How to suck carbon dioxide from the sky for fuels and more
Someday, the gasoline you buy might trace its heritage to carbon dioxide pulled straight out of the sky rather than from oil pumped out of the ground. By removing emitted carbon dioxide from the atmosphere and turning it into fresh fuels, engineers at a Canadian firm have demonstrated a scalable and cost-effective way to make deep cuts in the carbon footprint of transportation with minimal disruption to existing vehicles. Their work appears June 7 in the journal Joule.
“The carbon dioxide generated via direct air capture can be combined with sequestration for carbon removal, or it can enable the production of carbon-neutral hydrocarbons, which is a way to take low-cost carbon-free power sources like solar or wind and channel them into fuels that can be used to decarbonize the transportation sector,” says lead author David Keith, founder and chief scientist of Carbon Engineering, a Canadian CO2-capture and clean fuels enterprise, and a professor of applied physics and public policy at Harvard University.
Direct air capture technology works almost exactly like it sounds. Giant fans draw ambient air into contact with an aqueous solution that picks out and traps carbon dioxide. Through heating and a handful of familiar chemical reactions, that same carbon dioxide is re-extracted and ready for further use–as a carbon source for making valuable chemicals like fuels, or for storage via a sequestration strategy of choice. It’s not just theory–Carbon Engineering’s facility in British Columbia is already achieving both CO2 capture and fuel generation.

The idea of direct air capture is hardly new, but the successful implementation of a scalable and cost-effective working pilot plant is. After conducting a full process analysis and crunching the numbers, Keith and his colleagues claim that realizing direct air capture on an impactful scale will cost roughly $94-$232 per ton of carbon dioxide captured, which is on the low end of estimates that have ranged up to $1,000 per ton in theoretical analyses.
That price-point is low enough to use direct air capture to start tackling the roughly 20% of global carbon emissions that result from driving, flying, trucking, and other ways of getting people and goods around. “Electricity from solar and wind is intermittent; we can take this energy straight from big solar or wind installations at great sites where it’s cheap and apply it to reclaim and recycle carbon dioxide into new fuel,” Keith says, adding that “Making fuels that are easy to store and transport eases the challenge of integrating renewables into the energy system.”

The resulting fuels, including gasoline, diesel, and jet fuel, are compatible with existing fuel distribution and transportation infrastructure. Thanks to ultra-low life cycle carbon intensities, they are a promising route for reducing carbon emissions in heavy transportation and other sectors of the energy system that are demanding and difficult to electrify.
Centuries of unchecked human carbon emissions also mean that atmospheric carbon dioxide is a virtually unlimited feedstock for transformation into new fuels.
“We are not going to run out of air anytime soon,” adds Steve Oldham, CEO of Carbon Engineering. “We can keep collecting carbon dioxide with direct air capture, keep adding hydrogen generation and fuel synthesis, and keep reducing emissions through this AIR TO FUELSTM pathway.”
Keith and Oldham are optimistic that they have reduced scale-up risks by implementing direct air capture at reasonable costs using standard industrial equipment. That means that all the pieces are in place to move on to full-size plants capable of manufacturing 2,000 barrels of fuels per day– totaling over 30 million gallons per year across plants.

Commercialization of such plants would allow direct air capture to make a dent in transportation emissions by connecting low-cost renewable energy to low-carbon transportation fuels using Carbon Engineering’s AIR TO FUELSTM pathway.
“After 100 person-years of practical engineering and cost analysis, we can confidently say that while air capture is not some magical cheap solution, it is a viable and buildable technology for producing carbon-neutral fuels in the immediate future and for removing carbon in the long run,” says Keith.
###
In addition to funds raised by Carbon Engineering, this work was supported by the British Columbia Innovative Clean Energy Fund, Sustainable Development Technologies Canada, the Industrial Research Assistanceship Program, and the U.S. Department of Energy.
The paper:
Joule, Keith et al.: “A process for capturing CO2 from the atmosphere” https://www.cell.com/joule/fulltext/S2542-4351(18)30225-3
Okay, hook it directly to the national debt so it can be even more efficient as a perpetual motion machine.
The fuel produced will first have to power the engines which drive the generators that power the fans, pumps and compressors needed for the process. And breaking the O2 from the C is surely not a room temperature process? And if the end product is going to be a hydrocarbon, won’t we have to add hydrogen? Hydrogen could be got by electrolysis of H2O, but it’s usually gotten by “reforming” natural gas – it’s cheaper (more efficient), and the only byproduct is …. CO2.
