Guest post by Bob Fernley-Jones by Bob Fernley-Jones AKA Bob_FJ
CAUTION: This is written in Anglo-Oz English.
Here is the diagram as extracted from their 2009 paper, it being an update of that in the IPCC report of 2007 (& also 2001):
The unusual aspect of this diagram is that instead of directly showing radiative Heat Transfer from the surface, it gives their depiction of the greenhouse effect in terms of radiation flux or Electro-Magnetic Radiation, (AKA; EMR and a number of other descriptions of conflict between applied scientists and physicists). EMR is a form of energy that is sometimes confused with HEAT. It will be explained later, that the 396 W/m^2 surface radiation depicted above has very different behaviour to HEAT. Furthermore, temperature change in matter can only take place when there is a HEAT transfer, regardless of how much EMR is whizzing around in the atmosphere.
A more popular schematic from various divisions around NASA and Wikipedia etc, is next, and it avoids the issue above:
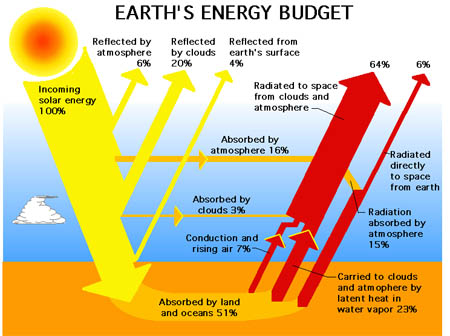
- Figure 2 NASA
Returning to the Trenberth et al paper, (link is in line 1 above), they give that the 396 W/m2 of EMR emitted from the surface in Fig.1 is calculated primarily by using the Stefan–Boltzmann law, and global year average conditions. Putting aside a few lesser but rather significant issues therein, it is useful to know that:
1) The Stefan-Boltzmann law (S-B) describes the total emission from a flat surface that is equally radiated in all directions, (is isotropic/hemispherical). Stefan found this via experimental measurement, and later his student Boltzmann derived it mathematically.
2) The validity of equally distributed hemispherical EMR is demonstrated quite well by observing the Sun. (with eye protection). It appears to be a flat disc of uniform brightness, but of course it is a sphere, and at its outer edge, the radiation towards Earth is tangential from its apparent surface, not vertical. It is not a perfect demonstration because of a phenomenon called limb darkening, due to the Sun not having a definable surface, but actually plasma with opacity effects. However, it is generally not apparent to the eye and the normally observed (shielded) eyeball observation is arguably adequate for purpose here.
3) Whilst reportedly the original Stefan lab test was for a small flat body radiating into a hemisphere, its conclusions can be extended to larger areas by simple addition of many small flat bodies of collectively flat configuration, because of the ability of EMR waves to pass through each other. This can be demonstrated by car driving at night, when approaching headlights do not change in brightness as a consequence of your own headlights opposing them. (not to be confused with any dazzling effects and fringe illumination)
4) My sketch below demonstrates how radiation is at its greatest concentration in the lateral directions. It applies to both the initial S-B hemispherical surface radiation and to subsequent spherical radiation from the atmosphere itself.
5) Expanding on the text in Figure 3: Air temperature decreases with altitude, (with lapse rate), but if we take any thin layer of air over a small region, and time interval, and with little turbulence, the temperature in the layer can be treated as constant. Yet, the most concentrated radiation within the layer is horizontal in all directions, but with a net heat transfer of zero. Where the radiation is not perfectly horizontal, adjacent layers will provide interception of it.
A more concise way of looking at it is with vectors, which put simply is a mathematical method for analysing parameters that  possess directional information. Figure 4, takes a random ray of EMR (C) at a modestly shallow angle, and analyses its vertical and horizontal vector components. The length of each vector is proportional to the power of the ray, in that direction, such that A + B = C. Of course this figure is only in 2D, and there are countless multi-directional rays in 3D, with the majority approaching the horizontal, through 360 planar degrees, where the vertical components also approach zero.
possess directional information. Figure 4, takes a random ray of EMR (C) at a modestly shallow angle, and analyses its vertical and horizontal vector components. The length of each vector is proportional to the power of the ray, in that direction, such that A + B = C. Of course this figure is only in 2D, and there are countless multi-directional rays in 3D, with the majority approaching the horizontal, through 360 planar degrees, where the vertical components also approach zero.
