Guest Post by Willis Eschenbach
Although it sounds like the title of an adventure movie like the “Bourne Identity”, the Bern Model is actually a model of the sequestration (removal from the atmosphere) of carbon by natural processes. It allegedly measures how fast CO2 is removed from the atmosphere. The Bern Model is used by the IPCC in their “scenarios” of future CO2 levels. I got to thinking about the Bern Model again after the recent publication of a paper called “Carbon sequestration in wetland dominated coastal systems — a global sink of rapidly diminishing magnitude” (paywalled here ).
 Figure 1. Tidal wetlands. Image Source
Figure 1. Tidal wetlands. Image Source
In the paper they claim that a) wetlands are a large and significant sink for carbon, and b) they are “rapidly diminishing”.
So what does the Bern model say about that?
Y’know, it’s hard to figure out what the Bern model says about anything. This is because, as far as I can see, the Bern model proposes an impossibility. It says that the CO2 in the air is somehow partitioned, and that the different partitions are sequestered at different rates. The details of the model are given here.
For example, in the IPCC Second Assessment Report (SAR), the atmospheric CO2 was divided into six partitions, containing respectively 14%, 13%, 19%, 25%, 21%, and 8% of the atmospheric CO2.
Each of these partitions is said to decay at different rates given by a characteristic time constant “tau” in years. (See Appendix for definitions). The first partition is said to be sequestered immediately. For the SAR, the “tau” time constant values for the five other partitions were taken to be 371.6 years, 55.7 years, 17.01 years, 4.16 years, and 1.33 years respectively.
Now let me stop here to discuss, not the numbers, but the underlying concept. The part of the Bern model that I’ve never understood is, what is the physical mechanism that is partitioning the CO2 so that some of it is sequestered quickly, and some is sequestered slowly?
I don’t get how that is supposed to work. The reference given above says:
CO2 concentration approximation
The CO2 concentration is approximated by a sum of exponentially decaying functions, one for each fraction of the additional concentrations, which should reflect the time scales of different sinks.
So theoretically, the different time constants (ranging from 371.6 years down to 1.33 years) are supposed to represent the different sinks. Here’s a graphic showing those sinks, along with approximations of the storage in each of the sinks as well as the fluxes in and out of the sinks:
Now, I understand that some of those sinks will operate quite quickly, and some will operate much more slowly.
But the Bern model reminds me of the old joke about the thermos bottle (Dewar flask), that poses this question:
The thermos bottle keeps cold things cold, and hot things hot … but how does it know the difference?
So my question is, how do the sinks know the difference? Why don’t the fast-acting sinks just soak up the excess CO2, leaving nothing for the long-term, slow-acting sinks? I mean, if some 13% of the CO2 excess is supposed to hang around in the atmosphere for 371.3 years … how do the fast-acting sinks know to not just absorb it before the slow sinks get to it?
Anyhow, that’s my problem with the Bern model—I can’t figure out how it is supposed to work physically.
Finally, note that there is no experimental evidence that will allow us to distinguish between plain old exponential decay (which is what I would expect) and the complexities of the Bern model. We simply don’t have enough years of accurate data to distinguish between the two.
Nor do we have any kind of evidence to distinguish between the various sets of parameters used in the Bern Model. As I mentioned above, in the IPCC SAR they used five time constants ranging from 1.33 years to 371.6 years (gotta love the accuracy, to six-tenths of a year).
But in the IPCC Third Assessment Report (TAR), they used only three constants, and those ranged from 2.57 years to 171 years.
However, there is nothing that I know of that allows us to establish any of those numbers. Once again, it seems to me that the authors are just picking parameters.
So … does anyone understand how 13% of the atmospheric CO2 is supposed to hang around for 371.6 years without being sequestered by the faster sinks?
All ideas welcome, I have no answers at all for this one. I’ll return to the observational evidence regarding the question of whether the global CO2 sinks are “rapidly diminishing”, and how I calculate the e-folding time of CO2 in a future post.
Best to all,
w.
