Note: This is a contentious subject, and I have often shied away from it because it often erupts in food fights. However, Mr. Gill is making a good-faith effort here, and asks some relevant questions that I consider worth discussing. His original essay was sans graphics, and I’ve added two relevant graphics to aid in the discussion. – Anthony
Do Wien’s Law and Quantum Physics 101 prove CO2 can’t warm anything?
Guest essay by Rod Gill
WUWT has happily demonstrated many ways CO2 fails to produce measurable warming. I’ve thought of another way. It’s so simple I must have missed something, but I simply can’t work out what. It goes like this…
Experts suggest there is a net down welling 2W/m2 of long wave infra-red radiation (LWIR) that is causing global warming. I suggest the quality of that 2W of radiation is crucial to determining whether or not it causes any atmospheric warming at all. First a few key points which I think are facts and not open to dispute.
My understanding of Thermodynamics and Radiation from CO2 is as follows:
In Thermodynamics, Temperature is the average kinetic energy of the particles in a body (solid or gas).
The temperature of a volume of air has nothing to do with the amount of radiation (sometimes mislabelled as heat by scientists) passing through it. Unless that radiation is at a frequency that can be absorbed by the air, its temperature is completely unaffected by the radiation (ignoring any convectional heating).For example at the top of Mount Everest, there is a lot of solar energy (long and short wave radiation) there when the sun is out but the temperature is still cold.
Different gases have different emission spectrums. For example Oxygen and Nitrogen do not absorb or emit Long Wave Infrared Radiation (LWIR) at all, so are not considered to be “Greenhouse” gases.
The temperature of a body (gas, liquid or solid) directly affects the wavelength of the radiation it emits and absorbs.
Wien’s Law defines the temperature – wave length relationship. The formula is Temperature (in degrees Kelvin) = 2898 / peak wave length in µm (micro metres). So for the average temperature of the Earth, lets call it 15C (=289 Kelvin), the wave length is 2898 / (15+274) = 2898 ÷ 289 = 10um.
The wavelength of the peak of the blackbody radiation curve decreases in a linear fashion as the temperature is increased (Wien’s displacement law). This linear variation is not evident in this kind of plot since the intensity increases with the fourth power of the temperature (Stefan- Boltzmann law). The nature of the peak wavelength change is made more evident by plotting the fourth root of the intensity. Source: http://hyperphysics.phy-astr.gsu.edu/hbase/wien.html
Carbon Dioxide’s absorption spectrum shows it absorbs LWIR at three different narrow wave lengths, sometimes called finger frequencies. Two of those wave lengths happen at temperatures too hot to exist in the atmosphere, the remaining wave length is 15um.
15um equates to 2898 ÷ 15 = 193K = -80C or -122F. In the atmosphere this temperature only occurs about 90-100Km high in the atmosphere.
Carbon Dioxide only emits and absorbs radiation at -80C from a narrow layer of atmosphere 90Km above the Earth’s surface.
So now we need to examine the quality of that 15um radiation and its ability to heat the lower atmosphere. To do this we need to understand basic Quantum Physics as taught in 101 classes to Physics and Engineering students at University. Confession: I’m an Engineer, but trained before Quantum Physics was introduced to University courses so I’m self-taught, hence my need for a sanity check. Which, dear reader, is where you come in.
The key points in basic Quantum Physics, regarding radiative heat transfer, are:
Molecules have one or more electrons circling them. Their orbital height is not variable, But fixed. The electrons only orbit at set altitudes, the closer to the molecule the lower the kinetic energy of the molecule and so the lower the molecule’s temperature.
For a molecule to “warm up” (have more kinetic energy) it needs its electrons to move to a higher, more energetic orbit. This can happen in one of two ways, get energy from a more energetic molecule via collision or receive energy via radiation.
For an electron to move to a higher orbit from radiation it must receive a photon with sufficient energy for an electron to reach that higher orbit.
Photons with too much energy raise the electron to the higher orbit then the molecule immediately re-radiates surplus energy.
Photons with not enough energy to raise the orbit of any of the electrons are either scattered or immediately re-radiated (effectively reflecting or scattering them) with no change to the molecule’s kinetic energy, or temperature.
The Photon must have a frequency that resonates with the molecule, otherwise the Photon is just scattered or reflected immediately with no temperature change to the molecule.
Carbon dioxide can only absorb Long Wave Infrared Radiation (LWIR) energy and radiate it at 15 micro metres, a fraction of the LWR spectrum.
Electrons orbiting molecules of a liquid or solid need more energy to boost an electron’s orbit than electrons in a gas, so require more energetic photons again to warm them.
Therefore it is my understanding that it is impossible for the LWIR emitted by a cold low energy CO2 molecule to have the energy required to warm any molecule in the atmosphere warmer than -80C and certainly no molecule in a liquid (EG water) or a solid body, as their electrons require even more energy.
LWIR from CO2 simply bounces around the atmosphere until it escapes into space and it causes no warming of the lower atmosphere at all. The energy level of that 2W of LWIR is too poor to have any affect. It needs to be closer to 10um to be energetic enough to warm anything.
So the idea of CO2 trapping heat in the atmosphere is all wrong. Yes LWIR from CO2 is retained in the atmosphere longer, but it simply bounces around until it escapes into space without causing any warming.
So am I right? I deliberately have not included any references because I want you to confirm or deny my understanding independently. If I gave you my references, which knowing the web may or may not be accurate, you might erroneously come to the same conclusions I have. However I have tried to limit my research to University papers and lecture notes hoping they are more reliable.
If I’ve got this right, CO2 caused global warming isn’t possible. If I haven’t got this right, then exactly how does LWIR radiated from CO2 warm anything?
Many thanks and please limit comments to specifics mentioned above. And if you disagree with the science above, please explain which sentences you disagree with and exactly how, at the Quantum Physics level, photons from a CO2 molecule at -80C can warm anything.
This would have been a great thread if one were allowed to reference those unmentionable scientists that deny that CO2 can warm the surface. Especially the astrophysicist who just published a week ago or so on this topic.
I hope there comes a time when luke-warmers here can debate with the skeptics. Perhaps some day.
TTY, “Both of course.” is a perfect response! Most of the comments are citing warming of a molecule, parts of them, or atmosphere of them, but in reality are MOSTLY talking about reducing heat already gained.
Fact: Energy can neither be created nor destroyed, it can only be changed in form.
1) CO2’s only defined mechanism to affect climate change is through absorbing and thermalizing LWIR between 13 and 18µ
2) Those bands are only a small fraction of the emitted LWIR, earth’s peak is 10µ
3) There is no way through thermalization for CO2 to warm anything above the temperature of the emitting body, the energy simply doesn’t exist. Absorbing a fraction can’t account for more than the whole.
4) Radiation could trigger combustion, but that isn’t the phenomenon that we are talking about
5) Conduction, convection, and radiation all act to move heat away from the earth. Claims that the GHG effect is responsible for all the warmth of the atmosphere ignore conduction and convection.
6) This is easily proven in a lab by shining 15µ IR light into a flask of CO2. It won’t warm because the room temperature is way above -80°C
Technically energy can be “destroyed.” If you observe the spectra from distant stars, you’ll note that the hydrogen emission lines are red shifted. When the photon left the star of origin, it had the normal wavelength (freq) of the emission lines of hydrogen. As it traversed the expanding intergalactic space, it’s wavelength grew longer (red-shift.) There is a calculable loss of energy in this red-shifted photon according to E=hc/l where delta-E = hc/delta-l.
…
Where did the ‘lost’ energy go?
I already did. The photon that was emitted a million light years ago, “lost” some of it’s energy. The frequency has decreased (increased wavelength.) Where is the “lost” energy?
it went into potential energy. for the gravitational red shift it was demonstrated by t Cranshaw at Harwell in the 1960s. a photon fired from the ground level to the top of a hanger was found to be red shifted. a photon fired downwards was blue shifted. it is like a ball being throw upwards it looses speed but that does not mean that energy is destroyed. Similarly with a photon
Gerontius, that does not explain it. There is no gravitational field in the million light years between the star that emitted the photon, and the spectrometer here on earth that measured it. The red shift is due to the expansion of space, not to a gravitational field.
PS Gerontius, photons coming from the opposite side of the universe display the same red shift. You would think that if you looked in the opposite direction of the red shifted photon, you’d observe a blue shifted one if your “theory” were correct.
The energy doesn’t change so much as the wavelength stretches out, changing its color. If the source were heading toward you the waves would bunch up. It’s all due to the Doppler effect.
A passing car is a good example. You hear the sound change as the car passes. Sound at sea level and standard conditions travels at about 761 mph. So if a car is traveling toward you at 30 mph, then the sound is travelling at 761 + 30 mph = 791 mph. After it passes, the sound you hear is traveling at 761 – 30 Mph = 731 mph. So the energy does not particularly change. Turbulence has a small effect, but the pitch goes from higher to lower just as starlight red shifts.
It’s all down to the observer’s or listener’s point of view or frame of reference. The sound wave or photon just keeps doing its own thing, while the looker looks on.
As for the effect of gravity, red shift or blue shift is not due to any change in frequency of the photons, but rather to the time rates being different for observers in different potentials. No energy change occurs. If a photon of a given frequency is created by some atomic transition in the vicinity of a star, then it will appear red-shifted compared with the energy of the same transition at some distance from the star, but it hasn’t changed frequency.
Similar considerations apply for red or blue shift due to relative motion; the energy is unchanged relative to the location at which the photon was emitted, but appears different from a moving frame. The situation isn’t quite so clear-cut about red shift due to cosmological expansion, because it depends on how you describe the expansion, but even in that case the frequency of the photon is unchanged relative to its original rest frame.
“The energy doesn’t change so much as the wavelength stretches out, changing its color. If the source were heading toward you the waves would bunch up. It’s all due to the Doppler effect.”
That is a great point, the total energy is the same, but energy/m is less. You are stretching the energy over a larger distance.
The energy is not conserved no even conceptually both QM and GR demand it to be so. This is one of the problems with classical physics it leads you to stupid answers.
It is not a problem with “classical physics”, but with your lack of understanding thereof and misguided thinking about it.
As CO2 observes above in response to my prior comment, energy is conserved, but just delivered at a different rate per unit of EM radiation travel. That’s one way of explaining the paradox which you imagine.
Clearly you don’t understand QM or “classical physics”, by which you mean “physics”.
How many times do I need to explain it to you?
The apparent change in color, ie wavelength and frequency, or a photon of visible light traveling along at its accustomed C, is due to the observer’s position. In the Doppler effect, sound waves don’t change their energy content.
Again, if photons don’t exist, then neither do any of the other elementary particles.
Oh here he goes again trying to obviscate the problem, we have cool lasers(tm) again. You make a statement then just want to wave hands ignore it and try to deflect to something else.
the gravitational red shift is real and explicable and demonstrated . in that the energy lost is conserved in potential energy
the velocity red shift is real and is due to energy conservation, and not an example of non conservation of energy. You cannot look at the photon in isolation and must look at where it came. There is a fairly simple classical (although with a bit of QM thrown in explanation). It is readily available on the web, although if you look you will always get the Kernoodle reply that is non conservational. You need however to be rigorous in accounting your energy balance . There is no need to invoke expanding universes only relative motion between observer and emitter. You then get the Doppler effect. Now the Doppler effect and the red shift in electromagnetic waves is well known and its energy conservation is not in doubt. And as I said before the energy balance of the effect demonstrates that the red shift is an effect of energy conservation
Robert I leave you to search the web, ps Cranshaw used the velocity red shift to measure the gravitational shifts. Now that’s a big clue as to where the missing energy goes.
None of you here has mentioned it in this blog, so ignorance is bliss.
co2islife November 19, 2017 at 4:12 pm
Fact: Energy can neither be created nor destroyed, it can only be changed in form.
1) CO2’s only defined mechanism to affect climate change is through absorbing and thermalizing LWIR between 13 and 18µ
2) Those bands are only a small fraction of the emitted LWIR, earth’s peak is 10µ
Since you claim to use Spectracalc try the following:
Using the Blackbody calculator set it for a BB at 288K
Set the lower limit of the band to 13 micron and the upper limit to 17 micron.
Perform the calculation.
You should find that the band radiance is 23.3 W/m2/sr whereas the total radiance is 124.2 W/m2/sr.
By my reckoning that means the CO2 absorption band accounts for about 19% of the total, hardly a small fraction.
Remember “Everyone of my comments can be verified by MODTRAN or SPECTRALCALC.”
“By my reckoning that means the CO2 absorption band accounts for about 19% of the total, hardly a small fraction.”
In an atmosphere of 100% CO2, that would prove a whole lot, but add H2O to the mix and you will see CO2 is meaningless in the lower atmosphere. You only see a CO2 signature at 15µ after H2O precipitates out of the atmosphere. BTW, that 19% is 19% of the very low energy end of the LWIR spectrum. Now compare the energy of visible light, of which H2O in the oceans absorb 100% of. That is where the energy is, not LWIR.
co2islife November 20, 2017 at 5:57 am
“By my reckoning that means the CO2 absorption band accounts for about 19% of the total, hardly a small fraction.”
In an atmosphere of 100% CO2, that would prove a whole lot, but add H2O to the mix and you will see CO2 is meaningless in the lower atmosphere. You only see a CO2 signature at 15µ after H2O precipitates out of the atmosphere. BTW, that 19% is 19% of the very low energy end of the LWIR spectrum. Now compare the energy of visible light, of which H2O in the oceans absorb 100% of. That is where the energy is, not LWIR.
Not true, that 19% is of the total of the LWIR you can check it out on Spectralcalc. Also the CO2 absorption band is near the peak of the energy spectrum, it is water that is near the tail.
The Blackbody radiance peaks at 550cm-1 for 288K (peak radiance 0.136 W/m2/sr/cm-1) whereas the CO2 absorption band is centered at 667cm-1 (radiance ~0.132 W/m2/sr/cm-1). So CO2 absorbs near the maximum of the earth’s BB energy distribution, by contrast water absorbs between 1500-2000 cm-1 (radiance ~0.02 W/m2/sr/cm-1).
You show the CO2 signature on the Modtran graphs on your own website from 1km, you also show the H2O signature off in the tail. If you do the calculations on Modtran comparing 0ppm CO2 with 400ppm CO2 you’ll find that ~10% of the total is removed by CO2.
“Also the CO2 absorption band is near the peak of the energy spectrum, it is water that is near the tail.”
Yep….
Dr. Deanster
November 19, 2017 4:13 pm
I see a lot of diagrams on radiation ,,,, incoming, outgoing, backwards. But all of this assumes that the majority of energy absorbed by the ocean is in some sort of magical equilibrium with the atmosphere. …. ie, a very large part of the incoming SW IR Is absorbed and NOT immediately released.
Exactly, but the ONLY outside source of energy to the earth, well, maybe some minuscule amount from plate tectonics, …. is SW, and it DOES penetrate the ocean surface. It doesn’t just warm the surface and immediately float back into the atmosphere.
Any equilibrium equation for the total earth can only be made n long term basis, as a good bit of energy is stored and released over time.
A close examination of USCRN soil data shows that solar IR hardly makes it 1 meter deep by conduction and then only during summer daylight which is the ONLY time the air is warmer than the ground.
Derek Colman
November 19, 2017 4:27 pm
My idea of an experiment is simple, maybe too simple. Take two identical sealed glass containers and fill each one with a mixture of gases which imitates the atmosphere. The only difference between them is that one has 0.028% of CO2 and the other has 0.04% of CO2. Both vessels should be fitted with highly accurate temperature probes. At the start ensure that both are at exactly the same temperature. Now radiate each with an IR lamp, tested to be identical in output. Now observe the temperature increase in the vessels. The data from this experiment can then be used to calculate and compare what happens to atmospheric temperature at pre-industrial and current CO2 levels. My guess is that the difference will be so small that even the accurate temperature probes can not distinguish it.
Sailboarder the earth is a closed system with the sun. Energy can enter but no mass. Also the video was done to correct numerous errors done for Al Gore by Bill Nye. Good video.
E=hv tells us how much energy is available to cause a CO2 molecule to increase its translation. And translation velocity is dependent on absolute temperature and molar mass. So heavier gas travels slower.
We know that Q = Cp m dT for how much energy is needed to raise a mass of something, air in this case.
I have yet to be able to show that hv * 400ppm CO2 = Cp m dT for a cubic meter air.
But a postulate of kinetic theory of gas says that all collisions are pure elastic and no energy is lost or gained, so if true CO2 cannot do this anyway.
That is all very interesting but it is a EM wave aka a radio wave. Just like your TV set the EM wave will only selectively react to certain frequencies. Do you really think the EM wave delivering your TV signal got absorbed and re-emited from every air molecule along the way or has anything to do with kinetic energy of molecules along the way?
I have no idea what GH theory works on I am not a climate scientist. All I am telling you is radiative transfer doesn’t occur in that manner described and energy can be lost and gained and attenuation can be measured. I am sure if you search Atmospheric Attenuation you can find the numbers for ll frequencies in the EM spectrum (from RF thru visible light up to X and Gamma rays).
LdB we have been shown graphs on this thread of CO2 absorbing at 15u. OK, then using E=hv we can calculate the amount of energy available to any CO2 molecule to increase its translation. Translation is directly related to temperature.
Due to the relationship of velocity to temperature we should be able to calculate the ability to raise a cubic meter of air via the energy in a 15u IR absorption.
Having taught basic radar theory and basic physics of underwater sound in the navy I have a idea of EM wave and attenuation etc. But that was many years ago. Thanks for your input.
