Guest opinion: Dr. Tim Ball
The central theme of purveyors of environmental and climate deception is to take normal events and present them as abnormal. Here are examples of the most frequently used stories involving ice in Antarctica. A 2017 New York Times (NYT) story titled “Miles of Ice ColIapsing into the Sea.” Or one from an agency that should know better but is among the worst for alarmist’s misinformation about ice. Without any evidence whatsoever they title an article, “Antarctic Ice Shelf Disintegration Underscores a Warming World.” It begins with the false assumption that CO2 causes global warming and extrapolates that to the claim of increased disasters.
The recently released Fourth US National Climate Assessment is a broader example. The historical records show no increase in natural disasters, but they point to recent hurricanes as evidence when they are all within any natural variability. As I said many times all this is possible because most people, including those in the media and environmentalists who exploit the stories, don’t know about or ignore the fundamental components of the natural world.
Few know that ice forms and behaves in many different ways and causes a variety of effects in the atmosphere and on the land. It was lack of knowledge about sea ice formation and variation that allowed Al Gore to exploit naturally changing amounts of Arctic ice. Add false claims of declining polar bear populations to create a full-blown alarmist deception.
In 1768 William Wales and Joseph Dymond, appointed by the Astronomer Royal, Nevil Maskelyne, took a scientific expedition to Churchill, Manitoba to measure the Transit of Venus. They were charged to make extensive measurements and answer important scientific questions of the day. One of those was a question many can’t answer today. Is sea ice salt or fresh?
The first form of ice is created when water freezes. People assume that it occurs at 32°Fahrenheit, 0°Celsius, or 273.15 Kelvin. Freshwater density begins to increase at 4°C, and ice forms as fusion occurs at 0°C. However, saltwater fusion occurs around -2°C, but that varies with the level of salinity. This means polar sea water can cool as much as 6°C before ice forms. However, the Arctic Ocean has the lowest salinity of any of the oceans because of low evaporation and high inflows of freshwater from massive rivers, like the Mackenzie, Ob, Yenisei, and Lena.
Sea ice forms from seawater. It expands when it freezes, so the volume of ice is greater than the water from which it is formed. It floats with most of the ice, about 7/8th underwater, so the volumetric changes between when it forms, and melt has virtually no impact on sea level. It is difficult to draw any conclusions about variations in volumes of sea ice from year to year because we only have satellite data since 1980. In addition, we only recently learned the effect of wind on moving the ice around and changing the melt rates in different regions. All the evidence is that sea ice variations are well within any normal range. All the fear about polar bears surviving and the disaster of loss of ice are given lie by the existence of a fossilized White Spruce (Picea Glauca) of considerable dimension some 100 km north of the current tree line radiocarbon dated to 4949 ± 140 years. This empirical evidence requires a world some 3°C warmer than today, yet the polar bear survived.
Most precipitation in middle and high latitudes begins as snow and melts into rain before it reaches the surface. A variety of stages and forms, some of them unique, occur while this is happening. For example, water can occur in all three phases, ice (solid) water (liquid) and water vapor (gas) at the same temperature. Water can also exist as a liquid well below the freezing point, a condition known as super-cooled water.
If the freezing level is at or very close to the ground, the precipitation will remain as snow. If it continues to accumulate it will add a positive feedback by changing the albedo that lowers the ambient air temperature. Most people are familiar with the direct freezing of water, however, all other ice, which is the majority on the planet, occurs in glaciers and begins life as snow. The process of transformation from snow to ice begins when a portion of the annual snowfall survives the summer melt. This occurs in two ways; the temperature of a region declines, or the amount of snowfall increases. It is surprising how close a large part of the world is to the impact of a temperature decline. For example, in 1816 snow stayed in the forest across central and northern Canada through the entire summer. Ice covered Lake Winnipeg until July 20th. Some areas are affected more than others. As the temperature cools, snow accumulates and survives more easily on the poleward side of high-altitude valleys.
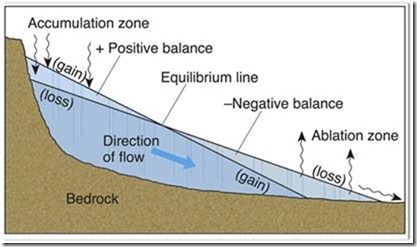
The diagram shows a simple cross-section through an Alpine glacier, Glaciologists distinguish these from continental glaciers like Greenland and Antarctica, but the formation and mechanisms are the same.
Snow accumulates in the zone above the Equilibrium Line. Some call it the permanent snow line. More snow falls than melts, so layers build up. The snow gradually changes to granules and the air is trapped in bubbles and eventually mostly expelled. For the air to become enclosed takes a long time, at least 70 years before the bubble is enclosed, which makes the claim that the layer in the ice represents a given year is nonsense. When snow accumulates to about 50 m, the ice layers begin to meld and become indistinguishable. At this point the ice changes from brittle to plastic, and from rigid to flowing. The “Direction of flow” arrow in the diagram illustrates the pattern.
Temperature affects the rate of melting in the Ablation zone, but overall the size and advance of the glacier is as much about changing snowfall in the accumulation zone, and that is true of all glaciers. With continental glaciers, especially the Antarctic, when the ice pushes out into the ocean it begins to float and create a unique ice feature known as Tabular ice shelves. They are at the center of most global warming alarmist stories like those in the first paragraph. When the glacier grows due to increased snowfall in the Accumulation zone, it moves downslope in “the Direction of Flow.” This occurs in the Antarctic and Arctic, but those in the Antarctic are much larger. The largest of these shelves in the world is the Ross Ice Shelf named after James Ross who wrote on February 5, 1775
“That there may be a Continent or large tract of land near the Pole, I will not deny,” “On the contrary I am of the opinion there is, and it is probable that we have seen a part of it. The excessive cold, the many islands and vast floats of ice all tend to prove that there must be land to the South.