APPENDIX: Many people confuse two ideas, the residence time of CO2, and the “e-folding time” of a pulse of CO2 emitted to the atmosphere.
The residence time is how long a typical CO2 molecule stays in the atmosphere. We can get an approximate answer from Figure 2. If the atmosphere contains 750 gigatonnes of carbon (GtC), and about 220 GtC are added each year (and removed each year), then the average residence time of a molecule of carbon is something on the order of four years. Of course those numbers are only approximations, but that’s the order of magnitude.
The “e-folding time” of a pulse, on the other hand, which they call “tau” or the time constant, is how long it would take for the atmospheric CO2 levels to drop to 1/e (37%) of the atmospheric CO2 level after the addition of a pulse of CO2. It’s like the “half-life”, the time it takes for something radioactive to decay to half its original value. The e-folding time is what the Bern Model is supposed to calculate. The IPCC, using the Bern Model, says that the e-folding time ranges from 50 to 200 years.
On the other hand, assuming normal exponential decay, I calculate the e-folding time to be about 35 years or so based on the evolution of the atmospheric concentration given the known rates of emission of CO2. Again, this is perforce an approximation because few of the numbers involved in the calculation are known to high accuracy. However, my calculations are generally confirmed by those of Mark Jacobson as published here in the Journal of Geophysical Research.

_Jim says:
May 6, 2012 at 12:35 pm
_Jim and Mydogs, please, this thread is about CO2 sequestration and the Bern Model. Please take the blackbody discussion to some other more appropriate thread.
Thanks,
w.
ferd berple says:
Your comment is awaiting moderation.
May 6, 2012 at 12:39 pm
Bart says:
May 6, 2012 at 12:32 pm
In the IPCC framework, that 1/2 dissolves rapidly into the oceans.
ps: when I said “nonsense” I was referring only to the IPCC framework or any other mechanism that suggests different absorption rates based on the age of the CO2 in the atmosphere.
Willis Eschenbach says:
May 6, 2012 at 12:33 pm
“No, the link you gave explains simple exponential decay from a mathematical viewpoint, which tells us nothing about the Bern model.”
No, that’s not what it explains at all. It is a statistical model in which the probability distribution is exponential, to be used in finding a solution of the Fokker-Planck equation. The “decay” he shows is actually 1/(1+a*sqrt(t)), the reciprocal of 1 plus a constant time the square root of time.
Sorry I cannot explain it better right now. Must go.
son of mulder asks:
“Any others?”
There is overwhelming evidence that the biosphere is expanding due to the increase in CO2. There is no doubt about that. Therefore, it is not in ‘equilibrium’. As ferd berple points out, more of the increase is absorbed every year.
In addition, the oceans contain an enormous quantity of calcium, which is utilized by biological processes to form protective shells for organisms. Those organisms require CO2. With more CO2 available, those organisms rapidly proliferate. When they die, they sink to the ocean floor, thus permanently removing CO2 from the atmosphere.
The planet is greening due to the added CO2, which is completely harmless at current and future concentrations. If CO2 increases from 0.00039 of the atmosphere to 0.00056 of the atmosphere, it is still a very minor trace gas. At such low concentrations plants are the only thing that will notice the change. And any incidental warming will be minor, and welcome.
I am particularly intrigued by the 17.01 year figure, I had no idea climate science was so precise.
son of mulder says:
May 6, 2012 at 12:36 pm
Any others?
================
bacteria…..the entire planet is one big biological filter
They are the most abundant…………..or we wouldn’t be here
Willis Eschenbach – “However, there is nothing that I know of that allows us to establish any of those numbers. Once again, it seems to me that the authors are just picking parameters.”
