To coincide with the upcoming “World Ozone Day 2014″ declared by the U.N. for September 16th, we have some “feel good” news coming out on the 25th anniversary of the Montreal Protocol
 On September 10, an article written by Seth Borenstein of the Associated Press trumpeted a claim of good news with the headline: “Scientists say the ozone layer is recovering.” The basis for Borenstein’s headline is a statistical analysis:
On September 10, an article written by Seth Borenstein of the Associated Press trumpeted a claim of good news with the headline: “Scientists say the ozone layer is recovering.” The basis for Borenstein’s headline is a statistical analysis:
For the first time in 35 years, scientists were able to confirm a statistically significant and sustained increase in stratospheric ozone, which shields the planet from solar radiation that causes skin cancer, crop damage and other problems.
…
From 2000 to 2013, ozone levels climbed 4 percent in the key mid-northern latitudes at about 30 miles up, said NASA scientist Paul A. Newman.
Later in the article, Borenstein cites this news as “one of the great success stories of international collective action in addressing a global environmental change phenomenon.”
Is it really?

Like many superficial claims made in the mainstream media, this one reveals a different story if you scratch ever so slightly below the surface. First, a bit of background on ozone depletion: ozone reduction in the upper atmosphere is said to be caused by a chemical interaction with the inert refrigerant chemical known as “chlorofluorocarbons,” or CFCs, which is found in the piping of millions of refrigerators and air conditioners worldwide. The loss of the ozone layer in the upper atmosphere will lead to normally reflected high-energy ultraviolet light reaching the Earth’s surface, causing more sunburns and skin cancer, disruption of ecosystems such as marine plankton and algae, and other photosynthetic biomass, with a large ripple effect.
The solution was to ban certain CFCs that were said to cause a loss of upper atmospheric ozone. Borenstein’s supposed “success story” hinges on a 1987 UN resolution called the Montreal Protocol:
The Montreal Protocol on Substances that Deplete the Ozone Layer was designed to reduce the production and consumption of ozone depleting substances in order to reduce their abundance in the atmosphere, and thereby protect the earth’s fragile ozone Layer. (Source)
The Montreal Protocol certainly seems rooted with good intentions. Yet, as with so many other things we see from the UN, the actual implementation — once the cocktail parties, the speeches, and the self-congratulatory claims are over — doesn’t quite fit the original intent or the claims of success. Just a few months ago on Dec 11, 2013, NASA issued a press release containing this statement:
More than 20 years after the Montreal Protocol agreement limited human emissions of ozone-depleting substances, satellites have monitored the area of the annual ozone hole and watched it essentially stabilize, ceasing to grow substantially larger. However, two new studies show that signs of recovery are not yet present, and that temperature and winds are still driving any annual changes in ozone hole size.
…
“We are still in the period where small changes in chlorine do not affect the area of the ozone hole, which is why it’s too soon to say the ozone hole is recovering,” Strahan said. “We’re going into a period of large variability and there will be bumps in the road before we can identify a clear recovery.” (Source)
Within the span of nine months, NASA issued statements claiming of atmospheric ozone that “signs of recovery are not yet present,” there is “large variability,” it is “stabilizing,” and now, that the ozone problem is “recovering”.
So which is it? The answer may lie in the relevant political science, not the atmospheric. The Montreal Protocol is 25 years old this year, having been entered into force in 1989. When such milestones are reached, there is always pressure to make some statement that the work of the UN actually made some sort of difference.
Importantly, neither China nor India was willing to or required to participate in the Montreal Protocol. That left them free to do whatever they wanted, and that is exactly what they did. In February 1989, the New Scientist reported that China had a plan in place to boost their production of CFCs up to 10 times the present level. And, it wasn’t until the summer of 2007 that China actually got around to banning the production of ozone-depleting CFCs. So there has not been much in the way of reduction from China. In fact, as reported by Walter Russell Mead in The American Interest, China used the UN-sponsored Kyoto Protocol to run an emission credits scam operation:
It appears that Chinese coolant manufacturers have been producing an excess of a harmful greenhouse chemical in order to dispose of it responsibly under the UN’s Clean Development Mechanism (CDM). By using incinerators to cleanly burn off the chemical, HFC-23, these manufacturers were earning emission credits that they would in turn sell to developed world companies in order to help them hit their targets under the Kyoto Protocol.
This chicanery didn’t go unnoticed, however: the European Emissions Trading Scheme banned trade in those credits in May, and other working climate exchanges have said they’re going to follow suit. A very lucrative business for Chinese manufacturers is drying up very quickly, and they’re not taking it sitting down.
