Guest post by Robert G. Brown
Duke University Physics Department
The Problem
In 2003 a paper was published in Energy & Environment by Hans Jelbring that asserted that a gravitationally bound, adiabatically isolated shell of ideal gas would exhibit a thermodynamically stable adiabatic lapse rate. No plausible explanation was offered for this state being thermodynamically stable – indeed, the explanation involved a moving air parcel:
An adiabatically moving air parcel has no energy loss or gain to the surroundings. For example, when an air parcel ascends the temperature has to decrease because of internal energy exchange due to the work against the gravity field.
This argument was not unique to Jelbring (in spite of his assertion otherwise):
The theoretically deducible influence of gravity on GE has rarely been acknowledged by climate change scientists for unknown reasons.
The adiabatic lapse rate was and is a standard feature in nearly every textbook on physical climatology. It is equally well known there that it is a dynamical consequence of the atmosphere being an open system. Those same textbooks carefully demonstrate that there is no lapse rate in an ideal gas in a gravitational field in thermal equilibrium because, as is well known, thermal equilibrium is an isothermal state; nothing as simple as gravity can function like a “Maxwell’s Demon” to cause the spontaneous stable equilibrium separation of gas molecules into hotter and colder reservoirs.
Spontaneous separation of a reservoir of gas into stable sub-reservoirs at different temperatures violates the second law of thermodynamics. It is a direct, literal violation of the refrigerator statement of the second law of thermodynamics as it causes and maintains such a separation without the input of external work. As is usually the case, violation of the refrigeration statement allows heat engines to be constructed that do nothing but convert heat into work – violating the “no perfectly efficient heat engine” statement as well.
The proposed adiabatic thermal lapse rate in EEJ is:

where g is the gravitational acceleration (presumed approximately constant throughout the spherical shell) and cp is the heat capacity per kilogram of the particular “ideal” gas at constant pressure. The details of the arguments for an adiabatic lapse rate in open systems is unimportant, nor does it matter what cp is as long as it is not zero or infinity.
What matters is that EEJ asserts that ![]() in stable thermodynamic equilibrium.
in stable thermodynamic equilibrium.
The purpose of this short paper is to demonstrate that such a system is not, in fact, in thermal equilibrium and that the correct static equilibrium distribution of gas in the system is the usual isothermal distribution.
The Failure of Equilibrium

In figure 1 above, an adiabatically isolated column of an ideal gas is illustrated. According to EEJ, this gas spontaneously equilibrates into a state where the temperature at the bottom of the column Tb is strictly greater than the temperature Tt at the top of the column. The magnitude of the difference, and the mechanism proposed for this separation are irrelevant, save to note that the internal conductivity of the ideal gas is completely neglected. It is assumed that the only mechanism for achieving equilibrium is physical (adiabatic) mixing of the air, mixing that in some fundamental sense does not allow for the fact that even an ideal gas conducts heat.
Note well the implication of stability. If additional heat is added to or removed from this container, it will always distribute itself in such a way as to maintain the lapse rate, which is a constant independent of absolute temperature. If the distribution of energy in the container is changed, then gravity will cause a flow of heat that will return the distribution of energy to one with Tb > Tt . For an ideal gas in an adiabatic container in a gravitational field, one will always observe the gas in this state once equilibrium is established, and while the time required to achieve equilibrium is not given in EEJ, it is presumably commensurate with convective mixing times of ordinary gases within the container and hence not terribly long.
Now imagine that the bottom of the container and top of the container are connected with a solid conductive material, e.g. a silver wire (adiabatically insulated except where it is in good thermal contact with the gas at the top and bottom of the container) of length L . Such a wire admits the thermally driven conduction of heat according to Fourier’s Law:

where λ is the thermal conductivity of silver, A is the cross-sectional area of the wire, and ΔT=Tb–Tt . This is an empirical law, and in no way depends on whether or not the wire is oriented horizontally or vertically (although there is a small correction for the bends in the wire above if one actually solves the heat equation for the particular geometry – this correction is completely irrelevant to the argument, however).
As one can see in figure 2, there can be no question that heat will flow in this silver wire. Its two ends are maintained at different temperatures. It will therefore systematically transfer heat energy from the bottom of the air column to the top via thermal conduction through the silver as long as the temperature difference is maintained.

One now has a choice:
- If EEJ is correct, the heat added to the top will redistribute itself to maintain the adiabatic lapse rate. How rapidly it does so compared to the rate of heat flow through the silver is irrelevant. The inescapable point is that in order to do so, there has to be net heat transfer from the top of the gas column to the bottom whenever the temperature of the top and bottom deviate from the adiabatic lapse rate if it is indeed a thermal equilibrium state.
- Otherwise, heat will flow from the bottom to the top until they are at the same temperature. At this point the top and the bottom are indeed in thermal equilibrium.
It is hopefully clear that the first of these statements is impossible. Heat will flow in this system forever; it will never reach thermal equilibrium. Thermal equilibrium for the silver no longer means the same thing as thermal equilibrium for the gas – heat only fails to flow in the silver when it is isothermal, but heat only fails to flow in the gas when it exhibits an adiabatic lapse in temperature that leaves it explicitly not isothermal. The combined system can literally never reach thermal equilibrium.
