Over at The Conversation Andrew Glikson asks Fact check: has global warming paused? citing an old Skeptical Science favorite graph, and that’s the problem; it’s old data. He writes:
As some 90% of the global heat rise is trapped in the oceans (since 1950, more than 20×1022 joules), the ocean heat level reflects global warming more accurately than land and atmosphere warming. The heat content of the ocean has risen since about 2000 by about 4×1022 joules.
…
To summarise, claims that warming has paused over the last 16 years (1997-2012) take no account of ocean heating.
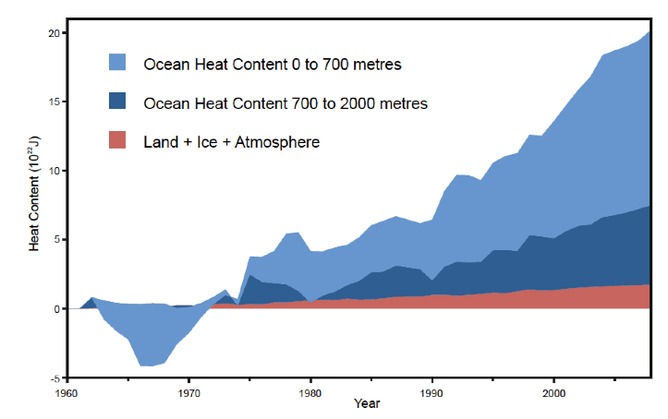
Hmmm, if “…ocean heat level reflects global warming more accurately than land and atmosphere warming…” I wonder what he and the SkS team will have to say about this graph from NOAA Pacific Marine Environment Laboratory (PMEL) using more up to date data from the ARGO buoy system?
Sure looks like a pause to me, especially after steep rises in OHC from 1997-2003. Note the highlighted period in yellow:

From PMEL at http://oceans.pmel.noaa.gov/
The plot shows the 18-year trend in 0-700 m Ocean Heat Content Anomaly (OHCA) estimated from in situ data according to Lyman et al. 2010. The error bars include uncertainties from baseline climatology, mapping method, sampling, and XBT bias correction.
Historical data are from XBTs, CTDs, moorings, and other sources. Additional displays of the upper OHCA are available in the Plots section.
As Dr. Sheldon Cooper would say: “Bazinga!“
h/t to Dr. Roger Pielke Sr. for the PMEL graph.
UPDATE: See the above graph converted to temperature anomaly in this post.
MiCro says: “Not when you compare cooling to how much the temp goes up the prior day.
What the paper behind the paywall is detecting is land use changes, ie UHIE.”
Now you’re moving the goalposts — earlier you said there was no reduction in how much it cools at night.
If you actually read the paper, you’d see that they wrote on the very first page:
“An important aspect of any surface-based land analysis
of temperature change relates to the potential impact of
growing urban heat islands. In this analysis the data used
for the USA (excluding the extremes) have been adjusted
for urban heat island biases using the procedures described
by Karl et al. [1988]. The station network we use in the
USSR is a rural network (no station in a city with population
of 10,000 or more). The PRC network consists of several
stations in and near large cities, but many of the stations
were also used by Jones et al. [1986] as described in Jones
et al. [ 1985]. In previous work [Jones et al., 1990; Wang et
al., 1990], we have compared the data in the PRC used by
Jones et al. [1986] in the eastern half of the PRC (the region
which contains the most urbanized stations) to various
networks comprised of only rural or urban stations. These
analyses indicate that urban heat island biases derived from
long-term stations in this portion of the globe are relatively
small (-0.1øC) over the time period we address in the PRC.
“It should also be noted that detailed station histories
have been compiled for each of the stations we use. There
is no evidence to suggest that either observational practices
or instruments may have introduced systematic
inhomogeneities in the data we analyze.”
http://onlinelibrary.wiley.com/doi/10.1029/91GL02900/abstract
@Mark Benson,
” Now you’re moving the goalposts — earlier you said there was no reduction in how much it cools at night.”
No I’m not moving the goal posts, and you’d know that if you went past the first paragraph, and looked at the data and procedures I used. But you would have to make an intelligent reply then that you can’t explain but cut and pasting of others.
There is no loss of night time cooling, not since 1950.
MiCro says: “Did you go look at my work yet?”
Yes — I already told you, it makes ridiculous assumptions about relationships between temperatures and energy balances.
@Mark Benson
Actually like I said, probably shouldn’t have used energy. But let’s talk about the energy.
The other night the sky was clear, with no winds. @9:00pm it was ~25F, and the temp dropped ~.9F/hour till ~7:00am to 16F, when the Sun came up.