I would point out a very important part of the link you referenced (http://unfccc.int/resource/brazil/carbon.html):
“All IRFs are obtained by running the Bern model (HILDA and 4-box biosphere) as used in SAR or the Bern CC model (HILDA and LPJ-DGVM) as used in the TAR.” – (IRF’s -> impulse response functions, time factors, and final percentages)
The percentages you quoted are the resulting partial absorptions of various climate compartments resulting from running the Bern model, which is described in Siegenthaler and Joos 1992 (http://tellusb.net/index.php/tellusb/article/viewFile/15441/17291). In short, those percentages are results, not the inputs, of running the Bern model – presented by Joos et al for use by other investigators if they wish to apply the Bern model to their calculations.
Also note the statement that “Parties are free to use a more elaborate carbon cycle model if they choose.” Again – the results of the Bern model were offered as an available computational tool for further work.
I hate to say this, but you give the impression you did not fully read the UN reference (with percentages) that you opened the discussion with…
“My thanks for your explanation. That was my first thought too, Nullius. But for it to work that way, we have to assume that the sinks become “full”, just like your tank “B” gets full, and thus everything must go to tank “C”.”
That’s where I was going with the following paragraph. The buffer ‘tank B’ doesn’t stop absorbing because it’s full, it stops absorbing because the levels equalise. If you keep pouring water into tank A continuously, the water level keeps going up in B continuously. The tanks have infinite capacity, but the ratios of their capacities are much smaller.
The partitioning is the equivalent of the ratio of surface areas in each tank. If A and B are of equal size, then half the water in A flows into B and half stays where it is. If B is a lot bigger than A, then the level in A drops more and the level in B only rises a tiny amount heightwise, although the changes in volume are the same. The atmospheric analogy to surface area is the derivative of buffer content with respect to concentration.
I just went through that post with Salby video (didn’t have time before). Amazing that people still misunderstand the natural CO2-rise argument (like by Salby). Again:
The rise in the atmospheric CO2 is caused by warming climatic factors. The source is anthropogenic CO2, because it’s available in the atmosphere, but the cause is the warmth. Without anthropogenic CO2, oceans would have to release the necessary CO2 to achieve the climatically driven atmospheric CO2.
rgbatduke says:
May 6, 2012 at 12:12 pm
Robert, if I understand you correctly, I can’t tell you how happy I am to see you write this. That was my first response when I read the linked description of the Bern Model, it made my head explode. Unfortunately, I didn’t (and don’t) have your familiarity with the underlying math, so I had to set up some separate sample exponential decay streams in Excel, combine them, and then calculate iteratively the joint time constant … all of which very clumsily lead me to the conclusion that you established with a few lines of math …
Since I did that, which was maybe five years ago, I’ve questioned it and people have given vague handwaving explanations for something that I, like you, consider to be “wrong, wrong, wrong” and without any physical justification.
w.
Willis, with all due respect, that is ALL I had (and have) time for; I have to ‘be somewhere’ shortly. Thanks. I ‘capeesh’/capisce/’savvy’ the expressed desire to stick-to-the-issue-presently-being-debated, too. Good luck with your present efforts, and with that I gotta run … 73’s
.
CO2 started increasing about 1750. Human emissions of CO2 more-or-less started at that time as well. Here is a chart of Human Emissions in CO2 ppm versus the amount of CO2 that actually stayed in the air each year (the airborne fraction – about 50%) since 1750.
http://img163.imageshack.us/img163/9917/co2emissandcon1750.png
Global CO2 levels only increased 1.94 ppm last year (to 390.45 ppm – a little lower than expected) while human emissions continued increasing to about 9.8 billion tonnes Carbon (about 4.6 percentage points of CO2).
The natural sinks of CO2 have been increasing gradually over time so that they are now over 224 billion tons Carbon versus 220 billion tons in 1750. (the actual natural sinks and sources level might be closer to 260 billion tons going by some recent estimates of plant take-up but none-the-less).
http://img233.imageshack.us/img233/1323/carbonnatsinks1750.png
The amount that the natural sinks absorb each year seems to be directly related to the concentration in the atmosphere. There is an equilibrium level of CO2 at about 275 ppm in non-ice-age conditions (this is the level it has been at for the past 24 million years).