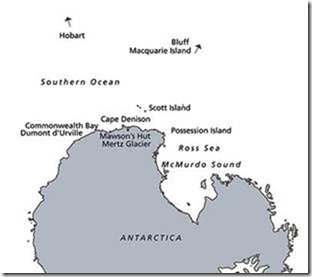
He is referring to the icebergs, large irregular blocks of ice that calve from glaciers all the time, but also the vast flat-topped ice islands that break off the Ice Shelf. These break away because of pressure from advancing ice behind, the up and down movement of the tides, the constant battering of large waves, and the extremely strong winds blowing out of the continent. These are called Katabatic winds and are the downslope flow from the height of Antarctica (3000 m in the center) and the density of the very cold air. At Cape Dennison on the edge of the continent, the mean wind velocity is 19.3 meters per second (43 mph) and an average for July (midwinter) is 24.5 m/s (55 mph). That pushes the ice away from the continent rapidly.
Proximity of Cape Dennison, “windiest place on earth” to Ross Shelf.
These floating ice islands are a navigational hazard south of 50° latitude. They also offer potential water supply. Australia, Saudi Arabia, and California all investigated towing these slabs, preferably carved out for easier towing. You then ground them along the shore, surround them with plastic curtains and pump the meltwater into onshore holding tanks. The California studies estimated a 50% melt in transit would still leave enough water for Los Angeles for a three-month supply. They made no mention of the ecologic damage as it melts in transit. What about the impact on local climate with a massive block of grounded ice and the fog that it would create? The French engineering company hired to look at the Saudi Arabia plan talked about wrapping the island in plastic to reduce the melt rate because of the higher air and water temperatures involved.
Meanwhile, the alarmists report these islands as a new phenomenon when they are perfectly natural and vary in size and frequency as a result of a growing Antarctic glacier, not a one that is melting.



“take normal events and present them as normal. ” huh?
MOD In the 1st line of text in article Please change the last word “normal” to “abnormal”
Pretty sure that is a typo. Should be “abnormal” me thinks. Which is exactly what they do.
Article needs to work on its “ab”s.
Has this post been peer-reviewed?
Curious George
You are doing peer review. That is, if you make any constructive contributions.
https://youtu.be/C9Pw0xX4DXI
Abby Normal
Typo in the first paragraph: “The central theme of purveyors of environmental and climate deception is to take normal events and present them as normal.”
I think that last “normal” should be “abnormal”.
There was a study that concluded that if all the ice in the Arctic melted the sea level rise would be only 20 mm (less than 1 inch).
of course that true … that ice has already displaced its water volume … ice cubes in a glass of water …
Not true for Greenland.
The discussion, I believe, is about sea ice, not land ice.
And how what’s the probability for ALL sea ice or even land ice to melt in its entirety? It approaches zero. All this talk about a thing that is so far outside the range of possibility has only one purpose: to sow angst.
Even if all of the Arctic sea ice were to melt by the end of Summer, it would be a very temporary condition, as once the Sun sets at the north pole and the circle of perpetual darkness gets wider for the next three months, it is gonna refreeze for sure, unless and until some mechanism transports a far larger amount of heat from the tropics up to the Arctic ocean and/or atmosphere than is now the case.
But imagine the calamity: No gigantic permanently frozen wasteland taking up a large portion of our planet!
Golly…all of the life that was snuffed out when the Earth went into an ice age might be able to re-establish itself!
Horrors and Heavens to Betsy!
Almost all of the of constant sea level rise over the last 250 or so years (well before any CO2 increase) since the end of the Little Ice Age has been due to thermal expansion so the cooling effect of this melting sea ice is a negative.
“All the fear about polar bears surviving and the disaster of loss of ice are given lie by the existence of a fossilized White Spruce (Picea Glauca) of considerable dimension some 100 km north of the current tree line radiocarbon dated to 4949 ± 140 years. This empirical evidence requires a world some 3°C warmer than today, ”
This corresponds to Greenland having lost 20 % of its ice mass over 3000 years from 8000 years ago to 5000 years ago.
Does that take into account a warmer ocean evaporating much more water into the air and hence increasing precipitation by a corresponding amount…including, one might surmise, the snow falling on an elevated ice sheet on Greenland?
Glacier mass is a balance between snow that falls and ice that melts as I am sure you are aware.
Perhaps what he was saying is that normal events are now touted as the “new normal” of catastrophic climate change
I tried at least twice to freeze oceanic salinity sea water for an introductory oceanography class. All students of ice should be required to attempt this experiment. Very instructive, not that I understood it. Pressure helped.
Perhaps consider, sir, that sea water is not lab-isolated, that sea water across our planet is not the same, varying with proximity to undersea eruptions, proximity to land and rivers, etc. Therefore, other components of sea water in addition to salinity affect ice formation.
I taught that too, conservative and non-conservative properties. Also showed pictures of Gulf of Mexico ice, mostly in lower salinities, 1989. Estuarine ice muddy, still surface froze at 10F, lots of heat capacity in water. Also taught the essential nature of field work, difficult for ice in the Gulf of Mexico. Experiments to be verified, modified.