For the temperature to drop, every square meter had to radiate ~120-125 watts, in those 10 hours 4.8 MJoules/m2 were lost through all that Co2. That’s not a ridiculous assumption about energy and temp, that’s science.
Temperature alone doesn’t tell you how much energy is in the system, but it can tell you how much radiates away.
D.B. Stealey says: “What they ‘found’ was another way to angle for some of that globaloney grant money.”
So since you can’t argue with the result, you dismiss them for other reasons.
There will always be some reason you’ll invent.
I think you’re dismissing results you find inconvenient for monetary reasons — you’re trying to protect your pocket book, afraid you will be made to pay for your carbon pollution.
I am astonished that anyone would be so credulous as to believe that a tiny 0.8º change in temperature, over a century and a half, has any measurable effect on plant life. Global temperatures are always naturally changing. The plants cope.
There is copious evidence in shifts already among plants of latitude, altitudes, and flowering and blossoming times. There is also copious evidence that the PETM, similar to the warming to come,if we don’t stop burning fossil fuels, led to extinctions in a significant fraction of plant and animal species.
Phobos says:
March 6, 2013 at 9:39 pm
“It turns out that there have been a couple of studies of deep ocean heat content changes. They, too, find warming:”
And, you think this is because of warming of the surface over the last 30-odd years?
Maybe you and James can form a tag team of comedy here. This should be fun.
Phobos says:
March 6, 2013 at 9:40 pm
You didn’t answer my question. How much longer does this have to not occur before you admit you are wrong?
Phobos says:
“you’re trying to protect your pocket book, afraid you will be made to pay for your carbon pollution.”
‘Carbon’ is not pollution any more than oxygen is pollution, and only a clueless dimwit would believe otherwise.
Bart says:
http://www.woodfortrees.org/plot/esrl-co2/derivative/mean:24/plot/gistemp/from:1959/scale:0.2/offset:0.075
Again, you have only obtained this plot by smoothing the heck out of the CO2 data, cherry picking the smoothing period.
Try this instead:
http://www.woodfortrees.org/plot/esrl-co2/derivative/mean:36/plot/gistemp/from:1959/scale:0.2/offset:0.075
Not nearly as nice, is it?
You still haven’t told us where the 11 GtC/yr humans are emitting is going, given that the atmosphere, oceans, and land are all gaining carbon.
D.B. Stealey says: “Carbon’ is not pollution any more than oxygen is pollution.”
Of course it is. The definition of a pollutant is a substance that, at certain levels in certain places, is harmful and undesirable.
By the way, the Supreme Court has ruled that CO2 is a pollutant. Are they all dimwits too?
Bart says: “You didn’t answer my question. How much longer does this have to not occur before you admit you are wrong?”
Wrong about what — that more than one variable determines surface temperature? That’s not wrong at all.
It *is* warming. The 15-yr trends for the surface are (with 2 sigma uncertainties)
GISS: 0.07 +/- 0.04 C/decade (stat sign = 99.9%)
HadCRUT4: 0.04 +/- 0.04 C/decade (stat sign = 95%)
Both are statistically positive at the 95% level or better. And this is with the anthropogenic influence of aerosols providing some cooling.
Phobos says:
D.B. Stealey says: “Carbon’ is not pollution any more than oxygen is pollution.”
Phobos asserts: “Of course it is. The definition of a pollutant is a substance that, at certain levels in certain places, is harmful and undesirable.”
What a clueless dope. By his own definition, water = pollution.
We’re done here. Arguing with dopes like Phobos, who is totally ignorant of basic science, is a wasted time sink. Where do the alarmist crowd get people like Phobos, anyway?
Bernard J. says:
March 6, 2013 at 6:20 pm
It’s both sad and frustrating to see that people still do not understand what “acidification” actually means.
———————————————————————————————————–
Sorry, Bernard, seeing as we’re all happy doing science here 9allegedly) I assumed that my “at a standard 25 deg C” was enough to acknowledge the effect of temperature on neutrality. It was, after all, a blog comment not a dissertation.
If you followed the thread of the argument, you’d also see that I wasn’t questioning the point that Phobos was obviously trying to make (the possible effect of changes in pH on some organsism). What I was pointing out was the inaccuracy and impropriety of using emotively hysterical terms like “increasing acidity” (increasing in caps mind you!) when claiming to be discussing science.
numerobis says:
March 6, 2013 at 8:25 pm
Joe: I clearly need to retake high school chemistry; you’re right I have forgotten some details (the constants I remembered must have been to convert to molarity from some other measure of concentration).