But after all that it’s all free energy, baby. If we just spend enough on research, propaganda and subsidies….
The fact that “scientists” are pushing this is proof positive that “scientists” will lie for money – they couldn’t possibly be stupid enough to think this will produce more energy than it consumes.
Or it can be free if you capture CO2 in the form of tettuce and potatoes.
The cleanest fuel is natural gas (methane) which is produced naturally by decomposition in low O2 environments (land fills, sewage treatment plants, swamps, forest floors, hog farm lagoons. ) I’m sure it would be a lot cheaper to use this renewable resource than trying to produce fuel out of thin air (sort of like central banks printing fiat money). Also. the polar oceans are already sucking up CO2 on a scale several magnitudes greater than is possible by their method.
Or, you can compel everyone to wear a CPAP like carbon capture device while they sleep and create a multi-trillion dollar utility infrastructure to pipe all the exhaled CO2 to a processing plant. This sounds just as practical, right?
You’ll need a connection in your tail pipe as well to capture the generated methane. Not sure you’ll get many who’ll willingly install one of those.
No doubt, the California state legislature is already working on a law to make that mandatory.
It worked for water recovery for the Fremen on Dune, why not CO2?
What could possibly go wrong. Once upon a time, citizens of planet earth were worried about Nuclear war destroying the planet. Have no fear, modern ‘scientists’ will outdo the nuclear menace by an order of magnitude.
The big sucking bird shredder is a laughable image. All to make a quart sized jar of fuel.
It looks to me like they have invented a very expensive way of making Moonshine liquor.
The market for these devices is innumerate consumers.
How much ya wanna bet that they have ignored construction and maintenance costs is their calculations?
….small correction:
The target market is innumerate policy makers and opinion leaders.
Artificially removing CO2 from the atmosphere is … dumb.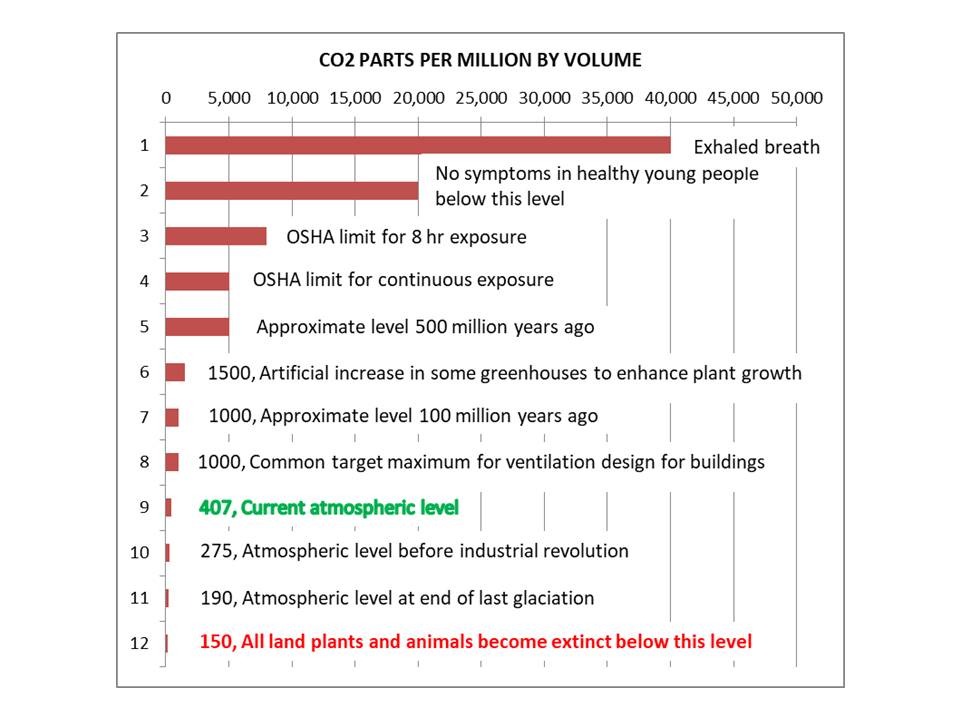
can you provide a source for the graph. Thanks
Right click, copy link address
Not sure about item #1.. 10k ppm level kills pests on plants via asphyxiation. Not sure how long a human could survive. Going from memory, 10,000 ppm, per docs I have, is deadly to humans within about an hour of exposure.
http://www.generalair.com/pdf/Safety%20Topics/Carbon%20Dioxide%20Asphyxiation.pdf
Ralph – Not sure where you’re going with that link. I assume you realize O2 storage is also dangerous. https://www.homecaremedical.com/services/training/oxygen-use-home-fire-safety-guidelines-storing-handling-oxygen-equipment/
The claim that 10,000 ppm (1%) was “deadly to humans within about an hour” didn’t look right.