Does anyone have a link to a research paper where 15µ IR was shined on a flask of CO2 and its temperature measured? Unless the CO2 spectrum is a lie, then the only contribution CO2 has to global warming is through the thermalization of IR at 15µ. That won’t warm anything. Simply focus on the basics. By what mechanism does CO2 affect climate change? Isolate that factor and test it. Has anyone tested the impact of CO2 thermalizing 15µ LWIR?
I too am working with this problem, I actually thought I had been ‘pipped to the post’, but after reading this, and the comments, nope – not even close. And Anthony, this may be ‘contentious’ but it is the key premise to greenhouse theory; without this, greenhouse theory collapses, so it is worth trashing out.
The way I see it the problem lies in the detectors of the greenhouse gases (see my youtube presentation below): they all use thermoelectric – Seebeck Effect transducers. What John Tyndall discovered In 1859 are really only the thermoelectric gases, N2 and O2 are not; nor is CO2s 1338cm mode. All thermograms and IR spectrograms are created using these detectors, but they discriminate the non thermoelectric modes and substances.
Greenhouse theory is 19th century science, pre Quantum mechanics . Quantum mechanics predicts and explains the vibrational modes, emission spectra, of all the molecules in the atmosphere including oxygen and nitrogen; It also says all matter radiates infrared. All matter! That oxygen and nitrogen are assumed not to radiate IR is a contradiction to QM. Either Quantum mechanics, or greenhouse theory is wrong: Quantum mechanics is not wrong. To observe the predicted Modes of oxygen and nitrogen – at 1556cm and 2338cm respectively – we must use 20th century technology, Raman spectroscopy (the complement instrument to thermoelectric IR spectroscopy) .
In the paper that I’m about to publish I will show the Raman laser technology is equivalent to Thermoelectric IR technology. Raman can do it all: it measures temperature, and the concentration of the gases. It is even used to measure the Keeling curve, and is the instrument of choice on solar system space probes for this reason. It can also show nitrogen and oxygen emit and absorb IR radiation, in total compliance with thermodynamic laws Stefan Boltzmann equation and Boltzmann constant. I can even show, by experiment and application, nitrogen radiates upon CO2 to heat the CO2 – figure that.
The following YouTube presentation is my rough beginnings – I have since developed my theory.
With oxygen and nitrogen being greenhouse gases, greenhouse theory collapses. https://www.youtube.com/watch?v=T0IHKKkOwdU&t=860s Reinterpreting John Tyndall’s 1859 Greenhouse Gas Experiment with Thermoelectric and Raman Theory
aGrimm
November 19, 2017 7:15 pm
Pretty much any wavelength of radiation can create the photons that we sense as heat. For example, the 662 kEv photons from Cs-137 when stopped by a lead shield will cause the lead to get hot if there are enough photons. Saw this with a cask of 55k Curies of Cs-137. The exterior of the cask was hot to the touch. Here is how I describe the conversion of the 662 kEv photons to heat EM. A loss of energy of the initial photon occurs as it impinges or interacts with an atom’s nucleus or electrons in a mass. There are number of processes by which the initial photon losses energy, but with each interaction of the photon with the orbiting electrons or the nucleus, some energy is “lost”. Of course the energy is not “lost” it is divided into lower or a different energy potential/form. One such loss might be when an orbital electron gains energy from the initial photon and ‘moves’ to a higher energy “excited” state. Generally, the electron does not want to stay in this state and will emit the excess energy as a low energy photon – sometimes immediately, sometimes after a passage of time. The process of the initial photon losing its energy can involve thousands of ever decreasing interactions. Another interaction might be where a photon strikes an electron and the electron is ejected from the atom. The initial photon loses energy equivalent to the binding energy of the electron. If the ejected photon received energy in excess of its binding energy, then that photon has energy to give to something else.
Because the eye is a measuring device for visible photons it is useful for describing the process of a photon being eventually degraded to heat. The eye has evolved to manage a reasonable level of visible photons without damage despite the fact that some of the dissipated energy becomes heat photons (whose energy is also dissipated). However, staring directly at the sun introduces too many photons and the heat dissipation mechanism cannot handle it and the retina will get burned. A burn is basically the rearrangement of a molecule, e.g. a protein molecule, due to the input of too much energy, whereby the new arrangement does not work biologically. Physics and biochemistry are fun fields to study in combination.
I have a few nit-picky points in Gill’s assessment, but overall I agree with his assessment.
Claude Harvey
November 19, 2017 7:42 pm
“This is a contentious subject, and I have often shied away from it because it often erupts in food fights.”
I think the stewed tomatoes now dripping from the ceiling have confirmed your reservations, Mr. Watts. I do hope Mr. Gill was properly attired for the occasion.
And for those who claim that gravity or atmospheric mass or some other phenomenon is responsible for the fact that the Earth is much warmer than we would expect given its distance from the sun, let me recommend my post entitled “A Matter Of Some Gravity“.
Or not, you could choose to ignore everything but the things that support your view …
Willis, I still see “The Steel Greenhouse” as one of the best articles I’ve ever read on the so-called “greenhouse effect.” In discussions on this site I’ve referred others to it several times. Thanks again.
The problem Willis is the way we still teach physics. Almost everyone who doesn’t get it has the image of a photon as something like a tennis ball flying thru the atmosphere crashing into every molecule in it’s path. They don’t understand or won’t accept it’s a radio signal that collapses to something we equate to a tennis ball.
“I think you might class the distance to that big shiny thingy in the sky …”
Exactly. FWIW, Venus receives about twice as much solar radiation per square meter as does the Earth. However the Venerian albedo is higher. More incoming radiation is reflected back to space. Also Venus rotates very slowly compared to Earth and Mars which probably alters the heat distribution considerably.
I considered adding solar radiation at ToA for each planet, but the differences are small compared to the enormous temperature differences. Naturally you’d expect Venus to be balmier and Mars chillier than Earth, all other things being equal, but of course other things aren’t. Not even close. The other things outweigh insolation.
Don,
Venus’ rotation is clearly another factor. But, even considering all other things, the fact that Venus and Mars both have high-CO2 atmospheres highlights the importance of density and surface pressure. The temperature at the point in the Venusian atmosphere with terrestrial surface pressure is about the same as on Earth.
Right on. The steel greenhouse ignores the pressure temperature effect.
The “pressure temperature effect”??? As I have shown, and as Dr. Brown has shown, that is a fantasy. READ THOSE LINKS, and then come back and tell us why Dr. Brown and I are wrong.
David, while a Chinook wind raises the temperature at the surface because the air is descending … what goes down must come up. So somewhere else the same amount of air is going up and cooling, so that there is no net gain or loss.
And from the tone of your comment, it is clear that you have NOT done as requested, which was:
As I have shown, and as Dr. Brown has shown, that is a fantasy. READ THOSE LINKS, and then come back and tell us why Dr. Brown and I are wrong.
If you want to argue with my ideas, at least do me the courtesy of reading them before starting in … both Dr. Brown and I have proven, not asserted but proven, that there is no “pressure temperature effect” capable of warming an entire planet on a continuous basis.
“Venus possesses a dense, hot atmosphere, composed primarily of carbon dioxide. A surface pressure of nearly 100 bars sustains the mean surface temperature of 740 K, which is essentially globally uniform except for topographic effects.”
Actually, Venus’ atmosphere is a little warmer than average Earth temperature at one bar, thanks to a bit more incident solar radiation. But close enough to make the point. For government work.
Put another way, IMO Boyle’s Law ought roughly to apply, since, while not a closed system, the atmosphere approximates one since incoming and outgoing radiation are close to in balance, while subject to minor perturbances.
Thus, greater pressure should yield higher temperature. The atmosphere expands and contracts under heating or cooling, thus lowering or increasing pressure and temperature at the margins to maintain a fairly constant temperature, until other factors such as Milankovitch cycles and albedo waxing and waning change the baseline. This process can be extrapolated out to even denser or yet lower pressures.
What would be the temperature of Venus at the distance of Mars from the Sun?
I’m sorry, but there are far too many unknowns in the thought experiment to even begin to say. FOR EXAMPLE, at the surface of Venus CO2 is a superfluid … would it still be a superfluid at the distance of Mars? And how would that change things?
Please explain why the temperature in Venus’ atmosphere at one bar is the same as on Earth. Thanks.
According to your graphic, the temperature of Venus’s atmosphere at one bar is on the order of 350K … which is 77°C or 170°F.
Look, my rule of thumb is that when someone starts talking about Venus to try to show something regarding the greenhouse effect, I stop listening. Venus is a hugely complex system about which we know little. Consider the trouble we have explaining the Earth, about which we know orders of magnitude more than we know about Venus …
Much as I respect Dr. Brown, “proof” is a strong word, appropriate for math, not technically applicable to science.
Say what? “Proof” is indeed a strong word, and I do not use it lightly. However, there are assuredly parts of science where things can indeed be proven, and this is one of them.
However, if you think that Dr. Browns proof is incorrect, how about you point out WHERE it is incorrect, and leave the grand overarching view of what “proof” is and isn’t for another time.
Note that I apply the same word to my proof regarding the same question, which takes a totally different path to the same conclusion. And again, I invite you to find something wrong with my proof.
Happens around 0.1 bar. on every planet with enough atmosphere.
angech
November 19, 2017 9:23 pm
“Once you have GHGs in the atmosphere, of course, some of the surface radiation can get absorbed in the atmosphere. In that case, the surface radiation is no longer constrained, and the surface is free to take up a higher temperature while the system as a whole emits the same amount of radiation to space that it absorbs.”
–
Thank you Willis.
I have never really understood you but this comment is great.
Since the same energy is going out that comes in there is never a real accumulation of energy in the system , just a change in the temperatures of the mediums of that system, if true.
More energy in the atmosphere less in the land and sea perhaps?
Without the CO2 the land under the sun would be a lot hotter during the middle of the day and would send out lots of IR at a higher temp than the atmosphere does.
Leo G
November 19, 2017 9:27 pm
“For a molecule to “warm up” (have more kinetic energy) it needs its electrons to move to a higher, more energetic orbit. ”
The kinetic energy of a gas is the sum of the energy of the translational motion of the centre of mass of each molecule. Electron energies are not involved.
Belief in AGW caused by CO2 = denying or ignoring the science of thermalization, Maxwell-Boltzmann distribution of molecule energy & quantum mechanics. The IR energy absorbed by CO2 is immediately (0.0002 microseconds) shared with surrounding molecules (thermalization) so, at low altitude, there is little chance for a CO2 molecule to emit a photon as a direct result of having absorbed one (relaxation time about 6 microseconds). Water vapor has many (>190) significant absorb/emit lines at substantially lower energy levels than the 15 micron absorb/emit band for CO2 and on average there are about 35 times as many WV molecules as CO2 molecules. At low altitude, energy absorbed by CO2 is effectively rerouted up via water vapor radiation and some convection. End result is CO2 has no significant effect on climate. http://globalclimatedrivers2.blogspot.com
Belief in AGW caused by CO2 = denying or ignoring the science of thermalization, Maxwell-Boltzmann distribution of molecule energy & quantum mechanics. The IR energy absorbed by CO2 is immediately (0.0002 microseconds) shared with surrounding molecules (thermalization) so, at low altitude, there is little chance for a CO2 molecule to emit a photon as a direct result of having absorbed one (relaxation time about 6 microseconds). Water vapor has many (>190) significant absorb/emit lines at substantially lower energy levels than the 15 micron absorb/emit band for CO2 and on average there are about 35 times as many WV molecules as CO2 molecules. At low altitude, energy absorbed by CO2 is effectively rerouted up via water vapor radiation and some convection. End result is CO2 has no significant effect on climate.
“Now my house is not heated with the insulator, but it surely is warmer because of them.”
Only because the heat source (furnace) puts out more heat than can escape because of the insulation. The “warmth” of your home is dependent upon the heat source and the air temp outside (heat loss). It’s possible for it to get so cold outside that the insulation cannot keep the heat in with the furnace going full blast. Hence the insulation’s role is to slow the rate of heat loss. It doesnt make your home warmer, it makes it less cold.
It doesn’t make it warmer, it makes it less cold??? That’s just a pointless semantic exercise trying to deny the fact that the earth is warmer with the atmosphere than it would be without the atmosphere.
J. Richard, if you asked someone who just put on their jacket “Are you warmer now?”, do you think they’d say “Yes”, or that they’d say “No, I’m not warmer now, but I’m less cold”?
If you think the latter, here’s a quick guide to things that exist on a spectrum:
J. Richard, if you asked someone who just put on their jacket “Are you warmer now?”, do you think they’d say “Yes”, or that they’d say “No, I’m not warmer now, but I’m less cold”?
Coming from a cold and damp (Scotland) part of the world a suitable response to that question would be “I’m not as cold as I was” 🙂
“It is only these 26 Wm^-2 that are effected by the amount of GHG in the atmosphere. Convection moves much more heat”
No,it’s not. The diagram is a budget. It records the fluxes that can be measured, and tests whether they are in balance. The flux that is affected by GHG concentration is the full 324 W/m2 downflux from air. The 390 W/m2 upflux at the surface responds to the temperature there. The difference, if you exclude AW, is small at 26 W/m2, but is pretty much locked. The 324 W/m2 is emitted from air near the surface, as you can tell from its magnitude. If the surface warms, that air warms too, and the flux difference remains near constant
Not true, according to CERES. As one example, for the Northern Hemisphere, as the average temperature swings from about 9°C to 22°C (a 4% change min to max), the net LW flux at the surface varies between about 52 and 60 W/m2 … since that is about a 15% change min to max, I’d hardly call it “near constant” W.R.T. temperature …
w.
(As a side note, CERES puts the upwelling LW at 398 W/m2, and the downwelling at 345 W/m2).
Willis,
The Trenberth budget deals with annual averages. That gives plenty of time for the air at DWLWIR emitting altitude to equilibrate with surface, if the (annual average) surface warms.
That makes no sense. You’ve said that “if the surface warms, that air warms too, and the flux difference remains constant”. You CANNOT determine that from a budget, whether Trenberth’s or someone else’s. A budget has no information at all about a “warming surface” …
Willis,
No, you can’t determine it from a budget. But you can reason about time scales. You are referring (I think) to a phase lag between surface and downward emitting layer temperature with the seasonal cycle. Averaging over a year is firstly a somewhat longer timescale, but also averages out the effect of such a seasonal lag. Basically the difference between upflux and down is determined by the temperature difference of emitting layer and ground temperatures, which is determined by lapse rate and GHG concentration. The latter fixes the altitude of the emitting layer.
Willis,
You might like to try this test with CERES (I am not so adept with it). Just look at different areas around the world. There will be big changes in upflux and down. But is it not always the case that upflux always slightly exceeds down, as they vary over such a range?
Willis,
You might like to try this test with CERES (I am not so adept with it). Just look at different areas around the world. There will be big changes in upflux and down. But is it not always the case that upflux always slightly exceeds down, as they vary over such a range?
Nick, interesting question. Here’s surface upwelling longwave minus surface downwelling longwave, in a couple of formats.:
As you point out, net flow is always from the surface upwards.
Willis,
Just adding some interpretation – I think the up-down difference is basically product of altitude of downward emitting layer and lapse rate. The dry lapse rate is higher than moist. And your map looks pretty much like a humidity map, except at poles, where flux in both directions is smaller.
Willis,
Further thought – altitude of downward emitting layer is dependent on GHG concentration – it is lower with more GHGs. So higher if water vapor is low. That adds to the difference being greater in dry places – it could be the main cause.
Martin A
November 20, 2017 12:20 am
The temperature of a body (gas, liquid or solid) directly affects the wavelength of the radiation it emits and absorbs.
I agree with your findings, but not for the same reasons. Retired now, I used to be a laser engineer, and instrument design engineer, including weather stations, and all manner of energy measuring devices. I created infrared measurement systems and laser energy measurement systems. Thus, I know a little about gas behavior and thermal characteristics.
So, my argument against CO2 ‘warming’ anything goes like this? If it DID increase the surface temperature, the surface would radiate to space at the 4th power of the increase, instantly cooling itself back down. We know that everything radiates and we have laws for quantifying it.
Experiment: Try to heat a stove with a flashlight. There are lots of photons, you can see them. Yet the stove does not warm.
Say what? If you shine a flashlight at a stove, it will end up warmer than without the flashlight. Not much warmer, but assuredly warmer. Look, John, a good chunk of the light from the flashlight will be ABSORBED by the stove, and the rest will be reflected.
Now, since energy cannot be either created or destroyed, what happens to the energy in the photons that are absorbed? Obviously, that energy must be changed into something … let’s see. It’s not converted to electricity. It’s not converted to chemical action. It’s not converted to mechanical energy … gosh, could it be converted to thermal energy?
And if (as you claim) the absorbed light is NOT converted to thermal energy … then just what are you claiming it is converted to?
You continue:
Why? Because for each photon that strikes the stove, the stove emits millions, maybe billions of photons itself. Those few photons from the flashlight are simply overwhelmed, and the result is immeasurable.
First, photons don’t get “overwhelmed” by photons going the other direction. If you are indeed an engineer you know that to be true. You can’t “overwhelm” a flashlight by shining fifty flashlights at it, that’s nuts.
Second, the resulting change in temperature in the stove is small. However, it is not “unmeasurable”, which you also know is true if you are an engineer.