So the natural sinks and sources are in equilbrium (give or take) when the CO2 level is 275 ppm or the Carbon level in the atmosphere is 569 billion tonnes.
The rise of the natural sinks over the past 250 years indicate the sinks will absorb down or sequester about 1.0% per year of the excess over this 569 billion tons or 275 ppm.
The last 65 years have been very close to the 1.0% level. It doesn’t matter how much we add each year. The plants and oceans and soils will respond to how much is in the air, not how much we add. And it is about 1.0% of the excess Carbon in the amtosphere each year – Bern model or no.
http://img580.imageshack.us/img580/521/co2absor17502011.png
It will take about 150 years to draw down CO2 to the equilibrium of 275 ppm if we stop adding to the atmosphere each year. Alternatively, we can stabilize the level just by cutting our emissions by 50%.
To attempt to clarify what I wrote in my previous post (http://wattsupwiththat.com/2012/05/06/the-bern-model-puzzle/#comment-978032):
The exponentials, percentages, and time factors in the link Willis Eschenbach provided are approximations that reproduce the results of running the Bern model – much as 3.7 W/m^2 direct forcing per doubling of CO2 is the approximation of running radiative code such as MODTRAN, allowing quick calculations without having to run the model over and over again. I.e., the percentages and time factors are shorthand for the model.
As Joos stated in that link (http://unfccc.int/resource/brazil/carbon.html), the Bern model approximations were offered as a tool for use by others, and “Parties are free to use a more elaborate carbon cycle model if they choose.”
OK, I’ve looked at the model details via the provided link. They are frigging insane. I mean seriously, one should just take the article’s provided advice and ‘use a more complex model’ if we like. I like. Here is a very simple linear model. Still too simple, but at least I can justify its structure:
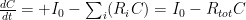
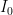 , that is completely independent of the concentration
, that is completely independent of the concentration 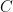 . Because the atmosphere is vast, the percentage of CO_2 in the atmosphere can be considered to be the amount of CO_2 added divided into the total where the latter basically does not vary, hence I don’t need to work harder and write an ODE that saturates at 100% CO_2 — we are in the linear growth regime of a saturating exponential and I can assume assume that the concentration increases linearly at a constant rate independent of how much is already there (true until a significant fraction of the atmosphere is CO_2, utterly true when 400 ppm is CO_2).
. Because the atmosphere is vast, the percentage of CO_2 in the atmosphere can be considered to be the amount of CO_2 added divided into the total where the latter basically does not vary, hence I don’t need to work harder and write an ODE that saturates at 100% CO_2 — we are in the linear growth regime of a saturating exponential and I can assume assume that the concentration increases linearly at a constant rate independent of how much is already there (true until a significant fraction of the atmosphere is CO_2, utterly true when 400 ppm is CO_2).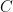 .
. 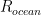 and
and 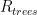 are themselves directly proportional to (or more generally dependent on) other sensible quantities — we might expect the former to be proportional to the total surface area of the ocean for example, or to be related to some function of its area, its local temperature, and the concentration of CO_2 in the water already (which MIGHT vary appreciably geographically, as seawater is not well-mixed and it has its own sources and sinks). We might expect the latter to be dependent on the total surface area of CO_2 scavenging tree leaves, or more simply to total acreage of trees, again leaving open a more complex model that couples in the further modulation by water availability, hours of sunlight, and so on. Still, averaging over these latter probably makes this simple model already pretty reasonable.
are themselves directly proportional to (or more generally dependent on) other sensible quantities — we might expect the former to be proportional to the total surface area of the ocean for example, or to be related to some function of its area, its local temperature, and the concentration of CO_2 in the water already (which MIGHT vary appreciably geographically, as seawater is not well-mixed and it has its own sources and sinks). We might expect the latter to be dependent on the total surface area of CO_2 scavenging tree leaves, or more simply to total acreage of trees, again leaving open a more complex model that couples in the further modulation by water availability, hours of sunlight, and so on. Still, averaging over these latter probably makes this simple model already pretty reasonable.