Ever heard of mouth-to-mouth resuscitation? That is done with 40,000 ppm exhaled air. The high CO2 is actually considered beneficial since it stimulates the breathing reflex.
Yep ! 10,000ppm will kill white-fly. HUMANS NOT AFFECTED at this level.
You breathe-out 4% to 5% CO2 ( 40,000ppm to 50,000ppm) without harm.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
“Submarine crew are reported to be the major source of CO2 on board submarines (Crawl 2003). Data collected on nine nuclear-powered ballistic missile submarines indicate an average CO2 concentration of 3,500 ppm with a range of 0-10,600 ppm, and data collected on 10 nuclear-powered attack submarines indicate an average CO2 concentration of 4,100 ppm with a range of 300-11,300 ppm (Hagar 2003).”
WUWT has an article on CO2
“Claim: CO2 makes you stupid? Ask a submariner that question
Anthony Watts / October 17, 2012”
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
CO2 is lethal at 100,000 ppm for about 5 to 10 minutes exposure.
That is 10% or about 250 TIMES as much as the present atmospheric concentration !!!
H2S however is lethal at 1000 ppm………ONE BREATH will do it !
Makes CO2 sound positively harmless !
Boy am I lucky.
On my boat I experienced about 1000 hours of 20,000 PPM CO2 while conducting Polaris patrols in the ’60s. Don’t know how I escaped certain death.
Don’t know how I escaped certain death
Would you know how had you not experienced such high-levels of CO2?
Earl
maybe you are dead and you just don’t know it
Wikipedia:
Inhaled air is by volume 78.08% nitrogen, 20.95% oxygen and small amounts include argon, carbon dioxide, neon, helium, and hydrogen.[16]
The gas exhaled is 4% to 5% by volume of carbon dioxide, about a 100 fold increase over the inhaled amount. The volume of oxygen is reduced by a small amount, 4% to 5%, compared to the oxygen inhaled. The typical composition is:[17]
5.0–6.3% water vapor
74.4% nitrogen
13.6–16.0% oxygen
4.0–5.3% carbon dioxide [40,000PPM – 53,000PPM]
[17] P.S.Dhami; G.Chopra; H.N. Shrivastava (2015). A Textbook of Biology. Jalandhar, Punjab: Pradeep Publications. pp. V/101.
You are wrong. Perhaps you made a decimal point error?
https://www.nap.edu/read/11170/chapter/5
“Submarine crew are reported to be the major source of CO2 on board submarines (Crawl 2003). Data collected on nine nuclear-powered ballistic missile submarines indicate an average CO2 concentration of 3,500 ppm with a range of 0-10,600 ppm, and data collected on 10 nuclear-powered attack submarines indicate an average CO2 concentration of 4,100 ppm with a range of 300-11,300 ppm (Hagar 2003).”
Why would we ever want to take CO2 out of the air? The atmosphere needs more CO2 NOT less.
Not all land plants and animals would become extinct below 150 ppm.
There would however be no trees, unless humans genetically modified them with C4 or CAM pathway genes. That would be even easier to do with C3 crop plants, as suggested for rice:
https://en.wikipedia.org/wiki/C4_carbon_fixation#Plants_that_use_C4_carbon_fixation
https://en.wikipedia.org/wiki/Crassulacean_acid_metabolism#Comparison_with_C4_metabolism
Could this end up being an actual global warming experiment. If they reduce CO2 levels down to pre-industrial times, we will be able to see what happens to temperatures.
If the effect of CO2 is as large as the IPCC needs to justify its existence, the presumed ‘forcing’ will force the planet into a 100 thousand year long ice age.
Since the warmists consider than increasing CO2 has caused global warming, surely they must agree that removing CO2 will cause cooling? And who can I sue as the level drops from 407 towards 150, thus endangering my life? Meanwhile, our NZ Socialist government has announced Green-influenced plans for a zero-carbon economy – no doubt in line with Green extremists’ wish to eliminate human life!
Jacinta’s hodgepodge of Marxist loons is really upsetting. $50 million given away to offset some bizarre climate guilt. and $80 million to blanket poison our forests, endlessly.
The greens do appear to hate human endeavour. I’m looking forward to the cognitive dissonance when they reveal Franz Josef Glacier grew this year
And since the increase in forcing is logarithmic decreasing the level of CO2 would be a very touchy exercise 30 or 40 PPM could be a disaster that would spiral us into a new ice age.
