Poste invité par David Middleton
Sacré bleu !
Trump Budget Attacks Montreal Protocol, Reagan’s Crown Jewel
May 24, 2017 David Doniger
The Trump FY18 budget proposal slashes funding to support compliance with the Montreal Protocol, Ronald Reagan’s treaty to save the ozone layer.
The cut—which appears to be on the order of 40 percent—welches on U.S. international commitments and will imperil the global phase-out of ozone-destroying chemicals.
The Montreal Protocol—widely considered the world’s most successful environmental treaty—was negotiated under President Ronald Reagan in 1987 and is his crowning environmental achievement. It has been strengthened repeatedly under both Republican and Democratic presidents.
[…]
This year will mark the 30th anniversary of the Montreal Protocol, the “treaty to save the ozone layer.” This treaty is often cited as the textbook example of a successful environmental treaty which literally saved the planet at a reasonable cost. In many ways it is a perfect model for global efforts to save the planet from climate change:
- Instrumental measurements of the phenomena do not date back far enough to establish natural variability.
- Measurable mitigation success won’t occur for decades.
- Unverifiable claims that things would be worse if we hadn’t acted are treated as evidence that the treaty was successful.
- The threat was hyped.
- The true economic costs have been blurred.
This excerpt from a Smithsonian Magazine article sums it up quite well:
[…]
Rumors of blind sheep—the increased radiation was thought to cause cataracts—and increased skin cancer stoked public fears. “It’s like AIDS from the sky,” a terrified environmentalist told Newsweek’s staff. Fueled in part by fears of the ozone hole worsening, 24 nations signed the Montreal Protocol limiting the use of CFCs in 1987.
These days, scientists understand a lot more about the ozone hole. They know that it’s a seasonal phenomenon that forms during Antarctica’s spring, when weather heats up and reactions between CFCs and ozone increase. As weather cools during Antarctic winter, the hole gradually recovers until next year. And the Antarctic ozone hole isn’t alone. A “mini-hole” was spotted over Tibet in 2003, and in 2005 scientists confirmed thinning over the Arctic so drastic it could be considered a hole.
Each year during ozone hole season, scientists from around the world track the depletion of the ozone above Antarctica using balloons, satellites and computer models. They have found that the ozone hole is actually getting smaller: Scientists estimate that if the Montreal Protocol had never been implemented, the hole would have grown by 40 percent by 2013. Instead, the hole is expected to completely heal by 2050.
Since the hole opens and closes and is subject to annual variances, air flow patterns and other atmospheric dynamics, it can be hard to keep in the public consciousness.
Bryan Johnson is a research chemist at the National Oceanic and Atmospheric Administration who helps monitor the ozone hole from year to year. He says public concern about the environment has shifted away from the hole to the ways in which carbon dioxide affects the environment. “There are three phases to atmospheric concerns,” he says. “First there was acid rain. Then it was the ozone hole. Now it’s greenhouse gases like CO2.”
It makes sense that as CFCs phase out of the atmosphere—a process that can take 50 to 100 years—concerns about their environmental impacts do, too. But there’s a downside to the hole’s lower profile: The success story could make the public more complacent about other atmospheric emergencies, like climate change.
[…]
Read more: http://www.smithsonianmag.com/science-nature/ozone-hole-was-super-scary-what-happened-it-180957775/#W5LRedAOT3ymcci1.99Give the gift of Smithsonian magazine for only $12! http://bit.ly/1cGUiGvFollow us: @SmithsonianMag on Twitter
Rumors of blind sheep!!! Drastic measures to eliminate CFC emissions!!! Thirty years on, the ozone hole has not significantly changed… Although it would have been worse without Montreal (wink, wink) and it will heal by 2050 (nudge, nudge). This is from NASA’s Ozone Hole Watch page:
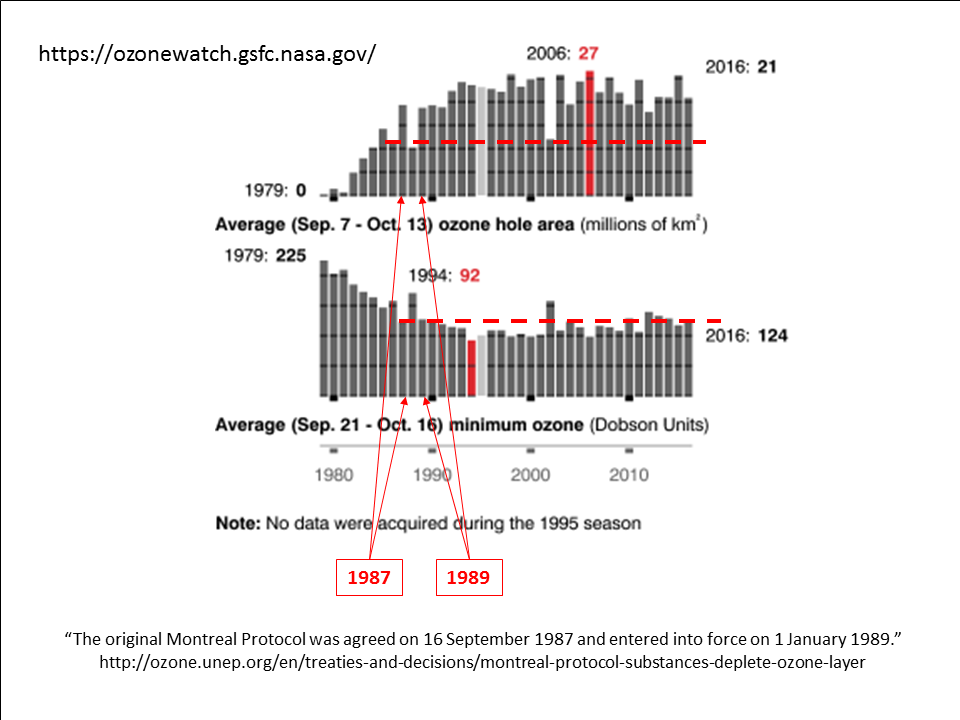
The annual thinning of the ozone layer over Antarctica has occurred during every Antarctic spring in which anyone was actually trying to measure it and continuous records only date back to 1986.