Finally, your explanation that if CO2 “DID increase the surface temperature, the surface would radiate to space at the 4th power of the increase, instantly cooling itself back down” makes no sense at all. According to that theory, nothing can increase the surface temperature, because if it did the surface would “instantly cool” …
Willis, you’re close to right there, when you try to haul the lower atmosphere up by its own bootstraps, a humourous definition of futility. Any warming would indeed cause expansion by molecules moving at c. 1km/sec in the gas phase. The gas laws do rule, so the IR catastrophe will not happen. Ditto the never-observed ghe.
Cold objects must warm hotter objects, or even more strongly stated, heat the warmer object
Is this true?
In this case the fire and the light bulb
However the general truth comes out that is if the two objects are thermally isolated from their surroundings heat only flows from the higher temperature because that is the definition of HEAT
For example
Two metal blocks A and B sit separated inside a vacuum filled adiabatic enclosure.
Adiabatic enclosure consists of a perfect reflector face surrounded by a perfect insulator
Initially both at the same temperature. The zeroth law of thermodynamics applies.
Both emit and absorb equal amounts of radiation.
Neither one is said to heat the other.
Both objects remain at the same temperature
One block (A) has a power supply which is now switched on causing the temperature of the block to rise.
This in turn means that it will emit more radiation.
A will now heat B causing its temperature to rise.
B will in turn emit extra radiation which is absorbed by A but this ‘back’ radiation is caused by A.
Now comes the clincher
If B were not there at all the temperature A would be even higher.
So B cannot be said in any meaning of the word as a cause of heating A
in the case of the furnace(Tf) and light bulb(Tb)
If Tf >Tb heat will flow from furnace to bulb
If Tf <Tb heat will flow from bulb to furnace
You whole answer is so crazy you do realize you aren’t dealing directly with heat but with EM wave. We call it a thermal emission not because it is “hot” or “heat” but because the source of emission is from a thermal source. Don’t think of the emission as heat it’s just EM energy.
Lets give you this with lasers which are the most precise EM frequencies I can generate.
So I fire a CO2 infrared laser and a red laser at each other.
We know the beams will pass thru each other without interacting and in fact on a laser cutting machine we often use this so you can see where the cutting in what we call that a red dot pointer. The IR frequency is invisible to us but you inject the red laser thru one of the mirrors so it follows the path of the IR.
So got it I have an IR laser one way and a red laser beam the other way.
What happens when each beam hits the other laser device (and they will hit)?
No Willis I am absolutely correct
Before the power switch on A was turned on block A and block B were at the same temperature.
The adiabatic walls confines all energy within that enclosure
Lets say each has a mass of one kilogram
With both A and B the power source has to heat 2Kilograms
With B removed all the power source has to heat one kilogram which implies the temperature of A will rise twice as fast as before
Any physicist will confirm that I am correct
Bryan, your case is so specific that it has almost nothing to do with climate.
“Adiabatic enclosure consists of a perfect reflector face surrounded by a perfect insulator”
so you have created a situation where every last bit of radiation leaving the blocks gets reflected back perfectly. This is the exact opposite of the earth’s situation, where every last bit of radiation that leaves from the top of the atmosphere disappears and never comes back. If no energy can leave, then the two blocks will reach some equilibrium where both have the same temperature.
“If B were not there at all the temperature A would be even higher.”
While strictly true, this is not really very interesting. Basically all you have claimed is that an isolated, insulated system with mass “m” warms faster than an isolate, insulated system with mass “2m”.
I would challenge you to re-analyze with a system where the enclosure has perfectly ABSORBING walls and is held at, say, 3 K. You will find that in this more useful system, Block A does indeed warm faster than without block B, and Block B ends up at some temperature between that of Block A and the cold walls.
Hi tjfolkerts
My post was to correct the impression that the presence of any object irrespective of its temperature must ‘warm’ some neighbouring object.
This piece of fiction has become so prevalent in the last 20 years that normally rational folk like Willes have been blindsighted by it.
My example was to remind people that the surroundings need to be specified before any progress is made in analysing the flow of heat.
My scenario would be found in opening chapters of a thermodynamics book where definitions of adiabatic and diathermic walls, heat transfer and so on are introduced.
I am sure that ‘warming’ by cold objects meme has been carefully fostered by global warming advocates.
Look at some earlier comments upthread .
Block of ice in sitting room with person
Torch heating a furnace
Surely the Earths atmosphere in which radiative resistance rather than reverse heating gives a rational explanation of the situation and should be encouraged.
Its up to people who should know better to get back within the framework of physics
I agree with your point that it is valuable to examine all details of a system, and to be precise about language (the word “heat” being perhaps the biggest language offender). Your scenario certainly could be worked out with some basic undergraduate thermodynamics.
My main issue with your scenario is that it is SO far removed from scenarios related to the thermodynamics global warming that it could easily give a false assurance to people who simply want a post to confirm their biases. For planets, the default condition is blackbody surroundings at 3K. This is almost exactly the opposite of what you hypothesized.
I think it would be more productive to directly address issues like “warming with ice”. The radiation from cold things still feels warmer (and will help keep you warm) when compared to radiation from even colder things. If I am facing a wall of dry ice @ur momisugly -79C and then someone interposes a wall of ice, then yes, I will feel warmer. A pot of water with an immersion heater will rise to a higher temperature when the radiation from the warmer ice replaces the radiation from the colder dry ice.
tjfolkerts
The block of ice in the sitting room example.
Everyday common sense tells people its not a good idea for keeping the house warm.
The walls floor and ceiling and air at around 20C (surroundings) will lose heat in melting the ice by conduction convection and radiation.
The fact that the ice block radiates to the wall is true but will not change the outcome .
If the surroundings were dry ice as you say it would be the ice block that would lose internal energy by heat transfer to the dry ice and so it would get colder.
Once the surroundings are specified it is much easier to keep track of the heat flow.
In reading posts on Global warming quite often only two items are mentioned
The hotter and colder object ( with no mention of surroundings).
Then comes an astonishing statement like….
Do you know that the cold object ‘warms’ the hotter object
It always seems that the main offenders are global warming advocates.
Also not all photons are created equal.
The ‘quality’ of the radiation was mentioned in the theme title but not discussed much any further.
Bryan says” In reading posts on Global warming quite often only two items are mentioned
The hotter and colder object ( with no mention of surroundings).”
Any decent discussion will include FOUR items
1) the warm object that is absorbing energy (eg the ground)
2) the source of energy (eg the sun)
3) a low temperature heat sink (eg 3 K outer space).
4) the object that is not absorbing energy from Item 2 but can absorb radiation from Item 1 (eg the atmosphere
Perhaps you were reading poor post where the authors did not really understand the GHE. Perhaps the authors figured the sun and outer space were simply givens that did not need specific mention. In any case, when all four items are included, it is very simple to see that adding Item 4 results in Item 1 getting warmer.
One place where intuition often goes astray is the “low temperature heat sink”. Normally our surroundings (around 20 C typically) are not all that cold. Then adding a wall of ice that is held at a steady 0 C (even colder than the surroundings) certainly will not warm you. But that would be like adding an atmosphere that is held steady at a temperature BELOW 3 K!
tjfolkerts: you state ” In any case, when all four items are included, it is very simple to see that adding Item 4 results in Item 1 getting warmer.” There is no atmosphere on the moon. Sunny side of it gets much higher temperature than the surface of the earth. How come?
esalil, this is a discussion of broad averages. The average surface temperature of the earth is MUCH higher than the average surface temperature of the moon. This despite the fact that the Moon absorbs a higher fraction of the incoming light.
Furthermore, the moon’s average temperature would rise if it had an atmosphere of GHGs (although the max temperature on the sunny side would probably drop a bit.
WUWT is an equal opportunity science “d@nial” site, except for “Sl@yers”. Those who d@ny gravity in favor of an imaginary “electric universe” are welcome here, along with those who d@ny EM radiation. Even creationists are permitted to comment, but not those skeptical of the GHE.
People skeptical of GHE are “not permitted to comment”? Say what???
READ THIS VERY THREAD. There are lots of people skeptical of the idea of GHE, and they are free to post, as are you. Your claim is a joke.
For me it looks like hopeless to explain the basic physics about the GHE for those who think that it is nonsense. I try another approach. If you think that the GHE does not exist, how do you explain the downward LW radiation at the surface which is about 345 W/2. Compare this to the direct solar radiation of 167 W/2 at the surface. (71 W/2 of solar radiation is absorbed by the atmosphere and therefore the outgoing LW radiation is totally 238 W/2). These figures are not nonsense, because they are both measurement based figures.We know that 99.97 % of the energy keeping the Earth warm originates from the Sun. Where this 345 W/2 comes from? How do you explain this, if there is no GHE?
It comes from the atmosphere (including clouds). The atmosphere radiates the energy absorbed from the sun and the surface. The energy from the surface is mostly nonradiative (evaporation and convection). This does not mean that the inceased atmospheric CO2 will result in a warmer surface.
There is a ghe, simply because there is an (insulating) atmosphere between the surface and the cold space. The atmosphere is in direct contact with the surface and is easily warmed by it, mostly by nonradiative processes. The bulk of the atmosphere (N2 and O2) plays a big role.
edimbukvarevic. Otherwise very good but you say that “The atmosphere is in direct contact with the surface and is easily warmed by it, mostly by nonradiative processes.” This is not correct: The upward LW radiation emitted by the surface is 396 W/2 (Excatly according to Max Planck’s formula). Latent heat flux 90 W/2, and thermal flux 25 W/2, together 115 W/2. According to basic mathematics 396 is more than three times greater than 115. Right?
Aveollila, the radiative heat flux surface-to-atmosphere is ~26 W/m2 (350 – 324). According to basic mathematics, that is less than 25% of the non-radiative fluxes (115 or 78 + 24 = 102 W/m2). It is only one fifth of the net surface-to-atmosphere heat flux (~128 W/m2).
edimbukvarevic You do not calculate the upward energy fluxes in the right way, because you subtract a donward flux from the upward fluxes (why you selected 324 and not the 168 ??). According to the energy balance of Kiehl an Trenberth (which is obsolete and based on the wrong atmosphere) the upward fluxes are 350 + 78 +24 = 492 and the downward fluxes are 168 + 324 = 492. That is called an energy balance. Your way of comparing fluxes is not based on the real world. Using your way, I can prove anything as I like.
Aveollila, I commmented on the atmosphere and its energy. In particular, the heat exchange between the atmosphere and the surface. The total heat transfer from the surface to the atmosphere is about 128 W/m2. Only a small part of this flux is radiative, about 20% (350 – 324 = 26 W/m2). This is not debatable, unless the Earth’s budgets are grossly inaccurate.
Furthermore, if we add solar radiation directly absorbed by the atmosphere (~67 W/m2), we get 195 W/m2. That means only ~14% of the total heat transfer to atmosphere is LWIR radiation.
Martin Mason
November 20, 2017 1:56 am
As always very confusing with so many different opinions, immovable positions and even contempt for anybody who doesn’t hold the same opinion. I’m an engineer but I’m constantly searching for an answer to how a cold body heats a warm body and how heat is apparently created from nothing.
Please allow me to ask a dumb question and hope that I can get an answer to at least help clear up some of my confusion. I can calculate solid surface temperature of the earth from incoming UV radiation (from a warmer body) and physical properties (absorptivity and emissivity of the surface). I can calculate a near surface air temperature based on conduction and convection. I can then calculate a lapse rate which gives me a vertical temperature profile which is pretty well matched by reality. I can do this with no reference to downwelling radiation. Why should the conclusion from this not be that this is because downwelling radiation doesn’t affect temperature and lapse rate?
Martin. Solar radiation is much more than UV radiation which has a wavelength shorter than the visible light.
I do not believe that you can show calculations that the solar radiation of 238 W/2 absorbed by the Earth produces the Earth’s surface temperature of 15 degrees Celcius. Why not? Because that absorbed radiation flux of 238 W/2 corresponds the surface temperature of -19 Celsius only as measured at TOA emitted by the Earth.
Nobody has shown that Max Planck’s equation is wrong. It shows excatly how the surface temperature produces an exact amount of radiation. The fact is that the surface has an average temperature of 15 C and it produces the radiation flux of 396 W/2. How it is possible that the Earth receives only 238 W/m2 but its surface can emit 396 W/? Perpetum mobile? Confirmed by measurements. Fighting against GHE like fighting against wind mills. You cannot win.
ave, the calculation for spot temperature is basic radiant heat transfer and is based on actual radiation not average hitting a flat plate and it doesn’t have an additive downwelling radiation component for it to work. The problem is that if you believe that downwelling IR is the only way that the surface can achieve it’s effective emitting radiation then you have to have 324 W/m2 of downwelling radiation that is higher than the sun gives us and a transfer of heat from cold regions to warmer ones.
If downwelling radiation is a key component in atmospheric temperature why is the lapse rate completely free of any influence from it. Why is Jericho warmer than Jerusalem or the temperature on a mountain lower than at the bottom.
Is it not so that any number of experiments of putting bodies of different temperature in proximity, the temperature of the highest temperature body is never increased by radiation from the lower temperature bodies (true?) no matter how many there are.
I believe that heat energy (as opposed to radiation) can never flow from a cold body to a warmer one at any level including quantum and once this is accepted, heating by downwelling radiation can’t be significant. No heat is added and none is trapped, no laws are broken
Maybe those who say that it is an atmospheric effect rather than a greenhouse effect could be correct. These wonderful gases though for sure moderate the temperature of the atmosphere but add heat to it?
To sailborder. Create a pressure of 100 atmosphere into a steel cylinder and leave it outside in the temperature of -10 degrees Celsius. What do think is the temperature after 2 hours. It is that -10 C degrees. A constant pressure cannot create heat into any system. Only if you do like in a diesel engine: apply a certain amount of mechanical work and turn it into heat.
“I believe that heat energy (as opposed to radiation) can never flow from a cold body to a warmer one at any level including quantum and once this is accepted, heating by downwelling radiation can’t be significant. No heat is added and none is trapped, no laws are broken”
What is so difficult to understand about the concept of slowed cooling?
Of course heat energy can impinge on a warm body from a colder one.
Photons from the colder object do not have any concept of the other object being hotter.
(and there will be photons unless it is at 0K)
And as a result the hotter body cools more slowly.
There is however no “Flow”, as in a net impingement of photons from the cold object such that it raises the temp of the warmer one.
It is like a tank loosing water at a rate of 10 gals/hr while at the same time you pour water from a bucket into the top at the rate of 1 gal/hr.
So the tank is actually leaking water at 9 gals/hr. It’s still leaking (cooling) but at a slower rate due to the small amount of water (energy) being added to it from the bucket (older object) leaking (cooling) at 1 gal/hr.
It is like a tank loosing water at a rate of 10 gals/hr while at the same time you pour water from a bucket into the top at the rate of 1 gal/hr.
So the tank is actually leaking water at 9 gals/hr. It’s still leaking (cooling) but at a slower rate due to the small amount of water (energy) being added to it from the bucket (older object) leaking (cooling) at 1 gal/hr.
I think this is probably the best visualization to explain what I’ve been talking about.
So, you have a big tank, with 2 holes in it. and over about 12 hr’s once a day, a big dump of water happens, pressure is a little higher, so it rushes out a bit faster at the peak level. One hole is the optical window, which is constant in size, and another that represents the energy through the other bands, part of this hole has grown slightly smaller from increases in GHG’s.
But together it’s a good size hole, and then the tank goes 12 hours between big dumps of water.
But, there’s a second smaller tank of water that is filled constantly, from far away.
And when the water level drops some, depending on the level of the second tank, some of the water in the second tank starts to flow into the bigger tank. This slows the drop in water level in the big tank, until the next big fill of water, where it shuts off till later.
If the second tank is really full, the big tank gets as much from it, as goes out the bottom once the valve opens, and the level doesn’t drop anymore, even though the main fill line is dry still.
This is why Co2 doesn’t have hardly any affect on the level. It’s the sheer size of the tanks compared to the tiny 3.7W.
“Unless the so called GHE is just the pressure density effect?”
Yes your bike tyre heats up as the air you pump into it increases in pressure.
But does it stay hot for you forever?
Obviously not.
Why?
You did the ‘work’ with the pump and then you stopped – then no work so the tyre cooled.
Now replace the ‘pump’ with gravity and the air by the Earth’s atmosphere.
(Hypothetically) if we could just switch on gravity from zero then yes the atmosphere would heat as it compressed.
But the work has then been done.
So it cools.
Gravity is NOT maintaining a surface temperature.
Full stop.
It does however set the LR by virtue of the -g/cp relation – the surface temp then being set by the GHE via raising the effective temp of Earth (255K) up to ~8km whilst the LR is maintained.
Rod
I believe that the simplest form of the question you ask is this:-
“Can something cold heat up something hot?”
To answer this question we need to establish the nature of collision:-
Can a slow moving object travelling behind ever catch up to and collide with a fast moving object travelling ahead of it and moving in the same direction?
Well of course not, only fast moving objects can ever catch up and collide with slow moving objects ahead. This is because collision is a vector process; fast objects collide into slow objects and not the other way round and so the transfer of momentum always proceeds from fast to slow and never from slow to fast (for objects moving in the same direction).
The issue of heating by “back radiation” is more subtle. The diagram that Anthony supplied demonstrates Wein’s displacement law and it shows the envelope curve for frequencies at different black body temperatures. In addition we have all seen the diagram that shows the effect of selective absorption of frequencies by greenhouse gases. What we have never seen is the diagram which shows an intensity mound in the low frequency tail above the envelope curve which is the absolute requirement for “cold” low frequency radiation to add energy to a high temperature body. This does not happen, it never has and it never will. Back radiation heating of the warm surface below by a cold surface above is a fiction and Wein’s displacement law proves this.