I got my introduction to freezing of seawater the hard way:
As crew on a US Navy nuclear attack submarine back in the 1970s. We did a “polar ice run”, and set the all time record (to that date, in 1976) for length of continuous time spent under the ice – 42 days. Aside from worrying over what we’d do if we had a ship’s emergency while submerged under the ice cap, where near the pole the polynas suitably thin for an emergency surfacing are pretty few and far between, I had a very good introduction to the effects of salinity on the freezing temperature of water. I distinctly recall being impressed at logging submerged seawater inlet temperatures of 28 degrees F, concurrent with a visible coating of ice in the freshwater in the bilges.
Ditto – but not under the ice cap, just above Murmansk in a USN boomer in 1972… hard to shoot a Polaris through the ice cap!
Same here, on a nuke fast attack in the 80s and 90s… Engine Room Lower Level, Sea Water Bay; 28F intake sea water temperature, and freshwater ice in the bilges.
We melted the ice by dumping hot water on it from the condensate wells, then pumping the bilges to the wastewater tank with a Randolph pump. I’m not sure about the older fast attacks, but the 688 Los Angeles class subs had a “dry bilge” order, where we weren’t supposed to let water build up in the bilges from watch to watch. So we spent a lot of time melting ice.
Engine Room Lower Level was so cold all the time that the cooks stored their food there on long ops. Strange how you can get used to the cold temperatures… I never wore a jacket when I was the ERLL watch.
ERUP (Engine Room Upper Level) was the opposite… hot. On station, just idling along at ~1 knot, it’d be around 90F… at ahead full or flank it could reach 130F.
Our MFP seals leaked so much there was never a question of whether the bilges were dry, only how many hours would pass before they needed to be pumped back out again.
By the way, port side, ERLL just inboard of the MSW coolers aft of the condensate bay but out of the way of inconvenient sight-seers, there is a convenient waist-high drain funnel …..
Didn’t realize so many old submariners here … that’s cool! I served on a 637 class attack boat.
I was a reactor operator. Among other memories was watching the throttleman to my left in Maneuvering continuously on the sound powered phones with the guys up forward on the rudder and planes, maneuvering slowly through the shallow Bering Strait in early March with thick pack ice above, and just a handful of feet clearance to the ice above the sail, and a handful of feet clearance to the bottom below the keel.
Thank you for your time standing watch on the wall.
I have questions. There have been several recent studies on western Antarctic glaciers melting at faster (unprecedented?) rates. A NASA study says warm ocean water is the cause. This sounds like pressure relief at the ablation zone. Isn’t some pressure is being added above in the accumulation zone due to new snow? What is the percentage of flowing ice being discharged from the continent vs static ice away from the edges. One article said the total flow out could surpass the accumulation of the total continent. Seems hard to believe.
And did NASA explain how that “warm ocean water” managed to cause the melting of land-based glacial ice?
But now iffen it was western Antarctic sea ice that was melting faster, that would be OK
According to this https://sealevel.nasa.gov/news/98/wind-warm-water-revved-up-melting-antarctic-glaciers , La Nina pushes warm water south. But that is not my interest. What I am curious about is if glaciers flowing into the ocean from Antarctica can raise sea level. It seems that most of the ice on that continent is stationary and only ice at the edges flow. Other stuff I have read says our warmer climate carries more moisture to the continent and the glacial flows from the continent are discharging slightly less so there is a slow net gain. (Reportedly, sublimation accounts for 10% of the total loss so is part of the equation but constant on moving and stationary ice.) But with glaciers speeding up, that trend could reverse. Whether its warm water or volcanoes or Al Gore’s bow mounted radar gun, isn’t most of the ice not going anywhere? It seems obvious. The ice caps are melting is a major talking point with Al and the gang. Seems like this myth should go away like the polar bear scare.
Isn’t it convenient that NASA ignores the likelihood that volcanism is melting those glaciers from beneath, and melt water is flowing from the land to the sea. Right concept, wrong direction, maybe. Someone needs to do a study measuring the temperature gradient and salinities from the mouth of those rivers into the the sea.
Facts don’t matter to acolytes of the Church of Gorebull Warmening. It’s all about good intentions, so even when the results directly contravene their stated beliefs and desires, they just move the goalposts.
The “Gorebal” thing has been overused to the point of trite tiresomeness. Yes, he is a boring hypocritical pedant, but please criticize him intelligently (really easy to do) rather than slide down to his level.
Sorry Tim, but I stopped correcting anyones “boo boo’s” because it takes 2 hours for my comments to post. (I was naughty back when I was young (last year), so I seem to be on permanent “moderation”) …But I’m pretty sure that all here understood it as “abnormal”
You still are…but we all like you that way….
“You still are…but we all like you that way….”
Naughty or young ? LOL
In my case, not only understood it that way, but read it that way until someone mentioned it. I failed the editor’s test.
Okay, I had to scroll all the way back up. Whew! Somebody (as in ctm, I’m sure) DID fix it before I saw it. I hate missing things…
I thought density increased as water cooled, 4C marked the point of greatest density. VOLUME increases as water drops below 4C as molecules lose kinetic energy and start to align.
MarkW
Yup! That’s my recollection, too…….water is at maximum density at ~4 deg.C and is slightly less dense (i.e. has greater volume) when it freezes……………………which is why ice floats. Perhaps, you can clarify this point or edit accordingly, Tim? Thanks!
Tim typically doesn’t respond to comments so good luck. He also says:
“Sea ice forms from seawater. It expands when it freezes, so the volume of ice is greater than the water from which it is formed. It floats with most of the ice, about 7/8th underwater, so the volumetric changes between when it forms, and melt has virtually no impact on sea level.