Of course this is nonsense. Any such system would quickly reach thermal equilibrium – one where the top and bottom of the gas are at an equal temperature. Nor does one require a silver wire to accomplish this. The gas is perfectly capable of conducting heat from the bottom of the container to the top all by itself!
One is then left with an uncomfortable picture of the gas moving constantly – heat must be adiabatically convected downward to the bottom of the container in figure 1 in ongoing opposition to the upward directed flow of heat due to the fact that Fourier’s Law applies to the ideal gas in such a way that equilibrium is never reached!
Of course, this will not happen. The gas in the container will quickly reach equilibrium. What will that equilibrium look like? The answer is contained in almost any introductory physics textbook. Take an ideal gas in thermal equilibrium:
![]()
where N is the number of molecules in the volume V, k is Boltzmann’s constant, and T is the temperature in degrees Kelvin. n is the number of moles of gas in question and R is the ideal gas constant. If we assume a constant temperature in the adiabatically isolated container, one gets the following formula for the density of an ideal gas:

where M is the molar mass, the number of kilograms of the gas per mole.
The formula for that describes the static equilibrium of a fluid is unchanged by the compressibility (or lack thereof) of the fluid – for the fluid to be in force balance the variation of the pressure must be:
(so that the pressure decreases with height, assuming a non-negative density). If we multiply both sides by dz and integrate, now we get:

Exponentiating both sides of this expression, we get the usual exponential isothermal lapse in the pressure, and by extension the density:
![]()
where P0 is the pressure at z=0 (the bottom of the container).
This describes a gas that is manifestly:
- In static force equilibrium. There is no bulk transport of the gas as buoyancy and gravity are in perfect balance throughout.
- In thermal equilibrium. There is no thermal gradient in the gas to drive the conduction of heat.
If this system is perturbed away from equilibrium, it will quickly return to this combination of static and thermal equilibrium, as both are stable. Even in the case of a gas with an adiabatic lapse rate (e.g. the atmosphere) remarkably small deviations are observed from the predicted P(z) one gets treating the atmosphere as an ideal gas. An adiabatically isolated gas initially prepared in a state with an adiabatic lapse rate will thermally equilibrate due to the internal conduction of heat within the gas by all mechanisms and relax to precisely this state.
Conclusion
As we can see, it is an introductory physics textbook exercise to demonstrate that an adiabatically isolated column of gas in a gravitational field cannot have a thermal gradient maintained by gravity. The same can readily be demonstrated by correctly using thermodynamics at a higher level or by using statistical mechanics, but it is not really necessary. The elementary argument already suffices to show violation of both the zeroth and second laws of thermodynamics by the assertion itself.
In nature, the dry adiabatic lapse rate of air in the atmosphere is maintained because the system is differentially heated from below causing parcels of air to constantly move up and down. Reverse that to a cooling, like those observed during the winter in the air above Antarctica, and the lapse rate readily inverts. Follow the air column up above the troposphere and the lapse rate fails to be observed in the stratosphere, precisely where vertical convection stops dominating heat transport. The EEJ assertion, that the dry adiabatic lapse rate alone explains the bulk of so-called “greenhouse warming” of the atmosphere as a stable feature of a bulk equilibrium gas, is incorrect.
“The real question is sensitivity.”
Yes indeed it is but the lapse rate issue goes to the heart of it.
If the lapse rate and thus the surface temperature is set by pressure and solar input alone then the effect of GHGs is zero.
GHGs do introduce more radiative and conductive energy around themselves by virtue of their thermal characteristics so, if there is more energy in the air but the surface temperature fails to change then something else has to give.
I propose a miniscule change in the surface air pressure distribution instead.
That would simply change the rate of energy flow through the system from surface to space and redistribute the energy at the surface as necessary for no global change in surface temperature at all.
“Heat is energy and hence by Einstein mass.”
I hate to even bring this up, but I am rather sure that gravitational red-shifting will indeed have a theoretical affect on things (but I don’t think in the way imagined by the earlier poster). A photon will be red-shifted when it rises from the surface. It would be a straightforward task to estimate the change in wavelength and hence the change in “temperature” for thermal IR photons arriving high above the earth from the surface. However, typically such effects are only noticeable very close to very massive objects. I am sure that the “relativistic lapse rate” would be microkelvins at most, and hence not important in this discussion.
But, hey, if anyone wants to calculate the actual “relativistic lapse rate” and its effects — go for it. As a warm-up, I would suggest calculating the time correction for GPS satellites to make sure you know enough to get started.
Ged says:
January 24, 2012 at 2:10 pm
@Willis,
Gravity doesn’t make the air warmer at the surface, it makes the air -colder- above the surface.
____________________________________________________
Bingo!
Ged says:
January 24, 2012 at 2:10 pm
“Gravity doesn’t make the air warmer at the surface, it makes the air -colder- above the surface.”
Yes. And if you remove the source of heat the column will cool and as it cools it shrinks and as it shrinks the molecules fall toward the surface and as they fall they gain back the kinetic energy they lost in making the ascent. If the temperature of the column drops enough the gas turns into an incompressible liquid or solid, completely collapses to the surface, and the gravity induced gradients are history.
@Willy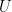 for an ideal gas is
for an ideal gas is
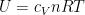
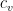 is the specific heat at constant volume,
is the specific heat at constant volume, 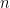 is the number of moles of the gas,
is the number of moles of the gas, 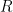 is the gas constant, and
is the gas constant, and 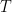 is the temperature. None of these quantities depend on volume or density.
is the temperature. None of these quantities depend on volume or density.