The EIA said an undercover investigation had shown that most of China’s non-CDM facilities were emitting HFC-23 already.“If all of these facilities [under the CDM] join China’s non-CDM and vent their HFC-23, they will set off a climate bomb emitting more than 2bn tonnes of CO2 equivalent emissions by 2020,” it said.
Thanks (in part) to financial incentives to destroy CFCs under the Kyoto Protocol, there are even more CFCs in existence (in China) than there were before. This might explain why, more than 15 years after the Montreal Protocol was put into effect by the UN, NASA reported in 2006 that the ozone hole over the Antarctic reached a record size:
“From September 21 to 30, [2006], the average area of the ozone hole was the largest ever observed, at 10.6 million square miles,” said Paul Newman, atmospheric scientist at NASA’s Goddard Space Flight Center. Newman was joined by other scientists from NASA and the National Oceanic and Atmospheric Administration (NOAA) in reporting that the ozone hole over the polar region of the Southern Hemisphere broke records for both area and depth in 2006. A little over a week after the ozone hole sustained its new record high for average area, satellites and balloon-based instruments recorded the lowest concentrations of ozone ever observed over Antarctica, making the ozone hole the deepest it had ever been. Source: NASA Earth Observatory

Or does it? Adding to the madness, now there is scientific uncertainty about the actual extent of the ozone problem as it relates to CFCs. More recent science has shown that the sensitivity of the Earth’s ozone layer might very well be 10 times less than was originally believed back in the 1980s when the alarm was first sounded. As reported in the prestigious science journal Nature, Markus Rex, an atmospheric scientist at the Alfred Wegener Institute for Polar and Marine Research in Potsdam, Germany, found that the breakdown rate of a crucial CFC-related molecule, dichlorine peroxide (Cl2O2), is almost an order of magnitude lower than the currently accepted rate:
“This must have far-reaching consequences,” Rex says. “If the measurements are correct we can basically no longer say we understand how ozone holes come into being.” What effect the results have on projections of the speed or extent of ozone depletion remains unclear. (Emphasis added)
One of the biggest issues with the Antarctic ozone hole is that it is not a year-round event. It peaks at its worst during the long, dark Antarctic winter. Observations show that it is highly correlated to weather patterns — more so than to actual atmospheric CFC content. The cold, the lack of sunlight to form new ozone, and the circular wind pattern in Antarctica all conspire to reduce ozone without any help from CFCs at all. Fellow of the American Meteorological Society and co-founder of the Weather Channel Joe D’Aleo says he thinks that the Antarctic ozone hole might simply be a permanent feature of the Earth that we only discovered when we went looking for the posited ozone reduction:
The data shows a lot of variability and no real trends after the Montreal protocol banned CFCs. The models had predicted a partial recovery by now. Later scientists adjusted their models and pronounced the recovery would take decades. It may be just another failed alarmist prediction.
Remember we first found the ozone hole when satellites that measure ozone were first available and processed (1985). It is very likely to have been there forever, varying year to year and decade to decade as solar cycles and volcanic events affected high latitude winter vortex strength. (Source)
With the claim in the AP story of “statistically significant” success being just a tiny improvement at higher latitudes, about 4%, while the Antarctic ozone hole continues mostly unabated, one wonders if the UN claim of success is nothing more than taking credit for simple natural variability.
The ozone hole may be a process that has been around for ages, which we only were able to notice as a result of recent technology.
This article originally appeared as a special report to PJMedia
Added: Ozone hole animation – watch the ozone hole form when Antarctic winter sets in.
Back when the ozone scam was at it’s peak, we didn’t have any means of measuring ozone directly. The only method was indirect, by measuring UV levels at the surface. At the time it was assumed that the sun was constant, therefore any changes in surface UV levels must be caused by changes in the ozone layer.
It wasn’t until years later when we put up a satellite to monitor the sun that we found out that the sun’s UV levels varied by as much as 10% over the course of a solar cycle.
BTW, did I mention that during the mid 80’s the sun was building up to a solar max.
The whole scare was based on blaming man for what the sun was doing.
Can’t forget that the patent for CFC’s was about to run out. However, the new, govt approved chemical has about 50 years left on it’s patent.
No patents run for 50 years. Seriously, you should do some elementary checking.
CFCs were long out of patent around the time of Montreal. Du Pont had a useful process patent, but the actual chemicals themselves could be made by anyone. The trademarks (such as Freon) were probably more valuable than the patents.
James Lovelock, 29 March, 2010
http://www.guardian.co.uk/environment/blog/2010/mar/29/james-lovelock
Lovelock, form the above:
the best CAGW headline i saw about this:
10 Sept: AFP: The ozone hole is recovering so well it’s adding to global warming
http://www.thejournal.ie/ozone-layer-improvement-1664716-Sep2014/
It seems to me the only people who have to worry about getting cancer from lack of ozone are those who live in live in Antarctica, and even then they would have to spend a lot of time outside, very few people do that.