Ozone in the upper atmosphere is created when UV radiation from the Sun strikes oxygen molecules. This leads to the creation of ozone. The ozone layer doesn’t work like sunscreen; it’s more like reactive armor.
Stratospheric ozone is created and destroyed primarily by ultraviolet (UV) radiation. The air in the stratosphere is bombarded continuously with UV radiation from the Sun.When high energy UV rays strike molecules of ordinary oxygen (O2), they split the molecule into two single oxygen atoms.The free oxygen atoms can then combine with oxygen molecules (O2) to form ozone (O3) molecules.
O2 + UV light → 2 O
O + O2 + M → O3 + M (where M indicates conservation of energy and momentum)
The same characteristic of ozone that makes it so valuable – its ability to absorb a range of UV radiation – also causes its destruction. When an ozone molecule is exposed to UV energy it may revert back into O2 and O. During dissociation, the atomic and molecular oxygens gain kinetic energy, which produces heat and causes an increase in atmospheric temperature.
During the Antarctic winter very little sunlight hits the upper atmosphere over Antarctica and the Antarctic polar vortex prevents much in the way of atmospheric mixing between the polar and higher lower latitude air masses. This leads to an annual depletion of Antarctic ozone from mid-August through mid-October (late winter to mid spring). As the Antarctic spring transitions to summer, there is more exposure to sunlight and the ozone layer is replenished.
This process has occurred since the dawn of continuous ozone measurements in 1986. NOAA’s Earth System Research Laboratory / Global Monitoring Division used to feature a very disingenuous chart on their ozone page.
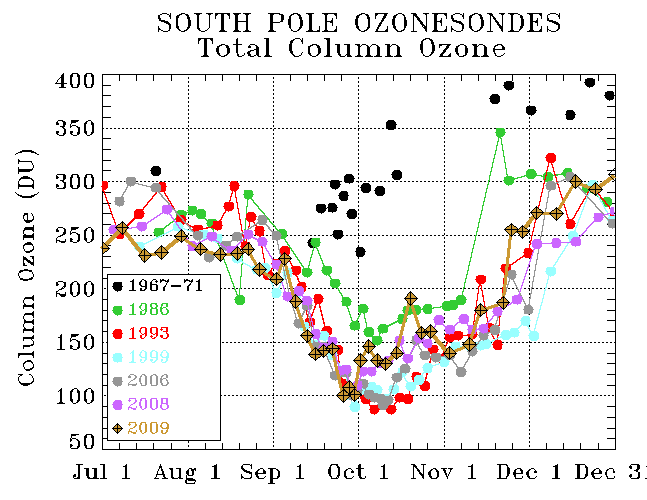
The image above has been replaced with the following:

The NOAA ESRL/GMD charts imply that the annual ozone hole did not exist during an earlier period of measurements from 1967-1971. This is wrong. The actual data from 1967-1971 clearly show that the annual ozone hole did exist. It may have been less pronounced at higher altitudes and it may have bottomed out in September rather than October; but it did exist. At low altitude (200 MB and 400 MB) it was nearly identical to the present-day…
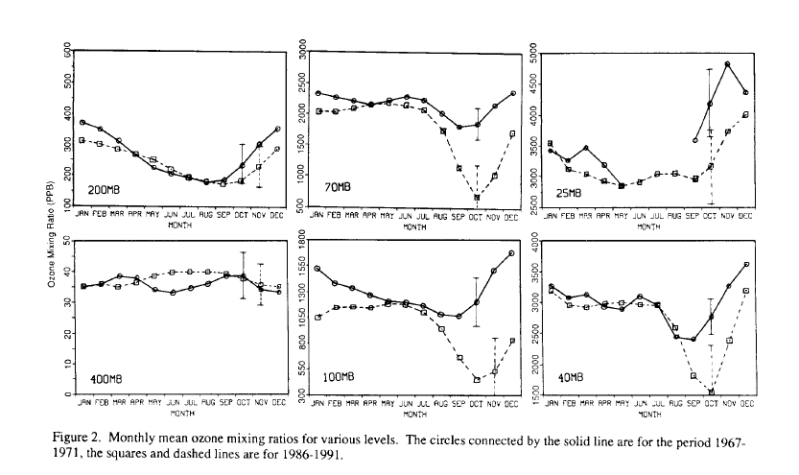
There are a lot of reasons why earlier measurements differ from the modern data:
- The older data were sparsely sampled (1/4 the number of profiles) and the earlier ozonesonde balloons rarely, if ever, reached higher altitudes (40 MB and 25 MB).
- Natural climate oscillations. 1967-1971 was during a period of global cooling. 1986-1991 was during a period of global warming.
- Fluctuations in the polar vortex. It has been demonstrated that fluctuations in the polar vortex can influence Antarctic ozone observations (Hassler et al. 2010).
