From MIT News:
Researchers combine the warm look of traditional light bulbs with 21st-century energy efficiency
Traditional light bulbs, thought to be well on their way to oblivion, may receive a reprieve thanks to a technological breakthrough.

Incandescent lighting and its warm, familiar glow is well over a century old yet survives virtually unchanged in homes around the world. That is changing fast, however, as regulations aimed at improving energy efficiency are phasing out the old bulbs in favor of more efficient compact fluorescent bulbs (CFLs) and newer light-emitting diode bulbs (LEDs).
Incandescent bulbs, commercially developed by Thomas Edison (and still used by cartoonists as the symbol of inventive insight), work by heating a thin tungsten wire to temperatures of around 2,700 degrees Celsius. That hot wire emits what is known as black body radiation, a very broad spectrum of light that provides a warm look and a faithful rendering of all colors in a scene.
But these bulbs have always suffered from one major problem: More than 95 percent of the energy that goes into them is wasted, most of it as heat. That’s why country after country has banned or is phasing out the inefficient technology. Now, researchers at MIT and Purdue University may have found a way to change all that.
The new findings are reported in the journal Nature Nanotechnology by three MIT professors — Marin Soljačić, professor of physics; John Joannopoulos, the Francis Wright Davis Professor of physics; and Gang Chen, the Carl Richard Soderberg Professor in Power Engineering — as well as MIT principal research scientist Ivan Celanovic, postdoc Ognjen Ilic, and Purdue physics professor (and MIT alumnus) Peter Bermel PhD ’07.
Light recycling
The key is to create a two-stage process, the researchers report. The first stage involves a conventional heated metal filament, with all its attendant losses. But instead of allowing the waste heat to dissipate in the form of infrared radiation, secondary structures surrounding the filament capture this radiation and reflect it back to the filament to be re-absorbed and re-emitted as visible light. These structures, a form of photonic crystal, are made of Earth-abundant elements and can be made using conventional material-deposition technology.
That second step makes a dramatic difference in how efficiently the system converts electricity into light. One quantity that characterizes a lighting source is the so-called luminous efficiency, which takes into account the response of the human eye. Whereas the luminous efficiency of conventional incandescent lights is between 2 and 3 percent, that of fluorescents (including CFLs) is between 7 and 15 percent, and that of most compact LEDs between 5 and 15 percent, the new two-stage incandescents could reach efficiencies as high as 40 percent, the team says.
The first proof-of-concept units made by the team do not yet reach that level, achieving about 6.6 percent efficiency. But even that preliminary result matches the efficiency of some of today’s CFLs and LEDs, they point out. And it is already a threefold improvement over the efficiency of today’s incandescents.
The team refers to their approach as “light recycling,” says Ilic, since their material takes in the unwanted, useless wavelengths of energy and converts them into the visible light wavelengths that are desired. “It recycles the energy that would otherwise be wasted,” says Soljačić.
Bulbs and beyond
One key to their success was designing a photonic crystal that works for a very wide range of wavelengths and angles. The photonic crystal itself is made as a stack of thin layers, deposited on a substrate. “When you put together layers, with the right thicknesses and sequence,” Ilic explains, you can get very efficient tuning of how the material interacts with light. In their system, the desired visible wavelengths pass right through the material and on out of the bulb, but the infrared wavelengths get reflected as if from a mirror. They then travel back to the filament, adding more heat that then gets converted to more light. Since only the visible ever gets out, the heat just keeps bouncing back in toward the filament until it finally ends up as visible light.
“The results are quite impressive, demonstrating luminosity and power efficiencies that rival those of conventional sources including fluorescent and LED bulbs,” says Alejandro Rodriguez, assistant professor of electrical engineering at Princeton University, who was not involved in this work. The findings, he says, “provide further evidence that application of novel photonic designs to old problems can lead to potentially new devices. I believe that this work will reinvigorate and set the stage for further studies of incandescence emitters, paving the way for the future design of commercially scalable structures.”
The technology involved has potential for many other applications besides light bulbs, Soljačić says. The same approach could “have dramatic implications” for the performance of energy-conversion schemes such as thermo-photovoltaics. In a thermo-photovoltaic device, heat from an external source (chemical, solar, etc.) makes a material glow, causing it to emit light that is converted into electricity by a photovoltaic absorber.
“LEDs are great things, and people should be buying them,” Soljačić says. “But understanding these basic properties” about the way light, heat, and matter interact and how the light’s energy can be more efficiently harnessed “is very important to a wide variety of things.”
He adds that “the ability to control thermal emissions is very important. That’s the real contribution of this work.” As for exactly which other practical applications are most likely to make use of this basic new technology, he says, “it’s too early to say.”
The work was supported by the Army Research Office through the MIT Institute for Soldier Nanotechnologies, and the S3TEC Energy Frontier Research Center funded by the U.S. Department of Energy.
Completely interesting!
Yep.
Not surprised at all, I drill holes in my light bulbs, fill them with the 2.2 grams of the mighty CO2 achieving concentration up to 0.9M ppm (this amount is available in one fresh can of Coke, well shaken but not stirred ); result is huge increase in efficiency from the CO2 radiative warming of the filament. Unfortunately the remaining flat coke taste lousy. Unless you believe in the AGW theory do not try this at home. (is /sarc required here?)
Vuk,
Just a minute. Along your line of thinking,,
Would it not be possible to drill a hole in the bulb and replace the argon with a gas that absorbs the heat and remits it as light? No need for a structure! Or better… dispense with the heat-making filament altogether and generate a voltage potential to ONLY directly from windmill electricity make the gas emit light?
I think I will call this invention fluorescence. Yes! Fluorescent light bulbs. I think we could raise a lot of seed capital on kick starter. (is /sarc required here also?)
So they want to cover the inside of the bulb with a material that is transparent to visible light, but reflects infra-red. I read about bulbs like this back in the 70’s.
Yep, used today especially in halogen lamps
MarkW
Top part of my light bulbs I cover with large number of photovoltaic diodes followed by layer of thermocouples all connected in series and fed as a DC voltage bias, AC goes in via capacitor of course. This method doubles the efficiency of my 75% efficient CO2 filed bulbs to nearly 150% ! !
There is drawback to this, since manufactures limit number of photons available in the filament, the bulb life expectancy is reduced.
p.s. manufactures are crafty lot and they don’t print number of photons available on the packaging, or on the bulb, it is worth asking shop assistant or writing to manufacturer for this essential info. /cras
“they don’t print number of photons available”
I thought that was what lumens are?
The Lumens is to distinguish it from the Luwomens, so people don’t go in the wrong door.
g
There was a restaurant named Bart’s Wharf East of Portland Or, at the Columbia River.
They labeled the doors “Inboards” and “Outboards”.
You mean “lupersons,” don’t you?
One big advantage of LEDs over both CFLs and incandescents is ther enormous lifespan – measured in decades. No mention here about the lifespan of these new bulbs.
Yup. A big problem. Tungsten evaporates. But the potential here in general (tuned wavelength nanomirrors) is still huge. Lots of possibilities if this can be commercially extended. A big nanotech issue is that this sort of stuff ‘doable’ in the lab at submillimeter scale is VERY hard at meter scale.
ristvan-
This is an old idea. GE commercialized a bulb in the 1990’s based on exactly this idea of recycling infrared back to the filament using a multilayer dielectric mirror on a small fused silica bulb surrounding the filament. I think they were able to demonstrate at least a factor of two improvement in lumens/Watt over a good incandescent bulb.
It is a daunting engineering challenge to meet all of the specifications (extreme temperatures, high transmittance in the visible, high reflectance in the near to mid IR) at a manufacturing cost of a few pennies per bulb. The MIT work has a long road to a commercially viable product.
Yes, and just think; 6.6% could easily be 40%.
Well what we do know for sure is that 6.6% IS 6.6%, not 40%
But it does sound like an interesting contraption.
And also LEDs are NOT between 5 and 15% efficiency. So these guys are lying in their teeth when they put that as a comparison figure.
White LED “light bulbs” achieve 100 lumens per watt today, and that number in research reports is rapidly approaching 200 lumens per watt.
And just for reference, the highest possible visibly white light source that is theoretically possible; no matter what, is 400 Lumens per watt. And just for kicks that theoretically possible 400 lumens per watt light source, would NOT be an acceptable illumination source for general lighting. The best you could do with it is make a bright flash light.
The reason is that the highest lumens per watt theoretically possible is made from a monochromatic yellow at around 585 nm and its blue complement which is about 472 nm.
You have a blue LINE source and a yellow LINE source, and NO OTHER COLORS. It has a CRI of close to zero.
Industry standards want a minimum of 80 CRI.
The internal quantum efficiency of the best LED chips is so close to 100% it is scary.
When you have a device that is consuming one watt of electricity, and putting out less than half a watt of heat, you wonder where the rest went. It went out to light up the rest of the universe.
But I’m always interested in new gizmos. So when can I buy one of these newcandescents from orchard Supply Hardware ??
G
this silica envelop sounds like the incandescent bulbs which are still available in europe.
This is worthy of Doug C’s ramblings. NO, a bit wire does NOT convert IR into visible light. Like the GHE, what happens here is that the extra layers simply slow down the cooling of the filament. This reduces the electrical power needed to maintain it at the desired emitting temperature, leading to a better overall efficiency in light output.
From the Wayback machine I recall the standard 500 watt light bulb that was used in virtually every 35 mm slide projector on the market. (I still have one that works).
And everybody also knows that those bulbs burn out frequently. It always happens when you are trying to have a slide show, and that is the least acceptable time to have that happen.
For starters it’s a royal pain trying to replace that bulb; the damn thing is red hot, so you have to wait for it to cool down, until you can pull it out without leaving your finger skin on the envelope.
Well then you have to plug in the new spare one: what do you mean you didn’t buy a spare !
Now while changing the light bulb you will notice that there is a nearly hemispherical mirror behind the light bulb, located so the center of the filaments of the light bulb lies right at the center of the mirror. Obviously they already knew about returning the heat to raise the Temperature of the filament.
Sorry ! bad assumption. It’s a dichroic mirror that reflects only visible light, and is highly transparent to anything beyond 700 nm wavelength.
You see, filament bulbs emit light in all directions; while 35 mm slides are flat and can only fit between the lamp and the projector lens, which only throws the picture in one direction. so without that mirror, more than half of the light emitted will never go through the slide, and on to the picture screen.
So the spherical mirror is there to return the backwards light to the forwards direction so it can help illuminate the slide.
So why not return the IR as well and heat the filament some more.
Did I forget to say up there that these bulbs burn out frequently ?? no they don’t need any IR assistance to burn out; they can do that by themselves.
