….what are the cliffs of Dover made of?…..when did those sediments form?…and what were CO2 levels then?
“This increases the tendency of calcium carbonate (shells etc) to dissolve…”
Yes, but it also increases the tendency of limestone rock to dissolve as well. The net effect is by adding CO2 to such a system is you actually INCREASE both the bicarbonate AND carbonate in solution. Simple experiment…. add finely powdered CaCO3 (limestone) to a flask of water. Bubble in CO2. Pretty much the only thing that happens it that the pH stays relatively constant until all of the CaCO3 has been dissolved with the net effect of increasing both HCO3- and CO2- concentration.
Nick Stokes…..As a regular visitor to WUWT, I am well aware of your position regarding CAGW but I consider your comments and input to be fairly reasonable, albeit biased. However, whenever you comment on “ocean acidification” it strikes me that you really don’t understand much about chemistry, particularly as it relates to biological systems & buffers. Furthermore, your repeated allegation that alkaline carbonate is being transformed into less alkaline bicarbonate is ridiculous. The concentration of carbonate ions in seawater is negligible because it is mostly present in the form of solid and relatively insoluble calcium and/or magnesium carbonates. These normally insoluble carbonates are in equilibrium with the bicarbonates and calcium/magnesium/hydrogen ions in solution. Any increase in H+ ions from CO2 dissolution will also generate bicarbonate ions. Any dissolution of solid carbonate will release the corresponding counter-ions (Ca2+/Mg2+) and the ultimate effect on the system as a whole will be controlled by the equilibria between the various ionic species and the solubility products of the respective solid phases (aragonite/calcite/magnesite, etc). It should also be noted that any slight fall in pH is accompanied by a decrease in CO2 solubility in the aqueous phase since there is an equilibrium between the gaseous phase and the bicarbonate/H+ pair in solution, too.
Bicarbonate ions are the principle buffering species of the (CO2/HCO3-/CO3- -) system in the natural world and this amazing buffering system is astonishingly good at maintaining pH in biological systems, including our own bodies, incidentally. All of this is perfectly explained by the Henderson-Hasselbalch equation which explains exactly how buffers work and why your arguments on this particular topic make no sense whatsoever.
In general, a buffer is capable of holding a system stable around the pK value of the buffering species despite a 10-fold increase in any of the species seeking to disequilibrate that system.
Phil,
“The concentration of carbonate ions in seawater is negligible because it is mostly present in the form of solid and relatively insoluble calcium and/or magnesium carbonates.”
That just isn’t true. Here for example is the Bjerrum plot of the equilibria, showing carbonate at about 10% of bicarb at normal sea-water conditions. It is usually quoted as 11%. This equilibrium must be obeyed in addition to the solubility equilibrium for CaCO₃. That is actually my point. We sit at the acid end of the HCO₃⁻/CO₃⁻⁻ buffer system, so each added CO₂ dissolves a molecule of CaCO₃. It’s true that that stabilizes the pH, but that isn’t the effect of interest.
“In general, a buffer is capable of holding a system stable around the pK value of the buffering species”
The system isn’t held stable. It is the pH that is stabilized. The other reagents are consumed while that is happening, and that is what matters here.
I have an active calculator here which solves the equilibrium equations to show how that works. There is more detailed explanation and data here.
Hi Nick!
Many thanks for taking the trouble to respond….much appreciated! I’m glad we agree that, despite much media hysteria & ridiculous alarmist comments, there is no risk of ocean acidification due to any credible future increases in atmospheric CO2. I understand the chemistry involved so none of this is new to me. However, I find your comments above (and your links) to be disingenuous to say the least.
There is an essentially infinite supply of solid calcium carbonate in contact with seawater and the presence of Ca2+/Mg2+ at significant concentrations (~450ppm & ~1250ppm, respectively) have a significant bearing on the various equilibria due to the low solubility products of the mineral species involved.
So, while there may be some limited dissolution of CaCO3, there will be no real “ocean acidification”. I’m sure you are also aware that marine life manufactures protective shells from soluble bicarbonate rather than the much less available carbonate ion at physiological pH.
Only true for arterial blood, Nick. Venous blood from exercising muscles easily reach 6.9 pH, resulting in cramps, especially when other factors are considered.
I am no underwater volcanologist, but I am pretty sure that the vent waters coming out of undersea vents has a mixture of chemicals other than CO2. If there is sulfur present, then it would greatly affect any pH measurements (assuming heat and come basic chemistry is lurking about).