Doesn’t bother to mention that the salt eventually gets expelled and that old seaice is fresh. If the seaice completely melts then there isn’t a change in sea level, however any subsequent warming of the seawater above the temperature in the presence of ice will cause a rise in sea level.
-4 is the temp of greatest density for fresh water, not for salty water.
Everything changes when you add salt.
We had a great discussion on this a few years ago, with some nifty graphs of the variances in the various properties of water as the salinity changes.
Sorry that should be plus 4 C for greatest density of fresh water.
Got ahead of my skis in my comment.
For seawater, it is much colder value for the temp of greatest density.
See comment and graph below.
The article and comments section I mentioned had a far ranging discussion of the entire very complex subject, and I posted a whole lot of physical chemistry graphs.
I think the original article was one by Willis E.
The saltiest seawater in the open ocean has a psu of 37 or so, and at this salinity, the temp of greatest density is somewhere around -4 C.
See this graph:
http://linkingweatherandclimate.com/ocean/figs/density2.png
Menicholas
Yes! It is a very complex subject and thanks for the graph…..much appreciated. Thanks also for the note about the earlier extensive discussion on this topic. I’ll have a look and see if I can find it since it’s always nice to learn things and gain a deeper understanding of how the world works.
Thanks again for taking the time to comment! Cheers!
Great chart! Can you also mark a line for the freezing point of water corresponding to each salinity line? BTW, can someone please define psu as used on that chart?
Red94Viper,
I meant to post the comment here, but it posted lower down my mistake.
Here is a link, with my reply to your questions (psu is parts per thousand, etc.):
https://wattsupwiththat.com/2018/12/03/the-difference-in-ice-types-causes-confusion-in-global-warming-story/#comment-2543488
Links to one of the charts posted which is not working in the original link, plus other versions of it.
This is freezing temp vs salinity of water:
http://www.fondriest.com/environmental-measurements/wp-content/uploads/2014/01/watertemp_salinity.jpg
http://mgh-images.s3.amazonaws.com/9780073376707/5925-5-2IATP1.png
http://www.fao.org/docrep/T0713E/T0713E0O.GIF
Great! Thanks. How does that vary with pressure?
Water is generally regarded as incompressible, at least at any pressures occurring naturally on the Earth or in the ocean, and the phase diagram of water shows a vertical line between the liquid and solid phases all the way up to about 10 mPa, where a bulge to the left shows that water will remain a liquid at a lower temp when it is under great pressure. This bulge peaks at a value listed as 209.9 mPa, and 251.165 K, or about -22C!
I do not know if the phase diagrams of salt water solutions similar to sea water have the same shape, mainly because phase diagrams showing the level of detail of the one for pure water are hard to come by. Water has been studied in great detail compared to most substances.
But we all know from common experience that liquid under pressure, like a two liter bottle of soda pop for example, will remain liquid at a temp far below it’s normal freezing point, in a supercooled state.
Unscrew the lid off of a two liter bottle of Diet Pepsi that has been in the freezer too long, and you can watch it rapidly freeze from the top down.
Same thing happens with beer.
How much of this is simply from pressure, and how much due to the presence of a large amount of dissolved CO2, I do not know. I have never put a bottle of pressurized plain water in the freezer to study what happens. Ditto for salt water.
But I do know that supercooled states are inherently unstable, and can rapidly freeze for any number of reasons, such as from a shock wave or even a light tap.
That two liter soda will eventually freeze and explode, so there is a limit to this effect which is somewhere above the -15 to -18C that most freezers are set at.
I got to wondering what would happen to bottom ocean water which is supercooled if it suddenly froze?
Would it spread rapidly to all of the supercooled water in the vicinity, or would it be forced upwards too fast for that to happen?
If it was a large mass of it, it could burst up to the surface and mix a gigantic amount of bottom water up to the top.
Big if…I know of no one who has suggested this could happen except me.
This ability of water to readily exist in a supercooled state under a wide range of conditions is very fortunate: If this was not the case in very cold clouds, the Earth would be a very dry desert. Unless some other process than the one that now operates came into play under conditions of very high humidity.
“These floating ice islands are a navigational hazard south of 50° latitude. They also offer potential water supply.”
True, and maybe some adverse environmental effects if towing around huge chunks of ice to LA or Saudi Arabia, although probably minimal in the scheme of things. I am surprised iceberg harvesting didn’t really take off, but that must say something about the usefulness of the enterprise.
If freezing salt water into ice makes the ice fresh water, why aren’t we harvesting frozen ice from somewhere like Hudson Bay and James Bay every spring, and utilizing that fresh water to sell to our American cousins? Maybe it is time to dust off the The Great Recycling and Northern Development (GRAND) Canal of North America or GCNA by damming off James Bay and floating all that annual ice from Hudson Bay into James Bay and converting it to a giant lake as large as one of the smaller Great Lakes. All the frozen ice could add immensely to the fresh water from inflows from the James Bay watershed, by just hauling in the floating ice through a level lock in the very shallow waters where the dam that would separate James Bay from Hudson Bay. The Grand Canal was envisioned by Newfoundland engineer Thomas Kierans to alleviate North American freshwater shortage problems.
https://en.wikipedia.org/wiki/Great_Recycling_and_Northern_Development_Canal
Water development was one of the main developments that produced the Roman civilization. I would certainly have no problem selling water to America, especially if we could ‘manufacture’ that water cheaply out of ice every winter. If the Greens think CO2 is bad, then let’s start advocating wholesale water diversion projects to the USA, and they can howl about that and maybe forget about CO2 being the devil for awhile.