And if you read the owners manual for your projector, you will see that it tells you that you need to adjust the reflector mirror.
So you turn the thing on without a slide, and you adjust the projector focus on the screen, until voilla, you get a sharp image of the light bulb filaments on the screen; except now there are twice as many filament strings, and they aren’t evenly spaced, and every other one is still out of focus.
So you have to loosen the clamp nut on the mirror mount, and then move the mirror axially until all of the filaments are sharply focused, and also laterally until the two sets of filaments are evenly spaced.
so you have to go out of your way, to make sure that the radiation returning from the mirror, does NOT get recaptured by the filaments, and forms a set of bass ackwards filaments to supplement the real ones, and create a more uniformly spread light source.
So now you can put in a slide, and refocus the picture on the screen; well after you tightened the mirror mount clamp nut of course.
And then you pray that you can get through your slide show, before the bulb burns out again. Failure to properly align the lamp reflector mirror, will ensure that you do not get through your slide show.
G
I once worked with a slide projector that had two bulbs. I never took one apart so I don’t know how it worked. But the idea was if one bulb burned out, you would turn off the projector, pull the knob until it locked in the new position, which would somehow position the new bulb in it’s proper place, then you could fire up and continue.
Yes, the LEDs last a long time. But the relatively complex assembly doesn’t. Voltage converters, solder joints, etc are still weak points. It’s ludicrous that a $10 tail lamp is now a $900 replacement. Unfortunately the problem will fall to the 3rd or 4th owner, who is most unlikely to be able to replace it. The $10 old school bulb may not last as long, but the Average person can afford to fix it.
um… leds are uber simple to use.
if you pay 900$ for it, it’s a tax for not knowing simple stuff
no sympathy here.
Where do you get these cost figures from? An old-school tail or brake or turn signal “bulb” of an industry-standard type (such as 1156, 1157, 3156, 3157) at an auto parts store is nothing like $10, more like $3. A failed solder joint is usually not a $900 problem. And electronics failures are generally repairable for a lot less than $900, unless the product is designed to be unrepairable by usual skills of an electronics repair technician.
The trouble is that designers are seduced by the “50,000 hour lifespan” so the light source changes from a replaceable item to an integral part of the whole assembly and $900 is not exceptional for some of those.
Of course it’s just that. The cheap, generic bulbs are now replaced with very model-specific items that are non repairable. You are tied the manufacturer for an over priced replacement.
Built-in obsolescence.
The 15-year old car with a failed LED tail-lamp will sprout an aftermarket lamp purchased at Halfords/Ebay/JcWhitney and bolted to the boot-lid or bumper.
I had a car that suffered from a failed grounding solder-joint on the local-oscillator for the “console area bus”. The dealer solution was to replace the whole gauge-cluster with new, at a cost of $900. Because the gauge-cluster contained the odometer memory, security cutouts make use of a secondhand-cluster require re-programming by an official dealer, which cost suspiciously close to $900. I found an electronics repair firm that would do the repair for $100 and warrantee the repair, but that required 15-days without the cluster, without which the car wouldn’t start. That $100 plus two-weeks of car rental would only be $775 – woohoo!. I ended up soldering it myself, but I don’t begrudge anyone choosing to go with the manufacturer’s suggested and supported repair procedure on something as expensive as a car.
Well so far as I know, the first red LED “tail light” was actually the first center high mount stop light; there never having been an incandescent center high mount stop light. It would be truly expensive to have to replace the filament bulb in a CHMSL (That’s what they are called). Seven square inches of illuminated surface, with a beam pattern specified by the SAE. Hewlett Packard made the first efficient basic LED for that lamp, and from that they evolved the first power LED device that could go into an actual red automobile tail light, including brake lights and rear lights.
The basic lamp dates from circa 1983, and the tail light first appeared in the Ford Thunderbird in I think 1996.
The only reason I know about those, is because I designed them, and my name is on the patents for them (now expired).
I told HP in 1983 that they couldn’t have seven square inches of uniform surface with the SAE radiation pattern from a single LED, that would be visible in sunlight. Well I might have told them that as early as 1980; but they didn’t believe me, so they tried to do it anyway, which is why they didn’t get one in 1980. I told them the best they could get was a group of bright spots, but most of the seven square inches would be dark.
I really enjoy pulling up behind modern all LED tail lighted cars, with their various arrays of bright spots in a mostly dark area. Yes the spots move around when you move your eyes, but it mostly stays dark.
There’s a couple of rules called the Optical Sine theorem, and the second Law of Thermo-dynamics that explains why it’s mostly dark.
A chap by the name of Rudolph Clausius derived the Optical Sine Theorem from the second law.
G
I have yet to come across an LED bulb that lasts anywhere near its rated lifetime.
They are rated at 30000 hours which means at 10 hours a day use they should last for over 8 years.
At the present moment I get about 6 months use before they fail. The last batch of ten I bought in February has already had 7 failures.
Fortunately the supplier has to replace any that fail within the first year which means that, with replacements, I’ll get about 18 months from something that should last over 8 years.
NOTE: I’ve bought many different LEDs from many different manufacturers ranging from cheap Chinese imports to the likes of OSRAM and Philips. They all fail well before their rated lifetime.
That’s wierd, I’ve never had any fail ever. 13 installed for up to 10 years replacing flourescents as they go too slow/dim.
Ditto – I’ve some LEDs approaching 4 years old, and have now replaced nearly all incandescents and most CFLs with LEDs – never had a failure.
Me 2. No failures yet after 4 years. Look at your electric supply.
All the failures I have had with LEDs have been with relatively cheap (probably Chinese) ones, or ones fitted with insufficient ventilation to keep the electronics cool. It is very difficult to buy attractive lighting units that have enough ventilation for LEDs. Philips LED bulbs that I have have yet to fail. With the cheaper ones the LEDs within a ‘bulb’ fail a few at a time, rather than the electronics failing. Our neighbour has seperate drive units for LED lighting, and these regularly fail rather than the LEDs themselves, presumably due to being installed with lack of ventilation
Most of the lights in my home are LED and have been for several years, all CREE or Philips brand. I have had 3 failures and 2 of those were used in fixtures that did not have very good (or any) ventilation. I have not dissected them yet, but when I do I expect to find that the voltage conversion modules have failed, not the LED emitters. This has been my experience with CFLs in the past.
At this point, if used in situations with proper airflow, I expect good name-brand LED A/C light bulbs to last about 10 years on average, and not the 25 or whatever ridiculous figure is always quoted. Even so, at the current bulb and electricity prices, they still make economic sense. When you consider the convenience factor of not having to replace them so often (especially in hard to reach places) they make even more sense.
I have to dis-agree. I installed an LED light bulb at my work years ago in a red light at a intersection inside the building. This light is on 24/7. The bulb cost $140 at the time and my boss had a conniption when he saw the bill. But I pointed out to him that the incandescent bulbs would not last a week in the fixture due to the heat and I changed this bulb at least once a week, if not more often, but because of it’s placement it took two people to change it safely and the charge out rate for my services was $85 per hour so it cost $170 plus the cost of the bulb to change it. This meant that if the led bulb lasted 2 weeks it paid for itself. My boss had to grudgingly agree. That bulb has lasted more than nine years. I have run in to a few LED fixtures that have failed but no more than the traditional fixtures we used to purchase.
If you are having problems with premature LED failures, there are two things to look at. One, already covered by other commenters, is inadequate ventilation to keep the LEDs cool. The other has to do with the quality of the power coming into your home. If there problems with voltage sags and surges,, or worse, high voltage transients (voltage spikes), in other words ‘dirty’ power, then it’s no wonder your LED lamps have been failing at a high rate. The driver circuitry in LED bulbs really don’t like those operating conditions, particularly transients. You might want to monitor the power quality for a while to see just how good or bad it might be.
I’ve been running almost 100% LED at my house for better than 5 years. I’ve never had a bulb go bad.
@TerryS
“NOTE: I’ve bought many different LEDs from many different manufacturers ranging from cheap Chinese imports to the likes of OSRAM and Philips. They all fail well before their rated lifetime.”
I have 27 LED bulbs (Phillips) in my restaurant ceiling. I have AC going…24 hours a day. The bulbs are on 24 hours. Most (all but two) have been in for 3 years (24,000 hours). One that didn’t make it was a Phillips and the other was ordered online. It was a Chinese product that was half the cost. It lasted two weeks.
True.
Also, LED bulbs are now very cheap. I get 11W (60W equivalent) LED bulbs at DollarTree for $1 each. It’s hard to see how anything else can hope to compete with that.
To me, the LED bulbs’ light quality seems pretty good. Colors seem truer than from CFL bulbs, and the “60W equivalent” LED bulbs actually seem brighter, to me, than either 60W equivalent CFLs or 60W incandescent bulbs.
ESL bulbs also exist, but they’re $15/bulb, and only about 50-60% as efficient as LEDs, and they have only half the rated lifespan of LEDs. They’re definitely not taking over the world, either: they’ve been out for five years, and I’ve never actually seen one.
The $1 DollarTree LED bulbs (made in China by a Canadian company) are labeled 2700 K, but they seem whiter than incandescents and most CFLs, to me. I have no way of knowing how much they’ll dim with age, yet, but they sure start out nice and bright.
As for whether a $1 11-watt 60W-equiv LED bulb from Dollar Tree is the best value available: I would compare this to an 8.5-watt 60W-equiv LED bulb of the Sylvania brand available at Lowes for about $2.60 when purchased in 3-packs. The $~2.60 Sylvania has rated life expectancy of 10,000 working hours. As for paying $~1.60 more to squeeze down power consumption by 2.5 watts more than if using the Dollar Tree one: Assuming 12 cents per kWh, which is $.00012 per watt-hour, this means $.0003 saved for each hour of using the Sylvania one instead of the Dollar Tree one. Using the Sylvania one instead of the Dollar Tree one pays for itself in 5,333 hours, and at 10,000 hours you get your money back plus 87.5%. Assuming 3 working hours per day, this happens in 9.125 years. Multiplying your money by 1.875 in 9.125 years is an annual rate of return of 7.1%. Where you use these LED bulbs more than 3 hours per day, the annual rate of return will be higher. And this annual rate of return is not taxed.
The Sylvania bulbs are only rated for 10,000 hours? The DollarTree no-name (“globe-electric.com”) bulbs claim 25,000 hours (“22.8 years at 3hrs/day”).
That might not mean the DollarTree bulbs will actually last longer. It might just mean that Sylvania is more honest/realistic.
The DollarTree/Globe bulbs claim to be dimmable (but I haven’t tried that).