6) Trenberth’s figure 1 gives that 65% of the HEAT loss from the surface is via thermals and evapo-transpiration. What is not elaborated is that as a consequence of this upward HEAT transfer, additional infrared radiation takes place in the air column by virtue of it being warmed. This initially starts as spherical emission and absorption, but as the air progressively thins upwards, absorption slows, and that radiation ultimately escapes directly to space. Thus, the infrared radiation observable from space has complex sources from various altitudes, but has no labels to say where it came from, making some of the attributions “difficult”.
DISCUSSION; So what to make of this?
The initial isotropic S-B surface emission, (Trenberth’s global 396 W/m2), would largely be absorbed by the greenhouse gases instantaneously near the surface. (ignoring some escaping directly to space through the so-called “atmospheric window”). However, a large proportion of the initial S-B 396 surface emission would be continuously lateral, at the Trenberth imposed constant conditions, without any heat transfer, and its horizontal vectors CANNOT be part of the alleged 396 vertical flux, because they are outside of the vertical field of view.
After the initial atmospheric absorptions, the S-B law, which applied initially to the surface, no longer applies to the air above. (although some clouds are sometimes considered to be not far-off from a black body). Most of the air’s initial absorption/emission is close to the surface, but the vertical distribution range is large, because of considerable variation in the photon free path lengths. These vary with many factors, a big one being the regional and more powerful GHG water vapour level range which varies globally between around ~0 to ~4%. (compared with CO2 at a somewhat constant ~0.04%). The total complexities in attempting to model/calculate what may be happening are huge and beyond the scope of this here, but the point is that every layer of air at ascending altitudes continuously possesses a great deal of lateral radiation that is partly driven by the S-B hemispherical 396, but cannot therefore be part of the vertical 396 claimed in Figure 1.
CONCLUSIONS:
The vertical radiative flux portrayed by Trenberth et al of 396 W/m^2 ascending from the surface to a high cloud level is not supported by first principle considerations. The S-B 396 W/m^2 is by definition isotropic as also is its ascending progeny, with always prevailing horizontal vector components that are not in the field of view of the vertical. The remaining vertical components of EMR from that source are thus less than 396 W/m^2.
It is apparent that HEAT loss from the surface via convective/evaporative processes must add to the real vertical EMR loss from the surface, and as observed from space. It may be that there is a resultant of similar order to 396 W/m^2, but that is NOT the S-B radiative process described by Trenberth.
~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~
ADDENDUM FOR AFICIONADOS
I Seek your advice
In figure 5 below, note that the NIMBUS 4 satellite data on the left must be for ALL sources of radiation as seen from space, in this case, at some point over the tropical Pacific. The total emissions, amount to the integrated area under the curve, which unfortunately is not given. However, for comparison purposes, a MODTRAN calculator, looking down from 100 Km gives some interesting information for the figure, which is further elaborated in the tables below. Unfortunately the calculator does not give global data or average cloud/sky conditions, so we have apples and pears to compare, not only with Nimbus, but also with Trenberth. However, they all seem to be of somewhat similar order, and see the additional tabulations.