Earthling2
My guess is that salt crystals and brine get trapped in the ice so that when the ice is melted the water has a lower salinity than the original sea water, but is probably still too salty to drink.
Yes, the salty brine tends to migrate downwards and is expelled to the bottom of the ice the more solid it becomes and begins to grow slushy type ‘stalagmites’ of half frozen brine/water that fall off the heavier they become. Multi year ice has less salt content but in this case of James Bay, the ice melts annually. The surface ice all fall, winter and spring also collects snow and rain, so additional fresh water is added to the top layers.
As you say, the water would still be too salty to utilize to drink or agriculture etc, so being at maybe less than 5%-10% original salt content, would be much easier to process through a mass Reverse Osmosis filter before shipped south. Since James Bay is also much less salty than the open ocean due to all the river inflow from the basin, that water would have to be RO’d too as James Bay was being converted to a fresh giant fresh water supply. James Bay is relatively shallow, so the water volume isn’t huge comparatively speaking. The fresh water from the river inflows could all be captured at the estuary interface around the basin so that could be piped directly into the canal. Of course, just to keep the Greens all happy with this plan, it would be powered by offshore Wind.
This link to a short video on ‘brinicles’ shows an interesting aspect of the process. I remember it being posted on WUWT a while back.
This brine which is expelled from freezing seawater is the apparently the source of all of the deep water in the world’s oceans.
The Arctic is, IIRC, the only place where water gets cold enough and salty enough, and hence dense enough, to sink all the way to the bottom of the ocean.
This is why the deepest layers of the ocean are so cold.
Earthling2
There is no shortage of freshwater lakes in Canada. I would think that harvesting lake water would get around the issues of any salinity at all.
Arctic explorers found that old ice was fresh enough to drink.
Wouldn’t that interfere with the huge hydroelectric dam up there? If the James Bay grows in height, where the turbines drain into?
Also sounds like it would cause another huge ecological disaster like the creation of the James Bay (or was it James River) Dams caused, where we had pictures of dead drowned caribou on the news for weeks.
No, I wasn’t proposing any change in the water level of James Bay, which is currently a shallow ocean bay connected to Hudson Bay. Just converting it to a large fresh water lake to be able to take the water in a canal for use in drier parts of North America. The hydroelectric dams on all the rivers would continue to operate normally since there would be no change in water level. A shallow water dyke would be built across James Bay where it meets Hudson Bay. This is already a fairly old proposal as the link to the Grand Canal proposal shows.
My only contribution to that plan was to take the mostly fresh water ice from Hudson Bay annually and tow it through a series of ‘locks’ in the dyke that separated the newly created fresh water in James Bay from the salt water in Hudson Bay. The additional mostly fresh water made by freezing salt water would add to the river inflow and all the fresh water in James Bay would be available to be utilized in North America through a canal to the Great Lakes. Water could then be withdrawn from some of the Great Lakes to pipe over to where it would be purchased. The profits from this would pay for the infrastructure to build it.
The answer to the implied question in the OP (if we could understand one type of ice, which would be the most important type to understand?) is of course ice Ih.
Coming in at a close, if obvious, second: ice-nine, which, if a critical nucleating volume of it could be synthesized, need only be dropped off a pier et violà! Sea level rise halted forever. Not THAT”S some Ocean Obamification (OO) we can all believe in!
But I rejected this answer primarily because scientists are still decades away from understanding, REALLY understanding, the themes of Breakfast of Champions. We must not let our technology advance too far ahead of our wisdom and humanity, for that way tattoos and IBM punch cards lie.
What do I win?
You win the Venus on the Half-Shell, Kilgore Trout memorial award.
put him on the Hooyuc
I thought the Breakfast of Champions was beer poured on Cheerios?
In my earlier days the Breakfast of Champions was 2 Goody powders and a cold Budweiser.
Still good for the ‘night after’ but as Hank Williams Jr sang “Hangovers hurt more than the used too”.
Thanks for the laugh. Hair of the dog….
Someone’s, apparently, a serious Vonnegut fan…
rip
Take the regular normal and present it as a new normal
Take science and present it as an attack on science
Take opinion columnists and present them as scientists
Take scientifically-illiterate English majors and present them as scientific-communications science communicators
Take lack of interest and present it as agreement
Take silence and present it as consent
Take consensus and present it as evidence
Reposition vague, innumerate speculation as hypothesis
Reposition hypothesis as theory
Reposition theory as fact
Plus ça change, plus c’est la meme meme…..
Brad Keyes: “Take scientifically-illiterate English majors and present them as scientific-communications science communicators”
More egregious for me is the English Majors that are semi-literate in Science. They control the dialogue with their ability to consume and produce large quantities of language. But when asked for clarification on the boundary conditions of their own assertions they deflect and claim that it’s all been well established ‘over there’. I’m left wondering why they don’t readily examine the boundary conditions of their assertion, and realize that they don’t actually think like a scientist even though they can produce ‘sciency’ sounding language in bulk.
Don’t forget the “climate communications specialists”, who feel sorry for us poor, deluded “deniers” and stand ready to unconfuse us with selected non-facts and assertions which will tug our heartstrings toward revelation. Cute penguins = Global Disaster! Fake polar bear pictures= The Total Loss of All Ecosystems!!
Believe! And you shall be healed!
Orestes is the worst of the worst. Have you seen her TED talk attacking the scientific method? Sickening. It sent Feynman spinning in his grave.
Ken
THANK YOU for understanding what’s at stake (no less than the scientific method) in Oreskes’ [sic] one-woman war on the Western mind.