I’ve gotten another brand of bulbs at DollarTree, in the past, which claimed to be 9W for 60W equivalent, but they didn’t have those the last time I bought bulbs, and I don’t remember whether those were dimmable or not, and I don’t remember what lifespan they claimed, or what color temperature they claimed.
None of the $1 LED “60W equivalent” bulbs I’ve bought have failed, so far, not even the one that I dropped. The glass broke, but it still works just fine. I broke off the remaining glass shards with pliers, screwed it into a fixture, and I’m still using it.
It really frustrates me that 60W equivalents are the smallest you can find at most stores. That’s too much light for my tastes. Most of my fixtures have one or two positions without functioning bulbs in them. (I hang onto burned out bulbs so I can put them into these fixtures so that I don’t have naked sockets.)
I’d put 25W and 40W in most of my sockets if I could find those values.
The exceptional Centennial Bulb has been lit for more than 100 years, more than a million hours, with brief interruptions. CentennialBulb.org as I recall.
I notice my led you beauts…seem to be a LOT duller after 2yrs
and I have heard others say the same so its not just MY eyes noticing it.
they still beat the hell outta the mercury twirly useless horrors though 😉
Individual LEDs do indeed last a long time. They tend to outlast their supporting electronics in fact. Often by a lot. LED flashlights are wondrous devices. Far superior to their incandescent predecessors. I’ve bought dozens over the years. Why so many? Not because the LEDs fail. But because the circuitry holding them fails. Just checked the LED flashlight I keep next to the computer. It works pretty well. … After I bang it on the table. And one of the 9 LEDs in its array of LEDs doesn’t light. Typical in my experience. Don’t know if it’s the LED or something else. Don’t really care.
I will probably switch over slowly from CFLs to LEDs. Already have in the couple of outside sockets because CFLs work poorly when the temperature is well below freezing. But I’m not in any rush because my experience is that the purported superior lifetime of LED devices is largely illusory.
Well the fact that your LED flashlight has nine LEDs including one dark emitting one, tells me that your flashlight is a piece of cheap crap.
Anybody can take 9 T1 3/4 5mm LEDs and mount them on a circuit board. Why is it nine instead of seven (six around one) ??
Well you only need one LED designed to be a flashlight LED light source.
the T-1 3/4 LED is just about the lousiest LED optical design ever put out on the market.
How do I know that ?? Well because I designed one of the very first of those, and plenty more since. I believe that Hewlett Packard, and Litronix came out with those at about the same time. Theirs had a cylindrical barrel with a hemispherical cap, and ours was slightly conical with an aspheric cap.
The problem is that they try to make a narrow angle beam with them, so the LED die is too deep in the lens. Yes you do get a narrow high axial intensity beam, but you also get a whole lot of TIR loss, which first of all gives you a close to 180 degree wide forward halo, and then you get a very large fraction of the flux comes right out of the back of the package, onto the PC board you mount it on.
Today you can buy (from the best camera stores even) a large array of a hundred or more T-1/3/4 LEDs for a vide light or such usage. They too are total crap, and I cringe when I go into my camera store and find that is the only kind of LED camera light I can buy.
The popular MR-16 quartz halogen spot lamps are a good example of a failure to properly LED convert. The quartz envelope is about 50 mm diameter, and the whole bulb is supposed to be about 35 mm maximum length including the pins. The quartz envelope has a dichroic reflective surface, that is designed to reflect both visible and especially infra-red radiation.
The object of those lamps is to get so hot (hence the quartz) that they cool themselves (as much as 50 watts) by radiating IR profusely out of the front of that quartz envelope.
The mounting pins and the flimsy fittings they plug into have ZERO capability of dissipating heat, so radiative cooling is what you have.
So now along comes a 4-H club optical designer, who is going to replace all of those bulbs which burn out frequently with his reliable LEDs, it’s a drop in replacement slot built to order.
Er, Um ! did I say somewhere those MR-16 fittings have zero capability of exporting heat out the back of the fitting, conducted there by the flimsy pins on the MR-16. (A newer design has more substantial studs). And no they still cannot conduct heat out of the back.
But a 50 watt equivalent LED bulb will only use maybe 12 watts of juice. Spot lamps are less efficient than ordinary A-19 light bulbs.
And no, a drop in slot MR-16 fitting is not capable of conducting or radiating 12 watts of heat out the back.
So LED MR-16s are a 50 mm diameter HEAT SINK designed to radiate forward, like a quartz halogen, and they only have room for about a 25 mm window for visible EM radiation to escape into your living room. So they put about three 8-10 mm power LEDs of some not unreasonable optical design in there, so they have not more than about 10% of the light emitting surface area as a 50 mm QH bulb has.
So yes they have maybe 10 times the luminance (brightness) of the original, so they have far too much glare.
If you use LED MR-16 replacements for spot lighting, you have to use them in locations, where they can illuminate away from the audience, because you cannot look directly at an LED MR-16 bulb head on.
Soraa has about the only reasonable 50 watt MR-16 replacement, and that is because their GaN LEDs are considerably more efficient than everyone else, so they get a 50W equivalent, with less real power than anybody else can do.
The solution is to scrap the MR-16 (for new designs and come out with a LED savvy replacement fitting, that can remove heat from the back of the fitting, and also that has a full 50 mm emitting aperture at the front, instead of the fly’s eyes junk.
Does it occur to anyone that designing an LED lamp to go into a fitting that is designed so that you can unscrew it when it craps out, makes no sense at all, if your LED lamp (properly designed is designed to not crap out.
Eventually it will happen, but the technology to do it already exists and is well known; at least by a few people. But getting light bulbs approved is no easier than designing a new standard toilet seat.
G
In my experience, the thing that always fails in a typical cheap LED flashlight is the “clicky” tail switch. They don’t use gold contacts in $2 flashlights!
It is generally possible to open one of these flashights up, knock/press out the switch assembly, pry apart the switch, clean the contacts & coat ’em with Stabilant, and reassemble, to make it reliable again, for a while. It’s probably not worth the trouble, though, unless you like taking things apart.
The twist-head switch of Thru-Nite flashlights is a lot better. I use mine just about every day. It works well with a single rechargeable LSD NiMH AAA battery; I swap batteries every couple of weeks. I bought it 2½ years ago, for $15, and it has lived on my keychain ever since. It’s gotten dunked in the water a few times, without harm. It’s difficult hard to get it to go into low-brightness mode, but I didn’t use that much, anyhow. The bright mode is nice and bright, though it uses up the battery pretty fast.
There is the CLAIM that LEDs last for decades, just as there was the CLAIM that CFC curly bulbs would last for years longer than incandescent bulbs, and the CLAIM that halogen bulbs will last for 10 years. But the curly bulbs never last as long as promised, and I have had 2 halogens in the last 6 months that died after less than 2 months each. So I’ll wait and see about the LEDs.
I suspect your voltage may be above normal. High voltage, even just a little high, greatly reduces lifespan of light bulbs of any kind. Heat is the enemy of CFL (Compact Fluorescent Lamp, not CFC, Chlorinated Fluoro Carbon or something like that, FREON in other words).
Operating CFL’s and LED’s in sealed enclosures allows heat to build up. Check the labeling on your LED’s in particular, most aren’t rated for sealed enclosures, some are okay.
Any ceiling fixture that can use a 40 or 60 watt incandescent light bulb, should be perfectly safe for installing an A-19 LED replacement. The 60 W ones I buy use only 9.5 watts for 800 lumens and they do not overheat in an ordinary ceiling fixture.
Same as CFLs (I have zero of those) it is the cheap junk hong kong fooey electronics in them that fails. Crappy capacitors and resistors.
Typical LED “ballasts” for fluorescent tube replacements, are built by the same people who make computer switching power supplies, and they think they are doing great at 80% efficiency.
With a fixed load (non dimmed) it is possible to get 95% efficiency in an AC-LED power supply, and they aren’t necessarily more expensive. Just lazy circuit designers.
It’s the same as the old “Japanese transistor radio” problem. Eliminate as many components as you can until it doesn’t quite quit working.
Today the Japanese transistor radio can be a work of genius. I have a couple of totally wonderful ones, that escaped the historic mold.
Today it is more likely a different -ese problem.
G
Michael 2 wrote to Monna, “I suspect your voltage may be above normal…”
Voltmeters are very inexpensive, these days. I have one of these little autoranging multimeters, and I like it a lot. They can be had for less than US$13 on eBay or Amazon. It is very accurate, rugged & portable, and the batteries seem to last forever.
>>One big advantage of LEDs over both CFLs and
>>incandescents is ther enormous lifespan.
Not so much with the compact fluorescents. The last one I bought lasted one week, before it exploded (pop and a big burn hole in the base). The second one lasted two weeks. The third is still going, three months later.
R
I have found CFLs to have a fairly good track record, and I have used them a lot ever since they became easily available. I typically got 5,000 working hours out of them. And LED lightbulbs don’t have starting-related wear, while most CFLs do and have less lifetime when their average on-time is less than something like 3 hours per start.
Lots of my decades long lifespan LED’s go dark after a few months, especially the ones bought in Australia. It all depends on what Chinese factory they come from I guess.
Yes. Enormous *theoretical* lifetime. But thecChinese crap available in the stores isade with such.poor manufacturing standards they usually don’t last more than a year. Does anyone else’s mileage vary?
Can’t they just tint the shells of the LEDs a little yellow to mimic incandescent light?
Nope. Read up.
You are rather missing the point.
No Jivey, there’s a small family of gases that don’t require a high voltage, or a high energy flashover to start glowing, and the yellow gases, don’t happen to be them.
The kinds of lights, sought to be replaced in efficiency are the metal vapor lamps, and they’re referred to this way because you use some kind of energy source to vaporize first, a conductive metal; and once this stuff starts conducting, – think mercury dosed into lamps so they’ll start at low energy levels – it creates the environment where the other gas in the bulb can start to fluoresce. And these things get hot, too. They burn at many hundreds degrees’ temperature, and even being near them will ignite stuff.
And yet Jivey these metal vapor lamps in their various colors, are the world’s most efficient low technology, widespread light sources for basically everything outdoors.
Even indoors. They’re used wherever this large heat load can be justified because somehow or other you can dump it to the outside.
Do you just make this stuff up as you go. Mercury vapor is just about the only yellow emitting “gas”, and has been sued to keep down sky light pollution in areas where big telescopes try to live. They emit only two narrow lines in the yellow a few nanometers apart, so it is easy to filter them out of photographic equipment.
g
george e. smith says “Mercury vapor is just about the only yellow emitting gas”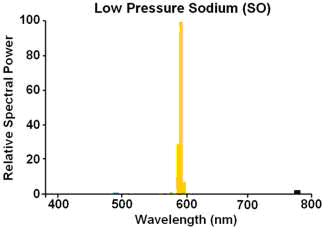
I vote for sodium vapor lamps as the most mellow yellow.