Still, you and Anthony seem to be arguing there’s a phase change at 10^-7 mol. This I certainly don’t remember — quite the opposite. There’s still protons (or H3O+) floating around in lye, which is why we can measure its pH. Similarly, the syndrome when your blood pH gets low is known as “acidosis”, not “slightlylessbasicosis”.
—————————————————————————————————————–
No worries, numerbosis, it’s not like it’s something many people worry about from day to day after they leave school 😉
I’m not arguing for a phase change as such but acids and bases do have different reactive properties and that change in properties happens either side of neutrality.
Acidosis in the case of blood is perfectly reasonable seeing as blood is acid to begin with. It’s the intentionally emotive language I have problems with. “Acidification” is borderline because it’s meaning is generally understood and accepted the scientific community now, but stretching that even further to “more acidic”, with the implication that it’s turning from vinegar to battery acid, is blatant scaresmanship and deserves challenging.
Phobos says:
March 6, 2013 at 10:42 pm
Yikes! You know nothing about filtering theory, do you? Oh, my, but that is comedic.
“You still haven’t told us where the 11 GtC/yr humans are emitting is going, given that the atmosphere, oceans, and land are all gaining carbon.”
Of course it is going into the atmosphere, oceans, and land, where it is being sequestered permanently, or at least for a very long time, by biological and mineral processes, and otherwise transported out of the surface system. But, it is only a small part of the flows, and the sinks do not discriminate on the basis of whether it is “natural” or “anthropogenic”.
You really cannot get past it with word games. If dCO2/dt = k*(T – To), and the data say it does, then you are wrong about the source of the rise in CO2.
Phobos says:
March 6, 2013 at 10:53 pm
“Both are statistically positive at the 95% level or better.”
Again with the bogus statistics bluster. We’ve been over, and over this.
“And this is with the anthropogenic influence of aerosols providing some cooling.”
Pure handwaving rationalization of a failed hypothesis. As I have shown, there is no divergence from pre-existing patterns in the temperature record.
The ~60 year cycle will continue its downward progression for perhaps 20-30 more years. So, if you think you are going to be vindicated in anything like the near term, you are going to be sorely disappointed.
Joe: “Acidosis in the case of blood is perfectly reasonable seeing as blood is acid to begin with.”
No, it’s not. Blood pH is 7.35-7.45. Any fall in pH is correctly described as acidosis, just as any decrease in ocean pH is acidification. It’s not emotive. It’s standard scientific terminology. /Shrug
If you followed the various threads linked in my comments above, and if you paid attention to other comments from people who actually understand chemistry, you’d understand that it’s not impropriety or hysterical emotion to refer to “increasing acidity”. In the Lowry-Brønsted context it simply refers to an increase in concentration of hydronium (H3+ ions.
An acid is simply a solution where the concentration of H3O+ is greater than the concentration of OH-. Conversely, a base is just a solution where the concentration of H3O+ is less than the concentration of OH-.
I will repeat – acidification is the process of increasing the concentration of H3O+), no matter the solution’s starting concentration with respect to ‘neutrality’. As acidity increases basicity decreases and conversely, as basicity increases acidity decreases. Acidification/acidity and bascification/basicity are qualitative descriptors: they are not quantitative descriptors which only differentially kick in with respect to a specific pH value.
If you are still struggling with the concept, consider acidity-basicity expressed as the ratio of hydronium to hydroxide ions – that is H3O+:OH-. When the ratio increases, acidification is occurring, when the ratio decreases, bascification is occurring. This is true regardless of whether the starting ratio is 0.5, 1.0, or 1.5.
Get it yet?
If not, consider this. Using your logic a man who weights 200 kilograms and loses 50 is not becoming lighter, he is merely becoming less heavy, because 150 kilograms is still heavier than most.
In truth though he is becoming lighter, whether or not he was light to begin and end with.
Phobos says:
February 25, 2013 at 1:30 pm
Phobos says:
February 25, 2013 at 4:04 pm
Phobos says:
March 6, 2013 at 9:50 pm
So, who’s making ridiculous assumptions about the relationship between temperature and energy?
It just goes to show Mr Scientist can read some papers, yet not actually understand a thing.
Mark Bofill writes: “lack of confidence in this 3.0 + / – 50% means we are speculating, speculation != science.”
It’s not speculation, it’s science, and science includes uncertainties. Observations of paleoclimates too put climate sensitivity at this level.
Besides, climate sensitivity is a lousy indicator of future changes, not only because no one knows the future path of GHG emissions, but mostly because it doesn’t completely include carbon sensitivity — changes in natural carbon sinks, and the effects of feedbacks between climate change and carbon uptake. A much better measure is the carbon-climate feedback function (CCR), which finds 1.5 C of greenhouse gas induced warming for every trillion tons of carbon emitted. (Matthews et al, Nature 11 June 2009). The 5 and 95% percentile limits are 1.0 and 2.1 C/TtC.