Even if they sequester the CO2 they extract, they won’t be able to put a dent in world-wide levels of CO2. They also won’t be able to make money on the process, so they will likely go broke unless the government funds them. However, if they make fuel out of the extracted CO2, they can sell the fuel and make some money. But then the process would only be carbon-neutral because burning the fuel will release the CO2 back into the air.
Atmospheric CO2 is also a practically unlimited feedstock for things called plants, which are also a bit more effective in converting it into polysaccharides. As CO2 is not a climate driver there is no need for elaborate schemes to do something which nature does perfectly well on its own. A solution for an imaginary problem.
I am sure that this atmospheric CO2 to fuel scheme would cost less if they made the fuel from trees, instead of directly from the air.
The irony is that this process is no more (and in fact no less) carbon neutral than fossil fuels.
Is this from the Onion?
Nope. Real. In my town. The interesting thing was when the enviros realized this was “just a way to perpetuate the fossil fuel economy”. We are all a bit perplexed at this plant – not sure if it is a real project , or just a way to keep the grant money flowing.
Bet on the latter.
Scanning through the paper, it appears no actual fuels were produced. Meanwhile, the forests and grasslands and crops in the northern hemisphere are doing the job nicely. When can we finally accept that CO2 is not a pollutant?
They should call it “Photosynthesis”……
Financynthesis
Cost effective? I’ve got a bridge in Brooklyn to sell ya….
We had London Bridge, but someone bought it.
They thought it was Tower Bridge.
Sorry Scot but please do not continue to promulgate that old urban myth. The buyers knew exactly what they were buying. They not only examined the bridge but took their time calculating the cost of disassembly and transport before putting in a bid.
Instead of doing this, you could just burn the money and use that fire for heat and energy. I am sure it would be more economical.
Indeed. Thermodynamically even more favorable if you add all of the money spent building the ‘big solar and wind farms’ needed to generate the intermittent and unreliable electricity they want to use to power the CO2 suckers!
There is a silver lining here, however…. If you devote all of the intermittent and unreliable solar and wind energy to solely power off-grid monstrosities like this proposal, the national electrical grid becomes immediately more stable, reliable, and the life cycle cost of grid based energy produced drops!
At least they give a cost estimate, but it is still rather question-begging as to global warming.
Yes, because we need to steal the food of all the plants on the planet.
I would generally expect to be reading someone making fun of an article like this on WUWT. Instead, WUWT is actually publishing this kind of stuff directly.
The US Department of Energy is still funding idiotic studies such as this?
I hear trees are pretty good at capturing “carbon”. Been doing it for eons.
Is it powered by 100% solar or wind ??
Unicorn flatus turbines.
The paper mostly describes a method to capture CO2 and turn it into solid CaCO3. For their CO2 to fuel process, they rely on hydrogen derived from electrolysis.
Hydrogen from electrolysis requires a separate power plant. The idea that the process will ever produce economic fuels is utter BS.
CaO + CO2 = CaCO3 … so where does the CaO come from?
Bake the CaCO3 at high temp, the CO2 is released. The CO2 in pure form is a sought after bulk commodity so that it can be processed into CO, which can then be converted by Fischer-Tropsch to synthetic methane, propane, gasoline, diesel, and wax.
The CaCO3 is converted back to CaO upon release of the CO2. The CaO is then used in the machine to absorb more CO2 from the atmosphere, thus the CaO is used in a loop as a CO2 capture mechanism. The purpose of this machine is an effective CO2 capture mechanism, compared to say liquefaction methods or biomass oxidation.
The Fischer-Tropsch synthetic fuels need CO+H2 as feedstock (called syngas). Over a simple iron-oxide catalyst, can make any synthetic liquid and gas fuel (from methane to octane to cetane to waxes). The process makes a very high quality ‘neat’ fuel, i.e. almost entirely paraffins. (Aside: most gasoline and diesel is a complex soup of paraffins, olefins, aromatics, and other complex organics. These other complex carbon compounds reduce the octane & cetane ratings, cause gumming, and other unwanted side effects of combustion performance.)
The CO2 to CO process can be done by ‘reverse water gas shift’, there might be a better method, I don’t know for sure. Anyway, RWGS also requires H2.
The H2 can be made by electrolysis sourced from hydroelectric, solar, wind, or nuclear.