Has she only given the one TED talk? Do you have a link? I have a queasy half-memory of it but I tend to watch these things late at night and I’m never sure, when it comes to Oreskes [sic], what rôle my horror-show hypnagogic hallucinations have to do with it. There are things I think she’s said about science but which are so evil that I know, rationally, she can’t possibly have said them unless she really was shat forth from Shaitan’s cloaca as the Medieval sources attest.
Now why would you wanna go and demean bird feces like that?
“Take scientifically-illiterate English majors and present them as scientific-communications science communicators”
Sounds like that climate communicator who has a PhD in Renaissance Lit. Her name’s not on the tip of my tongue.
I think you nailed it, Brad! Have you seen their playbook or are they just that obvious? Never mind answering. I think I know.
john,
I don’t mind answering at all.
Yes, towards the end of my year as a believer, by which time I’d been accepted into a fairly high echelon within the science, I was privileged to see a couple of pages of their playbook.
Monbiot was throwing one of his soirees that are famous for turning into matinees at his Volga dacha, so it wasn’t exactly the contemplative ambience of a library—nor had I thought to bring my white cotton gloves—or else I’m sure I would have sat down and read the playbook properly.
Nevertheless, the pages that lay open on Monbiot’s desk were more than fascinating enough. What was immediately clear was that this was the Tobacco Playbook—same strategies, same people—in a different dust-jacket.
Industry-specific terms had been updated to their climate analogues, apparently by a simple Edit > Find > Find And Replace. (A single misspelled word, tomacco, had slipped through the cracks and was now a part of official climate doctrine!’)
Perhaps I shouldn’t be telling you this because George makes all his guests sign an NDA. But on the other hand I did so in the name Michael J. Fox, which has been found (People v. Oregon Institute of Science and Medicine) not to be the name of a real person. SO it doesn’t “feel” binding. I’m not getting a binding vibe off the whole affair.
Dr. Tim Ball states, to wit:
No, no, no, ……. it was the increase in Global Warming temperatures caused by the increasing CO2 that caused the “Ship of Fools” to get trapped in the “melting” Antarctica sea ice, ……… t’weren’t it?
Ball of global confusion:
https://youtu.be/-9poCAuYT-s
Dr Ball: An enjoyable and informative article.
You wrote: ‘ It is difficult to draw any conclusions about variations in volumes of sea ice from year to year because we only have satellite data since 1980.’ I take issue with this statement on two levels: 1) Passive microwave and optical imagery of the North Pole began in the early 1970s, and was reported in the IPPC FAR, 1990, and the SAR, 1995, in graphical form. 2) IMO, several nondifficult conclusions can be drawn from the data from then through now. For me, principal among these is that there is measured year-to-year variation in Arctic Sea Ice extent, sometimes over a decade as much as +- 9×10^5 sq km.
Thanks for your articles. I always enjoy reading your perspective.
Just a correction:
Sir James Clark Ross discovered the Ross Sea, Victoria Land, and the volcanoes Mount Erebus and Mount Terror and several other places around 1840. He was the nephew of Sir John Ross, who was an arctic explorer. Sir James Clark Ross also was the first person to find the north magnetic pole and was trying to locate the south magnetic pole. It was Captain James Cook who made that statement quoted.
There is a new book out about the ship Erebus that is an interesting read and explains James Clark Ross’s trips to the Antarctic
https://en.wikipedia.org/wiki/James_Clark_Ross
https://www.theguardian.com/books/2018/sep/30/erebus-story-of-a-ship-review-michael-palin
“The first form of ice is created when water freezes. People assume that it occurs at 32°Fahrenheit, 0°Celsius, or 273.15 Kelvin. Freshwater density begins to increase at 4°C, and ice forms as fusion occurs at 0°C.”
This is a bit ambiguous. Freshwater density begins to increase at 4°C only if the water temperature is rising. As water temperature goes down, freshwater density begins to decrease until it is about 9% less at 0 °C.
Um…for pure water, 4C is the temp of maximum density.
It is all downhill from there.
It begins with the false assumption that CO2 causes global warming and
… And that’s as far as I read.
Ahhh.. the close the ears, close the eyes, response, hey ! 🙂
He’s got the hear no evil, see no evil parts down but then has no hand left to cover his mouth.
If you are correct Nylo, then the specific heat of air must have changed. Please let ,e know what the new value for air is.
Great article, but please: “For example, in 1816…”
The year without summer is a HUGE cherry to pick. Not exactly qualified as an example IMO.
Maybe, but hope/pray that doesn’t happen again when we are trying to feed 7.4 Billion on the planet and we only have a 2-3 month supply of basic foodstuffs on the planet. Unfortunately, it will happen again at some point, and we will never be happier that we were able as a human race to actually warm the planet a smidgon by maybe .8 C, assuming we humans are responsible for the whole amount. If another LIA were to occur now for 50-100 years similar to the worst in the 1690’s, it would certainly be a challenge.
Another little hiccup: The density of water DECREASES as the temperature drops below 4C. It does NOT increase. It is an easy mistake to make and in no way detracts from this excellent article.
My little science pocket book gives water densities as:
@ur momisugly 4C density -1.00000
@ur momisugly OC. “. – 0.99987
@ur momisugly-10 C. “. – 0.99815
It is a table and I have missed out the between temperatures. All regularly dropping.
I understand this is why ponds ice up on the surface as the colder water rises by convection.
The fish are glad about that.
Your table is correct for fresh water, not for saline water.
It is very complex, for numerous reasons.
As water freezes on the surface of the ocean, salt is expelled and increases the salinity of the surrounding water, and the density of the freezing stuff changes as the salt is expelled at the same time.