Sadly you are wrong. A quick trip to New Zealand will set you straight on that. An hour outside without your shirt on in summer and you will be lobster red and peeling. My parents once warned a black African that he shouldn’t be walking around without a hat. He scoffed, thinking his skin colour would protect him. He was wrong.
I’ve lived in Europe at about the same latitude and the difference is tremendous.
That’s not to say CFCs are the cause, but there is no doubt that the lack of UV protection is a major issue, and it would not be nice if it got worse.
While the effect you describe, Mooloo, might be real, you are, sadly, wrong about the cause. The Earth itself does not orbit the sun in a neat circle, but orbits in an ellipse. The distance from the Sun varies between 146.5 million km in January to 151.5 million km in July, giving a 7% variation in solar energy due to the `inverse square law’ of distance. NZ is simply closer to the sun in its summer as opposed to Europe in its summer thus explaining the effects you’ve observed.
When can we expect peter to grace us with his commentary?
The original claim that the ClOOCl absorption cross-section was lower than thought was from Stan Sander’s lab at JPL, Pope, et al. Rex only made a comment on the paper in Nature. About two years later it was shown that the Pope measurement was wrong by two groups, one in Taiwan using an elegant laser photolysis method and by Burkholder in Boulder using a better straightforward spectroscopic technique. The probable reason for Pope et al being wrong is that they overestimated the concentration of ClOOCl in their cell.
What a poor article. So many errors.
Trying to make out that ozone depletion might not be due to us. It continues the narrative that humans cannot possibly affect good old planet Earth.
Only we can – and have. The depletion of ozone over high latitudes north and south is due to our pollutants. A variety of chemicals containing chlorine and bromine react strongly and in complex ways to destroy ozone.
Its not in the winter that the depletion occurs, but largely in the spring and summer when long hours of sunshine return to high latitudes. We have had high UV alerts here in the UK in late spring specifically linked to abnormally low ozone concentrations.
This article also fits with the “how can a trace gas do that” narrative.
How can extremely low concentrations of pollutants possibly affect the ozone layer ? How can extremely low concentrations of ozone prevent most of the incoming solar UV from reaching the ground ? How can CO2 possibly have a significant warming effect when it is only 400 ppm ?
Well they all do.
The early signs of recovery in ozone is good news and if it continues will vindicate the international agreement to deal with a serious issue that affects far more than the sparse communities living on Antarctica.
A man with a clock knows what time it is. A man with two clocks is never sure.
Did you get your data from a reliable source you could link to, or do you just know you are right?
Like that kool aid eh? I’ve looked at hundreds of ozone graphs over both poles. It is NOT what you would expect from a ‘well mixed’ gas chemistry. It is full of blobs and splotches that vary hourly / daily. It is a radiative / magnetic / fast variance thing.
The solar UV / air layer movement /magnetic / particle pattern matches much much better.
IMHO Ozone was the test run for AGW and just as bogus, using many of the same techniques with the same problems.
James Abbott
“Well they all do.”
Well that’s it then, say no more, that proves it, game over.
But still waiting for those satellites to detect the warming from all that increased CO2
Black Pearl – Yep, those are accepted facts, as far as we can be sure. Based on science research going back decades.
I’ll ask you the same question I have often put on this site – and have never had an evidenced answer to…
If CO2 does not produce warming, then tell us what would happen if (model world of course) we take say half the CO2 out of the atmosphere ? Or all of it ? Presumably no change ?
The article has sources and links to graph of data. You pop up and say “Well they all do.”. Which do you think anyone reading will find more convincing?
Your main source of info seems to be what you see on the 2 min telly weather bulletins.
If you have some point to make back up by credible sources please try again.
Dumb assertions from someone too lazy to inform themselves has zero impact. Go back to watching telly.
If we remove half the CO2 from the air, we would get significantly less food. If we remove more, our plants will start to die. http://bit.ly/1weUbNZ
I recommend you should start this removal job right away to prove your point, as it is quite a job due to the existence of Henry’s Law causing the ocean to gas out CO2 to replace whatever you manage to remove.
Compared to plant death anything else happening, be it real or imagined, will pale into insignificance.
“The early signs of recovery in ozone is good news and if it continues will vindicate the international agreement to deal with a serious issue that affects far more than the sparse communities living on Antarctica.”
Could have just said that. After all, that WAS your real point wasn’t it ?
lawrence Cornell
That and the attempt in the article to undermine what is very widely accepted science as to the cause of ozone depletion.