- Chlorofluorocarbons (CFC’s). It is possible that CFC’s did exaggerate the Antarctic ozone hole. However, the data clearly show that CFC’s did not create it.
Anthropogenic CFC emissions may very well have contributed to the area and depth of the annual Antarctic ozone hole. Reducing CFC emissions was a good thing. However, there clearly is no evidence that this was a crisis which required immediate, drastic, global action. CFC’s could have easily been replaced gradually with substances less hostile to the stratospheric ozone layer. The panic was so severe, that no cost analysis was even required for the Montreal Protocol.
No estimate of the global cost of replacing CFC-based technology has been made, but the Environmental Protection Agency estimated that the cost in the United States alone would be some $3 billion, mostly for replacing equipment made obsolete or unusable by the ban.
Unsurprisingly, the Competitive Enterprise Institute estimated that the cost in the United States would be more than ten-times the EPA estimate.
The CFC phaseout may well be the single most expensive environmental measure taken to date. During the policy debate, the costs were underemphasized to the point that they never became an important factor. The impact on consumers was scarcely considered. It may be too late to reverse course on the CFC phaseout, but it can serve as a lesson for the future.
I have not been able to find any recent estimates of the costs imposed on the U.S. economy by the Montreal Protocol. However, I know the costs were not insignificant and all of the benefits are either: 1) model-based assumptions and/or 2) 50 years in the future.
The ozone hole panic cost many people a lot of money. Refrigerating fluids, particularly in automobile air conditioners, had to be replaced. If you were the owner of a 1980’s motor vehicle or in need of air conditioner repairs in the 1990’s, you may as well have traded your vehicle in; because the cost of repairs became almost prohibitive due to new environmental regulations related to CFC’s (I owned a 1983 Chevy Camaro back then). If your home HVAC system was manufactured before the CFC ban, you faced a similar dilemma. The elimination of CFC’s even drove up the cost of asthma breathalyzers inhalers. The elimination of CFC’s may have evened worsened AGW!
The economic cost of this particular chapter of environmental junk science was minuscule in comparison to that of the current environmental swindle (anthropogenic global warming)… But this should serve as a clear reminder that citizen scientists have a duty to always check the work of government and academic scientists when they start Chicken Littling about the latest environmental crisis du jour.
References:
[1] Data Visualization >> South Pole Ozone Hole >> South Pole Total Column Ozone
[2] Hassler, B., G. E. Bodeker, S. Solomon, and P. J. Young. 2011. Changes in the polar vortex: Effects on Antarctic total ozone observations at various stations. GEOPHYSICAL RESEARCH LETTERS, VOL. 38, L01805, doi:10.1029/2010GL045542
[3] Oltmans, S. J., Hofmann, D. J., Komhyr, W. D., Lathrop, J. A. 1994. Ozone vertical profile changes over South Pole. NASA. Goddard Space Flight Center, Ozone in the Troposphere and Stratosphere, Part 2, p 578-581
Featured Image:

“Defeated” Paris?
That’s hardly le mot juste.
We came, we saw, we bombed the City of Love back to the Renaissance.
Now let’s bring our boys home. They’re going to need some solid R&R before shipping off to fight Trump’s War on The Planet Earth Itself. Let me tell you, that’s one oblate spheroid that can put up a fight. It’s no cheese-eating surrender-SandPerson, that’s fer sure.
Just ask George Carlin…
Well… if he was still alive, you could ask him about the resilience of the planet.
La guerre climatique is enough to get anyone down, so if the constant toll of body-bags has all become a bit depressing, check out these uplifting, moving letters from the parents of our servicemen and servicepeople on the ground in gay Paris (that’s the Paris which is not in Texas). Booya!
It’s enough to make one proud of one’s lapsed US passport.
Yes it can. It’s called “Nature”. It can reform Ozone, absorb and transform Man’s relatively tiny bit of CO2 into plants and food.
Man can’t control “Nature”, let alone Global Temperature, but some men will do anything to control the people on the globe.
(George Carlin got it right.)
I still don’t understand how vastly heavier than air gasses like CFC’s, known to be decomposed rapidly by soil bacteria, make it into the upper atmosphere.
Nor do I understand how millions of square kilometers of natural, dried powdered halide salts are overlooked as a possible cause for the ozone thinning.. especially when my understanding is beyond modelling, all that’s being sampled up their is halide ions
The CFCs are very stable in the lower atmosphere so they disperse until they effectively behave as single molecules.
Molecules are being bounced around all the time very violently by brownian motion, so whilst molecules heavier than Nitrogen will fall they fall extremely slowly. Think how much faster a rock falls than a speck of dust, and then think at least a billion times lighter.
This means that even a very gently upward breeze will move CFCs with it, ready to get broken down by UV light and start destroying Ozone.
The altitudes we are talking about can support ice crystals in clouds which are much heavier than a CFC molecule.
I was thinking more of this “CORRECTED: in the everyday world, gases do not expand to fill their containers.”
‘if you squirt some carbon dioxide out of a CO2 fire extinguisher, it will not instantly expand to fill the room. Instead it will pour downwards like an invisible fluid and form a pool on the floor. It behaves similarly to dense sugar-water which was injected into a tank of water: it pours downwards, and only after a very long time it will mix with the rest of the water. “Mixing” is very different from “expanding to fill!”
The “Perfect” treaty that does nothing? I guess in politics that is a perfect treaty.
Did lots for AC chemical producers and patronage jobs….