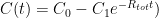
 and where
and where 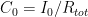 is the steady state concentration one arrives at eventually from any starting concentration, as long as
is the steady state concentration one arrives at eventually from any starting concentration, as long as  (see linearization requirement above).
(see linearization requirement above). 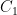 is a constant of integration used to set the initial conditions. If you started from no CO_2 in the air at all, you would make
is a constant of integration used to set the initial conditions. If you started from no CO_2 in the air at all, you would make 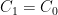 so that
so that 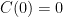 . We don't start from zero, so we have to choose it such that
. We don't start from zero, so we have to choose it such that 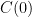 comes out right. At the steady state concentration, the sinks remove CO_2 at the rate
comes out right. At the steady state concentration, the sinks remove CO_2 at the rate 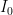 , balancing the sources.
, balancing the sources. and one approaches it exponentially with time constant
and one approaches it exponentially with time constant 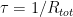 — you can’t get much simpler than that. In fact, if you know
— you can’t get much simpler than that. In fact, if you know 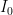 and can measure
and can measure  one can is done, no need for complex integrals over sums of exponential sinks times a source rate (what the hell does that even MEAN).
one can is done, no need for complex integrals over sums of exponential sinks times a source rate (what the hell does that even MEAN).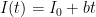 for example, describing source production that is increasing linearly in time, or
for example, describing source production that is increasing linearly in time, or  , a model that assumes source production is itself increasing towards an eventual peak at some rate with exponential time constant
, a model that assumes source production is itself increasing towards an eventual peak at some rate with exponential time constant  . The former suffers from the flaw that it increases without bound. The latter is probably not terrible, but I’m guessing CO_2 sources are bursty and that this equation is a pretty crude approximation of the industrial revolution and eventual saturation of production/sources. Both suffer from the fact that CO_2 production might depend on the concentration
. The former suffers from the flaw that it increases without bound. The latter is probably not terrible, but I’m guessing CO_2 sources are bursty and that this equation is a pretty crude approximation of the industrial revolution and eventual saturation of production/sources. Both suffer from the fact that CO_2 production might depend on the concentration 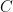 — although these production mechanisms can probably be handled with negative
— although these production mechanisms can probably be handled with negative  .
. depends on the temperature! A warming ocean can be a CO_2 source (or a heavily reduced sink as its uptake is reduced). A cooling ocean can sequester more CO_2, faster. But even this is too simple because part of the eventual sequestration involves chemistry and biology that depend on temperature, sunlight, animals activity, ocean currents and nutrients… so it is with all of the rates. They themselves might be — indeed, almost certainly are — functions of time!
depends on the temperature! A warming ocean can be a CO_2 source (or a heavily reduced sink as its uptake is reduced). A cooling ocean can sequester more CO_2, faster. But even this is too simple because part of the eventual sequestration involves chemistry and biology that depend on temperature, sunlight, animals activity, ocean currents and nutrients… so it is with all of the rates. They themselves might be — indeed, almost certainly are — functions of time!
Interpretation: We make CO_2 at some rate,
However, CO_2 is removed from the atmosphere by processes that literally have a probability of removing a CO_2 molecule per unit time, given the presence of a molecule to remove. They are all proportional to the concentration. If I double the concentration, I present twice as many molecules per second to the e.g. surface of the sea as candidates for adsorption or to the stoma of a leaf as candidates for respiration and conversion into cellulose or sugar or whatever. They are all independent; if some particular wave removes a molecule of CO_2 at 11:37 today, a leaf on a tree in my back yard doesn’t know about it. The removed CO_2 has no label, and the jostling of molecules in the well-mixed warm air guarantees that one cannot even meaningfully deplete the local concentration of CO_2 by this sort of process, so both remain proportional to the same total concentration
The nice thing about this is that it is a well-known linear first order inhomogeneous ordinary differential equation, and can be directly integrated just as simply as the previous one. The result is (non-calculus people can take my word for it):
where
This simple linear response model shows precisely how one expects the eventual atmospheric concentration of CO_2 to saturate as long as saturation is achieved at low net concentrations of the total atmosphere such that the total relative fractions of N_2 and O_2 were the same and are still much larger than CO_2 taken together. And it is a well known and easily understood one. Equilibrium is
Now as models go this one sucks — it is arguably TOO simple, but it is easy to fix. For example, the ODE is the same if one has a source rate that isn’t constant but is itself a function of time —
A bigger problem is that for the ocean
However, even if we put far more complex differential forms into this ODE, it remains pretty easy to solve without making any sort of formal approximation or decomposition. Matlab lets one program it in and solve it in a matter of minutes, and graph or otherwise present the results at the same time. Writing a parametric form and then fitting the parameters to past data in hope of predicting the future is also possible, although it is a bit dicey as soon as you have a handful of nonlinear parameters because then one is trying to optimize a possibly non-monotonic function on a multidimensional manifold, which is the literal definition of “complex systems” in the Santa Fe institute sense.