OOooPs !!
At Michael2. ??
Well I was just using the alternative spelling for Sodium, to see if you were awake.
Good catch there Michael2; you can be sure that won’t be the last fox pass I make.
The Sodium (preferred spelling) yellow pair is at 5890 and 5896 Angstrom units. usually they just say its 589.3 nm.
I’ll authorize you to check on that Michael just to be sure I didn’t screw up again.
G
What are these guys talking about. I just spent about an hour today, looking at the “white” 60 watt equivalent (800 lumen) LED bulbs, at Orchard Supply Hardware, and the biggest problem is that the vast majority of their LEDs were 2700 K , and those things DO look a sickly yellow color. They call them “warm white”. Evidently people like them in their homes. Even 3000 K LEDs are yellowish.
I settled for a couple of “60 watt” bulbs that cost be $6 for the pair, and they use 9.5 watts of juice each. Those were 5,000 K “daylight bulbs and are nice and white.
The 2700 K warm whites are less efficient, because of the red phosphor. The eye sees very few lumens per watt in the red. So warm (or soft white) suck in my book.
Yes my wife wants daylight over her bathroom mirror, not yellow jaundice color.
G
George,
Look for the high CRI CREE bulbs for your bathroom. They give you a much truer color than even the “daylight” bulbs which are too biased in the high end of the spectrum.
Most Americans and Europeans find 5000K light indoors at home-lighting levels as icy-cold, despite the color being about the same as that of noontime tropical direct sunlight. Incandescents generally achieve upper 2,000s K. As for less lumens from warmer color (lower color temperature): Just pay attention to the lumen ratings of the light bulbs. LED lightbulb manufacturers generally pay for higher efficiency grade LEDs in the warmer colors than in cooler colors because sales volume is higher with incandescent-like colors of 2700-3000 K, and LED manufacturers give price breaks for increasing purchase quantity.
For what it’s worth, I buy only daylight color illumination (5000k). Among other things I’m a photographer and I don’t like having the color temperature in my house vary room to room and from the computer monitor as well. With everything at 5000k, I can walk around without altering my eyes automatic color balance which would disrupt working on photographs.
Boss replaced multiple recessed CFLs with daylight LEDs in our lobby. It was unsettling… a bit ghastly. He swapped them out for “warm white” which was infinitely better. The 3000k bulbs might have been the best mix, but he didn’t ask. Either way, “daylight” only works in certain contexts in my opinion (or if you have blended light in a room with different color temperatures.)
Paul, CREE is one of the REAL people in the LED lighting business, who actually know what they are doing. They make their GaN LEDs on Silicon Carbide substrates, which is pretty whacked out material to deal with but they do it somehow. They are over in that research Triangle area, and their roots go way back in the LED technology history.
I would have no trouble recommending their products to anybody. Same as Phillips or Lumileds.
Soraa is very high tech, but so far not too many products out there. That is Shoji Nakamura’s doing. They are here in the Bay area.
I have only one sharp image functioning eyeball; the other one is MOMA ready. So I have to have a bright white light to be able to read. But if I could get a good 3500-4000 K LED, that would probably suit me. The brand I buy from Orchard Supply has an amazingly uniform smooth emitting surface. Too many LEDs are all patches of light. I see on the box the brand is TCP but they are made in China. Have NO idea who the heck they are just their bulb looks great to my eyes. Yes they claim 25000 hours and all that. They say suitable for damp areas, and totally enclosed housings. Look well made, but who knows.
G
You can buy LED’s with 2700K light which mimic incandescent. I’m looking at 2 right now.
Funny that you should mention lifespan. I have lived in this house for 30 years. All the incandescent light bulbs that were here when we moved in are still working. I don’t think you can say that about the curly light bulbs or the LED’s.
Eve, not credible. I worked in the lighting industry for over a year as a consultant starting in 1978. The design life spec world wide on ordinary tungsten incandescents was then/is now 1000 hours. Six sigma around that is 900-1100 hours. So either you live in the dark, or are only there for a fraction of a year, or… Now, halogens are ~4500 hours (the gas suppresses tungsten evaporation). I have those in my Chicago townhome. About the same as fluorescents, against which they were developed about four decades ago.
So please show some lifetime evidence rather than judt an assertion contradicted by all the global industry literature.
I have numerous bulbs in excess of 25 years in use in my house. Duty cycle is maybe 10%. Others have a <1 year life expectancy, most likely due to vibration. One in particular failed several times a year; I replaced it with a $30 LED lamp many years ago, still working.
I guess you would have to come to my house and see that the incandescent bulbs still light up. I am as surprised as you are. But that is what has happened. I will gladly tell you when I replace one of those bulbs, if I am still alive.
now they may be 240 volt bulbs in the US 110 voltage system. I would expect them to last decades under such conditions.
Agree with ristvan 100%.
It is not credible for incandescent bulbs to last thirty years, unless they are rarely used.
It does not matter where you live…as stated above, tungsten evaporates at a predicable rate at a given temperature.
Eve, I am reminded of the scene from the movie My Cousin Vinnie, when a guy was testifying that grits cooked faster in his house than they did for everyone else.
Besides, no matter how long they last, LEDs last many times longer, and use a fraction of the electricity and produce far less waste heat, both of which save money in the long run.
@Rud Istvan.
There is a documented case of a light bulb in a California fire station that was in continuous operation from 1901 to 2013, 113 years.
https://priceonomics.com/the-mysterious-case-of-the-113-year-old-light-bulb/
The “Centennial Light” at the fire station in Livermore CA is a 30-watt light bulb that produces as much light as a 4-watt 120V night light bulb. When a tungsten incandescent lightbulb is dimmed to the point of having energy efficiency so low, its life expectancy is likely to exceed a century.
No mystery.
The centennial light bulb runs at greatly reduced light output, about 4 watts.
The relationship between voltage, light output, power consumed, and lifetime of the filament is very well understood.
If you want to sacrifice efficiency for long life, you can do so, but you will spend a great deal more in electricity for a given amount of light output.
“Incandescent lamps are very sensitive to changes in the supply voltage. These characteristics are of great practical and economic importance.
For a supply voltage V near the rated voltage of the lamp:
Light output is approximately proportional to V 3.4
Power consumption is approximately proportional to V 1.6
Lifetime is approximately proportional to V −16
Color temperature is approximately proportional to V 0.42[114]
This means that a 5% reduction in operating voltage will more than double the life of the bulb, at the expense of reducing its light output by about 16%. This may be a very acceptable trade off for a light bulb that is in a difficult-to-access location (for example, traffic lights or fixtures hung from high ceilings). Long-life bulbs take advantage of this trade-off. Since the value of the electric power they consume is much more than the value of the lamp, general service lamps emphasize efficiency over long operating life. The objective is to minimize the cost of light, not the cost of lamps.[58] Early bulbs had a life of up to 2500 hours, but in 1924 a cartel agreed to limit life to 1000 hours.[115] When this was exposed in 1953, General Electric and other leading American manufacturers were banned from limiting the life.[116]
The relationships above are valid for only a few percent change of voltage around rated conditions, but they do indicate that a lamp operated at much lower than rated voltage could last for hundreds of times longer than at rated conditions, albeit with greatly reduced light output. The “Centennial Light” is a light bulb that is accepted by the Guinness Book of World Records as having been burning almost continuously at a fire station in Livermore, California, since 1901. However, the bulb emits the equivalent light of a four watt bulb. A similar story can be told of a 40-watt bulb in Texas that has been illuminated since 21 September 1908. It once resided in an opera house where notable celebrities stopped to take in its glow, and was moved to an area museum in 1977.[117]
In flood lamps used for photographic lighting, the tradeoff is made in the other direction. Compared to general-service bulbs, for the same power, these bulbs produce far more light, and (more importantly) light at a higher color temperature, at the expense of greatly reduced life (which may be as short as two hours for a type P1 lamp). The upper temperature limit for the filament is the melting point of the metal. Tungsten is the metal with the highest melting point, 3,695 K (6,191 °F). A 50-hour-life projection bulb, for instance, is designed to operate only 50 °C (122 °F) below that melting point. Such a lamp may achieve up to 22 lumens per watt, compared with 17.5 for a 750-hour general service lamp.[58]
Lamps designed for different voltages have different luminous efficacy. For example, a 100-watt, 120-volt lamp will produce about 17.1 lumens per watt. A lamp with the same rated lifetime but designed for 230 V would produce only around 12.8 lumens per watt, and a similar lamp designed for 30 volts (train lighting) would produce as much as 19.8 lumens per watt.[58] Lower voltage lamps have a thicker filament, for the same power rating. They can run hotter for the same lifetime before the filament evaporates.
The wires used to support the filament make it mechanically stronger, but remove heat, creating another tradeoff between efficiency and long life. Many general-service 120-volt lamps use no additional support wires, but lamps designed for “rough service” or “vibration service” may have as many as five. Low-voltage lamps have filaments made of heavier wire and do not require additional support wires.
Very low voltages are inefficient since the lead wires would conduct too much heat away from the filament, so the practical lower limit for incandescent lamps is 1.5 volts. Very long filaments for high voltages are fragile, and lamp bases become more difficult to insulate, so lamps for illumination are not made with rated voltages over 300 volts.[58] Some infrared heating elements are made for higher voltages, but these use tubular bulbs with widely separated terminals.”
https://en.wikipedia.org/wiki/Incandescent_light_bulb
“Menicholas October 25, 2016 at 8:16 pm
Agree with ristvan 100%.
It is not credible for incandescent bulbs to last thirty years, unless they are rarely used.”
That’s because they are made to fail. It’s called in-built fallibility and started with light bulbs in about 1922.
We used to own a house in France, and we brought some of the French incandescent 220v bulbs back to the US to install in our house here. They have the same base and work perfectly well, even though they are a bit dimmer. They will NEVER burn out, and are great for use in difficult-to-reach locations.
We used to have an apartment in a building where there was a hallway that had one 60w naked bulb. It was on 24/7 – there was no switch that we could discover. The bulb had to be replaced every 30 days (+-2 days). One of the other tenants claimed that it had to be replaced because “the wiring in the apt was bad”, but when you looked at the pacakaging, you found that it was rated for 750 hours of service. 30 days*24 hrs/day=720 hours. It operated for its design lifetime. After we left the apt CFL lights came out and they instaled on in this light. I have no idea how long it lasted.
Incandescent light bulbs will last a very long time if your voltage is low.
Yup. And give out very little light.