The radiative greenhouse effect is an attempt to explain how the low temperature isothermal stratosphere can heat a planet’s surface. It cannot. The troposphere (the weather layer) is a gigantic heat engine. It moves prodigious amounts of mass and energy all day and every day. Our atmosphere does work and it is powered by the heat of the sun and the speed of the Earth’s daily rotation. Like all heat engines it has an exhaust temperature, that exhaust temperature is observed in the isothermal shell of the stratosphere that surrounds our planet. Just as the exhaust temperature of an engine cannot be used to power that same engine, so it follows that the low temperature stratospheric shell aloft cannot provide back radiation “heat” to power the high temperature weather machine below.
So, Phillip, at night does a cloud overhead affect the temperature at the surface below and why as it is not cutting off any sunlight. Clouds are composed of GH effect water molecules at low temperature or do you have an alternate explanation?
angech,
I live in the British Isles. Almost every day somewhere here experiences the effect of warm moist cloudy air moving over night time cooled ground. My worst ever driving experience occurred a few years ago in Aberdeenshire when I lost control of my vehicle on black ice. One frosty winter’s evening I walked our dog alongside the River Don under crystal clear skies. The next morning, before dawn, the weather had changed to a mild 8C, with damp moist air, a thick haar (sea fog) and very poor visibility. As I drove south to work alongside the river I came to a small hill, the north facing slope of which was covered in black ice as moisture from the sea fog condensed onto the previously cooled road surface, so yes warm moist cloudy air can heat a cold ground. The question at issue however is can something cold heat up something hot?
I think that this is due to a change in wind direction rather than a change in the vertical temperature profile.
Certainly in Scotland we notice abrupt daily and hourly change.
If the wind is from south or west we have warmer weather.
From the north or northeast its much colder.
” so yes warm moist cloudy air can heat a cold ground. The question at issue however is can something cold heat up something hot?”
Yes and no.
I was meteorologist with the UKMO in a previous existence and one of our winter jobs was to monitor road temps over a wide part of the English Midlands for councils re gritting for ice/snow.
It is no mystery that cloud raises RST’s and hence air temps above.
It is added to the algorithms for RST prediction. On a otherwise frosty night it can make for a marked jump in road surface temp. In fact thin Ci cloud at 30,000ft at a temp of -40C will cause a road temp to either to halt or rise slightly when below 0C.
But it is NOT strictly raising it’s temp, as you will discover if you examine, say, a bridge in the same situation (air beneath).
What is happening is that the heat-flux from below the surface overcomes the surface emission to space (because of the cloud) and so the temp there rises.
In other words extra photons have impinged the road and added to it’s energy such that that entering from below is out of balance with the LWIR leaving.
It is though still a slowing of cooling and NOT cold heating up something hot.
Very interesting observation, thanks. I have often noticed that ground frost will form first on the metal skin of a car, before it sets into the pavement. Same principle I believe. I had never experienced black ice until that morning and had no idea how dangerous it can be, even with an overnight air temperature rise to well above freezing it is still possible to get into severe trouble on damp untreated roads.
I totally agree…… though I approach it from a heat capacity angle. The best the GHG can do is reach equilibrium with the ocean or ground …. simply because the GHG mass and heat capacity is minuscule compared to everything else.
Philip Mulholland November 20, 2017 at 6:52 am
It is standard knowledge that cloudy skies will be warmer than cloudless skies due to the blanketing affect of clouds. Surely it could also be that the air arrives already warm and clouds are part of the ‘package’? Here in the UK the majority of weather and associated temperatures come from somewhere else, either pre-heated or pre-cooled. However, we have cloudless days that can be very hot or very cold depending on the time of year. I understand the reason for this is that cloudless days tend to have low wind speeds as this is usually a high pressure weather system. In the summer when the sun is overhead the surface gets the maximum effect of the sun’s heat and the low wind speed does not remove this heat, much like a car can overheat if not moving. Conversely, with high pressure during the winter when the sun is week there is only slow out-flowing air and no in flowing warm air. Therefore, there are other considerations than just a cloudy sky keeps the temperature in.
Out of interest Adolf Galland gave four reasons why the Luftwaffe lost the Battle of Britain; number 1 was the unpredictable nature of Britain’s weather.
“The radiative greenhouse effect is an attempt to explain how the low temperature isothermal stratosphere can heat a planet’s surface.”
It doesn’t “heat it up”, you turkey, it reduces the rate of cooling!
Large amount of radiative energy going OUT. Smaller amount of radiative energy coming BACK. Result – less energy overall leaving.
So, with the Sun heating the ground, and the ground losing heat at a lesser rate than it would in vacuum, the ground gets warmer than it would in vacuum. It reaches equilibrium at a higher temperature.
Voila! Greenhouse Effect! Bleeding simple!
(No doubt someone is going to “debunk” this by nattering on about photons, or Wein, or by saying that the Earth is *never* in equilibrium. (Which it isn’t. And your point is?) Let someone else argue with them. Never debate with a fool, people might not be able to tell the difference.)
So, with the Sun heating the ground, and the ground losing heat at a lesser rate than it would in vacuum, the ground gets warmer than it would in vacuum. It reaches equilibrium at a higher temperature.
Here’s a thing; the maximum surface temperature measured on the Moon in a vacuum is 100C (373K) but on the Earth the maximum recorded surface temperate with an atmosphere of 100 kPa pressure is 58C (331K). The Moon orbits the Sun in tandem with the Earth at the same overall distance (Yes its absolute distance changes from New Moon to Full Moon and then back again) but, averaged over a year, the Moon experiences the same intensity of solar radiation as the Earth does.
So what do I conclude from this? That the sun heats more in a vacuum and that on Earth the atmosphere cools its surface.
Here’s a thing; the maximum surface temperature measured on the Moon in a vacuum is 100C (373K) but on the Earth the maximum recorded surface temperate with an atmosphere of 100 kPa pressure is 58C (331K).
The Moon orbits the Sun in tandem with the Earth at the same overall distance (Yes its absolute distance changes from New Moon to Full Moon and then back again) but, averaged over a year, the Moon experiences the same intensity of solar radiation as the Earth does.
So what do I conclude from this? That the sun heats more in a vacuum and that on Earth the atmosphere cools its surface.
Right facts, but I fear you’ve reached the wrong conclusion. It’s more complex than that. I discuss this question in a post called “The Moon Is A Cold Mistress” …
So, with the Sun heating the ground, and the ground losing heat at a lesser rate than it would in vacuum, the ground gets warmer than it would in vacuum. It reaches equilibrium at a higher temperature.
So, with the Sun heating the ground, and the ground losing heat at a lesser rate than it would in vacuum, the ground gets warmer than it would in vacuum. It reaches equilibrium at a higher temperature
Do you want to correct him or not?
I’m not sure what I have to do with this, as I haven’t commented on what Uncle Gus said. However, it is true that ceteris paribus, the surface with a GHG-containing atmosphere will indeed be warmer than it would be without said atmosphere … so it is not at all clear what you think I should “correct”.
so it is not at all clear what you think I should “correct”.
Try reading this:-
the ground gets warmer than it would in vacuum
Thanks for the reply, Philip. So your contention is that if we removed the earth’s atmosphere the surface would get warmer??? Seriously?
Stefan and Boltzmann disagree strongly with that … in a vacuum, the earth’s maximum possible temperature would be 5°C, assuming the earth were a superconducting sphere. Given that it is not a superconducting sphere, the temperature would be lower. For example, the moon, at the same distance from the sun as the earth, has an average temperature far below 0°C, because it rotates so slowly …
Willis,
One last time, I am talking about peak maximum temperature. In a vacuum, where there is no fluid (gas) to cool the surface the peak maximum temperature is higher under an equivalent radiation load.
Willis,
One last time, I am talking about peak maximum temperature. In a vacuum, where there is no fluid (gas) to cool the surface the peak maximum temperature is higher under an equivalent radiation load.
You may well have been talking about that, Philip.
But Uncle Gus, who we were discussing, definitely was NOT talking about that. You asked me, was there anything I would correct about the statement of UNCLE GUS, not about your statement … and he said nothing about max temperatures, he was talking about mean temperatures.
And what is with the “One last time …”, like you’re at the end of your tether and I’m too dumb to understand but you’ll give me one last chance. I don’t care which end of the tether you’re at, I was replying to your question about Uncle Gus.
w.
PS—the reason for the difference in max temperature of the earth and the moon is not the vacuum. It is a result of the fact that their rotation rates are different. Because the moon’s surface can cool or heat for a longer period, the cold side gets much colder and the warm side gets much warmer than the earth.
We know this because if the moon were spinning at say 1 rpm, where it would be evenly heated, given the moon’s albedo (0.12), the S-B equation says it would be at about -4°C. So clearly, the vacuum isn’t the reason for the high temperatures.
Willis,
Dinna fash. Here (I hope) is something we can agree on, slowly rotating planets heat more than rapidly rotating ones do.
pochas94
November 20, 2017 4:57 am
At thermal equilibrium, any time a molecule absorbs a photon, it will emit one (follows from Kirchoff’s radiation law.) There is an equal probability of emitting upward or downward. The greenhouse effect results because the sun / earth /space system is not in thermal equilibrium and an upward emitted photon may escape to space, while the ones emitted downward remain in the system. Missing from this discussion is convection, which controls surface temperature and indeed the whole temperature profile of the troposphere. Discussion of lapse rate and equivalent emission height is also missing. Also, the great source of stability of our climate, the Stephen-Boltzmann law which says that radiant emission goes as the fourth power of temperature, with the result that any increase in temperature is opposed by a massive increase in energy radiated to space.
Ian Macdonald
November 20, 2017 5:09 am
Infrared photons have far too little energy to change the orbit of an electron. The ‘greenhouse effect’ is entirely due to resonances in the elasticity of the chemical bonds between atoms in the gas molecule.
The 15um resonance is below the emission peak for typical ground temperature. However the photon distribution is wide enough that some will be in this range. Thus CO2 will have some effect on outgoing radiation.
When a CO2 molecule absorbs a photon, it may re-radiate it in a random direction, or it may transfer the energy to the bulk gas by way of colliding with a N2 or O2 molecule. Exactly which mechanism will predominate is hard to predict. The time interval of each is about the same, at 10ns.
N2 and O2 not being IR emitters, essentially have no way of liberating heat to space. However, if there is CO2 in the upper atmosphere, molecular collisions with this may provide a route for heat to escape to space, by way of exciting IR emission.
Hence the overall situation is far from simple, and it may even be that the presence of CO2 cools the upper atmosphere.
Perhaps more importantly there are numerous proofs of the fact that at sea level, CO2’s effect is already evident at very low concentrations, therefore further increases are unlikely to significantly increase its greenhouse effect. However at high altitudes this may not be the case. If so, adding CO2 to the atmosphere might actually cause planetary cooling rather than heating, since it will have little effect at sea level but will enhance the ability of the upper atmosphere to lose heat to space.
I’ve done a number of analyses of this on our website. For example:
Ian Macdonald November 20, 2017 at 5:09 am
Infrared photons have far too little energy to change the orbit of an electron. The ‘greenhouse effect’ is entirely due to resonances in the elasticity of the chemical bonds between atoms in the gas molecule.
The 15um resonance is below the emission peak for typical ground temperature. However the photon distribution is wide enough that some will be in this range. Thus CO2 will have some effect on outgoing radiation.
The 15 𝝻m resonance is very close to the emission peak at 288K.
When a CO2 molecule absorbs a photon, it may re-radiate it in a random direction, or it may transfer the energy to the bulk gas by way of colliding with a N2 or O2 molecule. Exactly which mechanism will predominate is hard to predict. The time interval of each is about the same, at 10ns.
The mean time between collisions in the lower troposphere is ~0.1ns the re-emission time is orders of magnitude longer.
When you say N2 and O2 cannot liberate ‘heat’ to space, are you saying they do not emit energy? I keep hearing that N2 and O2 do not absorb or emit IR, but I’m also aware that any matter above 0 Kelvin emits radiation at a rate determined by its avg temp. If an object or region of space emits radiation then it’s temperature decreases whether that energy is IR or not.
Given that N2 and O2 make up about 4950 Trillion tons of the total ~5,000 Trillion tons that comprise our atmosphere, it seems fairly obvious that the energy being emitted by N2 and O2 is where most of the energy is lost from the atmosphere radiating.
Steven Morris November 20, 2017 at 2:01 pm
When you say N2 and O2 cannot liberate ‘heat’ to space, are you saying they do not emit energy? I keep hearing that N2 and O2 do not absorb or emit IR, but I’m also aware that any matter above 0 Kelvin emits radiation at a rate determined by its avg temp. If an object or region of space emits radiation then it’s temperature decreases whether that energy is IR or not.
Does not apply to gases which can only emit at those wavelengths which correspond to allowed transitions between energy levels of the molecules. The blackbody relationship provides an upper limit to such emissions, there are no significant allowed transitions for either N2 or O2 within the range of frequencies emitted by a blackbody at the earth’s temperature.
Given that N2 and O2 make up about 4950 Trillion tons of the total ~5,000 Trillion tons that comprise our atmosphere, it seems fairly obvious that the energy being emitted by N2 and O2 is where most of the energy is lost from the atmosphere radiating.
Notice that the absorption/re-emission band of CO2 ranges from about 13 to 17 microns, corresponding to a temperature range of -51 to -103 degrees C, i.e., lower than is normally experienced at Earth’s surface (which corresponds to a range of about 9 to 13 microns), hence how could CO2 possibly behave like a greenhouse gas in the Earth surface environment? This is why infrared astronomer Michael Sanicola was able to report finding no back-radiation from CO2 that could interfere with his astronomical observations.
davidbennettlaing November 21, 2017 at 5:55 am
Notice that the absorption/re-emission band of CO2 ranges from about 13 to 17 microns, corresponding to a temperature range of -51 to -103 degrees C
There is no such correspondence. Blackbody radiation at the earth’s temperature peaks at 550cm-1 for 288K (peak radiance 0.136 W/m2/sr/cm-1) whereas the CO2 absorption band is centered at 667cm-1 (radiance ~0.132 W/m2/sr/cm-1). So CO2 absorbs near the maximum of the earth’s BB energy distribution (~97% of max).
By the way you shouldn’t believe anything you read on Steve Goddard’s site, not even the names!
There is no such correspondence. Blackbody radiation at the earth’s temperature peaks at 550cm-1 for 288K (peak radiance 0.136 W/m2/sr/cm-1) whereas the CO2 absorption band is centered at 667cm-1 (radiance ~0.132 W/m2/sr/cm-1). So CO2 absorbs near the maximum of the earth’s BB energy distribution (~97% of max).
When you look at the whole spectrum, being near the maximum, isn’t all that significant.
And remember water overlaps co2.
Here’s just co2
and Water
But, I think that as water condenses at night in the troposphere, that sensible heat, that IR will light up co2 @15u. And co2 might light up during the day, but IIRC that blocks about as much as it adds during the day. That why the focus on Min T. But they’re completely wrong about Min T, it just follows dew point, which follows the ocean cycles, and since air temps respond differently during the day over ocean than it does land, and the hemispheres are asymmetrical to land area, global Temp averages are going to cycle with the ocean cycles.
Then, what I found shows why co2 will never for the foreseeable oil reserves become to high for the atm water to not regulate Min T. If we doubled Co2, added another 3.7W/m^2, there’s just be ~3.7W/m^2 decrease in the complementary water vapor forcing. It condenses as much water vapor as it can, to try and stay above dew point. And it looks like it’s supplying 40 or 50W/m^2, That’s a lot of doubling.
Not to say 2,000ppm is acceptable, but even an absurdly high 800ppm, isn’t a temp problem for over heating the planet.
micro6500 November 21, 2017 at 8:50 am
“There is no such correspondence. Blackbody radiation at the earth’s temperature peaks at 550cm-1 for 288K (peak radiance 0.136 W/m2/sr/cm-1) whereas the CO2 absorption band is centered at 667cm-1 (radiance ~0.132 W/m2/sr/cm-1). So CO2 absorbs near the maximum of the earth’s BB energy distribution (~97% of max).”
When you look at the whole spectrum, being near the maximum, isn’t all that significant.
If you run it through Modtran you’ll find that about 10% of the total is absorbed by CO2.
And remember water overlaps co2.
If you’d run the spectra at a realistic resolution you’d find that both spectra are composed of discrete lines which in general are a different wavelengths and therefore don’t interfere with each other.
Run Spectralcalc between 600 and 700 cm-1 for both CO2 and H2O and you’ll see how sparse the H2O lines are compared with the CO2.
Phil; Who is “Steve Goddard?” I know that Wien temperatures can’t actually be inferred from the line spectra of gases, but my point is that even if you were to construct a continuous, bell-shaped, Planck-like curve from the radiation within the band of CO2’s output (13to 17 microns), the closest thing to a Wien temperature you’d get would be the most intense line, which is 14.95 microns. That would correspond to a Wien temperature of -79 degrees C, which is well below any temperature experienced at Earth’s surface. In fact, of course, gases acquire their temperatures not from radiation, but from the kinetic energy of their constituent molecules, which is a function of pressure, but sticking with the radiative argument on which greenhouse theory is based, the band within which CO2 can emit just doesn’t qualify the gas for greenhouse function within Earth’s atmosphere.
davidbennettlaing November 22, 2017 at 5:54 am
Phil; Who is “Steve Goddard?”
It’s the pseudonym of the host of the website where the bogus post purported to be by a non-existent IR astronomer Michael Sanicola, which you referred to, was posted.