Unless you know what you are doing — and few people do — you are likely to start the optimization process out with some set of assumptions and optimize with e.g. a gradient search to find an optimum that “confirms” those assumptions, ignoring the fact that a far better fit is available but is nowhere particularly near your initial guess. In a rough landscape, there might always be local maxima near at hand to get trapped on, and even finding the right neighborhood of the optimal fit can be challenging. Imagine an ant searching for the highest point on the surface of the earth by going up from wherever you drop them. Nearly every point they get dropped on will take them to the top of a grain of sand or a small hill. Only a teensy fraction of the Earth’s surface is mountains, a smaller one big mountains, a handful of mountains the highest peaks in a range, and one range the right range, one mountain the right mountain, one small area of slopes the right SLOPE, ones, that go straight on up to the top without being trapped.
There may be some way to formally justify the Bern model. Offhand I can’t see it — integrating E(t) is fine, but integrating it while multiplying it by that bizarre sum of exponential terms? It doesn’t even look like it has the right asymptotic form, implicit saturation. In other words, although it uses time constants, the time constants aren’t the time constants of a presumed exponential sequestration process that removes CO_2 at a rate proportional to its concentration, they are more like “relaxation times”. The expression looks like an unbounded integral growth in concentration modulated by temporal relaxation times that have nothing to do with concentration but rather describe something else entirely.
Just what is an interesting question, but this is not a sensible sequestration model, which is necessarily at least proportional to concentration. At higher concentrations, plants take up more of it and grow faster. The ocean absorbs more of it (at constant temperature) because more molecules hit the surface per unit time. More of the CO_2 that makes it into the ocean is taken up by algae and bound up, eventually to rain down onto the sea floor, removed from the game for a few hundred million years.
I cannot believe that there isn’t anybody out there in climate-ville that hasn’t worked all this out in a believable model of some sort, something that is a perturbation of the first order linear model I wrote out above. If not, shame on them.
rgb
In relation to sources and sinks, can Willis, or anyone else explain this image of global CO2 concentrations ?
Why don’t warm tropical oceans give high CO2 ?
Why is there a band of high CO2 around the 35S ?
How is the distribution over Africa and S America explained ?
Why does Antarctic ice appear to be such a strong absorber in parts and why such strong striation?
http://www.seos-project.eu/modules/world-of-images/world-of-images-c01-p05.html
Seems this is a mental image issue – like putting the cart in front of the mule. It isn’t the carbon dioxide in the atmosphere that controls the timing. You can buy paints, some are fast-drying. Some dry slowly. Eventually, they all dry. CO2 sinks (hundreds, not 6 or 3) do their thing in their own way, so other things equal (constant CO2 levels), a very slow process might take 371.3 years to sequester a unit of gas, a very fast process might take 1.33 years to do the same. Thus, the numbers (wherever they came from) might have meaning – just not that described. So, one should really call it the Bourne Model insofar as the identity of the processes is a mystery and no one is sure just what is going on.
Well, whatever the Bern model does, it must be correct. After all, once you match the results of 9 GCM models (except one outlier), you have matched them all. CO2 sensitivity was assumed to be 2.5 K to 4.5 K in the models.