The phrase “the false assumption that CO2 causes global warming” is incorrect. It is understood that CO2 causes warming — ask Judith Curry.
The issues have to with the magnitude and effects or warming.
john:
Appeal to Authority Fallacy:
Insisting that a claim is true simply because a valid authority or expert on the issue said it was true, without any other supporting evidence offered.
Want to try again?
John, please explain the mechanism by which CO2 causes warming.
Good post. Explains a lot. Nothing like this is ever simple. The Earth and its ecology is very complex. Add the Sun and atmosphere and all havoc breaks out. And that’s only for the weather. Add the component of climate variations with dozens and dozens of variables and super computers lock up. The KISS principle doesn’t even begin to explain it. There should be a Murphy’s Law that says “any thing that gives someone a massive headache, isn’t worth the thought or trouble”. My thoughts are that climate change and all of the political hype is a big nothing. Regards, retired mechanical engineer, physicist, astronomer and petroleum geologist (a REAL Scientist).
All Global Warmistas know that the fossilized White Spruce (Picea Glauca) 100 km north of the current tree line and radiocarbon dated to 4949 ± 140 years was deliberately planted there, recently, by Climate Deniers to cause doubt in the ranks of World Climateers
Whose initials are carved in the trunk?
Kilroy wuz there.
Alley Oop
For anyone interested, here is the WUWT article from May 15th 2015 by Willis.

The comments section had a conversation about how sea ice forms, and several details about the process of deep water formation and the progression from frazzle ice to sea ice, etc.
Here is one of my comments and a link:
“Janice,
Several things. The trench is 11 kilometers deep, not 11 miles, (Somewhat deeper than Mount Everest is high…about 6 miles) and it is only this deep in a very small portion of it.
And the trench does not contain the coldest water, perhaps because it is cut off from the thermohaline circulation and is warmed by heat flowing through the thin crust at the subduction boundary.
The coldest water is near Antarctica and in the Atlantic and Arctic…since the Bering Straits is too shallow to allow deep water to enter the Pacific there, there is no flow from pole to pole in the Pacific.
And yes, I was looking at that nose, and was wondering later why it is there, and what it signifies. I started wondering if the phase diagram of other substances is equally bumpy (for lack of a better word).
Certainly water has many unusual properties, and this seems to be one of them. The hiccup in density near the freezing point is well described and understood as being due to hydrogen bonding holding water molecules further apart in hexagonal ice, but the nose peaking at 205 mPa is very strange, and harder to find material on as well.
But note, the line is very straight and does not bend to the left until well up past 10 mPa, and only lowers 5 degrees or so at 50 mPa
http://upload.wikimedia.org/wikipedia/commons/thumb/0/08/Phase_diagram_of_water.svg/700px-Phase_diagram_of_water.svg.png
OK six hours later now. I have been reading a lot on this all day and evening, had had more to say, but wanted to find out the exact cause of this nose.
There is a lot of good information on the exact process by which surface water freezes, first into frazzle ice and some other forms with unusual properties, like being ale to bend without breaking around waves and such.
But, yet to find anything regarding why deep water will not freeze.
One thing is clear: It is incorrect to refer to the freezing point of water. Better to refer to the melting point of ice, because water can be readily supercooled, and is thus less dense than warmer water around it…being that the hump in the density curve remains, but peaks at the freezing point in sea water.
It is stated that supercooled water can remain so indefinitely, but normally only if undisturbed will it not freeze eventually.
Surface water in the process of freezing typically is supercooled a few degrees.
Another interesting tidbit is that the water at the bottom of the ocean is near freezing even in the tropics.
Here is what I wanted to be sure of…The vast majority of the ocean is no where near the pressure you mention. Only in the deepest trenches is it so, and these are tiny in relation to the size of the ocean
Also, notice the pressure scale is logarithmic on the phase diagram. The peak of the nose is at
over 200 mega pascals.
Most of the ocean bottom, the abyssal plains, is more like 6 kilometers and thus about 60 mPa.
Anyway, it seems likely that adding salt to water is only endothermic if the salt is in solid form when added. The change in enthalpy is due to breaking the ionic bonds when the salt is dissolved…so call off the runaway chain reaction from that cause.
And, making the remaining water saltier will continue to decrease the melting point.
But the water down there is near the freezing point, and if, if I say, a large quantity of water did freeze for any reason, it would rise to the top with tremendous force.
Seems like a coiled spring to have so much water so close the freezing point, and under pressure.
Could some event cause a sudden reduction in pressure…briefly but long enough to initiate rapid freezing of supercooled bottom water? Maybe related to methane clathrates? It is mostly water, those clathrates, and if dissociated from the methane, what are the thermal and salinity altering effects?
So many questions.
One thing I am more sure of than ever…there is more to be concerned about regarding a return to ice age conditions than some catastrophic warming of our frozen wastelands by a few degrees.
If you or anyone has reading material to link to regarding phase change of sea water under pressure, I would like to see it. The phase diagram is what it is. The question is, does salt alter the shape of those curves, or just shift them left and right? Might it smooth out the nose? Salt does alter the maximum density in relation to freezing point, after all.
This conversation gave me a reason to read a lot of material I was not about to read anyway, and I also found a whole lot of very interesting maps and graphs and charts. I learned a lot today, not sure how accurate it all is…much is official “climate science”. But the P Chem parts I am fairly certain are on the level.