Exactly right again.
Lower voltage will extend life because the filament is not getting as hot, and so the filament will evaporate more slowly.
They will use less electricity than stated on the label, and produce less light.
Patrick , I seen a doco some years ago about an East German or west German company (whichever was the communist side) I’m sure it was Narva but could be wrong , tried to sell their incandescent globes which had a claimed life span over 30 years but the main players in the industry mainly from the USA managed to stop them from exporting the globes .
I think the doco was called ” planned obsolescence ” .
Does sigma T^4 ring any bells with anybody ??
G
“Robert from oz October 26, 2016 at 1:01 am”
And Germans, being Germans, it was all written down.
I used to buy incandescent bulbs rated for 130V, and on 110V they typically lasted 10-15 years.
Reduced voltage works. That is why one of the tips for long incandescent bulb life is to use a dimmer. Even at full blast there is a couple of volt drop across the dimmer.
Many years ago, I saw an ad for a product that was supposed to greatly increase the life span of a bulb.
It was a small disk that that was somehow screwed into the bottom of the light bulb socket (Sorry, too many years to remember the exact method of mounting.)
The disk contained a thermistor. It’s resistance was 1 or 2 ohms when cold, dropping to less than a tenth of an ohm when it reached operating temperature.
The claim was that it would reduce the inrush of current when the bulb first turned on.
I live in the same apartment since 1974. There are two incandescent light bulbs on the veranda since then. The same is true for the summer house which is from 1980 and there are incandescent lights still working. Lifetime is the length of time they are lit , not the time of construction or acquisition. Also that the number of times they are lit also plays a role, as they put it on the new lamps.
if bulb fitting is attached to a wall or ceiling, rather than hanging on its cable of a chain, the life time expectancy is increased.
I had a garage door opener went through bulbs every few months. I rigged up a 1 foot extension cord that I could screw into the opener’s light socket so that the bulb hung down below the opener. 3 years later when I sold that house, the bulb was still working.
I suggest you get your supply voltage checked.
That’s sounds like 20% below rated voltage to me.
Eve,
I’m pretty sure if you unscrew one of those bulbs and check on the base, it is rated for 130V service. I you replace it with a bulb rated for 120V you will notice the new bulb is brighter, but it will not last as long. That is the trade-off.
I have read about this three times over the last two years.
Well, they are demonstrating a doubling of efficiency, so the claims of a possible 40% efficiency don not really matter except as a possible upside on development. The other thing is cost, which is very much not mentioned.
It should be. The incandescent light bulbs still burning in my house may have cost .30 cents each?
Ignorance is bliss Eve.
Ever looked at the efficiency comparison for LEDs over incandescent?
Hey, if you send me a shipping label, I will sent you a big box full of energy wasting incandescent bulbs, all sizes and types…I have hundreds that I replaced when I moved into my new house 3 1/2 years ago.
The LED and fluorescent replacements have already paid for themselves.
Menicholas,
if you have hundred of lights in your home, you should really be concerned about efficiency, unless you are a millionary.
Everyone should be concerned about efficiency and cost savings over time, IMO.
I am not a millionary , and never will be if I waste money
Including kitchen appliances, there are fewer than 30 sockets for light bulbs in my apartment.
The only bulbs I haven’t replaced are those that are on only a few minutes a day.
Efficiency is a good thing, however if payback is a couple of decades, I’m not interested.
If the quality of light is acceptable to you Eve, then you should stick with what you have. But if you ever get a failure, you could take a look at LEDs then. I don’t think CFLs are either aesthetically pleasing, or very functional , and I have had plenty of them fail with crappy electronics.
G
This is much like the designs of many of the current LED designs.
In order to achieve the 2700K color temperatures, many use a two-stage system of an UV LED that excites a secondary chunk of phosphorus, properly mixed to generate the desired color temperature.
Neo, not quite. A lot of the ‘warm’ flourescents and LEDs use this second warm stage phosphorous strategy. It has an efficiency cost plus an economic cost. Warm color temp alt bulbs cost more. Check Home Depot lest that be in question. There,is no free lunch.
In my experience, warm color and cool color fluorescents and white LEDs have the same number of phosphor stages – one. And warm color LED light bulbs and CFLs do not cost more than cool color ones.
Also, in usual white LEDs, the LED chip does not produce UV but blue. The phosphor in these absorbs some or most (but not all) of the blue light and converts it to a broad spectral band from mid-green to mid-red. The combination of the phosphor’s output and the LED chip’s unabsorbed blue output is white or warm-white, depending on choice of phosphor formulation.
Nonsense.
100% of “white” commercially available LED light bulbs (in USA at least) are actually BLUE LEDs made from Gallium Nitride, or Indium Gallium Nitride, and typically emit at 460-470 nm wavelength.
A common phosphor (not phosphorous) is Cerium Doped YAG (Yttrium aluminum Garnet) which absorbs (SOME) of the blue photons, and fluoresces in a broad yellow spectrum. The lower the percent of blue absorbed, the higher is the color Temperature, and the higher the lumens per watt. That combination is a patented process of Nichia in Japan. The process relies on the fact that Cerium doped YAG has a unique absorption spectrum, with just a single narrow absorption peak at 460 nm, and essentially nothing else. Every other possible rare earth doped YAG has a complex spectrum of absorption lines. There are other phosphor options to avoid the Nichia patent.
The wavelength shift from blue photon to longer wavelength is an loss called the Stokes Shift loss, and is the reason warm whites are less efficient, and is why fluorescents are even less efficient. They start with UV which is invisible so 100% of the visible light is Stokes shifted.
G
“That’s why country after country has banned or is phasing out the inefficient technology.”
I thought that it was all about mark up and regulatory capture. Incandescent bulbs are too cheap.
That is correct. They are too cheap. How do governments make money on this. Taxes on your light bulb.
Eve If I turn on every light in my entire house, including the one in the refrigerator, I use a total of 200 watts of electricity. My electric bill is around $65 per month, and we have an electric range.
G
The bulb is cheaper with incandescent, but the cost of LEDs is falling fast.
And when you account for the reduced electricity usage, and less heat produced which adds even more cost in an air conditioned space, and the LEDs are cheaper eventually.
Payback time is a couple of years. And since they last far longer, they are way cheaper in the long run.
My forays into Chinese-made LEDs for a yard light have been quite disappointing. After three of them failed in just a few months I dropped down the technology curve to a CFL. However, my experience with CFLs in my home have also been disappointing with respect to their longevity. They simply haven’t lived up to the expected/promised longevity.
Clyde,
Early LEDs were very bad, especially the ones made in China, but the technology is progressing very quickly.
If your early forays were more than a few years ago, I have good news…cost is way down and reliability is way way up.
I too have been disappointed in the life of CFLs, but they last longer for me since I learned why they fail, and avoid turning them off and then back on while still warm.
But I dislike them even more for another reason…they contain mercury, and if and when one breaks in the house, you have mercury vapor and dust all in your house.
No thanks on that.
Clyde Spencer, agreed – CFCs and LEDs just don’t last as long as claimed. And when a $3 bulb lasts only 2 months, you really aren’t saving anything on electricity.
sorry, that should be CFLs
Well, look at LED street lighting.
A UK town just borrowed £10 million to replace all street lighting with LEDs…
They will save £5 million each year on electricity to run their street lighting…
The city of Sunnyvale replaced every traffic light in the city with LEDs with a DOE grant, and then received a $750K bonus check from PG&E because of their electricity savings. And they don’t have drivers driving around in trucks all day replacing light bulbs.
They are using another DOE grant to replace all street lighting with LEDs.
The street lights aren’t so great. Plenty of light; but they are too damn bright to the eyes (looking at the lamp).
That’s because they don’t know how to design a good visible white street light. Same goes for auto LED head lights. They suck too.
Reason is simple. An auto headlamp is 8 inches in diameter. The LED ones are a bunch of half inch diameter spots, so their luminance is way too high. Same for the MR-16 spot lights. The quartz halogen ones are 50 mm diameter. The LED ones are a 50 mm diameter heat sink, with maybe three spots of 8 mm or so light.
And yes it is possible to do it right.
G
Don’t forget the guys you have to send around in the winter to clear the snow off because the LEDs don’t produce enough heat to melt it, the time wasted by drivers who can’t tell what colour the lights are because they’re covered in snow and have to slow to a crawl to make sure they’re not going to crash into anyone, and the cost of the crashes caused when people drive through red lights because they can’t tell what colour they are.
The IR reflectivity reminds me of TiNOX ,
http://cosy.com/Science/AGWpptTiNOX.jpg
, which has about the highest solar heat gain of any material yet created . I cited it in my Heartland presentation , http://climateconferences.heartland.org/robert-armstrong-iccc9-panel-18/ , because the ratio of Venus’s surface to orbital gray body temperature is even beyond TiNOX .
The idea that Venus’s surface temperature is due to some spectral “greenhouse” effect is beyond quantitatively absurd . The bottoms of atmosphere are not hotter than there tops due to an optical effect . If you dispute that statement , show us your equations and experimental demonstration . Neither exists .
When before has any physical “scientific” paradigm ever persisted for decades with disastrous real world consequences with no enabling quantitative testable equations ?
This sounds really interesting. In theory, the optical materials could be engineered to take heat from the environment and convert it directly to light without the aid of electric currents. Further, the light produced could be used to generate electric current. This could be a device that reverses entropy.
It is likely that materials will eventually be developed that will coat the outside of vehicles and homes and that would harness the energy for motion and home heating.
Are you kidding?
These things are not using ambient heat, but the IR that exists near a 2700 degree filament.
You need to add a sarc tag.
The relevance to global warming is that the photonic crystal reflects infrared back to its source allowing the source to become warmer than would otherwise be the case. This extra warmth shifts the spectrum up in frequency and more energy is emitted as light rather than heat OR more likely allows the same spectrum to be achieved with less energy.
The greenhouse effect, particularly “downwelling infrared”, achieves the same or similar outcome at least during the day when the sun causes the surface of the Earth to be the emitter. DWIR adds to the solar radiance; but because of its longer wavelength and considerably reduced intensity, doesn’t add much, but it adds something. If it had no effect on Earth, it also won’t have effect in a light bulb and by the same measure since the phenomenon works in a light bulb it must also work on Earth.
I do not say the photonic crystals warm the filament, it is the energy input that warms it. Rather, insulating the filament allows less energy (heat) to produce the same temperature (2700 C) OR same energy (heat), but higher temperature.
No there’s no green house effect making something warmer than the energy feeding it.
I knew it was a matter of very short time before someone else noticed the attempt to slip this in, and claim it proves that ‘such is possible in nature’ – giving off brighter light energy, than is coming in.
