Guest post by David Middleton
Introduction
My never-ending search for actual observational data that support the hypothesis of catastrophic anthropogenic ocean acidification (Chicken Little of the Sea) has taken me to offshore Hawaii and Station ALOHA.

The alarmists claim that anthropogenic CO2 emissions have lowered the average pH of the world’s oceans from 8.2 to 8.1 over the last 250 years; and that future emissions will lower the pH by 0.3 to 0.4 over the remainder of this century (Dore et al., 2009). What is the basis of this hypothesis? Systematic measurements of oceanic pH don’t go back much before 1990. Actual pH measurements are few and far between. The oceans have probably absorbed at least half of the anthropogenic carbon emissions of the last couple of centuries. The assertion of CO2-driven ocean acidification appears to he almost entirely based on the rising atmospheric CO2 level. Is this the only basis for the dire warnings of catastrophic ocean acidification? All other things being equal, the addition of CO2 should lower the pH of seawater… However, all other things are seldom equal.
Station ALOHA and the Hawaii Ocean Time-series (HOT)
The Station ALOHA data from the Hawaii Ocean Time-series are very extensive and easily accessible. These data were used by Dore et al., 2009 to demonstrate a ~20-yr trend of apparently CO2-related acidification offshore Hawaii…

At first glance, this appears to be a slam-dunk. The seawater at Station ALOHA have been acidifying in concert with rising atmospheric CO2 for ~20 years. But, looks can be deceiving. One of the tricky things about oceanic pH is that it is very dependent on water temperature, pressure and salinity. Most studies derive pH from Dissolved Inorganic Carbon (DIC or ΣCO2) and Total Alkalinity (TA). DIC is the sum of free CO2 ([CO2(aq)] & [H2CO3]), carbonate ([CO3–]) and bicarbonate ([HCO3-]). TA is the sum of carbonate (2x[CO3–]), bicarbonate ([HCO3-]), tetrahydroxyborate ([B(OH)4-]), hydroxide ([OH-]), hydrogen ([H-]) and other minor compounds. DIC and TA are “conservative quantities” – They are unaffected by pressure and temperature. The ratio of TA:DIC is a very robust measure os the alkalinity/acidity of seawater.
Most of the pH values in Dore et al., 2009 are not measured; they are derived from DIC and TA. While the calculated pH trend appears to show a continuous acidification trend, a plot of TA vs. DIC tells a slightly different story. The alkalinity of the seawater at ALOHA Station actually rose from ~2000 to ~2005, despite steadily rising atmospheric CO2 levels…
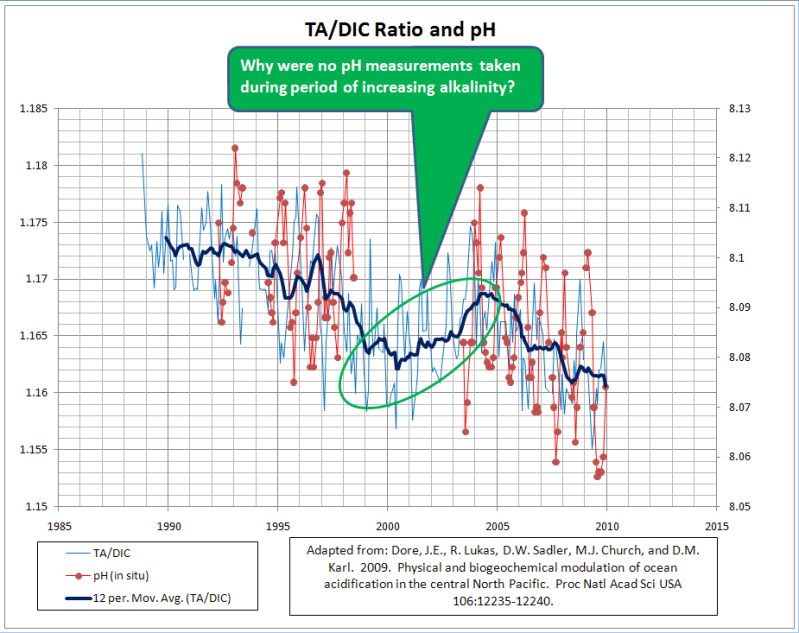
Why did the seawater become more alkaline from 2000-2005?