Thanks, and TTYL.
http://www.srh.noaa.gov/jetstream/ocean/images/layers.jpg
http://www.chartgeek.com/wp-content/uploads/2012/04/lakes_and_oceans_large1.png”
https://wattsupwiththat.com/
Menicholas
I just read somewhere (I forget where) that dissolving salt in water to create a saturated solution will increase the volume 3.5% (temperature unspecified). So, those ‘brinicles’ are adding salt to the underlying water and increasing the volume. The whole thing is very complicated!
Clyde Spencer, replying to Menicholas
I would strongly question that: Once dissolved, the former crystals are now independent ions, flowing between the water molecules themselves and so causing near-undetectable changes in density due to an expansion in volume. Unlike, say, a metal crystal, solid rust crystal or particle of dirt that does not change state when placed in water. Brine IS denser than pure water at the same pressure and temperature, and the saline brine DOES “fall” down to the bottom of a tank or lake with fresher, lighter (warmer!) water on top that is evaporating to the air.
A saturated solution of sodium chloride in water causes a decrease in volume of about 2.5%. It’s why one uses volumetric flasks to make up a known volume of solution, adding a solute to a known volume of solvent usually results in a change in volume.
Phil,
OK, that would increase the density of the water, which makes sense. That also suggests that the brine that is expelled from freezing sea ice contributes to an increase of the density of the water under the ice once it mixes.
Menicholas,
Thanks for the link to the phase diagram of water. I have spent way too much time looking at it over the years. Whereas you are concentrating on the liquid/freeze points as related to the topic of discussion, I was focused on the liquid/vapor relationship (that curve is pretty steep from the triple point to boiling at 1 atm) regarding clouds and decreasing pressure while receiving energy (heating) from LWIR from the ground to the base of the cloud . Anyway, much time can be spent when you start applying that chart to earth science. You probably got/need a couple more days to satisfy your curiosity! And then you still may not know and even more curious! But it’s a fun journey! LOL
psu is parts per thousand.
And sea water freezes at around -1.8 to -2.0 C at the salinity in the range of normal seawater, which is 35 to 37 or so.
I am reposting the link to the discussion from a few years back, although I see that some of the links no longer work.
Also, pictures which used to post as the photo no longer do so, which means one cannot see the graphs while reading the text of comments…a definite negative IMO.
In any case, here is that link and the comment I meant to attach (the one below was not the one I meant to copy here):
Here is the correct link to the article, Things in General, which had as one part of the comments section the discussion I was mentioning.
For some reason the link below posted as a link to WUWT home page.
https://wattsupwiththat.com/2015/05/15/things-in-general/
And here is another of the comments from that day/night:
“Crispin in Waterloo:
“The freezing temperature vs salinity curve is amazing. I never saw that before. I would like to pursue this further to see where water is and the condition and the influence of ice cycles.”
Yes, very interesting indeed. Boiling point elevation/freezing point depression is of course a very extensively covered subject in most chemistry and physics courses, and indispensable knowledge when covering physical chemistry.
It does seem to have been overlooked I many of these discussions, which, in retrospect, is odd.
I found one more chart that gives a better perspective, since the temp vs freezing point chart is in different units than the other chart.
Since ice does form at the surface, it must be the case, as you point out, that the process is rather more complicated, and that as the freezing progresses, salt is excluded gradually and allows the ice to remain on the surface.
(Unless one is to think that the ice forms at the bottom, or somewhere in the water column, and floats up to the surface, which I have seen no evidence to believe is the case)
Otherwise, water would sink before it froze and sea ice would have a hard time ever forming in open water, no matter the temp. It would seem that the whole water column would get very cold and then freeze all at once from top to bottom!
Here it is, and I agree that this needs a closer look.
http://www.fondriest.com/environmental-measurements/wp-content/uploads/2014/01/360x300xwatertemp_salinity.jpg.pagespeed.ic.yheIMC6nSN.jpg
*Somewhat off topic, but interesting:
I do know from various chemistry classes than getting certain substances to crystalize can be very difficult, and have heard some very strange tales of people attempting for years to get crystallization started.
As most students have experienced, some organic samples will tend to “oil out” instead of crystalizing.
I recall one technique was to scratch the glass vessel, which will sometimes get crystal growth started.
Sometimes a small seed crystal is needed, which upon being introduced the whole sample will abruptly crystalize.
One such anecdote involved a researcher who was unable to get the process going in a novel new compound, but heard that a colleague in another building had succeeded in doing so. He walked to this colleague’s office with the sample in hand and, as he entered building which housed the lab where a small amount of the crystalized sample was kept, the sample in his hand, which had resisted all attempts for a very long time (I do not remember how long, but I am thinking months or years), suddenly crystalized! Just from being in the same building!”
It was a wide-ranging discussion, and much of it is off topic, and many of the links are no longer working.
I will locate the missing graphs and charts and post in an additional comment.
Here is the phase diagram of saline solution, note that seawater is about 3.5% saline.
Thank you Phil.
I had found this chart, which plots a different parameter along the horizontal axis that a regular phase diagram.
I was hoping to come across a phase diagram of temp vs pressure with as much detail and range as the one for water we are all familiar with:
I have not been able to locate one.
This one is like the one you posted but smaller range with more detail:
http://slideplayer.com/slide/5740114/19/images/25/Water+-+NaCl+Phase+Diagram.jpg
Pure water has a constant melting point as pressure increases way up to a very high value, where it has a unique nose to the curve, which strangely bows the opposite way than might be expected. Most materials, such as metals, expand when they melt, so increasing pressure delays melting.
Water does this at all temps and all pressures, up to about 100 times atmospheric pressure…at least according to the standard phase diagram.
The phase diagram seems to be at odds with experience of how many things which are mostly water behave when frozen under pressure.