The output is a high quality traditional fuel that can be burned by traditional engines, and can be more efficient if optimized. They would then release the CO2 to the air. This machine would capture the CO2 from the air, and close the carbon cycle. It is not a perpetual motion machine. The actual energy driving everything is nuke, solar, wind, or hydro (for a non-carbon source), the synthetic fuel just repackages that primary energy in a convenient, easy to carry form.
It is a remarkable technology, especially considering that LENR equipment can be manufactured this year.
I am about to make a breakthrough, just need a million. Please.
Bunk. No LENR is coming this year or this decade.
Or ever.
this year. or next. We read this claim every year since ~2000.
I don’t know if LENR exist. I do know it isn’t understood, and far from real life application even if it exists. It takes decades to turn a proper science into a technology with real-life application for average Joe. Just look at electricity, nuclear energy, telephone, photoelectric effect, LED device, etc.
https://www.scientificamerican.com/article/cold-fusion-lives-experiments-create-energy-when-none-should-exist1/
I’m always amused by the terms “scalable and cost-effective” The cost of doing anything, at small scale, in an academic environment is negligible because the overhead and external costs are zero. Move this outside that environment, do the design and engineering work, and then come back to me with the notion of scalability and an objective cost-benefit analysis.
The plants already do that, why reinvent the wheel? Simply let the plants pull the CO2 out of the atmosphere and turn the plants into fuel. That process is called the Fischer-Tropsch Process and has been used for years. My bet it is far cheaper to scoop up lawn clippings and paper garbage than run that machine. Anyway, yes, converting existing CO2 into fuel is a great idea. We should be spending money developing Fischer-Tropsch and related technology refineries instead of wasteful wind and solar.
At about 240 kg of CO2 per barrel of oil, it takes 3.75 barrels of oil to produce 1 ton of CO2. Even at the overly optimistic cost of $200 per ton to extract, you still need to disassociate a lot of water to get the needed hydrogen to make fuel which adds another couple of hundred dollars per ton. The bottom line is that it still looks like it takes more fuel to make the fuel being produced. And of course, it always takes more energy to produce fuel than is released by burning the produced fuel.
Why is it that all these ‘remedies’ for a climate crisis that’s precluded by the laws of physics sound like perpetual motion machines? Perhaps it’s because perpetual motion is precluded by the same laws?
It’s too bad that lots of electricity consumers are already maxed out paying for massive windmills. We told you clowns that you have hundreds of years to see if this is really a problem and to come up with ways to address it, but no. You had to go and jump the gun and spend all your money on windmills. This is why we can’t have nice things.
Carbon dioxide can be removed from the air (if desired) by contacting air with certain chemicals, including caustic solution, which forms dissolved carbonates.
But trying to produce hydrocarbon fuels from carbon dioxide is counterproductive, and downright stupid. The First Law of Thermodyamics tells us that if an exothermic reaction, such as fuel + oxygen –> CO2 + steam, is run backwards, the backward reaction requires as much net energy input as the forward reaction releases.
The Second Law of Thermodynamics tells us that in any energy conversion process, some energy is lost to the surroundings as heat.
This means that more energy input is required to convert CO2 back to fuel as could be obtained by burning the fuel. Why should any sane person want to do this?
Steven
Wind turbines are a stupid idea. But they still went ahead with them.
I doubt that any of the authors have heard of, or if they understand any of the laws of thermodynamics. The first and second law of thermodynamics are the two laws before which every other law of physics must bow. Any proposed process which violates those laws just validates the PT Barnum Law (there is a sucker born every minute).
“Why should any sane person want to do this?”
Because electro-chemical batteries suck. Even Li-ion still suck compared to a high quality gasoline or diesel fuel, by an order of magnitude worse energy/volume and energy/mass density. Using this as one step in a syn-fuel process enables converting plentiful nuke (or other) energy to a more convenient, easy to carry form, that is a nearly trivial drop in replacement for the gasoline and diesel infrastructure.
If and when we run out of oil and gas, this could be really useful to perpetuate the ICE.
These people need to get out a little more. A visit to a gas station in the US or Brazil shows that capture of CO2 from the atmosphere and creation of fuel is already an industrialized process and motorists are already using ethanol from that process. Plants (not industrial plant – green growing plants) photosynthesize using CO2 from the atmosphere and create sugars which can then be used to create ethanol for fuels.
And industrial hemp is best for biofuels. Low moisture, easy to grow, doesn’t need a lot of water or fertilizer, pest resistant. The U.S. is slowly coming around to removing industrial hemp from the drug list, it should happen this year. This will be the next major cash crop for the U.S.
The more CO2 there is in the air, the more efficient this thing is going to be. So obviously we need to start boosting CO2 levels as fast as we can.