” After 80 years, the increase in global average surface temperature is 1.6 K and 2.4 K, respectively. This compares well with the results of nine A/OGCMs (excluding one outlier) which are in the range of 1.5 to 2.7 K”
KR says:
May 6, 2012 at 12:58 pm
Say what?
Look, KR, they used the box model to calculate the IRFs. The part you seem to be missing is that once they have used the box model to calculate the IRFs, then
claiming that the IRFs represent actual physical processes.
Next, you say the IRFs are the “resulting partial absorptions of various climate compartments”. This is similar to what the citation says, that they “reflect the time scales of different sinks”. Both of you are claiming that the IRFs have reflect real-world processes… so could you explain just how it is that the atmosphere is divided into “various climate compartments”, some of which decay quickly and some slowly?
You say ” the results of the Bern model were offered as an available computational tool for further work” … I understand that. What I don’t understand is the physical basis for what they are claiming, which is that e.g. 13% of the airborne CO2 hangs around with an e-folding time of 371.6 years, but is not touched during that time by any of the other sequestration mechanisms.
Perhaps you could explain to us how that works, that certain CO2 molecules are sequestered but some are immune to sequestration for hundreds of years.
I also note that there are no less than four different (and in fact very different) IFTs that have been used by the UN … hardly what one would expect if they actually are the “resulting partial absorptions of various climate compartments”. How are we supposed to pick one of them?
The SAR used five IRFs reflecting, according to you, five “climate compartments”, while the TAR used only three. What happened to two of the “climate compartments” between the SAR and the TAR … the IRFs just disappeared, what happened to the “climate compartments” that you claim they represent? Did they go out of business?
More to the point, why should we believe the model at all? The conclusion of your citation says:
The model calculates CFC-II well, but it misses on CO2. In other words, the model doesn’t work very well, and even the authors don’t understand why their model doesn’t work … not encouraging.
w.
Fascinating stuff, cheers Willis. So instead of trying to capture CO2 underground why not just dump into a high speed sink?
KR says:
May 6, 2012 at 1:19 pm
Both you and the citation I gave have said that the IRFs represent actual physical processes. You said the IRFs represent the “ resulting partial absorptions of various climate compartments”. My citation says that the IRFs “ reflect the time scales of different sinks”
Now you want to claim that this is just a way to approximate the model. Come back when you make up your mind and can explain why the citation is wrong.
w.
PS—See also Robert Brown’s comments above (rgbatduke) …
So … does anyone understand how 13% of the atmospheric CO2 is supposed to hang around for 371.6 years without being sequestered by the faster sinks?
Perhaps you need to look at this the other way around. We have often heard about the 800 year time lag between high temperatures and CO2 concentrations. Is it a coincidence that 371.6 is about half of 800? Is it possible that if the CO2 concentration were to suddenly drop, then various processes would act to raise the CO2? And that the part of the CO2 that is in the deep oceans may take 371.6 years to reach the atmosphere and add 13% to the overall increase in CO2 concentration?
RGB at duke says:
May 6, 2012 at 1:21 pm
…Please try to fix it for me if it looks bizarre.
Here is something bizarre that no one can do much about, let alone fix it.
http://www.vukcevic.talktalk.net/TMC.htm
Dr Burns says:
May 6, 2012 at 1:30 pm
Why does Antarctic ice appear to be such a strong absorber…
Antarctic is simply bizarre, see the link above
They may be discussing on-rate constants only. The concept might be equilibrium and saturation in different reservoirs. The on-rate for a given reservoir is the atmospheric concentration times the rate constant. The off rate is the reservoir concentration times the off-rate constant. If the on-rate equals the off-rate, then the reservoir is at equilibrium. No net uptake would occur.
rate(on) = k(on) * [conc(air)]
rate(of) = k(off) * [conc(res)]
No net uptake occurs when rate(on) = rate(off), that’s equilibrium.