Just at the time patent was running out on CFCs IIRC 😉
Actually a perfect treaty is one that cost the USA more in as many ways as possible than the other signatories. It is one that makes us less competitive and less able to produce wealth while the others grow their economies in order to pay for social welfare programs.
Why is it nobody mentions the fact that the Dupont Chemical Co. funded researcher, on whose research the entire Montreal Protocol was based, has admitted to having made up the “science” of how CFCs supposedly react with ozone?
Now, we know that it is solar radiation and N2 gas in the atmosphere that breaks down ozone, not CFCs.
The Ozone Scare was a scam perpetrated by Dupont Chemical and jumped on by a bunch of politicians and environmentalists looking for a cause. Ignorance prevailed.
Will I get compensated for the expensive HVAC package unit that had much more expensive design and components and essentially-required maintenance contract for the higher failure rate of the higher pressure coil units? This was not in the “pennies” of cost claim of the advocates when it was forced through public policy. I got the real story from the HVAC technicians on the back end of the policy fraud.
As did I back in the 1990’s… Every time my 1980’s vintage AC units had to be recharged and/or repaired.
My parents had a CFC chest freezer bought back in the 1940s. It had a very simple design and never did leak its CFC. It was still running in 2011 when my parents died and we sold the freezer to someone for ten dollars. For all I know it’s still running!
Congress was leaning against the Montreal Protocol when NASA published a finding that they had discovered an north hemisphere ozone hole and it was impacting how much UV reached the ground in the US. This news caused a number of senators who had been wavering to start supporting the treaty.
About 6 months after congress voted to join the protocols, NASA admitted that they had made a mistake and there was no northern hemisphere ozone hole after all.
Sounds remarkably like NASA’s Palmdale Bulge.
Yes, and the debate over the bulge and satelite measured land elevation was measured in 100s of mm!
Yet we claim almost perfect elevation change accuracy on the ever moving ocean surface with ever changing multi decadal lunar cycles.
Just because a computer spits out a precise number, it does not make it accurate.
It was probably just a coincidence that the patent for CFC’s was about to run out.
Dupont made a bundle on replacing CFC’s.
… while claiming the new mantle of “Green Company”
Exactly what I was thinking! The phrase “The ozone hole panic cost many people a lot of money.”
should be much more accurately written as “The ozone hole panic MADE many people a lot of money.”
specifically, large international corps like DuPont and other chemical corps, who were about to be undercut on price by cheap 3rd world competition.
DuPont, like all businesses, is in business to make money. The fact that DuPont turned this to their advantage is a *good* thing. It’s the silver lining to the dark ozone hold cloud.
Money does not disappear. When it “costs” someone. it means someone else is receiving it.
OK, the two of you can cut it out right now! CFC’s were patented back in 1932 or so and have been long off patent. Now, if you want to ague that the replacement compounds offered higher profit margins, have at it, but be sure to look up when the base compounds were patented too.
DuPont had two choices: 1) Go out of business or 2) Figure out how to profit from the Montreal Protocol:
http://onlinelibrary.wiley.com/doi/10.1002/(SICI)1099-0836(199711)6:5%3C276::AID-BSE123%3E3.0.CO;2-A/abstract
Unlike fossil fuels, CFC’s were relatively easy to replace and DuPont was well-positioned to market replacement chemicals.
DuPont held the patent for Freon until 1979, a year after it was banned in the U.S. …
https://en.wikipedia.org/wiki/Chlorofluorocarbon
DuPont didn’t just “figure out how to profit from the Montreal Protocol” – DuPont FUNDED the lawyers and activists who WROTE the Montreal Protocol, with the specific goal of creating something that would be intensely profitable for DuPont.
Nice try, David, but that’s a process patent. It’s a novel way to make existing compounds, not a new compound, and has nothing to do with anyone else making “Freon” by some other route. Thanks for playing.
I’m seeing a whole lot of banned in 1978, expires in 1979, on those documents.
Strange how suddenly when the eco fraud bubble is fixing to burst, now it’s Ronnie’s fault.
Reagan didn’t do this. More accurate to say they did this to him.
Here’s a view of the ozone hole from Mars.
http://btc.montana.edu/ceres/html/Polar/Images/marsnpc.jpg
Don’t get to see that angle a lot.
http://images.scienceworldreport.com/data/images/full/21458/martian-polar-ice-caps.jpg
here’s a cleaner pic. Notice how you can see dust storms right up to the edge of the hole.
We’ve been over this before. As far as I can tell DuPont’s patents had already expired. What did happen is that they had patents on replacements for CFCs that they could only sell if the government made CFCs illegal. Whichever way, the money trail ends at DuPont. WUWT
As many have pointed out R-12 (commonly called Freon) was well out of patent, the patents in question were process patents that DuPont held that likely helped them to make the compound better, faster, cheaper etc. But even with that they still would have been fine. Where their true advantage lie, however, was in not only the replacement but the replacement for the replacement. IIRC HFC-134A was the immediate replacement which was scheduled to be banned at a later date than R-12. At the time of passing the next generation refrigerants were still on the drawing board. That is where DuPont’s true advantage was. They had the resources and wherewithal to be on the leading edge of the replacement movement and could secure a majority stake in that market as it developed. The Montreal protocol took what was essentially a static market and put it back in play. The replacement chemicals still don’t have the thermal efficiency and thus the cooling capacity of the old R-12 systems. We get stuck with newer and better that is worse than what it is replacing. Which causes us to run our systems longer and use more electricity. That leads to burning more coal and voila, the cure to the last disease helped to cause the current one. Gotta love how that works. It’s the gift that keeps on giving.