I know that Wien temperatures can’t actually be inferred from the line spectra of gases, but my point is that even if you were to construct a continuous, bell-shaped, Planck-like curve from the radiation within the band of CO2’s output (13to 17 microns), the closest thing to a Wien temperature you’d get would be the most intense line, which is 14.95 microns. That would correspond to a Wien temperature of -79 degrees C, which is well below any temperature experienced at Earth’s surface.
So what? A blackbody at -80ºC emits its peak radiation at 15 microns, that has no relevance to the BB emission from the earth’s surface. The earth’s surface at 288K emits much more 15 micron radiation than a BB at -80ºC (193K), 5.8 W/m2/sr/µm vs 1.1 W/m2/sr/µm. What’s of interest is the IR emitted by the surface.
Phil; Michael Sanicola is apparently a pseudonym for Morgan Wright, who states “I’m an opticist, who specializes in optics and IR. I worked for GE’s infrared department and designed infrared telescopes for GE that were used by NASA in outer space. I invented the ambient temperature microbolometer.” By whatever name, the individual appears to know what he’s talking about. As to your contention that its all about Earth’s IR output, that’s just not the case. The important consideration is the radiation that CO2 gas is capable of re-emitting, and that lies between the limits of 13 and 19 microns, as I (and MODTRAN6) said.This band is represented in Earth’s BB spectrum, but it’s in the right hand, “cold” wing of the spectrum, corresponding to temperatures below those of Earth’s surface, where none of it can therefore influence Earth’s actual surface temperature range.
This would have been a great thread if one were allowed to reference those unmentionable scientists that deny that CO2 can warm the surface. Especially the astrophysicist who just published a week ago or so on this topic.
I hope there comes a time when luke-warmers here can debate with the skeptics. Perhaps some day.
First have to agree on whether atmosphere warms or cool the surface. Until then the debate will be infinite.
Both of course.
TTY, “Both of course.” is a perfect response! Most of the comments are citing warming of a molecule, parts of them, or atmosphere of them, but in reality are MOSTLY talking about reducing heat already gained.
Fact: Energy can neither be created nor destroyed, it can only be changed in form.
1) CO2’s only defined mechanism to affect climate change is through absorbing and thermalizing LWIR between 13 and 18µ
2) Those bands are only a small fraction of the emitted LWIR, earth’s peak is 10µ
3) There is no way through thermalization for CO2 to warm anything above the temperature of the emitting body, the energy simply doesn’t exist. Absorbing a fraction can’t account for more than the whole.
4) Radiation could trigger combustion, but that isn’t the phenomenon that we are talking about
5) Conduction, convection, and radiation all act to move heat away from the earth. Claims that the GHG effect is responsible for all the warmth of the atmosphere ignore conduction and convection.
6) This is easily proven in a lab by shining 15µ IR light into a flask of CO2. It won’t warm because the room temperature is way above -80°C
Technically energy can be “destroyed.” If you observe the spectra from distant stars, you’ll note that the hydrogen emission lines are red shifted. When the photon left the star of origin, it had the normal wavelength (freq) of the emission lines of hydrogen. As it traversed the expanding intergalactic space, it’s wavelength grew longer (red-shift.) There is a calculable loss of energy in this red-shifted photon according to E=hc/l where delta-E = hc/delta-l.
…
Where did the ‘lost’ energy go?
“Technically energy can be “destroyed.””
Prove that and you repeal Newton’s Law, and you will get a Nobel Prize.
I already did. The photon that was emitted a million light years ago, “lost” some of it’s energy. The frequency has decreased (increased wavelength.) Where is the “lost” energy?
it went into potential energy. for the gravitational red shift it was demonstrated by t Cranshaw at Harwell in the 1960s. a photon fired from the ground level to the top of a hanger was found to be red shifted. a photon fired downwards was blue shifted. it is like a ball being throw upwards it looses speed but that does not mean that energy is destroyed. Similarly with a photon
Gerontius, that does not explain it. There is no gravitational field in the million light years between the star that emitted the photon, and the spectrometer here on earth that measured it. The red shift is due to the expansion of space, not to a gravitational field.
PS Gerontius, photons coming from the opposite side of the universe display the same red shift. You would think that if you looked in the opposite direction of the red shifted photon, you’d observe a blue shifted one if your “theory” were correct.
They never get that, but you are being mean 🙂
The energy doesn’t change so much as the wavelength stretches out, changing its color. If the source were heading toward you the waves would bunch up. It’s all due to the Doppler effect.
A passing car is a good example. You hear the sound change as the car passes. Sound at sea level and standard conditions travels at about 761 mph. So if a car is traveling toward you at 30 mph, then the sound is travelling at 761 + 30 mph = 791 mph. After it passes, the sound you hear is traveling at 761 – 30 Mph = 731 mph. So the energy does not particularly change. Turbulence has a small effect, but the pitch goes from higher to lower just as starlight red shifts.
It’s all down to the observer’s or listener’s point of view or frame of reference. The sound wave or photon just keeps doing its own thing, while the looker looks on.
As for the effect of gravity, red shift or blue shift is not due to any change in frequency of the photons, but rather to the time rates being different for observers in different potentials. No energy change occurs. If a photon of a given frequency is created by some atomic transition in the vicinity of a star, then it will appear red-shifted compared with the energy of the same transition at some distance from the star, but it hasn’t changed frequency.
Similar considerations apply for red or blue shift due to relative motion; the energy is unchanged relative to the location at which the photon was emitted, but appears different from a moving frame. The situation isn’t quite so clear-cut about red shift due to cosmological expansion, because it depends on how you describe the expansion, but even in that case the frequency of the photon is unchanged relative to its original rest frame.
E= hv
E= Energy
h = Plancks constant
v = frequency
The formula begs to differ 🙂
“The energy doesn’t change so much as the wavelength stretches out, changing its color. If the source were heading toward you the waves would bunch up. It’s all due to the Doppler effect.”
That is a great point, the total energy is the same, but energy/m is less. You are stretching the energy over a larger distance.
You haven’t dealt with the issue that all your own formulas tell you that you lost energy.
Every real physicist will give you the same answer
https://physics.stackexchange.com/questions/21603/have-red-shifted-photons-lost-energy-and-where-did-it-go
https://physics.stackexchange.com/questions/7060/redshifting-of-light-and-the-expansion-of-the-universe
The energy is not conserved no even conceptually both QM and GR demand it to be so. This is one of the problems with classical physics it leads you to stupid answers.
LdB November 20, 2017 at 8:43 am
It is not a problem with “classical physics”, but with your lack of understanding thereof and misguided thinking about it.
As CO2 observes above in response to my prior comment, energy is conserved, but just delivered at a different rate per unit of EM radiation travel. That’s one way of explaining the paradox which you imagine.
What you are again showing is you don’t have a clue about QM.
So if you energy is coming in over time perhaps you would like to tell me how long your photon is and what time period 🙂
LdB November 20, 2017 at 11:45 am
Clearly you don’t understand QM or “classical physics”, by which you mean “physics”.
How many times do I need to explain it to you?
The apparent change in color, ie wavelength and frequency, or a photon of visible light traveling along at its accustomed C, is due to the observer’s position. In the Doppler effect, sound waves don’t change their energy content.
Again, if photons don’t exist, then neither do any of the other elementary particles.
Oh here he goes again trying to obviscate the problem, we have cool lasers(tm) again. You make a statement then just want to wave hands ignore it and try to deflect to something else.
Robert Kernoodle, Gabro,
Sorry for the tardy reply
the gravitational red shift is real and explicable and demonstrated . in that the energy lost is conserved in potential energy
the velocity red shift is real and is due to energy conservation, and not an example of non conservation of energy. You cannot look at the photon in isolation and must look at where it came. There is a fairly simple classical (although with a bit of QM thrown in explanation). It is readily available on the web, although if you look you will always get the Kernoodle reply that is non conservational. You need however to be rigorous in accounting your energy balance . There is no need to invoke expanding universes only relative motion between observer and emitter. You then get the Doppler effect. Now the Doppler effect and the red shift in electromagnetic waves is well known and its energy conservation is not in doubt. And as I said before the energy balance of the effect demonstrates that the red shift is an effect of energy conservation
Robert I leave you to search the web, ps Cranshaw used the velocity red shift to measure the gravitational shifts. Now that’s a big clue as to where the missing energy goes.
None of you here has mentioned it in this blog, so ignorance is bliss.
Robert Kernodle
in answer to your question
Please read Rev Mod Phys 4.87 1932 Enrico Fermi “Quantum Theory of Radiation”
It explains that red shift is an example of energy conservation!
co2islife November 19, 2017 at 4:12 pm
Fact: Energy can neither be created nor destroyed, it can only be changed in form.
1) CO2’s only defined mechanism to affect climate change is through absorbing and thermalizing LWIR between 13 and 18µ
2) Those bands are only a small fraction of the emitted LWIR, earth’s peak is 10µ
Since you claim to use Spectracalc try the following:
Using the Blackbody calculator set it for a BB at 288K
Set the lower limit of the band to 13 micron and the upper limit to 17 micron.
Perform the calculation.
You should find that the band radiance is 23.3 W/m2/sr whereas the total radiance is 124.2 W/m2/sr.
By my reckoning that means the CO2 absorption band accounts for about 19% of the total, hardly a small fraction.
Remember “Everyone of my comments can be verified by MODTRAN or SPECTRALCALC.”
“By my reckoning that means the CO2 absorption band accounts for about 19% of the total, hardly a small fraction.”
In an atmosphere of 100% CO2, that would prove a whole lot, but add H2O to the mix and you will see CO2 is meaningless in the lower atmosphere. You only see a CO2 signature at 15µ after H2O precipitates out of the atmosphere. BTW, that 19% is 19% of the very low energy end of the LWIR spectrum. Now compare the energy of visible light, of which H2O in the oceans absorb 100% of. That is where the energy is, not LWIR.
co2islife November 20, 2017 at 5:57 am
“By my reckoning that means the CO2 absorption band accounts for about 19% of the total, hardly a small fraction.”
In an atmosphere of 100% CO2, that would prove a whole lot, but add H2O to the mix and you will see CO2 is meaningless in the lower atmosphere. You only see a CO2 signature at 15µ after H2O precipitates out of the atmosphere. BTW, that 19% is 19% of the very low energy end of the LWIR spectrum. Now compare the energy of visible light, of which H2O in the oceans absorb 100% of. That is where the energy is, not LWIR.
Not true, that 19% is of the total of the LWIR you can check it out on Spectralcalc. Also the CO2 absorption band is near the peak of the energy spectrum, it is water that is near the tail.
The Blackbody radiance peaks at 550cm-1 for 288K (peak radiance 0.136 W/m2/sr/cm-1) whereas the CO2 absorption band is centered at 667cm-1 (radiance ~0.132 W/m2/sr/cm-1). So CO2 absorbs near the maximum of the earth’s BB energy distribution, by contrast water absorbs between 1500-2000 cm-1 (radiance ~0.02 W/m2/sr/cm-1).
You show the CO2 signature on the Modtran graphs on your own website from 1km, you also show the H2O signature off in the tail. If you do the calculations on Modtran comparing 0ppm CO2 with 400ppm CO2 you’ll find that ~10% of the total is removed by CO2.
“Also the CO2 absorption band is near the peak of the energy spectrum, it is water that is near the tail.”
Yep….

I see a lot of diagrams on radiation ,,,, incoming, outgoing, backwards. But all of this assumes that the majority of energy absorbed by the ocean is in some sort of magical equilibrium with the atmosphere. …. ie, a very large part of the incoming SW IR Is absorbed and NOT immediately released.
LWIR between 13 and 18µ doesn’t penetrate or warm the oceans.
Exactly, but the ONLY outside source of energy to the earth, well, maybe some minuscule amount from plate tectonics, …. is SW, and it DOES penetrate the ocean surface. It doesn’t just warm the surface and immediately float back into the atmosphere.
Any equilibrium equation for the total earth can only be made n long term basis, as a good bit of energy is stored and released over time.
A close examination of USCRN soil data shows that solar IR hardly makes it 1 meter deep by conduction and then only during summer daylight which is the ONLY time the air is warmer than the ground.
My idea of an experiment is simple, maybe too simple. Take two identical sealed glass containers and fill each one with a mixture of gases which imitates the atmosphere. The only difference between them is that one has 0.028% of CO2 and the other has 0.04% of CO2. Both vessels should be fitted with highly accurate temperature probes. At the start ensure that both are at exactly the same temperature. Now radiate each with an IR lamp, tested to be identical in output. Now observe the temperature increase in the vessels. The data from this experiment can then be used to calculate and compare what happens to atmospheric temperature at pre-industrial and current CO2 levels. My guess is that the difference will be so small that even the accurate temperature probes can not distinguish it.
Derek Anthony already did this with his two glass jar experiment. One filled with air the other with CO2. Search for it and watch the video.
A glass jar is a closed system. The earth is an open system. Thus a false experiment.
Sailboarder the earth is a closed system with the sun. Energy can enter but no mass. Also the video was done to correct numerous errors done for Al Gore by Bill Nye. Good video.
E=hv tells us how much energy is available to cause a CO2 molecule to increase its translation. And translation velocity is dependent on absolute temperature and molar mass. So heavier gas travels slower.
We know that Q = Cp m dT for how much energy is needed to raise a mass of something, air in this case.
I have yet to be able to show that hv * 400ppm CO2 = Cp m dT for a cubic meter air.
But a postulate of kinetic theory of gas says that all collisions are pure elastic and no energy is lost or gained, so if true CO2 cannot do this anyway.
That is all very interesting but it is a EM wave aka a radio wave. Just like your TV set the EM wave will only selectively react to certain frequencies. Do you really think the EM wave delivering your TV signal got absorbed and re-emited from every air molecule along the way or has anything to do with kinetic energy of molecules along the way?
To your question my answer is NO. But since the present GH theory is based on CO2 absorbing IR we should be able to reconcile these two items.
I have no idea what GH theory works on I am not a climate scientist. All I am telling you is radiative transfer doesn’t occur in that manner described and energy can be lost and gained and attenuation can be measured. I am sure if you search Atmospheric Attenuation you can find the numbers for ll frequencies in the EM spectrum (from RF thru visible light up to X and Gamma rays).
LdB we have been shown graphs on this thread of CO2 absorbing at 15u. OK, then using E=hv we can calculate the amount of energy available to any CO2 molecule to increase its translation. Translation is directly related to temperature.
Due to the relationship of velocity to temperature we should be able to calculate the ability to raise a cubic meter of air via the energy in a 15u IR absorption.
Having taught basic radar theory and basic physics of underwater sound in the navy I have a idea of EM wave and attenuation etc. But that was many years ago. Thanks for your input.
Does anyone have a link to a research paper where 15µ IR was shined on a flask of CO2 and its temperature measured? Unless the CO2 spectrum is a lie, then the only contribution CO2 has to global warming is through the thermalization of IR at 15µ. That won’t warm anything. Simply focus on the basics. By what mechanism does CO2 affect climate change? Isolate that factor and test it. Has anyone tested the impact of CO2 thermalizing 15µ LWIR?
Testing
I too am working with this problem, I actually thought I had been ‘pipped to the post’, but after reading this, and the comments, nope – not even close. And Anthony, this may be ‘contentious’ but it is the key premise to greenhouse theory; without this, greenhouse theory collapses, so it is worth trashing out.
The way I see it the problem lies in the detectors of the greenhouse gases (see my youtube presentation below): they all use thermoelectric – Seebeck Effect transducers. What John Tyndall discovered In 1859 are really only the thermoelectric gases, N2 and O2 are not; nor is CO2s 1338cm mode. All thermograms and IR spectrograms are created using these detectors, but they discriminate the non thermoelectric modes and substances.
Greenhouse theory is 19th century science, pre Quantum mechanics . Quantum mechanics predicts and explains the vibrational modes, emission spectra, of all the molecules in the atmosphere including oxygen and nitrogen; It also says all matter radiates infrared. All matter! That oxygen and nitrogen are assumed not to radiate IR is a contradiction to QM. Either Quantum mechanics, or greenhouse theory is wrong: Quantum mechanics is not wrong. To observe the predicted Modes of oxygen and nitrogen – at 1556cm and 2338cm respectively – we must use 20th century technology, Raman spectroscopy (the complement instrument to thermoelectric IR spectroscopy) .
In the paper that I’m about to publish I will show the Raman laser technology is equivalent to Thermoelectric IR technology. Raman can do it all: it measures temperature, and the concentration of the gases. It is even used to measure the Keeling curve, and is the instrument of choice on solar system space probes for this reason. It can also show nitrogen and oxygen emit and absorb IR radiation, in total compliance with thermodynamic laws Stefan Boltzmann equation and Boltzmann constant. I can even show, by experiment and application, nitrogen radiates upon CO2 to heat the CO2 – figure that.
The following YouTube presentation is my rough beginnings – I have since developed my theory.