Your statement that “the internal energy of an ideal gas is also directly proportional to density” is incorrect.
Internal energy, a term with a very specific meaning in thermodynamics, is proportional only to temperature for an ideal gas. For starters, see http://en.wikipedia.org/wiki/Ideal_gas , and in particular the section entitled “Classical thermodynamic ideal gas” where it states that the internal energy
where
Willis at 11.54am wrote:
Excellent insight, Wayne. That is exactly what happens. In an isothermal column of air, individual molecules at high altitude have more energy because of gravity. But for exactly that same reason, there are fewer molecules at high altitude. As a result, and as we would expect, in the isothermal condition the energy is spread out evenly through space (equal energy per volume) rather than equal energy per molecule as Hans Jelbring and Mr. Verne assert.
Although I agree with Dr Brown, I disagree with what Willis has said here. Consider a cubic metre of soil and a cubic metre of air above it, although both are at the same temperature, they certainly have different amounts of enegy due to differing densities.
equilibrate the total energy
This is a major misunderstanding. Thermal equilibrium does not equate the total energy. Read the equipartition theorem. Open a standard introductory physics textbook. Learn what temperature is. Then return.
Besides — and I’m going to make this a standard answer for all off topic replies. The presentation above challenges you to do just one thing. Tell me whether or not the system in figure 2 permits energy to flow in a circle forever. If you answer “no, of course not” you are quite right, and you have conceded that thermal equilibrium is isothermal, because the silver wire is just a proxy for thermal conductivity in the air itself that makes it clear why not (since detailed balance computations get confused when you add an utterly irrelevant process that you dream up involving gravity to them and then try to do them in your head without the faintest idea of how statistical mechanics actually works). If you answer yes, I’ve got this bridge in Brooklyn you might want to look at, right after I convince you to invest in the machine we can build that will turn heat into energy at the rate of 100%, because I could stick a heat engine into the thermal pathway of the silver conduction and the resulting system would convert 100% of any energy added to the fixed-lapse air on the left into work.
The choice is yours, of course.
rgb
My head hurts. Is someone keeping score? If so, who is wining, the For team or the Against team? Confused minds need to know.
@Willy
Note that I am *not* arguing that there *isn’t* a variation of density [or pressure] with height. It’s simply that once one knows the internal energy of a gas, one also knows its temperature, irrespective of either the density or pressure. Of course, the profiles of both density and pressure must still satisfy the ideal gas law, and as you intuitively expect both decrease with height [as does the temperature].
2) The silver wire will transport heat from warmer region to the cooler region, but in so doing it short circuits the transport of heat by convection. So with the wire present, convection will be less, but the net transport of heat will remain the same.
It won’t short circuit convection — in stable equilibrium there is no convection. Convection itself is a kind of heat engine driven by temperature differences that transports heat (on average) from a hot reservoir to a cold one. In stable equilibrium nothing moves, because there is always dissipation associated with movement that will slow it down, right down to the extreme quantum regime. I assume you aren’t talking about superfluid circulation and calling it “convection”.
There is no input energy, also, so there cannot be net transport of heat. That’s the bit about “violating the second law of thermodynamics” in spades. A system with interminable flow of heat in a circle is a textbook violation of the second law, and it permits one to design any number of perpetual motion machines of the second kind.
rgb
I thought metals had free electrons and electrons were matter and gravity accelerates all matter and metals conduct heat so well due to the free electrons and …. and how again is this proposed perpetual machine supposed to refute a lapse rate? Seems lifting the electrons against gravity in the metal bar from the warm to the cool would cancel if the gradient became -0.0098C/m. The only real thermal motion it seems would be with an isothermal air column, but that would just run, backwards to the arrows on the diagram, until all relevant potential energy was minimized. I think that point would be called the DALR.
http://en.wikipedia.org/wiki/Energy_minimization
We sure could use a proper experiment. I have already designed one that should work fine if anyone should ever want to consider doing one. Basically Graeff’s only designed correct this time with many sensors, multiple layers of insulation, large, and with air. Why he chose a liquid I’ll never understand.
“In the modern world the stupid are cocksure while the intelligent are full of doubt.”
~ Bertrand Russell
Clearly a non-isothermal dry adiabatic column isn’t a minimum energy state (as energy can be extracted, imagining an efficient thermal conduction system between top and bottom layers.
Convection requires that some parcel of gas is already at an elevated energy state (or it wouldn’t be rising *due* to convection), and thus convection doesn’t violate conservation or 2nd law. The adiabatic column is (ignoring radiation) the minimum energy state with differentially heated (cool top, hot bottom) boundary conditions.
I don’t have a clear picture how a heat engine connecting the top and bottom would look in T-S space, or what the minimum energy solution would look like were one to take an established dry adiabatic column, insulate top and bottom and run a heat engine within the column. It seems like it would have to go to an isothermal state as minimum energy
Willis, the short answer is “Yes”, the medium answer is “IMHO Robert Brown basically has got it right”, and the long answer is “The logical next step is discuss whether day-versus-night temperature swings alone — in the absence of GHG effects — suffice to mix the Earth’s atmosphere.”
Because that claim —foreseeably (and strictly IMHO) — is going to emerge as the primary fallback position of GHE skeptics.