So your point that 5,600 ppm CO2 did not create acidic water is made even more poignant – there was likely H(+)SO3(-) and H2(++)SO4(–) in the immediate area.
The natural ability of the oceans to buffer against a wide scale change in pH is huge.
Yes, the ability of seawater to act as a buffer is from ionic species already present. https://pubs.acs.org/doi/abs/10.1021/ac50076a029?journalCode=iecac0
What a great experiment! There’s no agenda that can be assumed, as this little experiment is what it is. A few thought to make it even more revealing.
– noticed your control was 450ppm. That probably due to out gassing due to temp increase. Control your temps, in fact …. see what happens as temp rises, or as temp cools.
– run you a control with nitrogen oxygen mix. … see if pH goes up in an atmosphere with no CO2
– run a series with added buffer, to see what happens if the biological system responds.
There’s all kinds of questions you could ask and somewhat answer.
so many of the data bases- like this one below on direst atmospheric measurements of CO2 are no longer available via the schripps institute, as they once were.
https://cdiac.ess-dive.lbl.gov/ftp/trends/co2/maunaloa.co2 –
this one (above) only goes up to 2008 – however – notice that the values change seasonally and what causes these changes is due to many contributing factors
temperature as already mentioned in previous posts for example- especially the inherent temperature of the ocean – and
no scientist living or dead knows all the factors that contribute to oceanic temperature changes.
photosynthesis amounts and rate – quasi periodic and very large oceanic circulation changes – etc, etc.
climate modellers attempt to put in as many variables as they can – they’ve been at it for decades now
the empirical data is clearly showing they’re not working very well – arguably because they don’t have much of a clue how all the variables interact – the dynamics of climate at this stage in the history of science cannot be adequately modelled
interesting – Dr of what – not to be rude – is my question?
the last time I looked
I enjoyed the article, and the comments. I have a slightly unrelated question: What is the chemical composition of hogwash? Have any scientific tests been run on it? With all the hogwash being emitted by certain politicians and candidates, shouldn’t we know its chemical makeup and how to neutralize any inimical effects? Just a thought . . .
my experiment relies on the theory that rising sea levels are partially the result of fishing quotas.
If the number of fish and whales taken from the sea is reduced, their numbers will grow. These animals live in the water and therefore displace their body volume in sea water. This naturally raises the sea level.
Obviously some animals, like whales , sometimes float around a bit, or even jump right out of the water so there is a modelling requirement, but the basic principle is sound.
My experiment involves filling a bath to the brim then inserting a space hopper, or other large fish-like simulation. According to the hypothesis, the water will overflow. If it does, I will write a paper with Michael mann and I will become as famous as he is. (but wetter)
I can only say Eureka!
obviously you haven’t learnt the “science” of “data adjustment” and of course, as with CO2 driving Globull Warming, CO2 itself has weak direct affect, so just add amplification factors, perhaps 6X, then show how that puts the World oceans on the “tipping point” that any additional CO2 will cause run away acidification. Data , schemata!
Hello,
Regarding this: “insufficient time to achieve pH equilibrium, which is found to take around 10 hours“. I do not see in your graphic anything reaching equilibrium. Where does that claim come from?
“A 2-litre “airtight” food storage jar contains seawater and air,”
How do you inject the CO2 if the jar is airtight?
“How do you inject the CO2 if the jar is airtight?”
A needle through the diaphram..
One-third marks, but in the correct direction for debunking “acidification”. See 2013 WUWT:
http://wattsupwiththat.com/2013/09/25/ipcc-on-acid-if-they-are-virtually-…
This experiment covers only the bicarbonate buffering system. Commenters added the carbonate (sea floor) buffering system. There is also a third borate buffering system that accounts for another third. See my comment in the 2013 WUWT article:
Neil Jordan September 25, 2013 at 2:00 pm
Re Sabertooth says: September 25, 2013 at 11:43 am
The pH ceiling of 8.3 is explained in Emerson & Hedges Chemical Oceanography, which also explains a pH floor of 7.6, also alkaline:
http://courses.washington.edu/pcc588/readings/EH_IV_CarbSys.pdf
This reference also includes borate buffering in addition to the carbonate and bicarbonate buffering that are customarily used to describe seawater
buffering.
According to Frankignoulle (1994):
http://www.co2.ulg.ac.be/pub/frankignoulle_1994.pdf
borate buffering accounts for 30% of the global buffering effect in seawater.
“This experiment covers only the bicarbonate buffering system.”
Really? Why so? Borate was not removed from the sea water, and its buffering remains in effect.