In the real case, you also have a loss rate for CO2 via other routes, e.g. diatom skeletons in the ocean, and leaves or grasses on land. Which means the reservoirs don’t necessarily saturate, and they can continue to take up more CO2. In some cases, the on-rate may depend on the rate of dropout loss in that reservoir since near equilibrium limits the net uptake.
d(CO2 air)/dt = k(off) * [CO2(res)] – k(on) * [CO2(air)]
d(CO2 res)/dt = k(on) * [CO2(air)] – k(off) * [CO2(res)] – k(dropout) * [CO2(res)]
If the reservoir is in dynamic equilibrium, and to a reasonable approximation the reservoir concentration doesn’t change. Then
-k(on) * [CO2(air)] – k(off) * [CO2(res)] = k(dropout) * [CO2(res)] ,
which means both sides of the equation are constant, since CO2(res) doesn’t change.
Since the dropout material doesn’t readily cycle back to the air via the reservoir, it only makes sense when the dropout material can be returned to the atmosphere via another route, e.g. biological digestion, fire, volcanoes, or burning fossil fuel (coal). Returning the dropout material to the atmosphere creates the Carbon cycle we think about.
Partitions in the atmosphere itself make no sense, unless you are silly enough to count flying birds (%P) <- that's an emoticon.
From 1976 to 1997, atmospheric 14CO2 was measured. The levels spiked after due to atmospheric testing of nuclear weapons that ended in the 1960s. These data show the 14CO2 half-life is about 11 years (see raw data here: http://cdiac.ornl.gov/trends/co2/cent-scha.html). In the first approximation, uptake mechanisms won't know the difference between carbon isotopes. It is quite safe to say half of the CO2 in the atmosphere is turned over in about 11.4 years. Five half-lives is about 57 years. That means only about 3% of the CO2 present in the atmosphere 57 years ago is still in the air.
It seems we have an idea of what the dropout rate for CO2 must be, and thus what the replenishment rate must be to keep the atmosphere in roughly steady state. In this simple model, a sudden change in atmospheric CO2 concentration could shift the equilibrium concentration in the reservoirs, and then establish a new constant uptake rate somewhat higher than the old one. If you think about it, perhaps that does make some sense in some cases, such as faster plant growth, or more alkalinty in the ocean (sorry catastrophists, biological action converts carbonic acid to bicarbonate, e.g. by nitrogen fixation).
Yes, models can be misused. It's up to you to decide when they are appropriate.
http://www.seos-project.eu/modules/world-of-images/world-of-images-c01-p05.html
That’s July, two months after annual peak.
Why don’t warm tropical oceans give high CO2 ?
Maybe they do, but you can’t evaluate “vertical” fluxes only on the basis of concentrations in a month (average). The horizontal transport of CO2 in the atmosphere is spatialy and temporarily very dynamic (sesonal). It could also be rain (CO2 scrubbing) in the tropics…
Why is there a band of high CO2 around the 35S ?
High sommer in NH?
How is the distribution over Africa and S America explained ?
Why does Antarctic ice appear to be such a strong absorber in parts and why such strong striation?
Others should speculate. Seasons, moisture, snow, sst, surface altitude, energy budget, mass budget…
Since you like differential equations…
Start with three variables A, B, and C. The volume of flow from A to B is k_AB (A-B), and the volume of flow from A to C is k_AC (A-C).
So
dA/dt = k_AB (B-A) + k_AC (C-A) + E
dB/dt = k_AB (A-B)
dC/dt = k_AC (A-C)
where E is the rate of emission.
Treat L = (A, B, C) as a vector, ignore E for the moment to get dL/dt = ML where M is a matrix of constants. Diagonalise the matrix to get two independent differential equations (the rows of M are not linearly independent), each giving a separate exponential decay with a different time constant. Transforming back to the original variables gives a sum of exponentials.
(I think the two time constants are -k_AB-k_AC +/- Sqrt(k_AB^2-k_AB k_AC +k_AC^2) but I did it quickly.)