I see two assumptions above:
1. It does not matter what the density of the gas is. It will equally conduct heat at the bottom into silver wire, as the wire will be able to conduct its heat into the gas at the top, even though the density at the bottom and top is different, due to the gravitational effect on the gas.
2. The cross-section of the wire will stay the same, which means the ability of the wire to conduct the heat, which depends on its cross-section, is the same at the bottom and top.
The gravitational field will actually pull down a considerable part of the mass to the bottom, making it far wider at the bottom then the top (depending on the length of the wire and its tensile strength), deforming it more into a tear drop shape.
With your setup you may be able to change the lapse rate, but I doubt that you achieve an isothermal state in this way.
It does not matter what the density of the gas is, or how good the contact of the silver with the gas is (as long as there is thermal contact, or how thick the wire is. The point is that if any heat enters at the top ever and the system spontaneously restores the lapse rate (which is constant, recall, for a container of fixed size independent of gas density so we can make the gas nice and thick with great thermal contact with the silver) then you’ve violated the second law, because any thermal pathway between the bottom and the top will deliver heat from the bottom to the top. All I’ve done with the wire is show you a pathway that is clearly completely independent of the supposed lapse rate in the gas, one that will conduct heat in any direction without prejudice, so that you can see why a lapse rate is impossible. If heat to the top goes back to the bottom because of “gravity”, and there is a pathway to the top that must conduct heat in the direction of a thermal gradient (the wire) you’re done. The system will circulate heat indefinitely.
But no system can circulate heat indefinitely, it’s absurd.
So the message is, stop trying to do statistical mechanics in your head without understanding it. Stick to thermodynamics. It is simpler, safer, and you can’t make mistakes with it as long as you remember TANSTAAFL. There ain’t no such thing as a free lunch. A nonzero thermal lapse is a Maxwell’s Demon working the free lunch counter for the suckers.
Here’s the deal, really. Every book I’ve ever read on statistical mechanics or thermodynamics is wrong — and you can safely assume I’ve read a few, since I did numerical simulations of both static and dynamic critical phenomena that actually were published in places like Physical Review, with referees and everything — or figure 2 above makes it clear that there is no possibility that figure 1 is correct. That is, assuming that you can’t believe the actual words of the second law that tell you that you can’t take a system and create a permanent thermal gradient in it without doing work to maintain it, because heat will flow from the cold side to the hot side to neutralize temperature differences otherwise.
That’s why you have to pay to refrigerate or air condition your house. It is why you can’t build a perfectly efficient heat engine. Tanstaafl, man.
rgb
I find the analysis quite reasonable – but it is so idealized as to be useless. A more interesting thought experiment has a spherical planet heated by a remote star, rotating on an axis roughly normal to the line to the star, with an atmosphere of non-greenhouse gases. The equator would be warmer than the poles, so there would be Hadley-type circulation that would cool the equator and warm the poles. Would there then be a vertical thermal gradient? I think there would be, but I’m sure someone would like to argue to the contrary.
Not at all. It is merely specific. I am specifically proving that EEJ, a specific paper written by Jelbring and published in a journal (God help the referees, absent that day on vacation or something), violates the zeroth but especially the second law of thermodynamics when it asserts that there will be a thermal lapse rate in an adiabatically isolated column of ideal gas in thermal equilibrium in a gravitational field.
No, there won’t.
Is it stupid to have to prove this? Sure, given that nearly any introductory physics textbook — I’m not talking about thermodynamics text, just things like Tipler and Mosca, or Halliday, Resnick and Walker — teach enough thermodynamics for one to be able to see that the spontaneous appearance of a stable thermal gradient in any system is impossible, because it is a direct violation of the second law, and indirectly the first, which more or less says that equilibrium is isothermal (in order to permit the definition of thermometry in the first place). If thermometers “work” to measure temperature, equilibrium is isothermal. Period.
rgb
So your wonderful assertion, is that the radiative forcing of Co2, occur after its entry into the thermostats of the tropopause, and that extra radiative forcing, causes that missing hot spot, increasing the temperature back through the stratosphere and down again through the thermostat of the tropopause.
Been there, done that.
No, my “wonderful assertion” is that EEJ, Jelbring’s paper, is obviously incorrect because a stable, isolated atmosphere cannot support a thermal gradient. There is no such thing, in other words, as “gravitational heating” for a system in static equilibrium, nor is there any such thing as “gravitational heating” for a system in dynamic equilibrium. There is such a thing as gravitational heating in a collapsing system, which is what raises the temperature of protostars to the ignition point and provides the heat outflow from brown dwarfs.
As the greenhouse effect is concerned, look at the IR spectrum from over the top of the atmosphere. I don’t care how you think heat gets to the top of the troposphere; the point is that one chunk of the outgoing spectrum observed from satellites comes from a gas that radiates in the CO_2 band that happens to be at top of troposphere temperatures. The net radiation in the water window therefore has to be higher (than it would otherwise be) in order to keep the Earth in detailed balance (on average). End of story. I don’t give a rat’s ass where the extra radiation comes from, or how it gets there, it is there. You can see it. It is emitted/transmitted at the blackbody temperature of the ground give or take a bit.