The ozone hole was first measured by Dobson back in 1950’s. We have no idea how long it appeared before that. CFC’s have little if anything to do with hole. Volnaco’s in Antarctica spew chlorine into air.
Volnacos are covfefe, IMO.
Well that’s not a very nice thing to say about volcanoes. Or maybe it is a very nice thing to say about volcanoes. Please let me know which.
“BallBounces on June 5, 2017 at 7:16 am
Volnacos are covfefe, IMO”
It’s 5.30am here and I’ve just had a great laugh to start the day – thanks BallBounces!
The treaty was applied after 1988. This NASA poster only goes to 2012, but things haven’t changed much.
http://www.rockyhigh66.org/stuff/ozoneposter_front.jpg
http://nineplanets.org/news/wp-content/uploads/2014/12/cassinijupiter.jpg
Here’s the hole from yet another angle. THis time from the Cassini spacecraft. As you can clearly see the hole doesn’t change much due to distance.
papiertigre (june 5,2017 at 4:22) your picture looks strangely like Jupiter. Is it???
That’s Jupiter with a big old ozone hole. Well probably a helium methane hole, but why quibble?
There’s a hole up top all right, but it’s not in the ozone layer.
Yes we need plug the money hole.
Beijing next?
https://www.nytimes.com/2017/06/04/world/asia/china-media-climate-trump.html?_r=0
On the Paris exit, one of the most asinine excuses for staying put in the Paris Accord was that the commitment was “voluntary & non binding”.
As opposed to what? A binding agreement under threat of enforcement by the UN or world court?
Was the US $uppo$ed to follow through voluntarily while naively expecting China, Russia and India would so the same when their time came many years later?
two statements
Of course the Chinese are against us leaving the accord. `1) we aren’t shooting ourselves in the manufacturing foot and 2) they won’t get more US money.
The ozone hole is not the issue. The issue is ozone depletion but without evidence of depletion.
https://papers.ssrn.com/sol3/papers.cfm?abstract_id=2748016
https://papers.ssrn.com/sol3/papers.cfm?abstract_id=2612810
My original home air conditioning condenser unit installed in 1985 was replaced a few years ago with a comparable unit that’s easily three times the size thanks to the Ozone Hole BS.
CFCs may be released by volcanoes in significant quantities: http://cfc.geologist-1011.net/
Also, there is a bit of a deafening silence about CFC impacts on ozone in the recent literature, would anybody dare to (or be able to) publish sceptical science on this issue?
Yep…
https://pubs.usgs.gov/of/1997/of97-262/of97-262.html
Well now that’s something I never knew. Thanks for that!
Won’t be long before he says “auf wiedersehen” to Bonn. Better take cover. Greenie greengoo head-explosion could be hazardous to your health.
At 2 – 8 ppm ozone must be more powerful than CO2 at 400 ppm or water vapor at 2,500 ppm.
Nicholas-San:
Water vapor is at around 30,000ppm, not 2,500ppm…
Vapor phase water over the tropical ocean surface is around 30000 ppm based on the saturation curve for tropical temperatures.
Any meteorology text shows total precipitable water is about 2.5 cm liquid water, about 2.5 kilograms per square meter.
2.5 kg divided by the total column weight of 10000 kilograms is about 0.00025 or 0.25 percent which is the same as 2500 ppm by weight.
Satellite measured total water vapor on a global basis averages 2500 ppm
http://www.climate4you.com/GreenhouseGasses.htm
That’s why some models use the 2500 ppm global average even though actual water vapor in any one location varies radically over and under that number.
Typical office building maintains 25C with 50 percent relative humidity which mean there is nearly 1 percent water vapor by volume, thats 10000 ppm wv.
Once upon a time, early in the game I was sent to question a climate modeler in public who was part of a dog and pony show traveling from state to state meeting elected officials and holding public workshops. After listening to his presentation and what our politicians were planning they ask if there were any questions. Note most of the people, several hundred, that were there were from organization environmental groups. There were only a handful of us that were not. I asked several questions and we spent an hour or longer discussing the whole topic. Two of my questions were as follows, (1) since water, especially oceans cover over 75% of the Earth, how it the interface being the ocean surface and the gaseous atmosphere used in the models? He explained that at the time basically all that was considered in the models was ocean surface and divided cubes of atmosphere directly above it. That led to a second question (2) So how is water vapor being considered in the models? The scientist laughed. His answer was “we are working on it.” Which led to a third question (3) Why is water vapor a significant component of the atmosphere not being modeled. He laughed again and said, “well for now considering all the ways water vapors presents itself in the environment, it is just too complex to model with any certainty that we would be close to right.”
China Receives US$95 Million Grant to End Production of Ozone-Depleting Global-Warming Gases to help Meet its Targets under the Montreal Protocol
http://www.worldbank.org/en/news/press-release/2013/05/02/china-receives-us-95-million-grant-to-end-production-of-ozone-depleting-global-warming-gases
…must be nice
The Chinese understand the art of the deal.
US negotiator: We want you to make the same costly change we are making.
Chinese negotiator: We won’t make the change unless you pay us first.
US negotiator: Make the change and we will pay you.
Chinese negotiator: You are the one wanting the change, pay first and we will change
US negotiator: OK, Here is the money
Chinese negotiator: Thanks, we will make change
US negotiator: When?
Chinese negotiator: We never agreed when, only to make change.