James Abbott, how and by how much can ozone depletion since, say, 1800 or 1900, be determined?
More appeal to authority trash.
“…what is very widely accepted science as to the cause of ozone depletion.”
Kinda rolls off the tongue so “authoritatively” like ‘ 97 percent consensus as to the cause of global warming’.
At least you admitted your primary point is political, but then, you don’t think you have, do you ?
A scientist [named] Dobson recorded large drops in the 1950’s too.
Why is there so much Ozone in the ring around the hole at 40S to 60S. These numbers are the highest for any part of the planet at any time of the year.
I wonder where all that extra Ozone in the stratosphere comes from. It couldn’t possibly be just moved away from that near-by hole I imagine.
——-
This year’s hole looks about the same as last year (and at September 9, we are not quite at the peak yet).
Ozone is the result of the protection not as much the protection. The hole is a result of the lack of sunlight.
Loss of Ozone in the upper atmosphere would just mean it would be absorbed at a lower altitude.
Ozone layer is at about 33 km, that means 33 km of oxygen it will have to pass through where ti could be absorbed the whole way.
Obviously this can only be called hearsay unless someone has access to source documents. I don’t recall details as well as might be desired but years ago, on an (anti-greens) environmental website, the claim was printed that the entire things was a scam, evidenced by an article in the 1920 edition (or some year near there) Encyclopedia Britannica which reported the ozone hole, discovered by an Antarctic expedition. No one at that time considered it any more than a curiosity.
The website author believed it was largely a product of the polar atmospheric conditions and Antarctic volcanoes. I don’t know enough about measuring such things to know if it was possible to detect such a thing with that era’s technology. Also, if it means anything, this author claimed to be a once well published environmental scientist who lost his university position by questioning some of the acid rain claims of that time.
“The cold, the lack of sunlight to form new ozone, and the [isolating] circular wind pattern in Antarctica all conspire to reduce ozone without any help from CFCs at all.”
= = = = =
&
Remove isolation of the polar vortex from the equation, and the ozone “hole,” now continuously replenished with enriched air from surrounding regions, will disappear, along with the ozone enriched accretion region surrounding it.
But remove CFCs, and the same depletion/accretion pattern will continue to emerge indefinitely, with seasonal variations in the size and shape of the polar vortex explaining and matching–exactly–any seasonal variations in the size and shape of the depletion/accretion regions:
http://apod.nasa.gov/apod/image/0210/ozone020924_toms_big.jpg
CFCs or Bipolar Vortex? — NASA, 2002
Yes, that’s the simplest explanation, i.e. the dynamical forces of the Polar Vortex displace the ozone, resulting in an Ozone “Surplus” around the Polar Vortex and associated Ozone “Hole”: NOAA – National Weather Service – Climate Prediction Center [/caption]
NOAA – National Weather Service – Climate Prediction Center [/caption] NOAA – National Weather Service – Climate Prediction Center [/caption]
NOAA – National Weather Service – Climate Prediction Center [/caption]
[caption id="" align="alignnone" width="449"]
It is primarily centrifugal force, i.e. “The walls of the polar vortex act as the boundaries for the extraordinary changes in chemical concentrations. Now the polar vortex can be considered a sealed chemical reactor bowl, containing a water vapor hole, a nitrogen oxide hole and an ozone hole, all occurring simultaneously (Labitzke and Kunze 2005)”
http://books.google.com/books?id=B93SSQrcAh4C&lpg=PA283&ots=d0-uBRjmyI&dq=%22water%20vapor%20hole%22%20polar%20vortex&pg=PA283#v=onepage&q=%22water%20vapor%20hole%22%20polar%20vortex&f=false
“The transport circulation is modified to simulate the vortex by preventing the exchange of air across the vortex boundary. The 03 distributions calculated using standard gasphase chemistry with and without this modification are shown in Figures 2a and 2b, respectively. Comparison of Figures 2a and 2b shows that modification of the circulation to eliminate transport of 03 into the vortex has the effect of increasing 03 (up to 10%) immediately outside the vortex wall and decreasing 03 (up to 20%) inside the vortex. In the following discussion the change in 03 due to the imposed 03 hole will be calculated relative to the 03 amount given in Figure 2b.”
http://onlinelibrary.wiley.com/doi/10.1029/JD094iD09p11521/abstract
By way of example:
“Structure of a Hurricane
In center: centrifugal force is very strong
Divergence and sinking air
Mostly clear skies in the eye of the hurricane”
http://www.indiana.edu/~geog109/topics/13_severe/13-Hurricanes_nf.pdf
“The hurricane’s center is a relatively calm, generally clear area of sinking air and light winds that usually do not exceed 15 mph (24 km/h) and is typically 20-40 miles (32-64 km) across. An eye will usually develop when the maximum sustained wind speeds go above 74 mph (119 km/h) and is the calmest part of the storm.