Here, let’s use our fingers and toes. Total outflow in all frequencies has to be the same. Outflow in one band of frequencies is smaller because it comes from colder molecules. In order to keep total outflow the same, the energy radiated in the other frequencies has to:
a) Go up.
b) Go down
c) Remain the same.
That’s it. Come up with any mechanism you like for heat absorption and transportation, they’re all the same to me. Just don’t forget the incontrovertible experimental IR spectroscopy data and the finger and toe arithmetic involved.
rgb
Lets try a slightly different explanation of this closed static system. Of
course, a closed static thermodynamic system is, per see a thought experiment
that can not exist in reality.
We have a quantity of gas in a sealed cylinder and the cylinder is a closed
system thermodynamically. That means energy can not enter or leave the cylinder.
The cylinder is *not* in a gravitational field. Therefore, the mean pressure of
the gas is the same through out the tube and the temperature of the gas is
isothermal. This is thermodynamic equilibrium and entropy can not change.
Now, we switch on our gravitational field. The gas settles into a pressure
gradient with higher pressure at the bottom and lower pressure at the top of the
cylinder. Because of the gas laws, the gas at the bottom heats up and the gas
at the top is cooler. We will also assume that the gas does not absorb or emit
at the frequencies associated with these temps ( i.e. we can ignore radiative
transfer).
So convection and conduction set in and start to mix the gas. The total energy
in the gas is still constant. As the gas mixes energy is transferred from the
warmer gas to the cooler gas and the temperature differential reduces.
convection slows. This continues until T.E ( Thermal Equilibrium ) is reached.
At the new T.E. there is no more convection, the gas is isothermal and the
pressure gradient remains. The gravitational field is no longer performing work
on the gas so it can not add energy to the system no matter what potential it
exerts on any particular molecule.
Just imagine if this were not the case and convection carried on at T.E.
We could have a wind turbine driven by the convection current and connected to
outside the system. We would now be removing a continuous source of energy
from at system at T.E. Good luck with your patent applications ;-).
/ikh
Also, a constant temperature with altitude means that particles at the top of the atmosphere have more momentum than particles at the bottom.
It means nothing of the kind. Momentum (magnitude) is p = mv. The distribution of v at the top and the bottom is identical — the Maxwell-Boltzmann distribution.
Oh, do you mean more total momentum (in any given general direction) at the bottom than at the top because there are more particles? Sure — that’s why the pressure at the bottom is greater than the pressure at the top in a compressible fluid (where there are more molecules at the bottom than at the top). See “Kinetic Theory of Gases” at your friendly wikipedia outlet:
http://en.wikipedia.org/wiki/Kinetic_theory
rgb
So again you post modern hotrod physicists please answer the question. This is the real world it IS going to happen.
The stupid people here, who believe in the Laws of Thermodynamiics, know what will happen in the universe ruled by the Laws. But do the clever people, who know that the Laws are wrong, know how it happens in the Jelbring universe.
Now don’t be shy, it doesn’t really matter if you show that you are a complete idiot more than once, does it?
A gas giant planet, like Saturn, radiates more than twice the amount of radiation than it receives from the Sun. Presumably this little gravity induced energy engine is at work here according to you.
How do these planets ever die, if gravity is constantly maintaining a heat gradient in the atmosphere? The Sun will die and become a cold white dwarf with no solar wind to blow any atmosphere away.
What is going to kill these gas giants?
If gravity is constantly maintaining hotter gases at the bottom then convection will move gases around and therefore we seem to have an everlasting living planet.
What kills it and when, if Jelbring is correct?
Folks – Fellow interested posters, I am going to attempt to have a conversation with many of /y’all & all at once, skim for your handle if interested in engaging. All in time sort.
Joules Verne says at 1/24 7:52am:
“The device in figure 2 doesn’t work…the gas will collapse to the surface as a liquid before it gets to absolute zero and this will shut off further extraction of energy because the cold side of the thermocouple no longer has any cold gas to cool it.”
Good one. Joules groks this stuff. Now by Robert Brown definition, the gas is isothermal, meaning the gas can’t cool – the Perpetuum Mobile w/isothermal gas & perfect insulator is born in Fig. 2. A neat design for Willis’ dream machine. Patent pending.
Eilert sats at 1/24 7:54am:
“…I doubt that you achieve an isothermal state in this way.”
This actually adds to the discussion in a good way. A good thought experiment is to turn up the gravity field and see what would happen. The silver rod could react in the manner you describe. Interesting.
Genghis says at 8:06am:
“Take a single gas molecule and put it at the top of the tube. It has zero kinetic energy and zero temperature. Let it fall and just before it hits the bottom it will have a lot of kinetic energy and heat.”
Very good one. At the top, the molecule also has lotsa’ PE. At the bottom, it also has 0 PE. No isothermal molecule down the tube, it is isoenergetic (I learned a new term) – where there is no wire!
Robert Brown says at 8:14am:
“Well then, by all means go patent your perpetual motion machine of the second kind or explain heat flow in the second diagram, Joules.”
Robert – YOUR machine beat Joules. That is your machine design, it is still running. It will run tomorrow. It will run forever w/isothermal gas & you just have developed the perfect insulator patent.
Go for it. The Perpetuum Mobile design, fig. 2 in the top post is worth A LOT!!
And I’m ever so sorry, but in an ideal gas the temperature IS determined by the total energy. I cite the 1st Law consistent with oth law in one reservoir and the 2nd law constant entropy.