| Compare MODTRAN & “Trenberth”, looking down from 2 altitudes, plus Surface Temperature | ||||
| Location | Kelvin | 10 metres | 100 Km. | (Centigrade) |
| Tropical Atmosphere | 300K | 419 W/m^2 | 288 W/m^2 | (27C) |
| Mid-latitude Summer | 294K | 391 W/m^2 | 280 W/m^2 | (21C) |
| Mid-latitude Winter | 272K | 291 W/m^2 | 228 W/m^2 | (-1C) |
| Sub-Arctic Winter | 257K | 235 W/m^2 | 196 W/m^2 | (-16C) |
| Trenberth Global | 288K ? | 396 W/m^2 | 239 W/m^2 | (15C ?) |
| Compare MODTRAN & “Trenberth”, looking UP from 4 altitudes: W/m^2 | ||||
| Location | From 10 m | From 2 Km | From 4Km | From 6Km |
| Tropical Atmosphere | 348 | 252 | 181 | 125 |
| Mid-latitude Summer | 310 | 232 | 168 | 118 |
| Mid-latitude Winter | 206 | 161 | 115 | 75 |
| Sub-Arctic Winter | 162 | 132 | 94 | 58 |
| Trenberth Global | 333 Shown as coming from high cloud area (= BS according to MODTRAN) | |||



Jose_X , Yes I am disagreeing with your observation that we each agree more CO2 will lead to higher temperatures of some amount and that to be precise we would have to perform calculations?
Firstly what do you mean by ‘some amount’?
>>
Jose_X says:
November 30, 2011 at 9:43 am
[Jim Masterson]>> I tried this argument on another thread
I don’t see what argument that was.
<<
The argument that per Wien’s law 15 microns is -80 °C. The surface has to be pretty cold for CO2 to absorb in this range.
>>
[Jim Masterson]>> thoroughly chastised (by alarmists on the thread) for not knowing that Planck’s equation gives different results in the frequency domain vice the wavelength domain.
Without seeing the argument I have no idea what those people attempted to say. Yes, where the speed is essentially speed of light, labeling the graph using frequency or wavelength can be done consistently and is really immaterial. It sounds to me that it would have been an odd argument.
<<
See the May 7 post by Ira (http://wattsupwiththat.com/2011/05/07/visualizing-the-greenhouse-effect-light-and-heat/). Specifically see Phil’s May 19, 2011, 8:19 pm response to me. That should be enough context to see if the argument was odd.
Be careful. This is where jae called me a “kook.”
Jim
Jim Masterson, I don’t know why he was talking about wanting to focus on 340K. I didn’t notice anything wrong with your reply (1-1 correspondence), but his response didn’t make much sense to me. I might have to read more, but I am not very interested :-).
Robert Stevenson, OK. You seemed to have acknowledged that the photons might find an absorber quicker and said that any gain (in temp) would likely be negligible. I pointed out that if you find CO2 quicker generally, that you’d get a larger number of photons bouncing around before leaving. Since the Sun is “constant”, this means we have a greater amount of energy in the atmosphere, which means higher temp somewhere. I figured you would agree and took your “negligible” comment to mean you’d accept that there might be some gain in temp.. say north of 0.00001 C. 1 C gain (purely due to CO2) for a doubling, to me, might be called negligible by some people.
Anyway, so you don’t think there would be anything resembling 1 deg C gain for CO2 doubling? I don’t think anyone can argue that one way or other without calculations.
And saying most photons from earth miss CO2 is not specific enough to say whether temperature may rise a lot or hardly at all.
Although the blanket example is not a broad analogy for the atmosphere “greenhouse effect”, it suggests that slowing down the escape of radiation leads to increased temperature over what we’d otherwise have.
An oven with the door closed is the same thing. We have very little heat leaving relative to the energy in the oven and the high temp.
Remember that the sun keeps firing photons at us nonstop. If the earth keeps radiating away and those photons take a long time to escape the atmosphere, that is a lot of kinetic energy among the molecules (think of the oven example). So slowing down the radiation escape means a greater concentration of energy.. higher temp.
Now, if you have no prob with greenhouse effect yet specifically think CO2 will have negligible effect, why would you say that without calculations?
btw, I know convection is what helps even the temps inside and outside quickly, but the analogy is useful to show that temp rises when we slow down the rate of loss of heat. This slowdown is what I am referring to.
>>
Jose_X says:
November 30, 2011 at 12:07 pm
I might have to read more, but I am not very interested.
<<
I don’t blame you. I wasn’t either.
Jim
With a little care, the issue of looking at graphs of [intensity vs wavelength] or [intensity vs wavenumber] doesn’t matter. It is not a question of “where the peak is” but rather “the total energy in the band”. Presumably calculations for the total energy absorbed by CO2 will be the same whether you deal with wavelength or wavenumber.