But why does an eye form? The cause of eye formation is still not fully understood. It probably has to do with the combination of “the conservation of angular momentum” and centrifugal force. The conservation of angular momentum means is objects will spin faster as they move toward the center of circulation. So air increases it speed as it heads toward the center of the tropical cyclone. One way of looking at this is watching figure skaters spin. The closer they hold their hands to the body, the faster they spin. Conversely, the farther the hands are from the body the slower they spin. In tropical cyclone, as the air moves toward the center, the speed must increase.
However, as the speed increases, an outward-directed force, called the centrifugal force, occurs because the wind’s momentum wants to carry the wind in a straight line. Since the wind is turning about the center of the tropical cyclone, there is a pull outward. The sharper the curvature, and/or the faster the rotation, the stronger is the centrifugal force.
Around 74 mph (119 km/h) the strong rotation of air around the cyclone balances inflow to the center, causing air to ascend about 10-20 miles (16-32 km) from the center forming the eyewall. This strong rotation also creates a vacuum of air at the center, causing some of the air flowing out the top of the eyewall to turn inward and sink to replace the loss of air mass near the center.
Radar image of hurricane Andrew showing eye, eyewall, and spiral bands. This sinking air suppresses cloud formation, creating a pocket of generally clear air in the center. ”
http://www.srh.noaa.gov/jetstream/tropics/tc_structure.htm
This explains why the Ozone “Hole” has it’s lowest concentrations at its center:
[caption id="" align="alignnone" width="449"]
And there are other dynamical influences that needs to be taken into account, i.e. “in the center of the Antarctic vortex. Air from very high altitudes descends vertically through the center of the vortex, moving air to lower altitudes over several months.”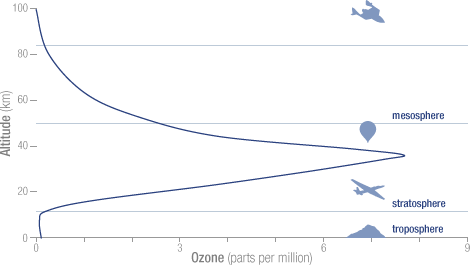 NOAA – National Aeronautics and Space Administration (NASA) Goddard Space Flight Center (GSFC) – Click the pic to view at source[/caption]
NOAA – National Aeronautics and Space Administration (NASA) Goddard Space Flight Center (GSFC) – Click the pic to view at source[/caption] Figure 2.14: Ozone Concentration vs. Height (Ajavon, et al., 2007)- Click the pic to view at source[/caption]
Figure 2.14: Ozone Concentration vs. Height (Ajavon, et al., 2007)- Click the pic to view at source[/caption] Figure 2.14: Ozone Concentration vs. Height (Ajavon, et al., 2007)- Click the pic to view at source[/caption]
Figure 2.14: Ozone Concentration vs. Height (Ajavon, et al., 2007)- Click the pic to view at source[/caption]
http://www.nasa.gov/centers/langley/news/factsheets/HALOE-Ozone.html
“In the NH vortex, air parcels which were initialized at 18 km on November 1, descended about 6 km by March 21, while air initially at 25 km descended 9 km in the same time period. This represents an average descent rate in the lower stratosphere of 1.3 to 2 km per month. Air initialized at 50 km descended 27 km between November 1 and March 21.
In the SH vortex, parcels initialized at 18 km on March 1, descended 3 km, while air at 25 km descended 5–7 km by the end of October. This is equivalent to an average descent in the lower stratosphere of 0.4 to 0.9 km per month during this 8-month period. Air initialized at 52 km descended 26–29 km between March 1 and October 31. In both the NH and the SH, computed descent rates increased markedly with height. The descent for the NH winter of 1992–1993 and the SH winter of 1992 computed with a three-dimensional trajectory model using the same radiation code was within 1 to 2 km of that calculated by the one-dimensional model, thus validating the vortex averaging procedure. The computed descent rates generally agree well with observations of long-lived tracers, thus validating the radiative transfer model.”
https://earthref.org/ERR/59278/
Air towards the top of the stratosphere and bottom of the mesosphere has lower concentrations of ozone;
[caption id="" align="alignnone" width="578"]
As such, when this “air from very high altitudes descends vertically through the center of the vortex” it displaces the air below it, decreasing the concentration of ozone within the Polar Vortex.