This result is found from both models, and from observations (which do include changes in clouds) over the last century, as Matthews et al show.
Given that we’re now emitting over 11 GtC/yr, we can expect between 0.11-0.23 C/decade of GHG induced warming — more if emissions increase, as they almost certainly will.
Both data and theory show that CCR definitely isn’t zero. Hoping it is — i.e. that uncertainties will go for you and not break even (let along go against you) — is gambling with the future well-being of civilization.
——————
‘Look you stupid bastard, you’ve got no arms left!’
Whoa there. Wasn’t it just yesterday that you were telling me that we’d got certainty that a doubling of CO2 leads to a new equilibrium with temperatures increases to +1.5C to +4.5C with +3.0C as a most likely value? Assuming a pre-industrial atmospheric CO2 level of 270 ppmv, we’re on track to double that by around 2083. (70 years * 2 ppmv/yr = 140 ppmv, + our current 400 = 540 ppmv). But today you’re telling me that we’ve got certainty that burning at our current rate we’re going to see .011 TtC * 70 years * 1.5 C = +1.16C by 2083, with a range of 0.77C to 1.62C This is substantially different than the figures you were certain of only just yesterday, and not consistent by any stretch with the ‘most probable value’ of 3.0C you offered. You DO understand what the word ‘certainty’ means, right? What are you going to tell me you’re certain of tomorrow? Next week? I hate to keep harping on this but you never seem to take the point in; you’ve got a serious methodology problem here. Can you imagine what your reaction would be if some shmuck made drastic decisions that substantially impacted your life based on ‘certainty’ that changed on a day to day basis?
And we’re not even examining the question about what impacts (if any) could be expected for a mere 1.1C increase.
Phobos, you should reevaluate your view on engineering. I don’t know, maybe you need to rethink the meaning of personal responsibility. You are being suckered by people telling you what you’ve decided you want to hear and are apparently making no effort to verify the claims. You are not a ‘babe in the woods’ and I don’t care how much you’re enjoying the ride, what you are doing by promoting this crap is reckless and irresponsible. Heck, you aren’t even sanity checking your own position, when what you’re telling me we’re certain of changes from day to day.
Now go forth and sin no more, you darn fool.
Phobos says:
March 6, 2013 at 8:01 pm
MiCro says: “There are lots of historical records that that have much of the Arctic open in the 30′s due to melting.”
Historical Arctic Maps
Bernard J. says:
March 7, 2013 at 5:52 am
“An acid is simply a solution where the concentration of H3O+ is greater than the concentration of OH-.”
I’m not a chemistry buff, but I cannot see how these things can co-exist in a well mixed solution. I would think they would necessarily react to reduce one another, until only one species survived or the solution became neutral.
Beyond any of this, however, it is blowing smoke to assert that “acidification of the oceans” is not an emotive phrase designed to elicit fear in the target audience.
MiCro,
Phobos never read your work, or he would have posted a detailed critique.
And thanks for your reminder of the historical Arctic ice maps. They prove beyond any doubt that the current situation is not unprecedented.
“I’m not a chemistry buff, but I cannot see how these things can co-exist in a well mixed solution.”
Looks like this thread has expired, but it’s been living on borrowed time anyway and I doubt many will mourn. In any case, to expand on this statement, I can imagine that H3O+ and OH- molecules are continually being created and destroyed in a virtual sense, and the balance is disturbed when a measurement is taken. But, the bottom line is that alkalines are proton receptors, and acids are proton donors, and it appears to me that there is a clear and fundamental line of demarcation at which a solution switches from one to the other.
Therein lies the problem.
Then it’s time to learn about Chemical equilibrium.
Or, in the context of this discussion, the dissociation of water.
.
No.
Again, read the above links. And more. And then some more after that.
Bernard J. says:
March 8, 2013 at 6:25 am
I think you are quibbling. Perhaps the pH measurement does not provide a definite line of demarcation, but that is a matter of convention. The balance of concentration still does provide a clear line. And, ocean waters are nowhere near that line.
As I stated before: ‘Beyond any of this, however, it is blowing smoke to assert that “acidification of the oceans” is not an emotive phrase designed to elicit fear in the target audience.’
If you disagree, then tell my why you prefer the term “acidification” over “reduction of alkalinity”. Is it mere brevity, or are you, in fact, inclined to play on lay-persons fears of anything with the word “acid” in it?