In one representation, CO2 might be near the peak, but have a narrow band. In the other representation, CO2 might be far from the peak, but have a broader band. If you want real numbers, you will have to integrate areas under curves. Presumably, if CO2 absorbs 20% of photons in when discussing wavelength, it will still absorb 20% when discussing wavenumber. Just looking at peak wavelength (or peak wavenumber) is insufficient.
The simplest analogy that springs to mind would be something like this. Suppose I have a whole bunch of speakers set to 20 Hz, 20.1 Hz, 20.2 Hz ….. 100 Hz. If I plotted (speakers per Hz) as a function of frequency, there would be a flat graph with (10 speakers per Hz) across the entire graph.
But If I plotted (speakers per meter) as a function of wavelength, I would find there are many more speakers with a wavelength between 4m and 5 m (~ 170) than between 16 m and 17 m (~ 12).
[Jim Masterson]>> The argument that per Wien’s law 15 microns is -80 °C. The surface has to be pretty cold for CO2 to absorb in this range.
I think what you are saying is that you have to have a fairly cold blackbody radiation if you want the “15 micrometer CO2 band” to end up centered at the max wavelength. However, the key point is area under curve and not whether a particular band is centered or not.
mkelly, I agree that “It will only be heated to the temperature of the highest radiator.” I disagree with the rest.
If I completely surround an object with 270 K ground/atmosphere/walls/clouds that emit ~ 300 W/m^2, the object will never get warmer than 270 K (absorbing ~ 300 W/m^2 if it is a black body).
And if I completely surround an object with 5780 K plasma (like the sun) that emits ~ 63,000,000 W/m^2, the object will never get warmer than 5780 K (absorbing ~ 63,000,000 W/m^2 if it is a black body).
If I partly surround the object with hot plasma @ur momisugly 5780 K and partly surround the object with 270 K ground/atmosphere/walls/clouds, the object will absorb somewhere between 300 W/m^s and 63,000,000 W/m^2, reaching a temperature somewhere between 270 K and 5780 K.
The energy from the cooler surroundings is still absorbed by the object, even though the object is warmer than the surroundings. The object does not magically know to absorb 4 um photons from the sun but reject 4 um photons from the surroundings.
Jose_X @ur momisugly November 29, at 12:22 pm
Of course the defences are not just made-up of the bowmen, but also politicians, green organisations, and the media, some of which are truly fearsome warriors.
The main Oz national broadcaster, the ABC, consistently breaches its editorial policies, and charter by act of parliament when it comes to AGW. For instance, I am not alone in noticing that they have been silent on the so-called Climategate II Emails. (as is also reportedly the BBC). Given that a significant majority of Australians are polled showing that they would have a strong interest in them, they have clearly breached their published editorial policy. It is particularly damming because of the timing of the release to coincide with that latest jamboree in Durban.
I recommend that you carefully read, as it contains some awful naughties between your heroes.
http://wattsupwiththat.com/2011/11/22/climategate-2-0/
The exchange between Severinghaus and Mann is rather telling, particularly since the infamous Hockey-stick, was so brilliantly applied to deceive the policy makers and media into thinking that recent warming, (prior to about the last decade), is unprecedented.
Oh BTW, unlike the Laframboise book at $4.99, it’s FREE
@Jose_X…
Two questions for you:
1. What is a photon?
2. How long a molecule of carbon dioxide retains the absorbed energy from a photon?
Thanks for answering.
Jose_X @ur momisugly November 30, at 8:51 am
In the context of elementary quantum theory:
You may be lacking in spatial perception when considering an absorbent atmosphere. For argument’s sake, let’s contemplate that the mean photon free path length from the surface is 10m. Let’s say your implied multiple sensors are uniformly distributed over a wide area at some height of less than 10m. Your suggested “sideways intercepts” of horizontal or near horizontal photons would need to be a great distance away from their source, but such photons cease to exist long before they could get there at only around 10m away from source. Therefore they cannot be detected in the direction that you suggest. Multiply these numbers by a factor of 10 if you like, but you get much the same story. Much the same situation exists in succeeding progenies of isotropic radiation into absorbent air at higher altitudes. (except of course in “transparent wavelengths”)
Jose_X,
Did you properly answer Robert’s November 29, at 4:53 am, particularly the bit I’ve bolded below?