With oxygen and nitrogen being greenhouse gases, greenhouse theory collapses.
https://www.youtube.com/watch?v=T0IHKKkOwdU&t=860s Reinterpreting John Tyndall’s 1859 Greenhouse Gas Experiment with Thermoelectric and Raman Theory
Pretty much any wavelength of radiation can create the photons that we sense as heat. For example, the 662 kEv photons from Cs-137 when stopped by a lead shield will cause the lead to get hot if there are enough photons. Saw this with a cask of 55k Curies of Cs-137. The exterior of the cask was hot to the touch. Here is how I describe the conversion of the 662 kEv photons to heat EM. A loss of energy of the initial photon occurs as it impinges or interacts with an atom’s nucleus or electrons in a mass. There are number of processes by which the initial photon losses energy, but with each interaction of the photon with the orbiting electrons or the nucleus, some energy is “lost”. Of course the energy is not “lost” it is divided into lower or a different energy potential/form. One such loss might be when an orbital electron gains energy from the initial photon and ‘moves’ to a higher energy “excited” state. Generally, the electron does not want to stay in this state and will emit the excess energy as a low energy photon – sometimes immediately, sometimes after a passage of time. The process of the initial photon losing its energy can involve thousands of ever decreasing interactions. Another interaction might be where a photon strikes an electron and the electron is ejected from the atom. The initial photon loses energy equivalent to the binding energy of the electron. If the ejected photon received energy in excess of its binding energy, then that photon has energy to give to something else.
Because the eye is a measuring device for visible photons it is useful for describing the process of a photon being eventually degraded to heat. The eye has evolved to manage a reasonable level of visible photons without damage despite the fact that some of the dissipated energy becomes heat photons (whose energy is also dissipated). However, staring directly at the sun introduces too many photons and the heat dissipation mechanism cannot handle it and the retina will get burned. A burn is basically the rearrangement of a molecule, e.g. a protein molecule, due to the input of too much energy, whereby the new arrangement does not work biologically. Physics and biochemistry are fun fields to study in combination.
I have a few nit-picky points in Gill’s assessment, but overall I agree with his assessment.
“This is a contentious subject, and I have often shied away from it because it often erupts in food fights.”
I think the stewed tomatoes now dripping from the ceiling have confirmed your reservations, Mr. Watts. I do hope Mr. Gill was properly attired for the occasion.
For those still confused about how the poorly named “greenhouse effect” works, please see two posts on the subject:
The Steel Greenhouse
People Living in Glass Planets
And for those who claim that gravity or atmospheric mass or some other phenomenon is responsible for the fact that the Earth is much warmer than we would expect given its distance from the sun, let me recommend my post entitled “A Matter Of Some Gravity“.
Or not, you could choose to ignore everything but the things that support your view …
w.
Willis, I still see “The Steel Greenhouse” as one of the best articles I’ve ever read on the so-called “greenhouse effect.” In discussions on this site I’ve referred others to it several times. Thanks again.
Seriously?
The problem Willis is the way we still teach physics. Almost everyone who doesn’t get it has the image of a photon as something like a tennis ball flying thru the atmosphere crashing into every molecule in it’s path. They don’t understand or won’t accept it’s a radio signal that collapses to something we equate to a tennis ball.
Neither of the first two bare the slightest resemblance to Earths’s atmosphere.
And everyone knows that an atmospheric pressure gradient, and related temperature gradient, exists on every known planet with an atmosphere.
Venus:
Atmospheric composition: 96.5% carbon dioxide, 3.5% nitrogen, various trace gases.
Surface atmospheric pressure: 93 bar
Temperature: Over 450 °C
Earth:
Atmospheric composition: 78.09% nitrogen, 20.95% oxygen, 0.93% argon, 0.04% water vapor (much higher at sea level), 0.04% carbon dioxide, other trace gases
Surface atmospheric pressure: 1.013 bar
Temperature: 15 °C
Mars:
Atmospheric composition: 95.32% carbon dioxide, 2.7% nitrogen, 1.6% oxygen, 0.08% carbon monoxide, 0.3% various trace gases.
Surface atmospheric pressure: 0.006 bar
Temperature: -60 °C
Thus, at first glance, mass of the atmosphere matters, but GHG concentration, not at all. No doubt there are extenuating circumstances.
I think you might class the distance to that big shiny thingy in the sky as an “extenuating circumstance”.
“I think you might class the distance to that big shiny thingy in the sky …”
Exactly. FWIW, Venus receives about twice as much solar radiation per square meter as does the Earth. However the Venerian albedo is higher. More incoming radiation is reflected back to space. Also Venus rotates very slowly compared to Earth and Mars which probably alters the heat distribution considerably.
Tty,
I considered adding solar radiation at ToA for each planet, but the differences are small compared to the enormous temperature differences. Naturally you’d expect Venus to be balmier and Mars chillier than Earth, all other things being equal, but of course other things aren’t. Not even close. The other things outweigh insolation.
Don,
Venus’ rotation is clearly another factor. But, even considering all other things, the fact that Venus and Mars both have high-CO2 atmospheres highlights the importance of density and surface pressure. The temperature at the point in the Venusian atmosphere with terrestrial surface pressure is about the same as on Earth.
Right on. The steel greenhouse ignores the pressure temperature effect.
sailboarder November 20, 2017 at 4:09 am
The “pressure temperature effect”??? As I have shown, and as Dr. Brown has shown, that is a fantasy. READ THOSE LINKS, and then come back and tell us why Dr. Brown and I are wrong.
w.
A Chinook demonstrates that you are wrong.
David,
How about Walla Walla winds, the reverse of a Chinook?
David Ball November 20, 2017 at 12:23 pm
David, while a Chinook wind raises the temperature at the surface because the air is descending … what goes down must come up. So somewhere else the same amount of air is going up and cooling, so that there is no net gain or loss.
And from the tone of your comment, it is clear that you have NOT done as requested, which was:
If you want to argue with my ideas, at least do me the courtesy of reading them before starting in … both Dr. Brown and I have proven, not asserted but proven, that there is no “pressure temperature effect” capable of warming an entire planet on a continuous basis.
w.
Willis,
What would be the temperature of Venus at the distance of Mars from the Sun?
Please explain why the temperature in Venus’ atmosphere at one bar is the same as on Earth. Thanks.
https://www.sciencedirect.com/science/article/pii/B9780124158450000141
“Venus possesses a dense, hot atmosphere, composed primarily of carbon dioxide. A surface pressure of nearly 100 bars sustains the mean surface temperature of 740 K, which is essentially globally uniform except for topographic effects.”
http://physics.uoregon.edu/~jimbrau/BrauImNew/Chap09/7th/AT_7e_Figure_09_17.jpg
Much as I respect Dr. Brown, “proof” is a strong word, appropriate for math, not technically applicable to science.
Actually, Venus’ atmosphere is a little warmer than average Earth temperature at one bar, thanks to a bit more incident solar radiation. But close enough to make the point. For government work.
Put another way, IMO Boyle’s Law ought roughly to apply, since, while not a closed system, the atmosphere approximates one since incoming and outgoing radiation are close to in balance, while subject to minor perturbances.
Thus, greater pressure should yield higher temperature. The atmosphere expands and contracts under heating or cooling, thus lowering or increasing pressure and temperature at the margins to maintain a fairly constant temperature, until other factors such as Milankovitch cycles and albedo waxing and waning change the baseline. This process can be extrapolated out to even denser or yet lower pressures.
“that there is no “pressure temperature effect”
Yet it exist on all known planets with an atmosphere.
hmmm !
Gabro November 20, 2017 at 12:57 pm
I’m sorry, but there are far too many unknowns in the thought experiment to even begin to say. FOR EXAMPLE, at the surface of Venus CO2 is a superfluid … would it still be a superfluid at the distance of Mars? And how would that change things?
According to your graphic, the temperature of Venus’s atmosphere at one bar is on the order of 350K … which is 77°C or 170°F.
Look, my rule of thumb is that when someone starts talking about Venus to try to show something regarding the greenhouse effect, I stop listening. Venus is a hugely complex system about which we know little. Consider the trouble we have explaining the Earth, about which we know orders of magnitude more than we know about Venus …
Say what? “Proof” is indeed a strong word, and I do not use it lightly. However, there are assuredly parts of science where things can indeed be proven, and this is one of them.
However, if you think that Dr. Browns proof is incorrect, how about you point out WHERE it is incorrect, and leave the grand overarching view of what “proof” is and isn’t for another time.
Note that I apply the same word to my proof regarding the same question, which takes a totally different path to the same conclusion. And again, I invite you to find something wrong with my proof.
Thanks,
w.
AndyG55 November 20, 2017 at 2:28 pm
“that there is no “pressure temperature effect”
Yet it exist on all known planets with an atmosphere.
Like the one where the pressure drops from 20,000 Pa to 110 Pa and the temperature increases from 210K to 270K?
You mean virtually a vacuum, where radiation starts to take over.
DOH !,
Phil, there has to be enough atmosphere to sustain the gradient
Do try to keep up.
Happens around 0.1 bar. on every planet with enough atmosphere.
“Once you have GHGs in the atmosphere, of course, some of the surface radiation can get absorbed in the atmosphere. In that case, the surface radiation is no longer constrained, and the surface is free to take up a higher temperature while the system as a whole emits the same amount of radiation to space that it absorbs.”
–
Thank you Willis.
I have never really understood you but this comment is great.
Since the same energy is going out that comes in there is never a real accumulation of energy in the system , just a change in the temperatures of the mediums of that system, if true.
More energy in the atmosphere less in the land and sea perhaps?
Without the CO2 the land under the sun would be a lot hotter during the middle of the day and would send out lots of IR at a higher temp than the atmosphere does.
“For a molecule to “warm up” (have more kinetic energy) it needs its electrons to move to a higher, more energetic orbit. ”
The kinetic energy of a gas is the sum of the energy of the translational motion of the centre of mass of each molecule. Electron energies are not involved.
Belief in AGW caused by CO2 = denying or ignoring the science of thermalization, Maxwell-Boltzmann distribution of molecule energy & quantum mechanics. The IR energy absorbed by CO2 is immediately (0.0002 microseconds) shared with surrounding molecules (thermalization) so, at low altitude, there is little chance for a CO2 molecule to emit a photon as a direct result of having absorbed one (relaxation time about 6 microseconds). Water vapor has many (>190) significant absorb/emit lines at substantially lower energy levels than the 15 micron absorb/emit band for CO2 and on average there are about 35 times as many WV molecules as CO2 molecules. At low altitude, energy absorbed by CO2 is effectively rerouted up via water vapor radiation and some convection. End result is CO2 has no significant effect on climate. http://globalclimatedrivers2.blogspot.com
Belief in AGW caused by CO2 = denying or ignoring the science of thermalization, Maxwell-Boltzmann distribution of molecule energy & quantum mechanics. The IR energy absorbed by CO2 is immediately (0.0002 microseconds) shared with surrounding molecules (thermalization) so, at low altitude, there is little chance for a CO2 molecule to emit a photon as a direct result of having absorbed one (relaxation time about 6 microseconds). Water vapor has many (>190) significant absorb/emit lines at substantially lower energy levels than the 15 micron absorb/emit band for CO2 and on average there are about 35 times as many WV molecules as CO2 molecules. At low altitude, energy absorbed by CO2 is effectively rerouted up via water vapor radiation and some convection. End result is CO2 has no significant effect on climate.
J. Richard Wakefield November 19, 2017 at 1:28 pm
It doesn’t make it warmer, it makes it less cold??? That’s just a pointless semantic exercise trying to deny the fact that the earth is warmer with the atmosphere than it would be without the atmosphere.
J. Richard, if you asked someone who just put on their jacket “Are you warmer now?”, do you think they’d say “Yes”, or that they’d say “No, I’m not warmer now, but I’m less cold”?
If you think the latter, here’s a quick guide to things that exist on a spectrum:
Less tall = shorter.
Less heavy = lighter.
Less dumb = smarter.
Less cold = warmer.
w.
Willis
Coming from a cold and damp (Scotland) part of the world a suitable response to that question would be “I’m not as cold as I was” 🙂
As I come from the “superhot” Arctic, I find this very amusing.
Nick Stokes November 19, 2017 at 5:32 pm
Not true, according to CERES. As one example, for the Northern Hemisphere, as the average temperature swings from about 9°C to 22°C (a 4% change min to max), the net LW flux at the surface varies between about 52 and 60 W/m2 … since that is about a 15% change min to max, I’d hardly call it “near constant” W.R.T. temperature …
w.
(As a side note, CERES puts the upwelling LW at 398 W/m2, and the downwelling at 345 W/m2).
Willis,
The Trenberth budget deals with annual averages. That gives plenty of time for the air at DWLWIR emitting altitude to equilibrate with surface, if the (annual average) surface warms.
That makes no sense. You’ve said that “if the surface warms, that air warms too, and the flux difference remains constant”. You CANNOT determine that from a budget, whether Trenberth’s or someone else’s. A budget has no information at all about a “warming surface” …
w.
Willis,
No, you can’t determine it from a budget. But you can reason about time scales. You are referring (I think) to a phase lag between surface and downward emitting layer temperature with the seasonal cycle. Averaging over a year is firstly a somewhat longer timescale, but also averages out the effect of such a seasonal lag. Basically the difference between upflux and down is determined by the temperature difference of emitting layer and ground temperatures, which is determined by lapse rate and GHG concentration. The latter fixes the altitude of the emitting layer.
Willis,
You might like to try this test with CERES (I am not so adept with it). Just look at different areas around the world. There will be big changes in upflux and down. But is it not always the case that upflux always slightly exceeds down, as they vary over such a range?
Nick Stokes November 20, 2017 at 2:25 am
Nick, interesting question. Here’s surface upwelling longwave minus surface downwelling longwave, in a couple of formats.:
As you point out, net flow is always from the surface upwards.
All the best,
w.
Willis,
Thanks
Willis,
Just adding some interpretation – I think the up-down difference is basically product of altitude of downward emitting layer and lapse rate. The dry lapse rate is higher than moist. And your map looks pretty much like a humidity map, except at poles, where flux in both directions is smaller.
Willis,
Further thought – altitude of downward emitting layer is dependent on GHG concentration – it is lower with more GHGs. So higher if water vapor is low. That adds to the difference being greater in dry places – it could be the main cause.
The temperature of a body (gas, liquid or solid) directly affects the wavelength of the radiation it emits and
absorbs.John November 19, 2017 at 7:06 am
Say what? If you shine a flashlight at a stove, it will end up warmer than without the flashlight. Not much warmer, but assuredly warmer. Look, John, a good chunk of the light from the flashlight will be ABSORBED by the stove, and the rest will be reflected.
Now, since energy cannot be either created or destroyed, what happens to the energy in the photons that are absorbed? Obviously, that energy must be changed into something … let’s see. It’s not converted to electricity. It’s not converted to chemical action. It’s not converted to mechanical energy … gosh, could it be converted to thermal energy?
And if (as you claim) the absorbed light is NOT converted to thermal energy … then just what are you claiming it is converted to?
You continue:
First, photons don’t get “overwhelmed” by photons going the other direction. If you are indeed an engineer you know that to be true. You can’t “overwhelm” a flashlight by shining fifty flashlights at it, that’s nuts.
Second, the resulting change in temperature in the stove is small. However, it is not “unmeasurable”, which you also know is true if you are an engineer.
Finally, your explanation that if CO2 “DID increase the surface temperature, the surface would radiate to space at the 4th power of the increase, instantly cooling itself back down” makes no sense at all. According to that theory, nothing can increase the surface temperature, because if it did the surface would “instantly cool” …
w.
Willis, you’re close to right there, when you try to haul the lower atmosphere up by its own bootstraps, a humourous definition of futility. Any warming would indeed cause expansion by molecules moving at c. 1km/sec in the gas phase. The gas laws do rule, so the IR catastrophe will not happen. Ditto the never-observed ghe.
“Ditto the never-observed ghe.”
Except it has been, and the link has been posted on this thread at least twice.
How about you read it?
https://phys.org/news/2015-02-carbon-dioxide-greenhouse-effect.html
The problem is that this only matters if all else is the same, and it’s not.
Proposition!
Cold objects must warm hotter objects, or even more strongly stated, heat the warmer object
Is this true?
In this case the fire and the light bulb
However the general truth comes out that is if the two objects are thermally isolated from their surroundings heat only flows from the higher temperature because that is the definition of HEAT
For example
Two metal blocks A and B sit separated inside a vacuum filled adiabatic enclosure.
Adiabatic enclosure consists of a perfect reflector face surrounded by a perfect insulator
Initially both at the same temperature. The zeroth law of thermodynamics applies.
Both emit and absorb equal amounts of radiation.
Neither one is said to heat the other.
Both objects remain at the same temperature
One block (A) has a power supply which is now switched on causing the temperature of the block to rise.
This in turn means that it will emit more radiation.
A will now heat B causing its temperature to rise.
B will in turn emit extra radiation which is absorbed by A but this ‘back’ radiation is caused by A.
Now comes the clincher
If B were not there at all the temperature A would be even higher.
So B cannot be said in any meaning of the word as a cause of heating A
in the case of the furnace(Tf) and light bulb(Tb)
If Tf >Tb heat will flow from furnace to bulb
If Tf <Tb heat will flow from bulb to furnace
You whole answer is so crazy you do realize you aren’t dealing directly with heat but with EM wave. We call it a thermal emission not because it is “hot” or “heat” but because the source of emission is from a thermal source. Don’t think of the emission as heat it’s just EM energy.
Lets give you this with lasers which are the most precise EM frequencies I can generate.
So I fire a CO2 infrared laser and a red laser at each other.
We know the beams will pass thru each other without interacting and in fact on a laser cutting machine we often use this so you can see where the cutting in what we call that a red dot pointer. The IR frequency is invisible to us but you inject the red laser thru one of the mirrors so it follows the path of the IR.
So got it I have an IR laser one way and a red laser beam the other way.
What happens when each beam hits the other laser device (and they will hit)?
Bryan, you were correct until you got to this statement:
Why on earth would that be? You’ve already said that B radiates energy to A. How can A be warmer when you remove that source of energy?