Robert continues:
“That’s an absurd idea, given that one can perform a gauge transformation — change the zero of the total energy — without changing any of the physics.”
The datum concept enables many good & proper science in many texts. Insert the thermometer at any datum to compute the invariant total energy value, say TE = C, & thereafter allow temperature and PE to vary with each other from that datum thermometer reading and you will be ok. Just place the thermometer at any different h. The temperature or mean kinetic energy of the molecules it will measure will be different: KE(h) = C – mgh.
Robert Brown says at 8:23am:
“Heat will definitely flow in the silver, right? It’s just a chunk of metal that’s an excellent conductor of heat.”
Right. This is why Robert Brown had to invent the perfect insulator design in fig. 2. How’s the patent pending process coming along Robert?
Robert Brown continues:
“You want to assert otherwise, you tell me what the equilibrium state is of figure 2.”
I have tried elsewhere. In fig. 2, let’s say white body starts with Twhite and gray body Tgray>Twhite. Cite 0th law that says heat will flow to a paler shade of gray, both bodies at single reservoir Tavg with silver tube! Or with moving jars, dipping bird, etc. At least while Robert’s perfect insulator patent is pending.
Robert Brown says at 8:28:
“No, I think you are generally quite right, and this agrees rather well with Caballero’s argument.”
Remember the Caballero text in the ref. given in Perpetuum Mobile thread makes an argument temperature is non-isothermal in a gravitation field section 2.3.
Caballero does write: 2.1 No gravity: “Note that pressure is due to only to the local properties of the gas and not to anything going on far away.”
2.3 w/gravity “Mean velocities (insert his defn. Temperature for mean velocities here) will be greater near the bottom of the box than near, the top: in other words, pressure decreases with height. We will now work out an equation giving the precise rate of decrease.”
Note Caballero defined “mean velocities” as temperature – I am just inserting for clarity, no meaning change. Thus temperature will be “greater near the bottom of the box”. This is not consistent with Robert Brown’s as yet unproven pronouncement that it is isothermal. Other than the fig. 2 w/perfect insulator inconsistent with 0th law driving an inconsistency with 1st Law.
Schodinger’s Cat says at 8:57am:
“Over time, I would expect the system to reach thermal equilibrium.”
Welcome back from the quantum world I see you survived after all. Yes, by gosh, the system in fig. 2 with real non-perfect insulator will reach thermal equilibrium over time by 0th law. The gas temperature will be non-isothermal.
Robert Brown says at 9:01am:
“No, it won’t have any “heat””
Right, but Genghis molecule will have the increased kinetic energy and per Caballero’s 2.3 ref. increased temperature. Rap Genghis on the knuckles for saying “heat”.
Robert Brown continues:
“But the basic point of my paper is that Jelbring is wrong not because of any possible microscopic description of a lapse rate. A lapse rate itself is wrong in thermal equilibrium, because figure 2 is very, very easy to understand.”
Robert Brown fig. 2 is wrong to have a perfect insulator which cannot exist by 0th Law. Jelbring cannot be proven wrong by fig. 2 which is not physical. At least until Robert Brown’s patent for the perfect insulator is granted.
Robert Brown says at 9:07am:
“…in figure 2 above. Which is violated — the heat equation in silver or your absurd assertion that gravity can stably sort out a gas into a hotter temperature and a colder one? One or the other.”
The heat equation in silver is not violated in fig. 2.
MDR says at 9:32am:
“This is an example of a system that is both in thermodynamic equilibrium and possesses a gradient in temperature.”
Good. This is consistent with 0th, 1st and 2nd thermo laws. Fig 2. is not & runs forever. Once fig. 2 has a non-perfect insulator admitted per 0th law, it will eventually be in equilibrium, non-isothermal gas, and not run forever.
Rober Brown says at 9:51am:
“Excuse me? I have no idea what you (Trick) could possibly be talking about.”
Believe me I grok this. You are smart though and the thermo master’s were right in the 0th, 1st, and 2nd law development. They have stood the test of time right up to fig. 2.
Robert Brown continues:
“heat will flow in the wire from the bottom to the top.”
Yes. Cite 0th Law.
“The point is that heat will flow in this system forever…”
Yes, fig. 2 is a Perpetuum Mobile. To win the patent for it though Robert Brown is going to first have to win the patent for the perfect insulator. If Robert cannot patent a perfect insulator, cite the 0th and 1st law tell us the system will stop flowing heat when it reaches thermal equilibrium and with 1st law non-isothermal gas (see Caballero 2.3 ref. telling us gravity results in non-isothermal gas column) thru the non-perfect conductor, this is less than forever but can be quite long because there are some quite close to perfect insulators.
“The real point is that you don’t need the silver wire to make this argument…..Gravity does no work in this problem, not in steady state.”
I whole heartedly agree. Wire is sort of useful to understand the concepts of the thermo laws. The ideal gas molecules in fig 2 lose just as much energy going up against gravity as energy they gain coming back down. Fig. 2 modified with real non-perfect rl insulator is ideal isentropic reversible process with non-isothermal gas (Per Caballero 2.3 – check it out).
“So what makes the heat go round and round?”
The perfect insulator. The perfect insulator is non 0th thermo law compliant. Your patent still pending?
“Of course. It doesn’t.”
Heat does stop flowing round and round with a patentable real non-perfect insulator. The gas will be non-isothermal per Caballero ref. 2.3.