I, and I guess so too Nasif, would be interested to hear your wisdom on this!
(although as I understand it, photons are rarely reemitted: it is a misnomer. Photons are typically annihilated upon absorption according to QT, and subsequent emissions depend on mutual transfer of KE in collisions, mostly with N2 and O2, and temperature and stuff).
Hi Nasif. Now I’m sure this is going to be quite interesting. I’ll just listen.
[wayne]>> Now I’m sure this is going to be quite interesting.
Well, I’m finding it to be interesting as well. I won’t know if I’m right without a fair amount of thinking and discussion. Whatever the outcome, it’s a free physics tutorial/lesson.
To revisit mkelly’s point, my reply considered that the multiple heaters were above ambient. No matter the case, a photon lost is energy lost. A photon gained is energy gained. Radiating is a cooling effect. I suppose I was also assuming the heaters were being driven by a source of energy that allowed them to remain at the elevated temperatures. For the planet case, we have the sun as a “constant” source of energy. So I think a better reply to:
[mkelly] >> The biggest problem with the KT graph is that you cannot add and subtract W/m^2 when talking about temperature. 10 W/m^2 of sunshine plus 10 W/m^2 of IR from atmosphere does not equal 20 W/m^2 of radiation to heat an object. It will only be heated to the temperature of the highest radiator.
would be:
(a) We don’t have enough information in general to make a determination about the last sentence based on arbitrary radiating bodies. We’d need to know the energy sources for the radiators and also the environment of the radiators. On earth, most radiators would wind down in energy if not constantly heated by the sun. The environment plays a major role. Meanwhile, we aren’t much affecting the sun, so that radiator can be assumed to be driven by an infinite reservoir of energy and radiate independently from the affects on earth.
(b) 10 W/m^2 from the atmosphere will lead to cooling of the atmosphere unless replenished. The earth and almost all bodies on it (to first approx, for the purposes of the earth system analysis) can also be assumed to be like the atmosphere in that, as they radiate and contribute energy to other bodies, they lose it and must have it replenished to preserve their temp (eg, by being radiated back).
(c) 10 W/m^2 from the sun is additional energy (and infinite stream essentially). If we on earth keep getting more of it and not radiating out into space (quickly enough), we will keep cooking — meaning on average getting hotter.
[Bob Fernley-Jones] >> The main Oz national broadcaster, the ABC .. For instance, I am not alone in noticing that they have been silent on the so-called Climategate II Emails.
From my perspective, calling climate science Oz is at this point in my understanding an assumption. I don’t see it that way. I will note that as pointed out by Jon Stewart (assuming he was correct), the BEST preliminary results — and generally we can say this about all the literature adding up monthly which supports climate science — were not covered by most media outlets. So I can easily call it a bias that the media is widely ignoring all the evidence and studies supporting current climate science.
I think a fair representation of climategate II is that the media jumped the gun before (CG I) and got burned to some degree. The media goes after what will attract eyeballs to sell ads. You can call conspiracy, but the fact remains that formal inquiries found nothing improper about climategate I (at least not beyond normal bureaucratic failures and politics that exists in all human organizations), and at least there is enough doubt about that doing any significant damage to climate science. Of course, that had an impact of sorts, but the science was not refuted because of it (although it obviously has attracted extra scrutiny).
http://mediamatters.org/blog/201111220013
>> The previous release of hacked emails triggered a storm of ill-informed media coverage in December 2009, with news outlets rushing to quote the documents without taking the time to research the context or ask experts to translate the scientific language
[Bob Fernley-Jones] >> between your heroes
I show respect to the growth of the science. I am certainly open to improvements. I really don’t know enough to take a strong position for or against any particular accepted climate specific theory, but the majority of attacks I see fall short IMO (so far of what I have experienced).