As LdB said, you are conflating heat with thermal emission, when they are two very different things.
Regards,
w.
Here is an RGB laser in operation and according to you each colour is at different temperatures 🙂
Hopefully you may care to rethink all that.
No Willis I am absolutely correct
Before the power switch on A was turned on block A and block B were at the same temperature.
The adiabatic walls confines all energy within that enclosure
Lets say each has a mass of one kilogram
With both A and B the power source has to heat 2Kilograms
With B removed all the power source has to heat one kilogram which implies the temperature of A will rise twice as fast as before
Any physicist will confirm that I am correct
Bryan, your case is so specific that it has almost nothing to do with climate.
“Adiabatic enclosure consists of a perfect reflector face surrounded by a perfect insulator”
so you have created a situation where every last bit of radiation leaving the blocks gets reflected back perfectly. This is the exact opposite of the earth’s situation, where every last bit of radiation that leaves from the top of the atmosphere disappears and never comes back. If no energy can leave, then the two blocks will reach some equilibrium where both have the same temperature.
“If B were not there at all the temperature A would be even higher.”
While strictly true, this is not really very interesting. Basically all you have claimed is that an isolated, insulated system with mass “m” warms faster than an isolate, insulated system with mass “2m”.
I would challenge you to re-analyze with a system where the enclosure has perfectly ABSORBING walls and is held at, say, 3 K. You will find that in this more useful system, Block A does indeed warm faster than without block B, and Block B ends up at some temperature between that of Block A and the cold walls.
Hi tjfolkerts
My post was to correct the impression that the presence of any object irrespective of its temperature must ‘warm’ some neighbouring object.
This piece of fiction has become so prevalent in the last 20 years that normally rational folk like Willes have been blindsighted by it.
My example was to remind people that the surroundings need to be specified before any progress is made in analysing the flow of heat.
My scenario would be found in opening chapters of a thermodynamics book where definitions of adiabatic and diathermic walls, heat transfer and so on are introduced.
I am sure that ‘warming’ by cold objects meme has been carefully fostered by global warming advocates.
Look at some earlier comments upthread .
Block of ice in sitting room with person
Torch heating a furnace
Surely the Earths atmosphere in which radiative resistance rather than reverse heating gives a rational explanation of the situation and should be encouraged.
Its up to people who should know better to get back within the framework of physics
Sorry (profuse apologies) should have written Willis rather than Willes
Bryan,
I agree with your point that it is valuable to examine all details of a system, and to be precise about language (the word “heat” being perhaps the biggest language offender). Your scenario certainly could be worked out with some basic undergraduate thermodynamics.
My main issue with your scenario is that it is SO far removed from scenarios related to the thermodynamics global warming that it could easily give a false assurance to people who simply want a post to confirm their biases. For planets, the default condition is blackbody surroundings at 3K. This is almost exactly the opposite of what you hypothesized.
I think it would be more productive to directly address issues like “warming with ice”. The radiation from cold things still feels warmer (and will help keep you warm) when compared to radiation from even colder things. If I am facing a wall of dry ice @ur momisugly -79C and then someone interposes a wall of ice, then yes, I will feel warmer. A pot of water with an immersion heater will rise to a higher temperature when the radiation from the warmer ice replaces the radiation from the colder dry ice.
tjfolkerts
The block of ice in the sitting room example.
Everyday common sense tells people its not a good idea for keeping the house warm.
The walls floor and ceiling and air at around 20C (surroundings) will lose heat in melting the ice by conduction convection and radiation.
The fact that the ice block radiates to the wall is true but will not change the outcome .
If the surroundings were dry ice as you say it would be the ice block that would lose internal energy by heat transfer to the dry ice and so it would get colder.
Once the surroundings are specified it is much easier to keep track of the heat flow.
In reading posts on Global warming quite often only two items are mentioned
The hotter and colder object ( with no mention of surroundings).
Then comes an astonishing statement like….
Do you know that the cold object ‘warms’ the hotter object
It always seems that the main offenders are global warming advocates.
Also not all photons are created equal.
The ‘quality’ of the radiation was mentioned in the theme title but not discussed much any further.
Bryan says” In reading posts on Global warming quite often only two items are mentioned
The hotter and colder object ( with no mention of surroundings).”
Any decent discussion will include FOUR items
1) the warm object that is absorbing energy (eg the ground)
2) the source of energy (eg the sun)
3) a low temperature heat sink (eg 3 K outer space).
4) the object that is not absorbing energy from Item 2 but can absorb radiation from Item 1 (eg the atmosphere
Perhaps you were reading poor post where the authors did not really understand the GHE. Perhaps the authors figured the sun and outer space were simply givens that did not need specific mention. In any case, when all four items are included, it is very simple to see that adding Item 4 results in Item 1 getting warmer.
One place where intuition often goes astray is the “low temperature heat sink”. Normally our surroundings (around 20 C typically) are not all that cold. Then adding a wall of ice that is held at a steady 0 C (even colder than the surroundings) certainly will not warm you. But that would be like adding an atmosphere that is held steady at a temperature BELOW 3 K!
tjfolkerts: you state ” In any case, when all four items are included, it is very simple to see that adding Item 4 results in Item 1 getting warmer.” There is no atmosphere on the moon. Sunny side of it gets much higher temperature than the surface of the earth. How come?
esalil, this is a discussion of broad averages. The average surface temperature of the earth is MUCH higher than the average surface temperature of the moon. This despite the fact that the Moon absorbs a higher fraction of the incoming light.
Furthermore, the moon’s average temperature would rise if it had an atmosphere of GHGs (although the max temperature on the sunny side would probably drop a bit.
Gabro November 19, 2017 at 5:45 pm
People skeptical of GHE are “not permitted to comment”? Say what???
READ THIS VERY THREAD. There are lots of people skeptical of the idea of GHE, and they are free to post, as are you. Your claim is a joke.
w.
For me it looks like hopeless to explain the basic physics about the GHE for those who think that it is nonsense. I try another approach. If you think that the GHE does not exist, how do you explain the downward LW radiation at the surface which is about 345 W/2. Compare this to the direct solar radiation of 167 W/2 at the surface. (71 W/2 of solar radiation is absorbed by the atmosphere and therefore the outgoing LW radiation is totally 238 W/2). These figures are not nonsense, because they are both measurement based figures.We know that 99.97 % of the energy keeping the Earth warm originates from the Sun. Where this 345 W/2 comes from? How do you explain this, if there is no GHE?
It comes from the atmosphere (including clouds). The atmosphere radiates the energy absorbed from the sun and the surface. The energy from the surface is mostly nonradiative (evaporation and convection). This does not mean that the inceased atmospheric CO2 will result in a warmer surface.
There is a ghe, simply because there is an (insulating) atmosphere between the surface and the cold space. The atmosphere is in direct contact with the surface and is easily warmed by it, mostly by nonradiative processes. The bulk of the atmosphere (N2 and O2) plays a big role.
edimbukvarevic. Otherwise very good but you say that “The atmosphere is in direct contact with the surface and is easily warmed by it, mostly by nonradiative processes.” This is not correct: The upward LW radiation emitted by the surface is 396 W/2 (Excatly according to Max Planck’s formula). Latent heat flux 90 W/2, and thermal flux 25 W/2, together 115 W/2. According to basic mathematics 396 is more than three times greater than 115. Right?
Aveollila, the radiative heat flux surface-to-atmosphere is ~26 W/m2 (350 – 324). According to basic mathematics, that is less than 25% of the non-radiative fluxes (115 or 78 + 24 = 102 W/m2). It is only one fifth of the net surface-to-atmosphere heat flux (~128 W/m2).
http://www.ipcc.ch/publications_and_data/ar4/wg1/en/fig/faq-1-1-figure-1-l.png
Pressure/temperature effect?
edimbukvarevic You do not calculate the upward energy fluxes in the right way, because you subtract a donward flux from the upward fluxes (why you selected 324 and not the 168 ??). According to the energy balance of Kiehl an Trenberth (which is obsolete and based on the wrong atmosphere) the upward fluxes are 350 + 78 +24 = 492 and the downward fluxes are 168 + 324 = 492. That is called an energy balance. Your way of comparing fluxes is not based on the real world. Using your way, I can prove anything as I like.
Aveollila, I commmented on the atmosphere and its energy. In particular, the heat exchange between the atmosphere and the surface. The total heat transfer from the surface to the atmosphere is about 128 W/m2. Only a small part of this flux is radiative, about 20% (350 – 324 = 26 W/m2). This is not debatable, unless the Earth’s budgets are grossly inaccurate.
Furthermore, if we add solar radiation directly absorbed by the atmosphere (~67 W/m2), we get 195 W/m2. That means only ~14% of the total heat transfer to atmosphere is LWIR radiation.
As always very confusing with so many different opinions, immovable positions and even contempt for anybody who doesn’t hold the same opinion. I’m an engineer but I’m constantly searching for an answer to how a cold body heats a warm body and how heat is apparently created from nothing.
Please allow me to ask a dumb question and hope that I can get an answer to at least help clear up some of my confusion. I can calculate solid surface temperature of the earth from incoming UV radiation (from a warmer body) and physical properties (absorptivity and emissivity of the surface). I can calculate a near surface air temperature based on conduction and convection. I can then calculate a lapse rate which gives me a vertical temperature profile which is pretty well matched by reality. I can do this with no reference to downwelling radiation. Why should the conclusion from this not be that this is because downwelling radiation doesn’t affect temperature and lapse rate?
Martin. Solar radiation is much more than UV radiation which has a wavelength shorter than the visible light.
I do not believe that you can show calculations that the solar radiation of 238 W/2 absorbed by the Earth produces the Earth’s surface temperature of 15 degrees Celcius. Why not? Because that absorbed radiation flux of 238 W/2 corresponds the surface temperature of -19 Celsius only as measured at TOA emitted by the Earth.
Nobody has shown that Max Planck’s equation is wrong. It shows excatly how the surface temperature produces an exact amount of radiation. The fact is that the surface has an average temperature of 15 C and it produces the radiation flux of 396 W/2. How it is possible that the Earth receives only 238 W/m2 but its surface can emit 396 W/? Perpetum mobile? Confirmed by measurements. Fighting against GHE like fighting against wind mills. You cannot win.
Unless the so called GHE is just the pressure density effect?
ave, the calculation for spot temperature is basic radiant heat transfer and is based on actual radiation not average hitting a flat plate and it doesn’t have an additive downwelling radiation component for it to work. The problem is that if you believe that downwelling IR is the only way that the surface can achieve it’s effective emitting radiation then you have to have 324 W/m2 of downwelling radiation that is higher than the sun gives us and a transfer of heat from cold regions to warmer ones.
If downwelling radiation is a key component in atmospheric temperature why is the lapse rate completely free of any influence from it. Why is Jericho warmer than Jerusalem or the temperature on a mountain lower than at the bottom.
Is it not so that any number of experiments of putting bodies of different temperature in proximity, the temperature of the highest temperature body is never increased by radiation from the lower temperature bodies (true?) no matter how many there are.
I believe that heat energy (as opposed to radiation) can never flow from a cold body to a warmer one at any level including quantum and once this is accepted, heating by downwelling radiation can’t be significant. No heat is added and none is trapped, no laws are broken
Maybe those who say that it is an atmospheric effect rather than a greenhouse effect could be correct. These wonderful gases though for sure moderate the temperature of the atmosphere but add heat to it?
To sailborder. Create a pressure of 100 atmosphere into a steel cylinder and leave it outside in the temperature of -10 degrees Celsius. What do think is the temperature after 2 hours. It is that -10 C degrees. A constant pressure cannot create heat into any system. Only if you do like in a diesel engine: apply a certain amount of mechanical work and turn it into heat.
“I believe that heat energy (as opposed to radiation) can never flow from a cold body to a warmer one at any level including quantum and once this is accepted, heating by downwelling radiation can’t be significant. No heat is added and none is trapped, no laws are broken”
What is so difficult to understand about the concept of slowed cooling?
Of course heat energy can impinge on a warm body from a colder one.
Photons from the colder object do not have any concept of the other object being hotter.
(and there will be photons unless it is at 0K)
And as a result the hotter body cools more slowly.
There is however no “Flow”, as in a net impingement of photons from the cold object such that it raises the temp of the warmer one.
It is like a tank loosing water at a rate of 10 gals/hr while at the same time you pour water from a bucket into the top at the rate of 1 gal/hr.
So the tank is actually leaking water at 9 gals/hr. It’s still leaking (cooling) but at a slower rate due to the small amount of water (energy) being added to it from the bucket (older object) leaking (cooling) at 1 gal/hr.
I think this is probably the best visualization to explain what I’ve been talking about.
So, you have a big tank, with 2 holes in it. and over about 12 hr’s once a day, a big dump of water happens, pressure is a little higher, so it rushes out a bit faster at the peak level. One hole is the optical window, which is constant in size, and another that represents the energy through the other bands, part of this hole has grown slightly smaller from increases in GHG’s.
But together it’s a good size hole, and then the tank goes 12 hours between big dumps of water.
But, there’s a second smaller tank of water that is filled constantly, from far away.
And when the water level drops some, depending on the level of the second tank, some of the water in the second tank starts to flow into the bigger tank. This slows the drop in water level in the big tank, until the next big fill of water, where it shuts off till later.
If the second tank is really full, the big tank gets as much from it, as goes out the bottom once the valve opens, and the level doesn’t drop anymore, even though the main fill line is dry still.
This is why Co2 doesn’t have hardly any affect on the level. It’s the sheer size of the tanks compared to the tiny 3.7W.
“Unless the so called GHE is just the pressure density effect?”
Yes your bike tyre heats up as the air you pump into it increases in pressure.
But does it stay hot for you forever?
Obviously not.
Why?
You did the ‘work’ with the pump and then you stopped – then no work so the tyre cooled.
Now replace the ‘pump’ with gravity and the air by the Earth’s atmosphere.
(Hypothetically) if we could just switch on gravity from zero then yes the atmosphere would heat as it compressed.
But the work has then been done.
So it cools.
Gravity is NOT maintaining a surface temperature.
Full stop.
It does however set the LR by virtue of the -g/cp relation – the surface temp then being set by the GHE via raising the effective temp of Earth (255K) up to ~8km whilst the LR is maintained.
Toneb, the pressure is constantly ON. Are you intentionally being obtuse?
Rod
I believe that the simplest form of the question you ask is this:-
“Can something cold heat up something hot?”
To answer this question we need to establish the nature of collision:-
Can a slow moving object travelling behind ever catch up to and collide with a fast moving object travelling ahead of it and moving in the same direction?
Well of course not, only fast moving objects can ever catch up and collide with slow moving objects ahead. This is because collision is a vector process; fast objects collide into slow objects and not the other way round and so the transfer of momentum always proceeds from fast to slow and never from slow to fast (for objects moving in the same direction).
The issue of heating by “back radiation” is more subtle. The diagram that Anthony supplied demonstrates Wein’s displacement law and it shows the envelope curve for frequencies at different black body temperatures. In addition we have all seen the diagram that shows the effect of selective absorption of frequencies by greenhouse gases. What we have never seen is the diagram which shows an intensity mound in the low frequency tail above the envelope curve which is the absolute requirement for “cold” low frequency radiation to add energy to a high temperature body. This does not happen, it never has and it never will. Back radiation heating of the warm surface below by a cold surface above is a fiction and Wein’s displacement law proves this.
The radiative greenhouse effect is an attempt to explain how the low temperature isothermal stratosphere can heat a planet’s surface. It cannot. The troposphere (the weather layer) is a gigantic heat engine. It moves prodigious amounts of mass and energy all day and every day. Our atmosphere does work and it is powered by the heat of the sun and the speed of the Earth’s daily rotation. Like all heat engines it has an exhaust temperature, that exhaust temperature is observed in the isothermal shell of the stratosphere that surrounds our planet. Just as the exhaust temperature of an engine cannot be used to power that same engine, so it follows that the low temperature stratospheric shell aloft cannot provide back radiation “heat” to power the high temperature weather machine below.
So, Phillip, at night does a cloud overhead affect the temperature at the surface below and why as it is not cutting off any sunlight. Clouds are composed of GH effect water molecules at low temperature or do you have an alternate explanation?
angech,
I live in the British Isles. Almost every day somewhere here experiences the effect of warm moist cloudy air moving over night time cooled ground. My worst ever driving experience occurred a few years ago in Aberdeenshire when I lost control of my vehicle on black ice. One frosty winter’s evening I walked our dog alongside the River Don under crystal clear skies. The next morning, before dawn, the weather had changed to a mild 8C, with damp moist air, a thick haar (sea fog) and very poor visibility. As I drove south to work alongside the river I came to a small hill, the north facing slope of which was covered in black ice as moisture from the sea fog condensed onto the previously cooled road surface, so yes warm moist cloudy air can heat a cold ground. The question at issue however is can something cold heat up something hot?
I think that this is due to a change in wind direction rather than a change in the vertical temperature profile.
Certainly in Scotland we notice abrupt daily and hourly change.
If the wind is from south or west we have warmer weather.
From the north or northeast its much colder.
Angech, are clouds not condensed water with no greenhouse effect?
” so yes warm moist cloudy air can heat a cold ground. The question at issue however is can something cold heat up something hot?”
Yes and no.
I was meteorologist with the UKMO in a previous existence and one of our winter jobs was to monitor road temps over a wide part of the English Midlands for councils re gritting for ice/snow.
It is no mystery that cloud raises RST’s and hence air temps above.