Robert Brown says at 9:58am:
“Are you crazy?”
No Joules Verne is eminently sane, Robert Brown misses the other poster (Joules Verne) said the work extracted was not from the system but from the ideal gas column with the perfect insulator (patent pending).
Robert continues:
“In the real world, the system will evolve to an isothermal state precisely as I described it because it is in equilibrium.”
Not isothermal unless you think Caballero is crazy too (my view Caballero is not crazy), since his reference 2.3, disagrees with you. Caballero shows ideal gas column is non-isothermal in the presence of gravity. Caballero also shows ideal gas column is isothermal w/o gravity.
JKrob says at 10:24:
?In moving the heat from the bottom of the tube to the top is causing the lapse rate to become **more stable** – cool at the bottom with warm air above is an inversion which inhibits vertical mixing!! THAT is why the engine will not work as it is set up.”
A good .02 added viewpoint. Thanks for de-lurk.
D.J. Hawkins:
“Now I can construct a heat engine which extracts useful work based on the temperature gradient and gravity will continue to organize the air column forever and my heat engine will never run out of “fuel”? Really??”
Robert Brown will demand a royalty for using his patent pending perfect insulator. Other than that cost, yeah fig. 2 works for free.
Graeme W says at 11:50am:
“I don’t understand the theoretical derivation well enough…”
In the WUWT Perpetuum Mobile thread at the top post, there is a Rodrigo Caballero link cited by Robert Brown to the theory of ideal gas without and with gravity field. In 2.3 gravity is added to a non-isothermal gas column and this condition is theoretically justified – it is pretty simple derivation, I recommend it. Fig 2 perfect insulators needed only for a Perpetuum Mobile.
Robert Brown for some reason does not grok Caballero 2.3 even though he cites it as good ref.
Willis Eschenbach says at 11:54am:
“In an isothermal column of air, individual molecules at high altitude have more energy because of gravity.”
Bzzzt! Isothermal column of air is for no gravity case ref. Chapter 2.1 Caballero link in your Perpetuum Mobile post.
Bzzzzt! “…because of gravity”. Ok, with gravity Rodrigo Caballero (bless him) tells us in 2.3 same link the air column becomes non-isothermal.
Bzzzzt! Fail 1st law: individual molecules must have constant energy. Note the period. Willis “more energy” fails. More energy is not constant energy.
Willis tries to knock down Jelbring paper with these fails. Not possible to knock down Jelbring with these 3 arguments.
Willis Eschenbach says at 11:57am:
“It is the formal disproof of Jelbring’s theory. It is idealized by its very nature.”
By Willis use of “it”, my view Willis means top post where Willis summarizes his understanding of “it” at 11:54am. Willis at 11:54am fails any disproof on all 3 points of his points, see right above. Gosh, I hope Robert Brown groks Caballero & 1st law energy conservation quickly.
Willis Eschenbach at 12:09pm (? to another poster):
“Does heat flow forever or not?”
Heat flows forever in fig. 2. The reason is a grand master thermal law is broken, the easiest one, the zeroth law. There can be no perfect insulator. Body A placed in contact with body B will always eventually equilibrate temperature. This is so easy. Geez. If Robert Brown can patent a perfect insulator, I will change my view. Patent granted: Perpetuum Mobiles will be possible.
Until the patent is granted, heat will NOT flow forever in fig. 2 b/c there is no perfect insulator. Willis should grok this on his own (read Caballero slowly). Robert Brown is going to take longer I think, to reach a state of equilibrium grokness. LOL, I feel better my head cold is receding faster.
Willis Eschenbach at 12:09pm:
“Folks, a lot of you here don’t seem to get it.”
No kidding. Including Willis viz:
Will heat flow in the silver wire forever?
“Me, I say no, and I say Roberts thought experiment elegantly proves that the answer is no.”
For fig. 2 silver wire heat will flow forever like Robert Brown says it will in top post, his words:´ One is then left with an uncomfortable picture of the gas moving constantly.. At least until poster Joules Verne situation is reached and the gas cools & liquefies under a vacuum. That’s good thinking but beyond the intention of top post.
My view is the insulator in non-perfect to be 0th law compliant and in that cas heat will not flow forever and gas per Caballero 2.3 will be non-isothermal.
Willis Eschenbach at 1:04pm:
“I hate it when my perpetual motion machines are poorly designed …”
That fig. 2 is a WELL designed Perpetuum Mobile w/isothermal air & perfect insulator. Just gotta’ pay royalties for those: (patents pending).
– – – – – – –
Out of breath and w/blisters on my fingers I stopped at 1:04pm. I will happily rengage if more interested parties appear thereafter. Typo’s are possible in this post. Probable really.
Robert Brown and Willis: You will never convince some people. These same people are quite sure that the reason we don’t have machines capable of giving free energy forever is due to “poor engineering”, or its a conspiracy by Big Oil or Big Gubmint to fleece us all. If only the shackles of these evil doers were thrown off we could live a life of luxury forever, with energy too cheap to meter, made in our own basements be a nice perpetual motion machine – yours for $499 from your local hardware store. But of course, us incompetent engineers and physicists wouldn’t want that… line us up against wall now because clearly thats what we deserve.
/sarc
Great theory. Baloney, but very entertaining.