The most likely answer is that the seawater concentration of [CO3–] (carbonate) increased while the concentration of [CO2] decreased during that time interval. This happened despite the fact that atmospheric CO2 levels continued their inexorable rise over that same time period. The ocean at Station ALOHA was actually a net source of atmospheric CO2 for about 5 years.

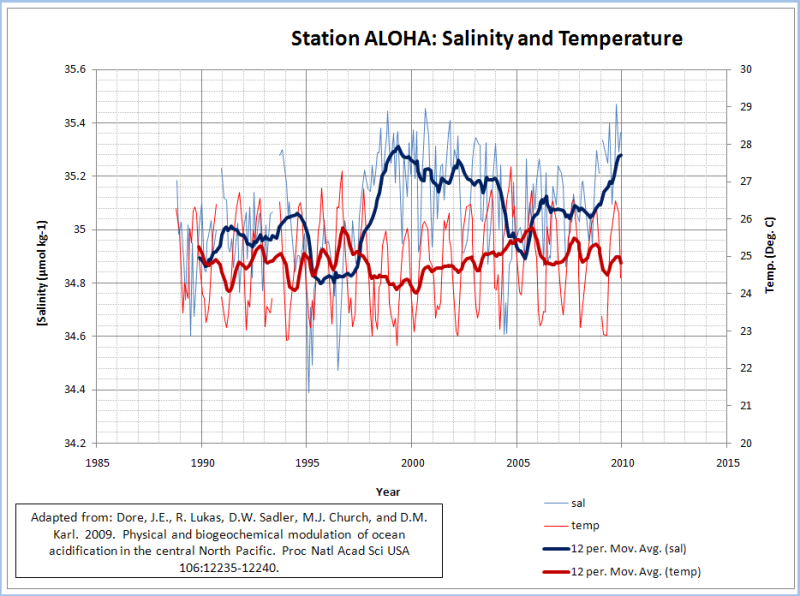
The reason for the carbonate concentration increase and [CO2] decrease from 2000-2005 is probably related to the slight rise in water temperature over that period and the elevated salinity. The change in water temperature and salinity was also the most likely reason that the 5-year trend of increasing alkalinity was not reflected in the calculated pH trend.
So, the data do support the notion that rising atmospheric CO2 levels can contribute to a lowering of oceanic pH; but that other processes can periodically raise the pH despite the rise in atmospheric CO2.
How Dangerous is Chicken Little of the Sea?
Ocean acidification can only occur if Dissolved Inorganic Carbon (DIC) is rising faster than Total Alkalinity (TA). This nomogram demonstrates the relationship of TA & DIC to pH…
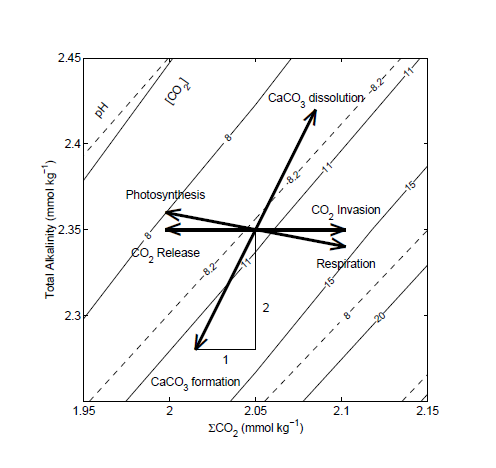
According to Dore et al., 2009, “Over the past 250 years, the mean pH of the surface global ocean has decreased from ≈8.2 to 8.1… This acidification of the sea is driven by the rapidly increasing atmospheric CO2 concentration, which results from fossil fuel combustion, deforestation, and other human activities. Models predict that surface ocean pH may decline by an additional 0.3–0.4 during the 21st century”… A total pH decline of 0.4 to 0.5 (8.2 to 7.7 or 7.6).