It is added to the algorithms for RST prediction. On a otherwise frosty night it can make for a marked jump in road surface temp. In fact thin Ci cloud at 30,000ft at a temp of -40C will cause a road temp to either to halt or rise slightly when below 0C.
But it is NOT strictly raising it’s temp, as you will discover if you examine, say, a bridge in the same situation (air beneath).
What is happening is that the heat-flux from below the surface overcomes the surface emission to space (because of the cloud) and so the temp there rises.
In other words extra photons have impinged the road and added to it’s energy such that that entering from below is out of balance with the LWIR leaving.
It is though still a slowing of cooling and NOT cold heating up something hot.
“I was meteorologist with the UKMO”
roflmao.
And you actually “advertise” that…. that’s funny !
Toneb,
Very interesting observation, thanks. I have often noticed that ground frost will form first on the metal skin of a car, before it sets into the pavement. Same principle I believe. I had never experienced black ice until that morning and had no idea how dangerous it can be, even with an overnight air temperature rise to well above freezing it is still possible to get into severe trouble on damp untreated roads.
I totally agree…… though I approach it from a heat capacity angle. The best the GHG can do is reach equilibrium with the ocean or ground …. simply because the GHG mass and heat capacity is minuscule compared to everything else.
Philip Mulholland November 20, 2017 at 6:52 am
It is standard knowledge that cloudy skies will be warmer than cloudless skies due to the blanketing affect of clouds. Surely it could also be that the air arrives already warm and clouds are part of the ‘package’? Here in the UK the majority of weather and associated temperatures come from somewhere else, either pre-heated or pre-cooled. However, we have cloudless days that can be very hot or very cold depending on the time of year. I understand the reason for this is that cloudless days tend to have low wind speeds as this is usually a high pressure weather system. In the summer when the sun is overhead the surface gets the maximum effect of the sun’s heat and the low wind speed does not remove this heat, much like a car can overheat if not moving. Conversely, with high pressure during the winter when the sun is week there is only slow out-flowing air and no in flowing warm air. Therefore, there are other considerations than just a cloudy sky keeps the temperature in.
Out of interest Adolf Galland gave four reasons why the Luftwaffe lost the Battle of Britain; number 1 was the unpredictable nature of Britain’s weather.
“The radiative greenhouse effect is an attempt to explain how the low temperature isothermal stratosphere can heat a planet’s surface.”
It doesn’t “heat it up”, you turkey, it reduces the rate of cooling!
Large amount of radiative energy going OUT. Smaller amount of radiative energy coming BACK. Result – less energy overall leaving.
So, with the Sun heating the ground, and the ground losing heat at a lesser rate than it would in vacuum, the ground gets warmer than it would in vacuum. It reaches equilibrium at a higher temperature.
Voila! Greenhouse Effect! Bleeding simple!
(No doubt someone is going to “debunk” this by nattering on about photons, or Wein, or by saying that the Earth is *never* in equilibrium. (Which it isn’t. And your point is?) Let someone else argue with them. Never debate with a fool, people might not be able to tell the difference.)
Gus,
Here’s a thing; the maximum surface temperature measured on the Moon in a vacuum is 100C (373K) but on the Earth the maximum recorded surface temperate with an atmosphere of 100 kPa pressure is 58C (331K). The Moon orbits the Sun in tandem with the Earth at the same overall distance (Yes its absolute distance changes from New Moon to Full Moon and then back again) but, averaged over a year, the Moon experiences the same intensity of solar radiation as the Earth does.
So what do I conclude from this? That the sun heats more in a vacuum and that on Earth the atmosphere cools its surface.
Earth’s atmosphere and oceans move the heat around. On the moon, it stays put, except for moving through the ground.
The moon is thus also colder than earth ever gets. Our air and water both cool and warm the surface.
Philip Mulholland November 20, 2017 at 1:57 pm
Right facts, but I fear you’ve reached the wrong conclusion. It’s more complex than that. I discuss this question in a post called “The Moon Is A Cold Mistress” …
w.
Willis,
This is what Uncle Gus wrote:-
Do you want to correct him or not?
Philip Mulholland November 20, 2017 at 10:17 pm
I’m not sure what I have to do with this, as I haven’t commented on what Uncle Gus said. However, it is true that ceteris paribus, the surface with a GHG-containing atmosphere will indeed be warmer than it would be without said atmosphere … so it is not at all clear what you think I should “correct”.
What am I missing here?
w.
Willis
Try reading this:-
Gus thinks that a surface in the presence of an atmosphere can be warmed to a higher temperature than a surface in a vacuum can be.
The invention of the vacuum flask
Philip Mulholland November 21, 2017 at 1:34 am
Thanks for the reply, Philip. So your contention is that if we removed the earth’s atmosphere the surface would get warmer??? Seriously?
Stefan and Boltzmann disagree strongly with that … in a vacuum, the earth’s maximum possible temperature would be 5°C, assuming the earth were a superconducting sphere. Given that it is not a superconducting sphere, the temperature would be lower. For example, the moon, at the same distance from the sun as the earth, has an average temperature far below 0°C, because it rotates so slowly …
w.
Willis,
One last time, I am talking about peak maximum temperature. In a vacuum, where there is no fluid (gas) to cool the surface the peak maximum temperature is higher under an equivalent radiation load.
Philip Mulholland November 21, 2017 at 11:14 am
You may well have been talking about that, Philip.
But Uncle Gus, who we were discussing, definitely was NOT talking about that. You asked me, was there anything I would correct about the statement of UNCLE GUS, not about your statement … and he said nothing about max temperatures, he was talking about mean temperatures.
And what is with the “One last time …”, like you’re at the end of your tether and I’m too dumb to understand but you’ll give me one last chance. I don’t care which end of the tether you’re at, I was replying to your question about Uncle Gus.
w.
PS—the reason for the difference in max temperature of the earth and the moon is not the vacuum. It is a result of the fact that their rotation rates are different. Because the moon’s surface can cool or heat for a longer period, the cold side gets much colder and the warm side gets much warmer than the earth.
We know this because if the moon were spinning at say 1 rpm, where it would be evenly heated, given the moon’s albedo (0.12), the S-B equation says it would be at about -4°C. So clearly, the vacuum isn’t the reason for the high temperatures.
I go into this in some detail in “The Moon Is A Cold Mistress“, which you might enjoy.
Willis,
Dinna fash. Here (I hope) is something we can agree on, slowly rotating planets heat more than rapidly rotating ones do.
At thermal equilibrium, any time a molecule absorbs a photon, it will emit one (follows from Kirchoff’s radiation law.) There is an equal probability of emitting upward or downward. The greenhouse effect results because the sun / earth /space system is not in thermal equilibrium and an upward emitted photon may escape to space, while the ones emitted downward remain in the system. Missing from this discussion is convection, which controls surface temperature and indeed the whole temperature profile of the troposphere. Discussion of lapse rate and equivalent emission height is also missing. Also, the great source of stability of our climate, the Stephen-Boltzmann law which says that radiant emission goes as the fourth power of temperature, with the result that any increase in temperature is opposed by a massive increase in energy radiated to space.
Infrared photons have far too little energy to change the orbit of an electron. The ‘greenhouse effect’ is entirely due to resonances in the elasticity of the chemical bonds between atoms in the gas molecule.
The 15um resonance is below the emission peak for typical ground temperature. However the photon distribution is wide enough that some will be in this range. Thus CO2 will have some effect on outgoing radiation.
When a CO2 molecule absorbs a photon, it may re-radiate it in a random direction, or it may transfer the energy to the bulk gas by way of colliding with a N2 or O2 molecule. Exactly which mechanism will predominate is hard to predict. The time interval of each is about the same, at 10ns.
N2 and O2 not being IR emitters, essentially have no way of liberating heat to space. However, if there is CO2 in the upper atmosphere, molecular collisions with this may provide a route for heat to escape to space, by way of exciting IR emission.
Hence the overall situation is far from simple, and it may even be that the presence of CO2 cools the upper atmosphere.
Perhaps more importantly there are numerous proofs of the fact that at sea level, CO2’s effect is already evident at very low concentrations, therefore further increases are unlikely to significantly increase its greenhouse effect. However at high altitudes this may not be the case. If so, adding CO2 to the atmosphere might actually cause planetary cooling rather than heating, since it will have little effect at sea level but will enhance the ability of the upper atmosphere to lose heat to space.
I’ve done a number of analyses of this on our website. For example:
https://iwrconsultancy.co.uk/science/modtran

https://iwrconsultancy.co.uk/science/computed
https://iwrconsultancy.co.uk/science/co2maths
That is a very interesting graphic.
Ian Macdonald November 20, 2017 at 5:09 am
Infrared photons have far too little energy to change the orbit of an electron. The ‘greenhouse effect’ is entirely due to resonances in the elasticity of the chemical bonds between atoms in the gas molecule.
The 15um resonance is below the emission peak for typical ground temperature. However the photon distribution is wide enough that some will be in this range. Thus CO2 will have some effect on outgoing radiation.
The 15 𝝻m resonance is very close to the emission peak at 288K.
When a CO2 molecule absorbs a photon, it may re-radiate it in a random direction, or it may transfer the energy to the bulk gas by way of colliding with a N2 or O2 molecule. Exactly which mechanism will predominate is hard to predict. The time interval of each is about the same, at 10ns.
The mean time between collisions in the lower troposphere is ~0.1ns the re-emission time is orders of magnitude longer.
Ian – Where did the 10ns time come from? I calculated approx 0.2 ns average time between contacts using the calculator at http://hyperphysics.phy-astr.gsu.edu/hbase/kinetic/frecol.html and found relaxation time of 6000 ns at 210 K at http://onlinelibrary.wiley.com/doi/10.1002/qj.49709540302/abstract. Other refs allowed deternination of effect of temperature resulting in approx 5300 ns at 280 K.
When you say N2 and O2 cannot liberate ‘heat’ to space, are you saying they do not emit energy? I keep hearing that N2 and O2 do not absorb or emit IR, but I’m also aware that any matter above 0 Kelvin emits radiation at a rate determined by its avg temp. If an object or region of space emits radiation then it’s temperature decreases whether that energy is IR or not.
Given that N2 and O2 make up about 4950 Trillion tons of the total ~5,000 Trillion tons that comprise our atmosphere, it seems fairly obvious that the energy being emitted by N2 and O2 is where most of the energy is lost from the atmosphere radiating.
Steven Morris November 20, 2017 at 2:01 pm
When you say N2 and O2 cannot liberate ‘heat’ to space, are you saying they do not emit energy? I keep hearing that N2 and O2 do not absorb or emit IR, but I’m also aware that any matter above 0 Kelvin emits radiation at a rate determined by its avg temp. If an object or region of space emits radiation then it’s temperature decreases whether that energy is IR or not.
Does not apply to gases which can only emit at those wavelengths which correspond to allowed transitions between energy levels of the molecules. The blackbody relationship provides an upper limit to such emissions, there are no significant allowed transitions for either N2 or O2 within the range of frequencies emitted by a blackbody at the earth’s temperature.
Given that N2 and O2 make up about 4950 Trillion tons of the total ~5,000 Trillion tons that comprise our atmosphere, it seems fairly obvious that the energy being emitted by N2 and O2 is where most of the energy is lost from the atmosphere radiating.
Not true as it’s based on your false premise.
Notice that the absorption/re-emission band of CO2 ranges from about 13 to 17 microns, corresponding to a temperature range of -51 to -103 degrees C, i.e., lower than is normally experienced at Earth’s surface (which corresponds to a range of about 9 to 13 microns), hence how could CO2 possibly behave like a greenhouse gas in the Earth surface environment? This is why infrared astronomer Michael Sanicola was able to report finding no back-radiation from CO2 that could interfere with his astronomical observations.
davidbennettlaing November 21, 2017 at 5:55 am
Notice that the absorption/re-emission band of CO2 ranges from about 13 to 17 microns, corresponding to a temperature range of -51 to -103 degrees C
There is no such correspondence. Blackbody radiation at the earth’s temperature peaks at 550cm-1 for 288K (peak radiance 0.136 W/m2/sr/cm-1) whereas the CO2 absorption band is centered at 667cm-1 (radiance ~0.132 W/m2/sr/cm-1). So CO2 absorbs near the maximum of the earth’s BB energy distribution (~97% of max).
By the way you shouldn’t believe anything you read on Steve Goddard’s site, not even the names!
When you look at the whole spectrum, being near the maximum, isn’t all that significant.
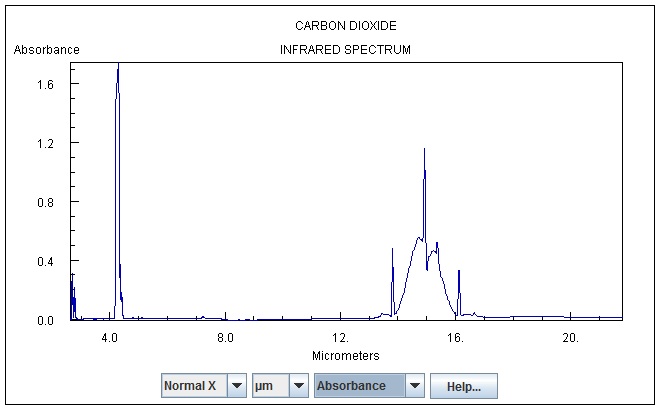
And remember water overlaps co2.
Here’s just co2
and Water
But, I think that as water condenses at night in the troposphere, that sensible heat, that IR will light up co2 @15u. And co2 might light up during the day, but IIRC that blocks about as much as it adds during the day. That why the focus on Min T. But they’re completely wrong about Min T, it just follows dew point, which follows the ocean cycles, and since air temps respond differently during the day over ocean than it does land, and the hemispheres are asymmetrical to land area, global Temp averages are going to cycle with the ocean cycles.
Then, what I found shows why co2 will never for the foreseeable oil reserves become to high for the atm water to not regulate Min T. If we doubled Co2, added another 3.7W/m^2, there’s just be ~3.7W/m^2 decrease in the complementary water vapor forcing. It condenses as much water vapor as it can, to try and stay above dew point. And it looks like it’s supplying 40 or 50W/m^2, That’s a lot of doubling.
Not to say 2,000ppm is acceptable, but even an absurdly high 800ppm, isn’t a temp problem for over heating the planet.
micro6500 November 21, 2017 at 8:50 am
“There is no such correspondence. Blackbody radiation at the earth’s temperature peaks at 550cm-1 for 288K (peak radiance 0.136 W/m2/sr/cm-1) whereas the CO2 absorption band is centered at 667cm-1 (radiance ~0.132 W/m2/sr/cm-1). So CO2 absorbs near the maximum of the earth’s BB energy distribution (~97% of max).”
When you look at the whole spectrum, being near the maximum, isn’t all that significant.
If you run it through Modtran you’ll find that about 10% of the total is absorbed by CO2.
And remember water overlaps co2.
If you’d run the spectra at a realistic resolution you’d find that both spectra are composed of discrete lines which in general are a different wavelengths and therefore don’t interfere with each other.
Run Spectralcalc between 600 and 700 cm-1 for both CO2 and H2O and you’ll see how sparse the H2O lines are compared with the CO2.
Phil; Who is “Steve Goddard?” I know that Wien temperatures can’t actually be inferred from the line spectra of gases, but my point is that even if you were to construct a continuous, bell-shaped, Planck-like curve from the radiation within the band of CO2’s output (13to 17 microns), the closest thing to a Wien temperature you’d get would be the most intense line, which is 14.95 microns. That would correspond to a Wien temperature of -79 degrees C, which is well below any temperature experienced at Earth’s surface. In fact, of course, gases acquire their temperatures not from radiation, but from the kinetic energy of their constituent molecules, which is a function of pressure, but sticking with the radiative argument on which greenhouse theory is based, the band within which CO2 can emit just doesn’t qualify the gas for greenhouse function within Earth’s atmosphere.
davidbennettlaing November 22, 2017 at 5:54 am
Phil; Who is “Steve Goddard?”
It’s the pseudonym of the host of the website where the bogus post purported to be by a non-existent IR astronomer Michael Sanicola, which you referred to, was posted.
I know that Wien temperatures can’t actually be inferred from the line spectra of gases, but my point is that even if you were to construct a continuous, bell-shaped, Planck-like curve from the radiation within the band of CO2’s output (13to 17 microns), the closest thing to a Wien temperature you’d get would be the most intense line, which is 14.95 microns. That would correspond to a Wien temperature of -79 degrees C, which is well below any temperature experienced at Earth’s surface.
So what? A blackbody at -80ºC emits its peak radiation at 15 microns, that has no relevance to the BB emission from the earth’s surface. The earth’s surface at 288K emits much more 15 micron radiation than a BB at -80ºC (193K), 5.8 W/m2/sr/µm vs 1.1 W/m2/sr/µm. What’s of interest is the IR emitted by the surface.
Phil; Michael Sanicola is apparently a pseudonym for Morgan Wright, who states “I’m an opticist, who specializes in optics and IR. I worked for GE’s infrared department and designed infrared telescopes for GE that were used by NASA in outer space. I invented the ambient temperature microbolometer.” By whatever name, the individual appears to know what he’s talking about. As to your contention that its all about Earth’s IR output, that’s just not the case. The important consideration is the radiation that CO2 gas is capable of re-emitting, and that lies between the limits of 13 and 19 microns, as I (and MODTRAN6) said.This band is represented in Earth’s BB spectrum, but it’s in the right hand, “cold” wing of the spectrum, corresponding to temperatures below those of Earth’s surface, where none of it can therefore influence Earth’s actual surface temperature range.