The reason the silver thingy won’t generate perpetual motion is that the exposed ends will assume whatever the air temp is at that altitude. A temperature difference of 1 degree will not move any heat in a silver rod 100 meters long.
adiabatically isolated column of gas in a gravitational field cannot have a thermal gradient maintained by gravity.
And yet we see this stratification in the atmospheres of Venus, Earth and Jupiter. In the real world, it’s hotter at the ground because the air is heated from the ground up and rising air columns draw this warmer air up… you know, the thermopiles birds ride up instead of having to beat their wings furiously. Imagine that, it’s colder on top of a mountain than at it’s base. It’s called the triumph of empirical science over clever explanations that claim otherwise.
The details of the arguments for an adiabatic lapse rate in open systems is unimportant, nor does it matter what cp is as long as it is not zero or infinity.
What matters is that EEJ asserts that in stable thermodynamic equilibrium.
Which does not occur in the real world. That’s why we call it weather. Sorry, no sale. This is just as unacceptable as the nonsense about CO2, a trace gas having more effect than water vapor on the planet’s temperature.
I hasten to add that the lapse rate that does prevail at equilibrium is much smaller than that for which Jelbring contends, so Jelbring is still wrong.
If you want to bet against the laws of thermodynamics, I wouldn’t advise it.
I do not care about what generates the lapse rate. If the lapse rate is stable, so that heat delivered to the top redistributes to maintain a constant equilibrium temperature lapse between the top and the bottom — the sole case examined in the article above — then it violates the second law of thermodynamics.
Let me put it bluntly. If somebody presents a statistical mechanical computation that suggests that the second law is violated, I would knee jerk assume that the authors had made a terrible mistake unless and until proven otherwise, especially if I “could not understand” everything that they did.
Even then I would be doubtful.. To be honest, I would be doubtful if I did the work myself. I think that the paper you link has the right idea, and you will note that on other threads I propose precisely the same experiment. Show me, in other words. I’m a theorist, but I’m no fool. Experiments trump theory every time, but the Earth’s atmosphere does not exhibit a DALR:
* Uniformly. See “troposphere”, “stratosphere”, “thermosphere” etc. Why exactly doesn’t the DALR extend to the top of the exosphere again?
* Ubiquitously. See “thermal inversion”, or for extreme cases “thermal profile of the atmosphere above Antarctica in July” (there’s an IR spectrum in Caballero with the data you need). Hmmm, thermal inversion, with the upper troposphere hotter than the ground? Could that be, maybe, because the ground is cooler than the air instead of warmer? Regardless, it is nearly impossible to explain if there was an intrinsic DALR that didn’t depend on differential heating.
Nor is the atmosphere ever even crudely static. Air is always moving up, down, sideways. Even when it is “windless” down near the ground, there are damn few places on Earth where the air isn’t moving up and down and sideways from a kilometer on up, on any given day. So it isn’t really all that surprising that the air (a decent insulator and hence quasi-adiabatic a parcel at a time) has a DALR when it is differentially heated and moving all of the time, keeping it crudely “well-mixed”.
Thermal static equilibrium is something else. No mixing. Conduction matters — it can be slower, but there is plenty of time to reach equilibrium. That’s why the thermodynamic argument is so powerful — it is very difficult to explain how heat delivered to the top of any truly equilibrated air column would spontaneously redistribute to maintain a vertical lapse rate and not enable a heat loop and/or any number of PMM2Ks.
But show me the “high precision” experimental result, done with a dewar in a centrifuge filled with maybe Xenon gas at a G value such that there is sufficient pressure at the top of the vessel to justify the thermodynamic assumptions, with recording high-precision, carefully calibrated thermometers.
Just bear in mind that if it works, and there is a lapse rate, the second law itself is done. I’ll stick a thermocouple between the top and the bottom of said gas, and it will sit in there spinning forever.
rgb
Dr Brown is not correct with his explanation of Fig2
In a cylinder with gas at the usual DALR, all that his conducting wire will achieve is a infintesimally thin layer or hotter gas at the top plus an infintesimally thin layer of cooler gas at the bottom.
Both of these would reverse the lapse rate ( inversion) and thus no further heat can be exchanged without adding work to the cylinder (His statement that the system would reorganise itslf into an adiabatic column is wrong)
This situation would also apply to an attempt to use a themocouple to derive any work from the system. (Or any other heat engine)
It is thus clear that the adiabatic column cant be used as a perp -motion device so the proof that it breaks thermo laws has gone away.. The reverse holds true for an isothermal column where the bottom must be cooler than a surrounding DALR atmosphere with the opposite at the top– In this case heat can be removed at the top and added at the bottom. This would comprise a real-perp motion machine as the column would then reoganise itself into the isothermal state
Clearly it is the isothemal condition that breakes the 2nd Law
PS Much has been said about the need in the real atmosphere to maintain a DALR by pumping from below with energy from the Sun. In the real world the column is subject to all sorts of energy lossed and gains. The list is very large as Anthony has enumerated is is recent post– The water cycle is one of the most important
Most contributors seem to have ignored the fact that in the DALR cylinder all the heat losses and gains have been elininated and thus it takes an infintesimally small amount of energy to lift a parcel of air fron the bottom to the top hence an infintesimally small amount of energy will maintain the DALR.
We just need to overcome conductive and radiative transfers which have already been assumed to be zero