Additionally, “Throughout the winter, air from the upper stratosphere moves poleward and descends into the middle stratosphere. In the SH lower to middle stratosphere, strongest descent occurs near the edge of the polar vortex. The NH shows a similar pattern in late winter, but in early winter strongest descent is near the center of the vortex. Strong barriers to latitudinal mixing exist above =420 K throughout the winter. Below this, the polar night jet is weak in early winter, so air that descends below that level mixes between polar and middle latitudes. In late winter, parcels descend less, and the polar night jet moves downward, so there is less latitudinal mixing. The degree of mixing in the lower stratosphere thus depends strongly on the position and evolution of the polar night jet.”
“Diagnostics of mixing show a strong barrier to mixing at the edge of the polar vortex in the lower stratosphere, even into December in the SH, In the middle stratosphere, stratospheric warmings cause mixing to increase, and the vortex edge, as identified by a minimum in mixing, to move to higher PV values. This is consistent with previous studies showing the shrinking of the vortex and weakening of PV gradients during stratospheric warmings. As expected, considerably more mixing is seen in the NH than in the SH, In the lower stratosphere, in early winter the ,po]ar night jet usually does not extend significantly below =400 K, and parcels that descend below this level experience little barrier to mixing into mid-latitudes. As the winter progresses, the polar night jet moves downwards, and the parcels descend more slowly. Consistent with this, our simulations show considerably less mixing of polar air into mid-latitudes in late winter than in early winter in both hemispheres. In the presence of this polar night jet structure in the lower stratosphere and the sub-tropical jet in the upper troposphere, the degree of mixing of between polar and mid-latitude air in the lower stratosphere is strongly dependent on the amount of descent.”
http://trs-new.jpl.nasa.gov/dspace/bitstream/2014/33809/1/94-0540.pdf
Here is a simple view of the Ozone Layer;
[caption id="" align="alignnone" width="451"]
here is what happens to Column Ozone when the Polar Vortex descends into and through the Ozone Layer:
[caption id="" align="alignnone" width="451"]
and here is an animation of it occurring in 2013:
http://www.esrl.noaa.gov/gmd/dv/spo_oz/movies/index.html
Simple dynamical effects appear to explain the existence of Ozone “Holes”.
If the Antarctic polar vortex is to blame due to its ‘tight circulation’ then the reason for the ‘recovery’ of the ozone ‘hole’ could be that, like the Arctic polar vortex, the jetstreams in the antipodes has moved equatorward and become latitudinal with huge Rossby waves.
See: http://earth.nullschool.net/#current/wind/isobaric/250hPa/orthographic=-90.99,-89.01,486
The polar vortex around the South pole now reaches almost to the equator.
Ian W September 13, 2014 at 10:53 am PhysicalGeography.net – Click the pic to view at source[/caption]
PhysicalGeography.net – Click the pic to view at source[/caption]
If the Antarctic polar vortex is to blame due to its ‘tight circulation’ then the reason for the ‘recovery’ of the ozone ‘hole’ could be that, like the Arctic polar vortex, the jetstreams in the antipodes has moved equatorward and become latitudinal with huge Rossby waves.
The most likely explanation for any observed “recover” of the “ozone hole” is that the stratospheric polar vortex is breaking down earlier, i.e.:
“it is clear from the TOMS data that ozone transport and especially the timing of the vortex breakdown can have a large influence on the monthly mean total ozone, especially for October and November, when the vortex breakdown usually takes place. When the vortex breakdown occurs early (late) the October mean ozone is high (low). Figure 1 in Farman et al. also suggests that extreme high ozone values are associated with early breakdowns, while extreme low values are found in years with weak and late breakdowns. The vortex breakdowns of 1980-1982 were weaker and later than for 1978 and 1979. Thus, it is possible that the observed long-term ozone changes are in part the result of changes in planetary wave activity and the stratospheric circulation.
http://geotest.tamu.edu/userfiles/213/GL013i012p01193.pdf
“several studies (including Waugh and Randel 1999; Waugh et al. 1999; Karpetchko et al. 2005; Black and McDaniel 2007) have indicated a trend over the 1980s and 1990s toward a later vortex breakdown.”
http://www.columbia.edu/~lmp/paps/waugh+polvani-PlumbFestVolume-2010.pdf
See: http://earth.nullschool.net/#current/wind/isobaric/250hPa/orthographic=-90.99,-89.01,486
The polar vortex around the South pole now reaches almost to the equator.