I used a linear regression to estimate TA and DIC at ~275 and ~550 ppmv…
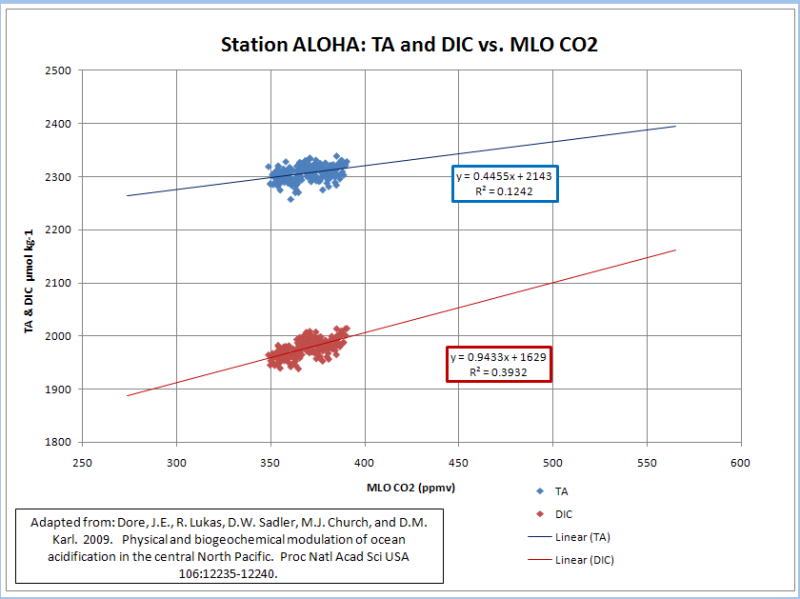
If I plot their in situ TA vs in situ DIC and extrapolated it as above (red curve), I get a very strong correlation (R^2=0.72); but I don’t get anything close to a 0.5 to 0.6 pH decline from a doubling of pre-industrial CO2 levels. I get a total decline of 0.16 (8.30 to 8.14) due to a doubling of pre-industrial atmospheric CO2 levels.
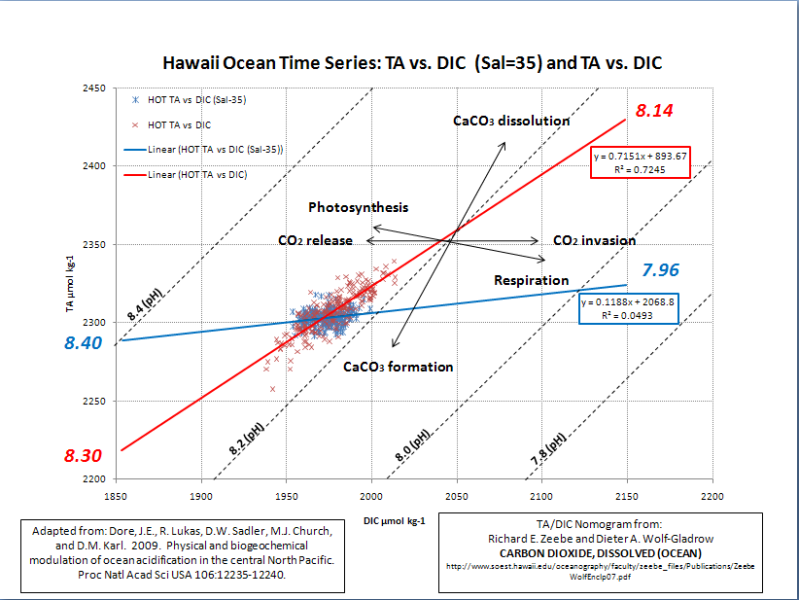
The only way I get a pH decline comparable to 0.4 to 0.5 is when I use the TA and DIC values that were normalized to a salinity of 35 (blue curve). This yields a pH decline of 0.44 (8.40 to 7.96); but it is a horrible correlation (R^2=0.05). TA and DIC are highly correlated to salinity(R^2=0.88, 0.74). DIC has a moderate correlation (R^2=0.39) and TA has a weak correlation (R^2=0.12) to atmospheric CO2…
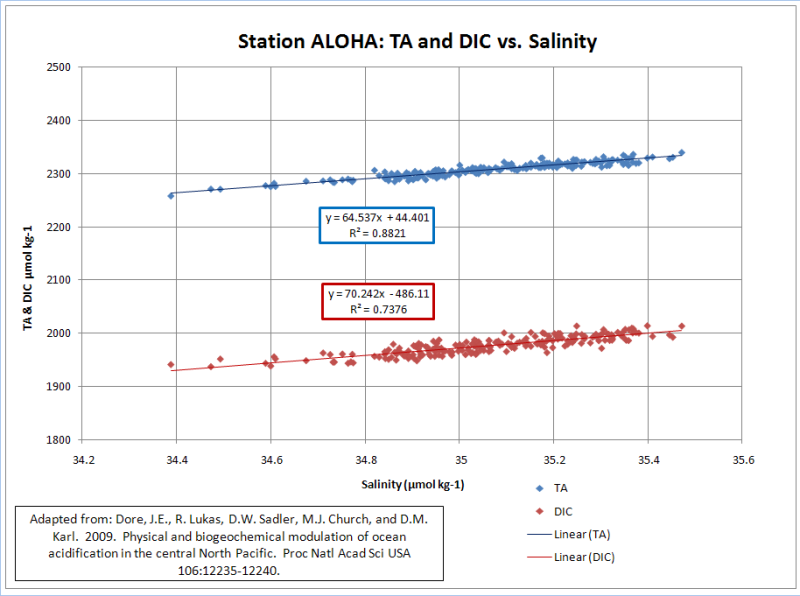
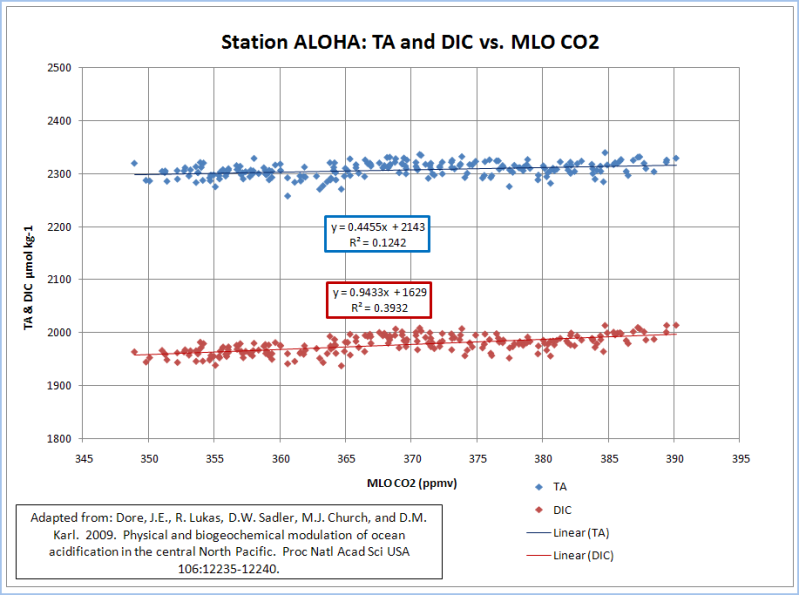
The normalization of TA and DIC to a constant salinity subdues the buffering provided by salinity; while amplifying the acidification effect of increasing CO2. A realistic treatment of salinity, yields an insignificant lowering of pH from a doubling of pre-industrial CO2. Chicken Little of the Sea does not appear to be very dangerous.
Will Chicken Little of the Sea Wreak Havoc on Marine Calcifers?
The Hawaii Ocean Time-series included ~20 years worth aragonite saturation data for Station ALOHA; so we can estimate the effects of atmospheric CO2 changes on aragonite saturation. Ries et al., 2009 experimentally demonstrated the effects of aragonite saturation changes on marine calficers under several CO2 scenarios. So, we can combine Ries’ experimental results with the observational data from Station ALOHA.
I plotted Ries’ CO2 vs. aragonite saturation assumptions and found that a power function trend-line was the best fit. I then plotted the Station ALOHA aragonite saturation vs. MLO CO2 and applied a power function trend-line to extrapolate the aragonite saturation out to about 2900ppmv CO2.