The green house effect is a con. CO2 and the green house gases are standard, passive, refractory materials suspended within a larger overall bath of compressible fluids.
They shield the planet from light from the sun, in a spectrum where their presence reduces surface energy 20% right off the top.
These bulbs are not utilizing some sort of magic filter making more light come out of something, than went in. They’re simply making a smaller object warmer with the same power source, then allowing the light to leak out in highly controlled spectra so they trap anything that isn’t highly responsive in the human eye.
We’ll all be having to watch this one make the believer rounds for years, having to explain it all again and again.
There’s no such thing as a green house warming effect, the gases are passive refractory material in a bath. They reduce the amount of sunlight to the surface, therefore less infrared light comes out of the surface, and the more of them there are, the more source light is blocked.
That’s how it happens that total GHGs in the atmosphere, with water about 25,000ppm almost all of it water – about 400 of that’s CO2 –
reduce the earth’s surface energy by 20%. Green House Gases are the only gases that block significant sunlight. Furthermore Water’s solid and liquid phases as clouds, snow, and low angle liquid refraction, are responsible for another 20% across the board reduction in all light to the surface: those phases of water are almost white as far as earth albedo are concerned.
Wou see a large amount of light in any chart, not reaching the planet’s surface, but not being designated, ‘reflected to space by water in non vapor phase,’ that’s almost all water.
When you check a chart showing light at the top of atmosphere vs the surface, you see clearly that the only really significant energy reduction to the planet is by green house gases.
Oxygen comprises about three percent of the light that doesn’t make it to thermalize on the surface, with it’s daylight blue-sky refraction
If there was a green house effect due to these gases, the fundamental gas calculations that regulate everything on earth, would include it. They do not.
Matter of fact there is a separate law of thermodynamics written specifically for compressible fluids; and in the energy specified for fractional gas components of an overall atmospheric mix, -they all get identical energy except one, water.
And the water’s different energy level is due to the quantity of energy the two hydrogen S-orbital electrons’ prodigious energy handling, before they start emitting light as they take more in.
The green house effect is a scam. The atmosphere is not a heater, and the part of the atmosphere that makes less and less light arrive on the surface, are the green house gases, and practically speaking, it’s them alone. (There’s the oxygen mentioned)
And this surface energy reduction is almost 40%, by the GHGs before it’s over.
*In their gaseous state, operating as refractory compounds in the greater nitrogen/oxygen bath, they stop 20% of total sunlight arriving on the surface.
There’s nothing special about them, you can’t block light to an object and thus cause more light to come out of it. It’s a scam.
All frigid baths doped with refractory materials lower temperature of the light-warmed objects immersed in them,
and the more light not arriving due to rising refractory media levels,
the less light is emitted. Just like in all real physics.
There’s no one on earth who has a fluid that they dip things into, which creates a condition of a larger light stream coming out of the objects immersed in it
as more and more fractional refractory components are added to the overall mix,
blocking source light, to the source-dependent, light emitting rock/spherical rock/planet.
E.T. wrote “No there’s no green house effect making something warmer than the energy feeding it.”
Yes, there is. You (and all of your kind) conflate energy and temperature. Energy is power, temperature is pressure. You can have a LOT of pressure (voltage) with nearly zero power; static electricity being an example. At high altitudes the air is “hot” because of highly excited molecules, but so few of them that there’s little heat. Water is the opposite example; it can hold a lot of heat while not changing temperature much (or at all at the “triple point” of 0 C at sea level pressure).
Any time you have a continuous input of energy (heat) and you prevent some or all of the heat from escaping by any means whatsoever, temperature will rise, and it will rise as a direct consequence of preventing energy (heat) from escaping. Temperature is pressure, it is the desire of the heat to leave its source. A little bit of heat tightly confined is going to have a strong desire to leave. In electricity this is voltage, in heat it is temperature, in air compression it is air pressure.
Compressing air does not add heat but it certainly increases its temperature (ignoring frictional heat from the mechanical process of compression).
In the case of the light bulb, preventing some of the energy leaving necessarily raises the temperature of the filament, which would destroy it, so the energy input is reduced. You get the same visible light with less electricity as a result.
In the case of Earth, if you partially prevent heat energy from leaving the surface, by any means whatsoever, and you do not reduce the energy input, the temperature must rise. Anyone that does not comprehend this most basic of facts is probably beyond possibility of having a meaningful conversation on the topic. Of course where Earth is involved it is very complex since the energy input IS sometimes reduced (cloud cover for instance) but the theory is simple enough.
“I knew it was a matter of very short time before someone else noticed the attempt to slip this in.”
And I knew it was a matter of very short time before someone physics challenged would step in. Not you of course, but “someone” 🙂
E.T., you are confused. But “greenhouse gas theory” is pretty simple, in its fundamentals — maybe even simple enough for me to explain it.
You might not realize that the Earth emits about as much radiant energy into space as it receives from the Sun. Well, it does. However, the incoming and outgoing radiation are at very different wavelengths.
The incoming radiation is mostly shortwave: near-IR & shorter (visible, UV, etc.). The spectral peak for radiation from the Sun (i.e., for energy absorbed by the Earth) is around 500 nm. Here’s what the emission spectrum from the Sun looks like:
https://www.google.com/search?q=emission+spectrum+from+the+sun&tbm=isch
The outgoing radiation is almost entirely far-IR & microwave (i.e., longwave). The spectral peak for radiation from the Earth is nearly 20,000 nm. Here’s a good WUWT article showing emission spectra from the Earth:
http://wattsupwiththat.com/2011/03/10/visualizing-the-greenhouse-effect-emission-spectra/
So, anything in the atmosphere which has a different effect on different wavelengths has the potential to either warm or cool the Earth. If it blocks shortwave radiation but passes longwave radiation it will have a cooling effect. If it blocks longwave radiation but passes shortwave radiation it will have a warming effect.
Very roughly, for most wavelengths longer than about 4000 nm, the Earth emits more than it absorbs, and for wavelengths shorter than that the Earth absorbs more than it emits. Conventionally, 4000 nm is near the boundary between near-infrared and mid-infrared.
If you “tint” the atmosphere with a colorant which blocks more incoming short wavelength (4000 nm) radiation, the planet will cool. If you tint the atmosphere with a colorant which blocks more outgoing than incoming radiation, the planet will warm: that’s the so-called (but poorly named) “greenhouse effect.”
Greenhouse gases (GHGs) are colorants. They tint the atmosphere, but in the far-infrared, rather than the visible, part of the spectrum. Carbon dioxide and other GHGs act as dyes in the atmosphere, which “color” the atmosphere in the far-infrared (in the case of CO2, around 15 µm).
There’s no legitimate dispute about this. We know how it works, and what it does, and we can measure the direct effects (such as downwelling IR). The only legitimate arguments are secondary: e.g., whether greenhouse warming is amplified or attenuated by feedbacks (and by how much), whether it is benign or dangerous, and what, if anything, should be done about it.
Here’s a good article:
http://barrettbellamyclimate.com/page8.htm
Since nearly all of the energy emissions from the Earth are in the far infrared & longer wavelengths, but over half of the incoming energy (from the Sun) is at shorter wavelengths (near infrared, visible & UV), tinting the atmosphere in the far infrared has a differential effect. Since there’s more outgoing than incoming far infrared, GHGs absorb mostly outgoing radiation, preventing it from escaping into space. That causes warming. (It’s not how actual greenhouses work, but it’s still a real effect.)
Greenhouse warming of the air, in turn, warms the ground, by several mechanisms, including increased infrared back-radiation from the air.
It doesn’t take much of a trace gas to have a substantial effect on the absorption and emission spectra of the atmosphere. If you add dye to a clear gas or liquid, and then shine through it a light which contains wavelengths that are absorbed by the dye, the gas or liquid will warm due to absorption of the light (compared to its temperature without the dye). Even a few ppm of dye is sufficient to have a substantial effect.
Compare it to the effect of food coloring on water: one drop of food coloring added to 10x10x10 cm (one liter) cubic jar of water (or 57 drops in a 15 gallon fish tank) will noticeably tint the whole liter, but one drop is only about 0.05 ml, so one drop in one liter is 0.05 / 1000 = 0.00005 = just 50 ppm.
But consider: although the atmosphere is less dense than liquid water, it is miles thick. The full thickness of the atmosphere is about the same mass as a 30 foot deep layer of water. Your cubic jar of colored water is only about four inches thick. So to get an equivalent thickness to the Earth’s atmosphere, you’ll have to stack up 90 of those jars of colored water in a 30-foot-long row.
Now, if you were to look through (or shine a light through) the row of 90 jars of colored water, imagine how deep the color would be, from just 50 ppm food coloring.
That’s why just a few ppm of a trace gas can significantly affect the spectrum of the light which passes through the Earth’s atmosphere, and have a potentially significant so-called “greenhouse” effect.
Except at the fringes of carbon dioxide’s absorption bands, there’s so much carbon dioxide in the air that the atmosphere is already very nearly opaque to the IR wavelengths which carbon dioxide mainly blocks. So adding additional carbon dioxide has only a small effect. (MODTRAN Tropical Atmosphere calculates that just 20 ppmv of carbon dioxide would have fully half the warming effect of the current 400 ppmv.) But additional carbon dioxide still does have an effect, primarily on those wavelengths corresponding to the far fringes of carbon dioxide’s absorption bands, where carbon dioxide is nearly-but-not-quite transparent.
The best evidence is that the amount of warming in prospect from anthropogenic CO2 & CH4 is modest and benign. But it’s not zero. (Aside: atmospheric physicist Wm Happer has discovered evidence that it is probably overestimated by about 40%, in which case there’s even less to worry about.)
dave, my only quibble is that you don’t mention that other gases in the atmosphere absorb most of the same bands that CO2 does. This serves to decrease the impact of more CO2 in the atmosphere, since many of these bands are already pretty much saturated.
That is a good point, MarkW.
In fact, I think that’s one of the reasons for so-called “polar amplification.” At extreme latitudes, the air is cold and therefore dry. Less water vapor means there’s less masking of CO2’s 15 µm absorption band by water vapor, so additional CO2 has greater warming effect. (Conversely, closer to the equator, the air is so moist that CO2’s µm absorption band is largely masked by water vapor, so additional CO2 has less warming effect.)
(So “global” warming is actually mostly “polar warming,” because less warming is expected near the equator.)
Nothing to do with the so called ‘greenhouse effect’.
It indicates that the unwanted low frequency radiation is REFLECTED back to the source.
The greenhouse effect is absorption and re-emission which is a totally different kind of physics.