Let me put this in simple terms, as nothing else seems to be working:
1) Hydronium ions (H3+ are the Lowry-Brønsted ‘acid’ moiety in the context of ocean pH.
2) H3+ ions are present even in basic sea water.
3) The concentration of H3+ ions in sea water is increasing. This increase is occurring as a direct consequence of the increase in atmospheric CO2 resulting from human emissions, and which dissolves into sea water and forms carbonic acid.
4) This increase in the concentration of acid H3+ ions in sea water, a process referred to by chemists, physicists and biologists as acidification (see, pH and neutrality don’t even need to be mentioned…) has a direct impact on carbonate solubility in sea water, which in turn directly impacts on the ability of calcifers (organisms that depost calcium shells) to successfully form their calcium cabonate support/defence structures.
5) If humans persist in increasing in the concentration of acid H3+ ions in sea water for more than a couple more decades, there will be profound ecological consequences. Humans will not escape the impacts of these consequences.
Bernard J. says:
March 9, 2013 at 1:00 am
“2) H3+ ions are present even in basic sea water.”
So what? It’s not going to make it eat through your hand, because there are plenty of OH- around to keep them busy.
“4) …a process referred to by chemists, physicists and biologists as acidification…”
Or as neutralization. Why not use that term?
“…which in turn directly impacts on the ability of calcifers…”
But, by how much? Agenda driven studies notwithstanding, what is the real effect of a pH drop from 8.25 to 8.14 over more than 200 years? This is the problem with agenda driven science – after too many false alarms, nobody believes you. Ever read the fable of The Boy Who Cried “Wolf!”? A responsible scientist must be very cautious about issuing categorical proclamations without conclusive evidence to back it up.
“5) If humans persist in increasing in the concentration of acid H3+ ions in sea water…”
Are we increasing it? Possibly, but I have my doubts, as we aren’t increasing it in the atmosphere. I know this to be true. I have laid out the evidence above. And, you are straying into my bailiwick if and when you assert otherwise. Yes, I am more competent in the science of feedback systems than climate scientists in general, and no, I am not a crackpot, though crackpots are a dime a dozen on both sides of this tendentious issue. The evidence is quite clear, and feedback systems do not behave according to common intuition. And, that is why these simplistic scenarios do not work out as most, who are not familiar with the dynamics of feedback systems, expect.
“Neutralization” does not describe the process as accurately as acidification. You are scrambling for a euphemistic fig leaf to avoid the fact that the concentration of acid H3+ in sea water is increasing, as a direct consequence of human carbon dioxide emissions.
Lowry-Brønsted neutrality is simply a descriptor of concentrations of hydronium and of hydroxide where each chemical species occurs in solution at the same concentration, that is, where the ratio H3+:OH- is 1. The concentration (and thus the solution’s pH) of these species at neutrality is highly variable, contingent on many physiochemical parameters, and for this reason alone “neutralization” is an ambiguous term.
Further, the significance of an increase in the concentration of H3+ is directly reflected in aragonite saturation, which is critical for calcifer carbonate deposition.
Neutrality is an inconstant point on the pH scale. In a complex buffer system with multiple chemcial species neutrality is even more tenuous a concept, because of the manner in which buffers maintain equilibria. However, when the concentration of an acid moiety increases, semantics won’t change the fact. This increase is acidification, and no amount of playing with words and with arbitrary definitions changes the fact of the increase in acid concentation.
You can deny the phenomenon, but it doesn’t change the chemistry. The laws of nature are stubborn like that.
1) Humans are increasing the amount of carbon dioxide in the atmosphere. This is a trivially obvious fact.
2) A significant portion of the CO2 emitted by humans dissolves in the oceans, where it reacts with water to form carbonic acid. These too are trivially obvious facts.
3) This formation of carbonic acid includes the formation of hydronium (H3+) ions. This is a painfully, trivially obvious fact.
4) By deductive logic your prevarication about admitting that humans are the cause of the increase in hydroniumin sea water is shown to be unfounded. By your own admission you are not up on chemistry, so it is unsurprising that you are having difficulty understanding that humans are causing this increase in sea water hydronium concentration.
The discussion is not about the increase of H3+ in the atmosphere. This is a classic straw man argument – a logical fallacy.
As noted above, humans are increasing the amount of CO2 in the atmosphere, which increases the amount of CO2 in the oceans, which increases the amount of carbonic acid in the oceans, which represents an increase in the amount of H3+ in the oceans. Your diversion of the amount of H3+ in the atmosphere is completely irrelevant, totally spurious, and avoids the painful (for you, apparently) fact that humans are increasing the concentration of acid moiety in the oceans.