The Station ALOHA data demonstrate a significant difference in the actual effects of CO2 on aragonite saturation relative to the Ries et al., 2009 scenarios…
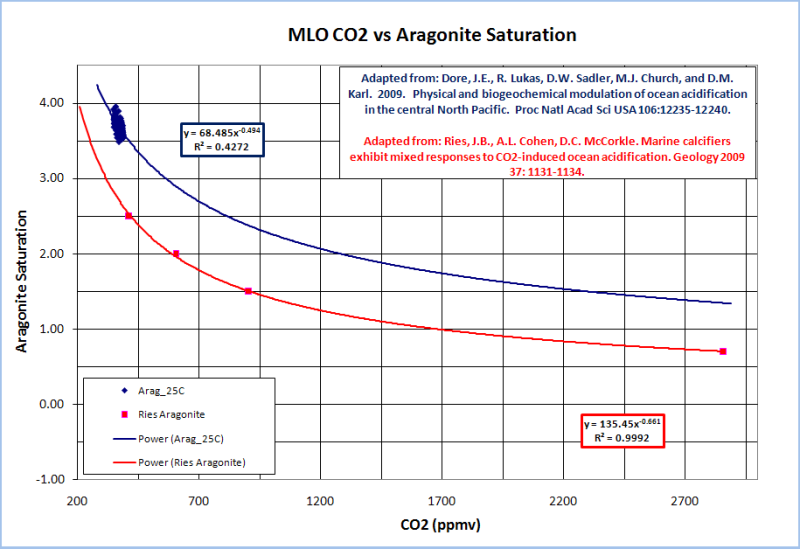
The observational data show that rising atmospheric CO2 is less detrimental to aragonite saturation levels than was assumed in the Ries experiment.
The Station ALOHA data indicate that marine calcifers have a much higher threshold CO2 level at which the calcification rate drops below its current range than that indicated by Ries…
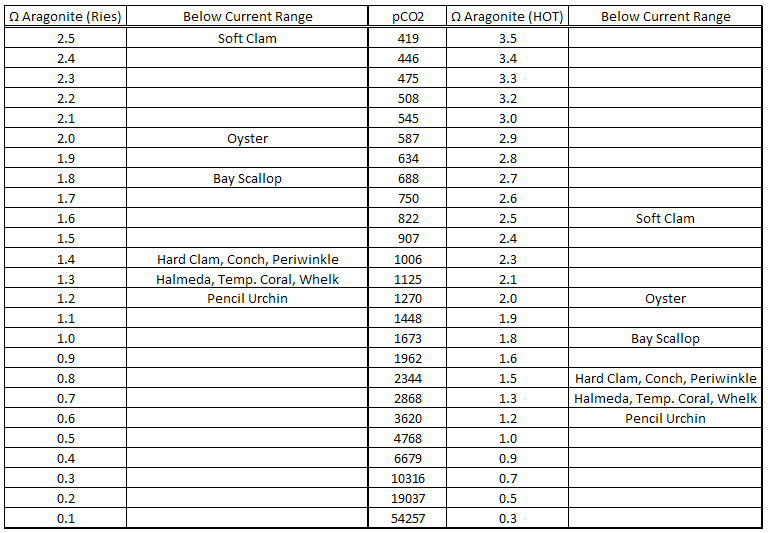
The observed relationship between atmospheric CO2 and aragonite saturation from Station ALOHA indicates that even the most vulnerable marine calcifer in the Ries study (soft clam) was not adversely affected by CO2 levels below 800ppmv.
References:
Richard E. Zeebe and Dieter A. Wolf-Gladrow
CARBON DIOXIDE, DISSOLVED (OCEAN)
Hawaii Ocean Time-series (HOT)
Station ALOHA Surface Ocean Carbon Dioxide
Dore JE, Lukas R, Sadler DW, Church MJ, KarlDM (2009)
Physical and biogeochemical modulation of ocean acidification in the central North Pacific.
Proc Natl Acad Sci USA 106:12235–12240.
Abrupt Decrease in Tropical Pacific Sea Surface Salinity at End of Little Ice Age
Ries, J.B., A.L. Cohen, D.C. McCorkle. Marine calcifers exhibit mixed responses to CO2-induced ocean acidification. Geology 2009 37: 1131-1134.
Great post David.
I wonder how many of the acidification Chicken Littles actually understand the chemistry. Precious few, I’ll bet.
How far can you use a linear regression with pH, given that it’s a log function?
A single location. A single depth. Measuring pH (w/ some procedural risk) in Nature’s most buffered solution…. Hmmm.
Call me a skeptic…..
@Z,
Its perfectly legitimate to regress to pH, since the biological effects are also logarithimic as far as pH is concerned.
Most biological systems use a logarithmic response: your ear, your eyes, your nose.
It is the most efficient way to cope with a wide range of data: it makes sense! Literally!