For instance the ‘reflection effect’ can be achieved by selective coating such as the ‘blooming’ of lens surfaces.
This is fully explained by classical optic theory.
This is how the thermal efficiency of solar flat plate collectors is improved.
http://www.almecogroup.com/en/pagina/16-solar
The article further proposes optimising the device to preferentially emit at wavelengths maximising at the part of the eye most sensitive to light to further increase the apparent efficiency.
https://www.nde-ed.org/EducationResources/CommunityCollege/PenetrantTest/Introduction/lightresponse.htm
Its good that the old tungsten incandescent lamp is having a revamp.
“It indicates that the unwanted low frequency radiation is REFLECTED back to the source.
The greenhouse effect is absorption and re-emission which is a totally different kind of physics.”
But same result. How exactly were you going to tell me whether a photon was emitted or reflected?
Well a reflected photon is just that, and is the same wavelength. But a photon emitted after absorption is at a different wavelength and also is emitted in an isotropic distribution pattern, so at least half of it is still directed upwards to space. So the physics is quite different.
And clouds don’t “reflect” at least not more than 2-3%. The rest is refractively scattered, unless it is absorbed. Again different physics, with a different result.
G
george e. smith writes “Well a reflected photon is just that, and is the same wavelength. But a photon emitted after absorption is at a different wavelength”
With respect to your obvious qualifications, I suggest that the emitted photon might be at a different wavelength or the same. Emission spectra and absorption spectra tend to be identical for such substances as can do either. CO2’s emission spectra is also its absorption spectra depending on other factors.
But the elephant in the room is that at STP (standard temperature and pressure) CO2 will absorb the photon and will transfer that energy (to nitrogen probably) by mechanical collision much sooner (on average) than it has a chance to re-emit that photon. Of course, this mechanical energy can come right back to it and *then* be emitted as a photon.
But my point remains; nobody has a way to know whether a particular photon of any wavelength had been emitted or reflected particularly at the longwave IR being discussed. If you have a photon of visible wavelength then you can be pretty sure it wasn’t emitted by a room-temperature molecule of CO2. Not quote 100 percent sure but as close as makes no practical difference.
Consequently, for purposes of comparison with global warming, whether CO2 *reflects* some IR back to Earth, or absorbs and re-emits back to Earth, really doesn’t make a difference; near as I can tell. Also, the selectivity by which CO2 can do this mimics the function of a dichroic mirror’s selective reflectivity.
George writes “And clouds don’t “reflect” at least not more than 2-3%. The rest is refractively scattered, unless it is absorbed.”
Pedantically true; well done! So long as half that refraction goes “up” back into space it accomplishes the same goal of preventing some photons from hitting my zucchini plants.
Michael 2 wrote, “…at STP (standard temperature and pressure) CO2 will absorb the photon and will transfer that energy (to nitrogen probably) by mechanical collision much sooner (on average) than it has a chance to re-emit that photon…”
That is true! Atmospheric physicist Wm Happer graciously explained this to me a couple of years ago.
1. At low altitudes, the mean time between molecular collisions, through which an excited CO2 molecule can transfer its energy to another gas molecule (usually N2) is a little over 1 nanosecond.
2. The mean decay time for an excited CO2 molecule to emit an IR photon is on the order of 1 second (a billion times as long).
So, after a CO2 (or H2O) molecule absorbs a 15 micron IR photon, nearly 99.9999999% of the time it will give up its energy by collision with another gas molecule, not by re-emission of another photon.
So the very widely repeated description of GHG molecules absorbing infrared photons and then re-emitting them in random directions is only correct for about one or two absorbed photons in a billion. Here’s an example from the NSF, with a lovely animated picture, which even illustrates the correct vibrational mode:
http://www.sealevel.info/Happer_UNC_2014-09-08/Another_question_files/co2_absorb_emit_infrared_anim_320x240.gif
http://scied.ucar.edu/carbon-dioxide-absorbs-and-re-emits-infrared-radiation
That illustration is wrong for about 99.9999998% of the photons which CO2 absorbs in the lower troposphere.
Prof. Happer told me, ” It is this extreme slowness of radiative decay rates that allows the CO2 molecules in the atmosphere to have very nearly the same vibration-rotation temperature of the local air molecules.”
Michael 2 wrote, “…nobody has a way to know whether a particular photon of any wavelength had been emitted or reflected particularly at the longwave IR being discussed…”
True. Photons of the same wavelength and polarization are fungible.
Michael 2 wrote, “Consequently, for purposes of comparison with global warming, whether CO2 *reflects* some IR back to Earth, or absorbs and re-emits back to Earth, really doesn’t make a difference; near as I can tell…”
There is one difference.
If the photons were reflected by CO2, or if that NSF web page & illustration were right, then the amount of IR emitted by CO2 in the atmosphere would depend heavily on how much IR it received and absorbed. If more IR was emitted from the ground, then more IR would be re-emitted by the CO2 molecules, back toward the ground. But that’s wrong.
Since ~99.9999998% of the IR absorbed by atmospheric CO2 is converted by molecular collisions into heat, the amount of ~15 micron IR emitted by atmospheric CO2 depends only on the atmosphere’s temperature (and CO2 partial pressure), not on how the air got to that temperature. Whether the ground is very cold and emits little IR, or very warm and emits lots of IR, will not affect the amount of IR emitted by the CO2 in the adjacent atmosphere (except by affecting the temperature of that air).
“The relevance to global warming is that the photonic crystal reflects infrared back to its source allowing the source to become warmer than would otherwise be the case.”
This article reminds me of Willis Eschenbach’s classic “Steel Greenhouse” article on WUWT:
https://wattsupwiththat.com/2009/11/17/the-steel-greenhouse/
Sunlight mimicking broad spectrum would be a welcome relief from the current designs, but suspect light bulb marketing ploys still lurk in the shadows.
http://spectrum.ieee.org/geek-life/history/the-great-lightbulb-conspiracy
Wonderful idea and keep working on it, but I am totally sold on LEDs. They can take the stress of a fan or car ride, never flicker, produce perfect spectrum light, little heat, and actually do last unlike CFLs which are extremely sensitive to power surges, shaking, etc. and whose light spectrum changes as it ages.
The big thing that wrecks the lifespan of the CFLs is turning them on and off frequently. If they are not allowed to cool off completely before being turned back on, it ruins them quickly.
I am also sold on LEDs…they are getting pretty cheap, and use a tiny trickle of current compared to the old kind of bulbs.
And there are getting to be a lot of choices regarding directionality and wattage. You can get light that is pointed just where you want it, or ones that spread out evenly.
But these bulbs have always suffered from one major problem: More than 95 percent of the energy that goes into them is wasted, most of it as heat.
To me it is not wast heat, when we use our regular light bulbs in the winter that heat is used, it does help “heating” the house and that leads me to question that has me bothered for a while
We have a few desk lamps that plug in the wall goes through a transformer at the base of the lamp and lights a 13 watt “energy saving” bulb. The base of the desk lamp is always warm after a few minutes of being on, to me that is waste heat as well so where does the “energy savings” kick in?
Those used to be called “high intensity” lights.
The transformer is to lower the line voltage, typically to 12 V, a factor of 10. To make a low wattage bulb that works at 120 V, you’d need a thin, long filament to boost the electrical resistance of the filament. That means a delicate filament and one that produces a pulsating light. Not too obnoxious in most settings, but quite usable as a strobe light for checking LP turntables. Before low voltage lights came back, a lot of Christmas tree lights used 7.5, 120 V bulbs. When I was growing up, I took on the job of replacing the burned out ones before we strung them on our tree.
At 12 V, the filament in your lamp can be thicker and shorter, so more sturdy and has a longer life.
The energy savings is that you have a 13 watt bulb instead of a 40 or 60 watt bulb. The transformer uses a few watts.
A more efficient transformer would generate less heat and waste less energy. It would be heavier, larger, and more expensive.
I’m often surprised at how small the transformers are in consumer grade equipment. For the same watts, the equivalent transformer in industrial grade equipment is much larger and heavier.
Small transformers are designed to operate at some maximum temperature, limited by the specific materials; insulation and such. They are designed so that the amount of resistive loss in the copper wire is the same as the eddy current and hysteresis losses in the iron core. You can prove that is the highest efficiency condition. So you reduce the core size, the flux and core losses increase. You put more turns of finer wire the copper losses increase, but the flux goes down so they core losses reduce. It’s a well understood design problem, and they are designed to be as hot as they get. You make them bigger, and they weigh more and cost more for iron and copper, but they will run cooler.
G
It’s interested me in the way that modern, ‘cooler’ lights do not heat the things (
People, walls, other objects) like incandescent lights do.
What,is the,comparison in price between a watt of IR and a watt of warmer air from a gas furnace?
What you get from incandescent bulbs is basically the same as electrical resistance heating.
The comparison between electricity and natural gas depends on the price of each. Here’s an article that has electricity costing about three times as much as gas. Where I am, right now, it’s even worse.
You mentioned IR. That’s not much more efficient for raising the indoor temperature of your house. In that case a BTU is a BTU. We use IR on the patio because it warms the people without having to heat the great outdoors.
asybot.
Good point. Those dumb fools in the DOE don’t realize that there are situations where the heat from a light bulb may add to comfort and is not necessarily wasted. The room we spend the most time in our house is poorly heated by our forced air heating system, being on a concrete pad, having 3 outside walls and only one register to deliver warmed air from the furnace. Early on I tried everything to improve the heat distribution including installing a larger fan capacity, cutting out the shutters in the vent cover, adding insulation, replacing paneling with dry wall, and playing with the poorly designed butterfly in the basement to try to redistribute the air.
While some improvement was noted, I did ultimately install overhead lighting using 4 incandescent flood lights which do a good job in the winter. Since the bulbs are on a dimmer switch, I can minimize the extra heat in the summer when minimal lighting is needed.
FYI these bulbs are just beginning to burn out after about 25 years so don’t talk to me about short life.
Sure the heat from the electricity is more expensive than the heat via natural gas, but incremental heating can’t be that expensive.
We should be given a choice and not have ignorant people in Washington declare mandates with foolish regulations.
Incandescents have their already-low energy efficiency greatly decreasing when they are dimmed. When an incandescent is dimmed so as to consume half its normal wattage, its light output is around 20% of its normal light output. This is not the case with LED and CFL light bulbs (when dimmable ones are used – some LED and most CFL light bulbs are not designed to be compatible with dimmers).
Catcracking, as DLK points out, your dimmed flood lights last a long time, but are using a lot more power per lumen than if you just had fewer lamps lit.
For heating, using a ceramic heater right where you are sitting may be much more efficient than having most of it way up near the ceiling, too.