US negotiator: We need change by 2020
Chinese negotiator: That will cost extra
US negotiator: OK, Here is the money
Chinese negotiator: Thanks, we will make change 2020
2020:
US negotiator: Where is change?
Chinese negotiator: We made change
US negotiator: Nothing has changed
Chinese negotiator: We replaced old with new
US negotiator: New has same problem.
Chinese negotiator: Money was to fix old, not fix new
US negotiator: We want you to make the same costly change to new as well.
Chinese negotiator: We won’t make the change unless you pay us first.
US negotiator: Make the change and we will pay you.
Chinese negotiator: You are the one wanting the change, pay first and we will change
US negotiator: OK, Here is the money
Chinese negotiator: Thanks, we will make change
US negotiator: When?
Chinese negotiator: We never agreed when, only to make change.
Oh, this is a thing of beauty, and absolutely true!!!
The Chinese always get the better of those foolish Foreign Devils that think they can control what the Chinese do.
I can’t recall the source, but the phrase was priceless:
“A fool lies here who tried to hustle the East.”
Magnificent.
“The Chinese always get the better of those foolish Foreign Devils that think they can control what the Chinese do”
We got em hooked on opium.
Yes the British, not the USA, got them hooked on opium. The USA was there at the time but played a very minor role, we were yet a major power. The reason it could be done is that China was going through a period of somewhat disarray and confusion at their highest levels. Some of their leadership running the show was corrupt. However, the Chinese rose up and started a couple of wars because of it all. They lost but the Chinese have long, very long memories, so today it is all part of their “DNA” when negotiating with the West.
It took the English Empire to do it.
It was Kipling, “The Naulahka.”
Nah.
Obama could never talk so long without talking about Obama.
Which brings up a thought. Some culture place great store on proper bargaining.
Don’t or won’t bargain properly and you’re just not worth anything.
The biggest damage done by the treaty was the requirement to phase out CFC fire suppressants on ships. There is no suitable fire suppressant available which is A safe and B either takes up little space or is of low mass. Carbon dioxide is unsafe and has killed many engine room staff, even on naval vessels – it is NOT toxic, but the amount released into the engine room has to be so large to put the fire out that there is then insufficient oxygen for life. It cannot be released unless all personnel have evacuated the ER. Elsewise those left are dead. Steam used to be used but it has a similar effect – it drives out the air from the ER.
The only ‘safe’ system is believe to be that using Inergen. From the Monroe Extinguisher blurb: https://www.monroeextinguisher.com/inergen-clean-agent-fire-suppression-system/
INERGEN Agent – INERGEN agent is a mixture of three inerting (oxygen diluting) gases: 52% nitrogen, 40% argon, and 8% carbon dioxide. INERGEN gas extinguishes fire by lowering the oxygen content below the level that supports combustion. When INERGEN agent is discharged into a room, it introduces the proper mixture of gases that still allow a person to breathe in a reduced oxygen atmosphere. It actually enhances the body’s ability to assimilate oxygen. The normal atmosphere in a room contains 21% oxygen and less than 1% carbon dioxide. If the oxygen content is reduced below 15%, most ordinary combustibles will cease to burn. INERGEN agent will reduce the oxygen content to approximately 12.5% while increasing the carbon dioxide content to about 3%. The increase in the carbon dioxide content increases a person’s respiration rate and the body’s ability to absorb oxygen. Simply stated, the human body is stimulated by the carbon dioxide to breathe more deeply and rapidly to compensate for the lower oxygen content of the atmosphere.
The major problem with the system for a ship is that the cylinders are extremely large and heavy. This is because of the very large volume required to be inerted. The CFC system used far less gas, and was safe.
Dive support vessels use to use Halon systems and according to the online dictionary
halon
ˈheɪlɒn/Submit
noun
any of a number of unreactive gaseous compounds of carbon with bromine and other halogens, used in fire extinguishers, but now known to damage the ozone layer.
In the mid-1970s, Chrysler Corporation discovered that Halon was the only effective fire suppressant for use in their very popular Dodge vans – hence the great popularity, in the 70s and 80s, of Van Halon.
Don’t inhale the Halon, Michael.
Edward Lodewijk Van Halen is a Dutch-American musician, songwriter and producer. He is best known as the lead guitarist, occasional keyboardist and co-founder of the American hard rock band Van Halen.
Some designers use localised water mist systems.
See, for example, this Investigation report: –
https://www.ntsb.gov/investigations/AccidentReports/Reports/MAB1624.pdf
But it is crucial that these are triggered early, but working fire-detection systems, properly located, and big enough, to get enough water onto the location of the fire, should ensure that.
And because the mist is comparatively localised, people can survive elsewhere in the same compartment.
Auto
I would like a knowledgeable explanation of just how CFCs are supposed to reach the ozone in the first place. The ozone is high in the atmosphere, and forms from Oxygen. I can see oxygen and nitrogen being high in the atmosphere, the make up, what, 95+% of it, and CFCs are profoundly heavier molecules, and a miniscule concentration. This has never made sense to me.
Sand and dust particles are heavier than air and much heavier than CFC’s; yet they can be driven into the air and across the globe by winds. Wind and atmospheric circulation can lift lots of heavier-than-air particles into the upper atmosphere.
Diffuse gasses mix. The stratosphere also exchanges with lower atmosphere across the tropopause. There is also diffusion. https://water.usgs.gov/lab/chlorofluorocarbons/background/
Even solids can mix and diffuse into gasses. Clouds are massive, but don’t fall out of the sky. Whe tobacco is burned, the smoke particles are heavy, but don’t fall to the ground.