Excellent.
It is nice to do the debunking early.
Bookmark this folks we may need it in the near future!
Totally OT, but zeebe as in “Richard E. Zeebe and Dieter A. Wolf-Gladrow” means wolf.
The very fact that the Pollo Pequeños use “acidification” as a term for the decrease of alkalinity is telling in itself. Use of the term implies that the ocean has become acidic, lending alarm to the equation, when, in reality, the water is still firmly alkaline: in other words, nothing to worry about.
I experiment a couple of decades ago with CO2 injection in brackish water aquariums ( I also experimented with decomposing vegetation, something I would not recommend for armatures). I have not been able to find my data since my storage unit was broken into. But, I do not remember any appreciable change in PH non-correlated with temperature and time of day. OTOH, I do remember a significant difference between my morning and evening measurements.
I see someone has already gotten onto the “pH is more than 7, so a decrease in pH is not acidification” thing. Can we all agree that lowering pH is acidification and raising it is alkalinization. There are enough issues with this area of science without descending into pedantry. It is very difficult to search for a definition of “acidification” because there is so much out there on ocean acidification. Alkalinization on the other hand is not popular, so definitions are easy to find. Most of them are along the lines of “The process of BECOMING alkaline”. Hence by those definintions, only a base can be acidified and only an acid can be alkalized.
I’m fairly certain that I didn’t regress pH.
I wonder whether the sulfate concentration is changing at all. If burning S-rich coal is producing H2SO4, perhaps that might have some effect in some locations on the surface. I don’t buy the CO2 connection. There is a lot of C exchange with the atmosphere, and deposition as sediment. It seems although CO2 may be involved in a fluctuating equilibrium, other drivers would be more important than atmospheric CO2. Also, there is no discussion about N fixation and the resulting increase in alkalinity. What are the ratios of C, N, and P? I’d worry more about N and P imbalance than CO2. I would expect toxic effects are more likely to occur with excess P. Of course, that sort of imbalance is more likely close to estuaries near urban areas. Algal blooms and low DO can be very unpleasant if you have to live in the sea (that is, if you are a fish, etc.).
Typo? “hydrogen ([H-])” Should it not be hydrogen ([H+])?
@Werner Brozek
[H-] is a typo.
Ask any mermaid
you happen to see
what’s the best loonie
chickens**t of the sea.
The ocean contains about 50x as much CO2 as does the atmosphere, so even if we add enough CO2 to the atmosphere to double the atmospheric CO2 level, as the extra CO2 dissolves into the ocean it will still have very little long term effect on the oceans’ acidity. Dr. Jack Barrett has good info on ocean acidification here:
http://www.barrettbellamyclimate.com/page50.htm
What is with this daja vu? Are the oceanographers making the same mistakes as the atmospheric people did 30 years ago?
DesertYote says:
November 24, 2011 at 3:58 pm
“OTOH, I do remember a significant difference between my morning and evening measurements.”
Dynamic patterns and ecological impacts of declining ocean pH in a high-resolution multi-year dataset
J. Timothy Wootton1, Catherine A. Pfister, and James D. Forester, 2008
http://www.pnas.org/content/105/48/18848.full
“Examination of 24,519 measurements of coastal ocean pH spanning 8 years (Fig. 1) revealed several patterns. First, in contrast to the historical perspective that the ocean is well buffered, pH exhibited a pronounced 24-hour cycle, spanning 0.24 units during a typical day (Fig. 1A). This diurnal oscillation is readily explained by daily variation in photosynthesis and background respiration: water pH increases as CO2 is taken up, via photosynthesis, over the course of the day, and then declines as respiration and diffusion from the atmosphere replenish CO2 overnight (16). Second, pH fluctuated substantially among days and years, ranging across a unit or more within any given year and 1.5 units over the study period. Finally, when the entire temporal span of the data was considered (Fig. 1B), a general declining trend in pH became apparent.”
http://www.pnas.org/content/105/48/18848/F1.large.jpg
Fig. 1. Patterns of ocean pH through time at Tatoosh Island (N = 24,519). (A) Daily cycle of pH arising from photosynthetic uptake of CO2 by algal primary producers. Colors indicate month that the data were collected (blue, April; black, May; red, June; green, July; purple, August; yellow, September). (B) pH readings as a function of date and time taken between 2000 and 2007. The decline is significant (P < 0.05).