I also like the ones that heat up a tank of oil and are shaped like a old fashioned radiator.
But the ceramic ones with a small fan built in are very good for heating a small area if you spend a lot of time in one place, lie watching TV or one the computer.
Of course, down hear in Florida, most of the year the AC is cranking, so bulbs that get hotter are doubly wasteful.
Catcrackin,
Another option you might try is overhead infrared heat lamps. Years ago my father was asked to come up with a way of providing a more comfortable work environment on the floor of an old textile mill that had very high, maybe 15′ ceilings. He had them install infrared heat lamps hanging from the ceiling, about 10′ above the floor between the florescent light fixtures. He took me in to show me the difference after one of the rooms had been converted. The air temperature was still around the high 50s to low 60s but the work area was warm and toasty. According to the client it saved them quite a bit on heating bills.
My main point is that it is a lie to claim the heat is wasted since the bulbs heat a particular room in my house in the fall/winter. I should be able to choose based on my decision what bulbs are best for me in a particular situation not a bureaucrat in Washington. I once tried the wonderful CFD bulbs in table lamps, and their life was not as claimed and the light color annoying. I am well aware of the of the potential benefits of LED lights, but will not throw away perfectly good incandescent bulbs and have to address the need for supplemental winter heat at this stage. Space heaters are not too attractive to me. Thanks for comments.
not wast heat, sorry, waste heat.
Various links from January 2016:
https://energy.mit.edu/news/a-nanophotonic-comeback-for-incandescent-bulbs/
The original.
http://www.nature.com/nnano/journal/v11/n4/full/nnano.2015.309.html
A different original. Paywalled.
http://www.pri.org/stories/2016-01-31/can-we-recycle-light-and-can-it-help-us-fight-global-warming
I like the headline.
Now the hordes of wannabelievers are gonna come out of the woodwork and claim this is the ‘same principle as the green house effect in action!’
LoL
E.T. writes “Now the hordes of wannabelievers are gonna come out of the woodwork and claim this is the ‘same principle as the green house effect in action!’ “
Followed by hordes of wannanotbelievers with nothing substantial to contribute. Keeping the emitter hot is the operating principle of this new light bulb and is also the principle (or result) of the greenhouse effect.
And how can these be reconciled with sitting in our caves around a dung-fueled fire if the Greens get their way?
so is the 100 year bulb a lie? http://centennialbulb.org
4 watts and uses a lot of power (proportionally speaking) for that small amount of light.
The energy is only wasted in warm months, and the spectrum is annoying. So this whole scheme is another boondoggle.
By the way, the low-flow toilets our “representatives” force US to use are so weak they have to be flushed at least twice, and they can’t even remove a piece of toilet paper on the side of the bowl. So you can bet THEY don’t use them in their many mansions and offices, which we pay for.
Politicians need to keep their noses out of everyone elses business.
http://ecx.images-amazon.com/images/I/518msm8IDDL.jpg
The most powerfully-flushing toilets I ever saw in houses and other residential buildings are low flow ones.
Larry,
“By the way, the low-flow toilets our “representatives” force US to use are so weak they have to be flushed at least twice…”
I got one not long ago, though I wouldn’t call it low-flow, so much as low total volume of water per flush . . so I added a small section of plastic tubing that fit snugly into the one it came with, which allows the tank to fill more than it did at first . . and it flushes quite well now. (You can still get a minimal flush if appropriate, by letting go of the handle quickly). Very easy fix.
Well, I for one am very keen to see the return of incandescent light bulbs. The light given out by the ugly curly bulbs is horribly unfriendly – let’s face it, that’s why in the past fluorescent tubes were used for commercial premises not in people’s living rooms. LEDs are brilliantly efficient but their light too is horrible for the home. I deeply resent that my government made incandescent bulbs illegal, thus allowing the makers of the curly rubbish and LEDs to gouge enormous prices for their products. The capital cost of these was such that it could never be recouped by reduced electricity usage. Well, it couldn’t, that is, until the green left started on their push into unreliables thus pushing up the cost of electricity.
So, please allow me to choose for myself whether to use incandescents (and pay for the electricity usage), and please allow the electricity providers to choose freely from all possible electricity sources. It’s no fun being sane in an insane society.
A 10,000 hour LED lightbulb with spectrum good enough for it to be used in some restaurants and consuming 8.5 watts to produce 60-watt incandescent-equivalence is now available at Lowes for about $8 per 3-pack.
Would, “some restaurants” be those who don’t actually want you to see what’s on your plate? 🙂
In a kitchen renovation my wife replaced all the CFL ceiling light bulbs with (moderately) inexpensive Cree lamps. Cree has always produced bulbs with a high CRI (Color Rendition Index or something like it) and has been producing inexpensive stuff lately. I already had one of their older lamps over the stove, I had bought it partly to see how it operated and also so egg yolks wouldn’t look sickly pale.
I work for a company that manufactures lake fountains, and we are moving ahead with LEDs as an option on lighting.
The early Chinese ones from a few years back were a disaster…not very bright, and they did not last.
Newer ones made domestically in the US and Canada are very good, and very efficient.
The payback time is a couple of years for submersible fixtures that use about 55 watts to produce equivalent illumination to the old standard 500 watt PAR56 MFL. And that is just the electricity payback. The old PAR 56 500 watt bulbs last about two years or so, depending on how late the fountain stays on at night.
For home lighting, the situation has changed rapidly in the past few years, and many color temps are now readily available.
And prices are falling very fast.
What was true in years past is no longer true.
Epic sentence. Is it yours? Can I use it?
First they banned the 75 watt bulb so I bought plenty of them to store away. Then they banned the 60 watt bulb. I don’t use them. Then they banned the 40 watt bulb which is the bulb I use the most. I bought so many that they will last me a lifetime. That was to be the end of the incandescent bulb. Then I stopped at a “poor person’s” store in an area that I haven’t been to in years. I had gotten a job in the area. To my gleeful surprise, they sold 38 watt incandescent bulbs, four for $2.19. Ha, ha! My faith in mankind was restored.
There is a store in my home state, New Jersey, that sells incandescent bulbs by mail. They found a loophole in the federal legislation. They sell a modified, improved type of bulb.
http://www.newcandescent.com/
The CREE high CRI LED bulbs (True Color I think they call it) produce a very nice, natural color. Take a look. I have had good luck with the CREE bulbs lasting a long time, as long as they get decent airflow. They will pay for themselves in electricity costs even if they only last 10 years. And it’s nice not having to replace bulbs every year or two.
Well I’m all for your freedom too Mike; we don’t need the gummint choosing for us.
But as to your primary objection; the alternatives to your beloved Edison screw filament bulbs. What makes you think these newcandescents will be any less ugly than the CFLs or LEDs. So even if they worked and became available you probably still wouldn’t like them. That’s whay they still make bare filament lamps and flickering filaments and pointy lamps.
People think they are decorations. Fine if that is your thing. For me thay are purely utilitarian.
I go into a room, I turn the light on (unless it is daytime) and when I leave the room, I turn the light off, and probably close the door too, if I want to conserve the heat pattern.
Just the way I’m built, I guess.
G
A lot of my paintings don’t work with this new inflorescent lighting. If they were painted with incandescent light vs daylight. As An example:
http://www.purecolorartist.com/night–day.html
J Philip Peterson…
“More than 95 percent of the energy that goes into them is wasted, most of it as heat. That’s why country after country has banned or is phasing out the inefficient technology. ”
NO, that was no why they banned the traditional light bulb. GE got tired of selling cheap, affordable product and quite often the heat produced went to heat the room it was in. The released heat was far from all bad. GE lobbied Congress to ban the bulb so that they could force the public to buy these heinous devices
AND, the CFLs are much more expensive, can burst into flame, usually take time to get bright, and do not last anywhere as long as claimed, and are environmental hazards if broken and in disposal. They are as ungreen for lighting as wind turbines are for energy. They both suck big time.
Ridiculous.
GE had a large market share in incandescent bulbs.
They had a far smaller share of the CFL market.
And they are not a major manufacturer of LED lighting at all.
So that makes zero sense and is likely a crock of BS.
GE now has some LED bulbs on the store shelves. And no they have nothing to crow about compared to the competition. Same as Sylvania. They have a product to hold their place on the shelves, and people will just pick the known brand. I have NO GE or Sylvania lighting.
G
And how exactly does a light bulb with no flammable parts “burst into flames”?
It cant, but that is not an accurate description of CFLs. They do have inflammable parts.
CFLS are simply pants. They never achieved the efficiency or longevity of separate ballast style fluorescents, because packaging the electronics in a small bulb was simply a bad idea.
LEDS, which can in the limit run with a lossless capacitor to limit current, run cooler. start faster and can be tailored to a much kinder spectrum.
CFL’S didn’t cut the mustard. LEDS do.
I have had the small ballast in the base of a cheap CFL burn out and fuse the base to the socket such that I had to replace the light socket. It must have generated a lot of heat to do that, so I suppose it is possible that it could start a fire. But “burst into flames” is a bit of hyperbole, if you ask me. They do contain small amounts of mercury though, so the “environmental hazards” part is accurate. The other problem with CFLs is that they don’t work well (or at all) if they get too cold, making them impractical for outdoor lighting north of the frost line.
Bursting into flames is a well-known failure mode among electricians. I hang out at a blog where several have shared pictures of the remains. Basically the ballast fails, shooting out a small flame at the base of the bulb. UL considers this an “acceptable” failure mode <:-O.
“But these bulbs have always suffered from one major problem: More than 95 percent of the energy that goes into them is wasted, most of it as heat”
Well heat is often quite useful,, depending on where you live and the cost of gas heat.
I never looked at the heat from light bulbs as waste. They just nudge the thermostat thingy a little to the warm side and save me some natural gas.
Of course deep summer is a different story, but we turn out the lights anyway.
Resistive electric heat costs more than heat from natural gas (or oil) in most of the US. This has been true most of the time at least since 1975.
Sure, but the price difference is small, especially where I live it’s about $0.10/kWhr.
Small enough that the LED bulbs that don’t dim properly and have narrowband light aren’t worth the extra cost.
“Did you know that the lifetime of light bulbs have been reduced on purpose to just 1000 hours?
Or that your printer may ‘break’ after a predefined number of printed pages determined by an electronic counter?
Or that Apple batteries lasted only 18 months which meant that you had to throw away your iPods and buy another?
What about Dupont stockings that were strong enough to tow a car, before they went back to design a stocking that would wear out more quickly!”
Or bais ply tires that got replaced by radials that last much longer and have lower rolling friction.
Uh, wait….
Lets not forget the 100mpg carburetor, or the perpetual motion machine.