Thunderstorms can lift parachutists upwards, and snap the wings of unwary aircraft that fly through them. It’s called convection and its way more powerful at the molecular scale than little old gravity. Thunder clouds regularly punch up into the lower stratosphere.
Smaller scale uplift by convection means air is pretty well mixed and gravitational fractionation is trvial by comparison.
Did I not read a study not long ago that the Cl and F required for the O3 “destruction” equation could easily come from ocean evaporation?
“… come from ocean evaporation?”
Sort of. A rough ocean surface (spray) lifts the mixture of water and other compounds into the air. As water evaporates the other compounds, including Halogens, will remain to be lofted into the atmosphere. (It has been awhile since I read about these things.)
See David’s comment at 7:45.
I began reading WUWT in September 2008.
A post about that time — October 25, 2008 — is, I think, the earliest of the many you will find if you search for ‘ ozone ‘ via the box provided along the right side of the page, above. I’m not going to re-read all of them, plus all the other things about ozone I found during those earlier years.
proves that if there is a solution, you secondly have to create a problem. thirdly convince the gov’t to throw
money at unending and perpetual amount of research to pay for a house, kids through college and retirement. The bureaucratic’s American Dream.
It is interesting to not that the Ozone scare was, again, only based on model assertions and lab chemistry, and not conclusively proven at all.
The dangers of smoking are stated to be based on a strong statistical correlation, but it is surprisingly hard to find find any academic critical examination of this, and no one has yet provided a mechanism for the link between cancer and cigarettes.
It is surely the mark of flourishing science when early findings are constantly rechecked, and moribund science when they are never revisited, Kuhn points out that progress from an obsolete paradigm to a better one are driven by the re-examination of discrepancies in the pre-existing theories which are usually ignored as ‘exceptions’ by the vast bulk of regular scientists….
The problem with tobacco is that you can’t perform controlled experiments on people. You have to rely on statistical evidence.
A controlled experiment would take a group of identical twins. Have one twin from each pair start smoking at a specific age and the other would never inhale tobacco smoke. Keep all other environmental factors identical. Then measure the differences in respiratory, heart and cancer ailments. The odds are that the smoker twins would have much higher cancer, hear and lung disease rates… But you can’t perform this sort of experiment on people.
This is the only manner in which AGW and tobacco are similar. There is no “Planet B” to use as an experimental control.
… Have one twin from each pair start smoking at a specific age and the other would never inhale tobacco smoke. Keep all other environmental factors identical….
This is complete nonsense. There is no known way to keep all ‘other’ environmental factors identical – especially when you have those ‘environmental’ factors might be. Identical twins develop differently in all sorts of different ways – I have a pair myself, and have watched them.
You don’t find out anything new by random ‘controlled’ experiments unless it’s by chance – that was the sort of ‘scientific’ mistake that the ‘curio collectors’ of the 18th century made, and it’s also the well-known debating fallacy that environmental activists like to present as an argument for suppressing all opposition to their demands. You need to FIRST find a mechanism, and THEN test it using experimental controls.
Without a mechanism, statistical correlation remains an interesting curiosity, and one which, as we have seen from the AGW fraud, can readily be manufactured whenever more grants are needed.
There is a “known way to keep all ‘other’ environmental factors identical.” There just isn’t a practical, legal or ethical “way to keep all ‘other’ environmental factors identical.”
Science doesn’t start with the mechanism. It identifies the mechanism via the scientific method.
Smoking beagles .. “the failure of many investigators to induce experimental cancers, except in a handful of cases, during fifty years of trying, casts serious doubt on the validity of the cigarette-lung cancer theory.”
Of course, Richard Doll’s affable doctors study, weakly ‘researched’ was taken as proof positive over any other research, even when he was now known to be on the payroll of Monsanto and other companies, and even today he’s defended by scientists as an authority figure despite undeclared earning as high as 70 times the UK weekly income for being a ‘consultant’.. in secret.
Doll also said dioxin, 24D, Agent Orange and other nasties were all perfectly safe.>/a>
The dangers of smoking are stated to be based on a strong statistical correlation
======================
rural non-smoker – 0.1 % lung cancer
rural smoker – 1% lung cancer
urban non-smoker – 1% lung cancer
urban smoker – 7% lung cancer
What is interesting is that smoking is about as bad as living in a city as far as lung cancer goes. With all the hype about banning smoking, if we are really concerned about lung cancer the solution is to ban cities as well.
..The dangers of smoking are stated to be based on a strong statistical correlation..
It’s the ‘stated’ bit that I have problems with. I would like to see that investigated, rather than simply being treated as doctrine…
Lung X-rays show this graphically. Non-smoker city dwellers have lungs that resemble those of light smokers. Rural non-smokers have clear lungs. I’m sure that micro-inspection of alveoli in lung tissue from autopsy or biopsy shows the same.
Dodgy Geezer,
Same with the ‘saturated fats are bad for you’ mantra originally created with the 1970 Seven Countries Report (think I got that right). Diet doctors made a fortune with diets replacing everything with olive oil and avocado oil, etc. The Mediterranean Diet. The this diet. The that diet. Fat doctors making wild claims but millions with books that recommended high carb diets instead of meat, cheese, and butter (which the skinny French eat). I bought the whole enchilada.