The authors place most of their emphasis on the down trend, but what I always found most interesting is the extreme range of pH values they measured over a fairly brief time period and a very small area. Given that rather large variability, it seems to me that if life in the oceans was as sensitive to minor changes in pH as the catastrophists claimed most of it would have disappeared long long ago.
On a side note, at about the time this paper came out, I was chasing links on this topic one day and I came across a couple papers that suggested a multidecadal oscillation in the mean pH of the oceans over a 0.4-0.5 range and a 30yr or 60 yr timescale as I recall. Unfortunately I got distracted and didn't bookmark them and I haven't been able to find them again. Has anybody else seen these papers? If so, I'd appreciate a link.
What a good post! Thank you.
Seems the ocean is more resilient than the team.
According to my text books on oceanography the pH varies between 7.9 to 8.4 in ocean surface waters. So this small change is within the natural variation!?! What is the problem.
Any pH change round Hawaii is more probably due to volcanic gasses which at the black smokers on the oceanic ridge system, divergent boundaries, the pH has been measured at 4.5. In spite of this acid molluscs seem to thrive and have done for millions of years.
@Dave Wendt on November 24, 2011 at 11:15 pm
The paper you are trying to recall is Pelejerso’s pH reconstruction of Flinder’s Reef. The [I]Science[/i] paper is available with a free AAAS membership. The data are available from NOAA’s paleoclimatology library.
Pejejero demonstrated a ~0.5 pH oscillation with a ~50-yr period, highly correlated to the PDO.
Coastal and coral reef waters exhibit wide variations in pH on times scales ranging from diurnal to multidecadal. Open ocean waters generally exhibit less variability.
In the opening graph from Dore at al 2009, why is pH at 30 m depth about 8.08, and then (after a data gap), pH 8.07 at 235 m depth? Given that one cannot place any confidence in a pH change of 0.01, why is there an “inconvenient” data gap from 30m to 135 m depth?
At what depth at this location is the unconformity between near-surface waters with pH around 8 and deeper waters around 7.6 or so? I realize that there is said to be little mixing of waters either side of this horizon, based on salinity, conductivity etc measurements, but surely it would take only a little mixing to wipe man-made atmospheric CO2 largely out of contention, partly because of the logarithmic mathematics for pH in mixing. Like, equal volumes of pH 7 and pH 8 do not mix to make pH 7.5.
pH is the negative logarithm of the hydrogen ion activity.
@Dave Burton on November 24, 2011 at 8:58 pm
Twenty years of actual data from Station Aloha demonstrate that the models (like the one posted by Dr. Barrett) are not reflective of the real world (see Fig. 8 in this post).
I remember reading a paper on the chemistry of meltwater from an alpine glacier and there was a significant diurnal trend.Bearing in mind the complexity of the chemistry of the sea close to shore( within continental shelf), there is going to need a vast amount of data with regard to spatial , temporal and determinants to ensure valid conclusions are arrived at. Lagoons and bays in tropical latitudes have extremely variable chemistry. Inputs from sewage, soil run off , algal blooms churning of sea bed by dredging, etc , etc are all going to make near shore chemistry very difficult to analyse.
John Eggert says:
November 24, 2011 at 5:07 pm
I see someone has already gotten onto the “pH is more than 7, so a decrease in pH is not acidification” thing
While I understand the point you are making, I can’t help but feel that if you were stood outside at the south pole about now and noticed that the temperature had risen from -40°C to -35°C , you wouldn’t be saying that it was getting hotter.
further to davidburton’s comment, I did a ‘back of the envelope’ calculation on the assumptions that half of human emissions are partitioned into the hydrospehre and marine biosphere, and that the ocean is generally reasonably well mixed (i.e. an assumption that may not be applicable for barred basins, lagoon areas and lakes but probably applies for the vast majority of the ocean). The mass of the oceans mean that the change in dissolves CO2 would be less than 1 ppm, and so the consequence on pH by the ‘increase’ in carbonic acid would be undetectable.
There are possible causes of acidification of near shore waters because of run-off from fertilisers and other local pollution sources, and these should be dealt with as far as reasonably possible, but I honestly don’t see CO2 as a significant problem in this regard.