No, you are at the wrong altitude. The stratospheric”polar vortex extends from the tropopause (the dividing line between the stratosphere and troposphere) through the stratosphere and into the mesosphere (above 50 km). Low values of ozone and cold temperatures are associated with the air inside the vortex.” NASA
[caption id="" align="alignnone" width="578"]
250hPa is approximately 10,000 feet and thus the Stratospheric Polar Vortex rarely reaches that far down and is if so it is only its tail. To see the Stratospheric Polar Vortex, you need to look here;
http://earth.nullschool.net/#current/wind/isobaric/70hPa/orthographic=-90.99,-89.01,486
and here:
http://earth.nullschool.net/#current/wind/isobaric/10hPa/orthographic=-90.99,-89.01,486
or alternately, you can review the new WUWT Southern Polar Vortex page I am working on:
http://wattsupwiththat.com/reference-pages/climatic-phenomena-pages/polar-vortex/southern-polar-vortex-page/
Ian, further to your statement that;
“The polar vortex around the South pole now reaches almost to the equator.”
“The ozone hole is in the center of a spiraling mass of air over the Antarctic that is called the polar vortex. The vortex is not stationary and sometimes moves as far north as the southern half of South America, taking the ozone hole with it.”
http://www.nasa.gov/centers/langley/news/factsheets/HALOE-Ozone.html
Just The Facts September 13, 2014 at 10:54 pm
Thanks for the link — a little later it also states:
“Effects on lower latitudes
HALOE also has returned evidence that the effects of the Antarctic stratospheric vortex extend well beyond the South Pole during the Antarctic spring, when the vortex is most vigorous. The HALOE data show that Antarctic-type air, identified by low levels of ozone and other trace chemicals, reaches as far north as 40 degrees south latitude, covering part of the South American continent (Fig. 1b). The effects of the vortex are also extended by air currents into the tropics, to 25 degrees south latitude.
During the 1992 Antarctic Spring, air with low levels of ozone, which also contained chemicals that help ozone destruction, spread northward into latitudes well beyond the Antarctic continent (Figs. 1a and 1b).”
Are the lower and upper jets related?
Are the lower and upper jets related?
It is all related, i.e. the atmosphere is rotating due to the Coriolis Effect, i.e.:
“The Coriolis effect is caused by the rotation of the Earth and the inertia of the mass experiencing the effect. Because the Earth completes only one rotation per day, the Coriolis force is quite small, and its effects generally become noticeable only for motions occurring over large distances and long periods of time, such as large-scale movement of air in the atmosphere or water in the ocean. Such motions are constrained by the surface of the earth, so only the horizontal component of the Coriolis force is generally important. This force causes moving objects on the surface of the Earth to be deflected in a clockwise sense (with respect to the direction of travel) in the Northern Hemisphere and in a counter-clockwise sense in the Southern Hemisphere. Rather than flowing directly from areas of high pressure to low pressure, as they would in a non-rotating system, winds and currents tend to flow to the right of this direction north of the equator and to the left of this direction south of it. This effect is responsible for the rotation of large cyclones (see Coriolis effects in meteorology).”
http://en.wikipedia.org/wiki/Coriolis_effect
When the Stratospheric Polar Vortex becomes split or displaced, it can have major impacts on circulations and temperatures at lower altitudes and far from the Arctic, i.e.:
http://wattsupwiththat.com/2014/02/01/a-displaced-polar-vortex-and-its-causes/
http://wattsupwiththat.com/2014/02/08/when-does-a-displaced-polar-vortex-becomes-a-split-vortex/
“The loss of the ozone layer in the upper atmosphere will lead to normally reflected high-energy ultraviolet light reaching the Earth’s surface, causing more sunburns and skin cancer, disruption of ecosystems such as marine plankton and algae, and other photosynthetic biomass, with a large ripple effect.”
Sunburns and skin cancer are caused by sunbathing rather than ozone hole. Only a few scientists live in Antarctica and they don’t sunbathe there. UV-B hardly penetrates beyond one meter deep in the Southern Ocean around Antarctica. The tropical seas get the most UV-B penetrating up to 20 meters deep. Plankton and algae are blooming in the tropical seas. Apparently UV-B is not killing them.
The ozone layer is NOT recovering from our CFCs. The claim that they hurt the ozone layer was a Dupont Chemical-created scam designed to get an out of patent CFC banned so that they could step up with a patented more expensive replacement. Twenty years later, the scientist that claimed that the CFC was bad admitted that he had fabricated the results.
Now we know that ozone is broken down by nitrogen gas and solar radiation. Now, that the sun is going quiet, the ozone layer may be able to grow. Do not forget that we have never seen the planet without an ozone hole.
I had not heard of this scam until today, but the forged data claim corresponds to what Lovelock says. Do you have reference for this thing about 80% forged data?
I second the motion – provide the evidence!