[Disclaimer: I used to know the details of this whole thing cold three years ago, but I’m working off a sloppy memory that wasn’t interested in keeping these facts uppermost. So if I upset anyone by skewing minor details, my apologies. Feel free to correct me.]
A geeky researcher at the National Institutes of Health (NIH) about a decade ago looked up the original data claimed about saturated fats being bad for you just for s**ts and grins and decided to replicate it. He did not have the slightest intention of disproving the theory. No suspicions. It never entered his mind. It was just something that he liked to do during his off-hours. Replicate and verify medical theories. Check them out for the helluvit. Literally for the joy of research and nerdy stuff.
The long and short of it is that he discovered 80% of the data was falsified, and in 2014 the original doctor who made these claims had his papers removed from all medical journals and databases and his credentials revoked by the institutions that issued them. Disgraced. The story was so embarrassing to the NYC doctors who made a fortune for over four decades screaming about the evils of butter, pork fat, and beef tallow that the NYTimes ran only one complete story on it. (I have a copy on my storage drive, but no interest in searching for it right now.)
As my doctor said to me–he is an eat-your-saturated-fats guy now–you ever see skinny black women who have lived to be 108? What have they been cooking with all their lives? From chicken to collard greens. Pork lard. What about Jewish grandmas who are still charging up and down the stairs at 100, he said, going concerns who still boss me around like I’m 10? What have they been cooking with all their lives? Chicken fat. And he told me that the only real thing that protects your brain as your age, particularly after the age of 65, is fat. And his claim is that it has to be saturated animal fats. Butter. Butter. Butter. Pork and beef lard.
Your appetite is satiated with animal fats and you eat less, btw. Ask the French. I now buy duck fat from dartagnan.com in NJ because you can freeze it and unfreeze it and re-freeze it, and reuse it after cooking over and over and over again, and it is a true luxury in the taste department. Not to mention that it protects your heart.
Olive and avocado oils are full of saturated fats, and the Mediterranean diet is also a (mostly) saturated fat diet (and can easily be made low carb). So if these things were in vogue when sat fats were considered bad, and are in vogue now when trans fats have been discovered to be the enemy, they must be doing something right with their marketing.
DG, not completely true. There are a number of chemicals in cigarrette smoke that are known carcinogens. Granted, animal studies and statistics. But the carcinogenic pathways for these carcinogens are quite well known.
Ristvan, I did look into this and OHS guidelines for all of those chemicals were shown to be nowhere near the levels smokers experience – that is to say they’re thousands to tens of thousands of times lower.
The main carcinogen was benzene, and again filling your fuel tank at a gas station was measurable exposing people to 10,000 times the benzene smokers got in a day. Benzene’s also supposed to induce leukemia – and smoker leukemia rates are unremarkable.
I know we’re having chemical names hurled at us to induce fear, but never quantities ‘cigarette smoke contains ammonia!’ – yeah, about 100,000 times less than we exhale an hour. ‘Cyanide’ yup, a pack a day smoker would take in as much cyanide from cigarettes as they’d get from eating one whole apple every 3 weeks. It’s all rather silly considering many plants we eat contain carcinogens and toxins.
The funding cut “slashes funding to support compliance with the Montreal Protocol”. So what? If we have successfully phased out most CFC usage in the US, would there not be a reduced need for compliance checking?
They now want to phase out HFC’s and other CFC replacements.
More legislative obsolescence masquerading as enviro protection.
President Trump needs to take on the Green Mafia:
Funniest. Ad. EVAH!
+++ Thanks!
David–thanks for the charts. I’ve spent decades trying to explain that when O2 is in the shade, it can’t be ionized to O3.
Then there is the thickness part. High solar activity (ultra-violet changes the most) ionizes more O2, which makes the ozone layer thicker. Less active sun reduces ionization and the thickness. The modern industrial world has little to do with it.
David,
Back in the early ’80s I was struck by the observation that all the articles in the Media talked about how ozone was declining and UV “might” or “could” increase and cause problems for the biosphere. It was all speculation without any hard evidence showing that there was any trend in UV matching the trend in O3. Yet, academics were jumping on the bandwagon with research papers on such things as how increasing UV might be responsible for the decline in amphibian populations, or some such things.
Therefore, about 1984 I used my then-new Amiga computer to write a simulation in BASIC to predict what the surface UV might actually be. I based the simulation on Total Ozone Mapping Spectrometer ozone levels. I took into account the variation in TOA UV with sunspot cycles and also the eccentricity of the Earth’s orbit. I modeled the change in sunlight intensity at a given latitude with the seasons, taking into account both the longer path length and increased footprint for low sun angles.
What I found was that there was an apparent slow upward drift in UV intensity during the Winter over a period of years, when levels were so low as to be inconsequential. However, during the Summer (when we get our sunburns) the UV flux was flat. That is, those organisms that had evolved protective mechanisms against UV were adequately prepared to deal with the highest levels, which were essentially static.
Incidentally, David, if you look at the typical September TOMS ozone maps, just before the ozone ‘hole’ breaks up in the Spring, there are anomalously high concentrations of ozone just outside the circumpolar vortex because most ozone is created in the tropics and migrates polewards. So, when the polar vortex does break up, that ozone-rich air is able to mix with the ozone-depleted air and rapidly restore the ‘missing’ ozone.
This whole AGW alarm is strangely familiar, like a rerun of the old ozone science fiction movie.
Al Gore was also involved in ozone-hole hype.
I seem to remember that the Nobel Prize issued for the Ozone Problem/Solution said something about saving the earth sort of thing. Even if saving us was true, this was not science.