There is a good discussion of the events and Du Pont’s strategy here:
https://eng.ucmerced.edu/people/awesterling/copy_of_ESS141.2010/Assignments/DuPont
I cannot vouch for any accuracy but it is detailed.
From a Guardian interview with James Lovelock in 2010
http://www.theguardian.com/environment/blog/2010/mar/29/james-lovelock
The increasing Ozone in the NH can’t stand the heat of AGW and so takes a winter sojourn to the SH. Sorted.
I’m still not sure why CO2 hasn’t been able to take that same sojourn south of the equator, seeing it’s a well mixed gas and all.
Thompson and Solomon 2008: “Understanding Recent Stratospheric Climate Change”
http://journals.ametsoc.org/doi/pdf/10.1175/2008JCLI2482.1
This paper looks at changes in lower stratospheric temperatures and ozone concentration. Figure 1c shows the drop in ozone was concurrent with the two major eruptions in that period and slowly recovered from a low in 1993 after Mt Pinatubo until about 2004. There was a short recovery in between the two events
http://climategrog.files.wordpress.com/2014/09/ozone_msu4_solomon.png
Much of the “alarming” destruction of the ozone layer seems to be a result of natural events as does the recovery, though it is not possible from that data to rule out a possible additional anthropogenic effect.
I have already shown how this cooling of the stratosphere is reflected by warming of the troposphere.
http://climategrog.wordpress.com/?attachment_id=902
In short both the “alarming” global warming and the “alarming” loss of ozone seen in the period 1980-2000 were mainly cause by volcanoes.
The subsequent lack of volcanoes and slow return towards earlier conditions explains the famous “pause” in global warming and the “recovery” of the ozone layer.
The UN is trying to puff up claims that it’s last treaty actually did something in an attempt to convince us to engage in another one, renounce democracy and let them run the world.
It is a coincidence that they are shouting about this in the run up to the meeting of ( some ) heads of state at the end of the month?
Stratospheric temps as above ( here inverted ) overlayed on changes in southern hemisphere SST.
http://climategrog.files.wordpress.com/2014/07/tls_icoads_70s-20s.png
Volcanoes are a large cause of the late 20th. c warming. Contrary to mainstream climatology that only acknowledges the initial, short-lived, cooling blip.
Note the rise SST takes several years because of the thermal inertia of the ocean’s surface layers. The stratosphere curve ( mauve line ) gives a fairly instantaneous indication of the changes in atmospheric composition.
NASA not a drop of 5%-8% in ozone due to Mt Pinatubo but try to make that the fault of CFCs anyway. Despite admitting having NO data to support the idea they say that the earlier eruption in 1963 “probably” did not cause ozone reduction.
They never let data ( or lack of it ) get in the way of science.
http://earthobservatory.nasa.gov/Features/Volcano/
Methane + chlorine = chlorinated hydrocarbons?
Seawater + sunshine generate free chlorine. We titrated seawater back in high school in1962 by means of classic wet chemistry and gravimetry, using silver nitrate (soluble) turning into silver chloride (insoluble).
I have often wondered why the presence of free chlorine in the oceans is not mentioned when discussing dioxines in seafood.
“Within the span of nine months, NASA issued statements claiming of atmospheric ozone that “signs of recovery are not yet present,” there is “large variability,” it is “stabilizing,” and now, that the ozone problem is “recovering”…So which is it? “
No, NASA said that “signs of recovery [of the Antarctic Ozone hole] are not yet present,” but the ozone hole stabilized, as measured by size of hole. This article says that the ozone layer “in the key mid-northern latitudes” is recovering, as measured by ozone concentration. They are not at all the same.
The list of nations ratifying the Montreal Protocol is here. China ratified in June 1991, India in June 1992.
Check out “Another Day Another Dollar – CFC’s And The UN”
http://scienceandpublicpolicy.org/originals/another_day_another_dollar.html
“The CFC story is a parallel for the CO2 story and was another EPA “cause celebre”. Claims of dramatic changes to the atmosphere were made; time was running out, the world was in danger and it could only be saved by “Global Action”.”
For me, the interesting thing about the ozone hole is that it’s never actually done anyone or anything any harm. Not only is it over the most barren and unpopulated part of the planet, the Sun’s rays come in at such a steep angle down there that UV is scarcely a problem.
I remember one time when the usual suspects were bigging up this issue, a child in Tierra del Fuego came down with a bad case of sunburn (one child, mind you), and it made international news! There were always serious problems with justifying the cost of this boondoggle.
What puzzles me is; what would these turkeys do if there ever was a real, preventable global problem with an obvious cause? (Like, endemic war, povery, starvation, disease… I’ll get me coat.)