Can current results be hacked? Well, I know little enough that from that limited perspective I would say of course I think that would be a possibility. This is one reason I have taken interest in hearing the objections.
[Bob Fernley-Jones] >> The exchange between Severinghaus and Mann is rather telling, particularly since the infamous Hockey-stick, was so brilliantly applied to deceive the policy makers and media into thinking that recent warming, (prior to about the last decade), is unprecedented.
Can you point to a precise discussion? The link you provided had a load of stuff.
Also, when you say “telling”, what do you think is the conclusion? That result x or y are wrong? What specifically?
[Bob Fernley-Jones] >> unlike the Laframboise book at $4.99, it’s FREE
I likely wouldn’t be looking at it if it wasn’t free. I really wasn’t trying to deceive you. I have not bought reading material in a long time (I think the last time I bought reading material was $20 for an electronic ansi standard years ago). I also don’t make a lot of money.
[Nasif Nahle] >> 1. What is a photon?
What point are you trying to get at?
I am not an expert in photons. I have taken some quantum mechanics courses. I can research. I am more than willing to consider your definition.
In short, if you want a response form me in order to get a clue about my background and biases and lapses, a photon is a massless entity with a fixed amount of energy. All “entities” I think, potentially exhibit particle and wave behavior to some degree (even if almost immeasurable for large complex entities).
[Judging from a different comment I reply to further down, let me add that a photon effectively is created when it is emitted and then destroyed when “absorbed”. I think theory considers these particles to exist at a given quanta of energy and not to exist any longer once they are “absorbed”. The theory isn’t that a photon gets re-emitted later after being absorbed, no; however, I am not sure that a model where photons do live forever would necessarily be incorrect, but it appears it would simply lead to a more complex theory and potentially less intuitive. Tomorrow, however, we may find a new model if we appear to discover that there are 3 centillion photons in the universe, not one more or less, and they hide within other particles and change their energy levels when they leave the other particles, etc.]
[Nasif Nahle] >> 2. How long a molecule of carbon dioxide retains the absorbed energy from a photon?
I don’t know the specifics (haven’t set up the model or done the math or read it). The duration in time falls out of the wave function in some way. The actual time for any molecule is expected to vary, but we can calculate expectation values if we know enough about the system. Whether that event is likely to happen before the molecule reacts with another molecule, I am not sure.
On a related note: I think I heard that the time between absorbing photons (not sure in what part of the atmosphere this applies) is perhaps 2 orders of magnitude longer than between encountering and interacting with another molecule (ie, having it’s kinetic energy and momentum changed is likely to happen before another photon is absorbed).
[Bob Fernley-Jones] >> but such photons cease to exist long before they could get [to the sensor]
I don’t see your point.
If the photon hits something less than 10m (which is where you placed the hypothetical sensors), then it imparts energy. That energy didn’t disappear. It arose from the planet at some point X and was absorbed at some other point at least slightly above it.. regardless that it went laterally or straight up. So the point is that (the energy associated with) all photons created within some surface m^2 eventually make it higher up. Diagramming that as 396 “up” sounds perfectly reasonable (assuming the values are correct of course).
[Bob Fernley-Jones/Robert S]>> The subsequent retention time of these photons as they are re-emitted may be slightjy altered but not to affect the overall heat balance.
I addressed this before by saying that the concluding part of this statement is qualitative opinion. It isn’t quantitative. I am sure many people have many opinions here. It’s even ambiguous what “slightly” would mean. Yes, I addressed this. If you want more, we need numbers and/or calculations (or, if you think you can measure it, measurements).
You also mentioned contact of these molecules with other molecules (exchange of KE). I am not sure what point you want to communicate.
Also, I know that photons don’t bounce around. It is energy transfer essentially. My point is that if the odds of absorption of a photon are greater because of more molecules, then this leads to a longer time period before the energy radiated at ground level dissipates. The energy contribution of a given photon produced at ground level has to go through more intermediaries (via energy exchanges, whether absorption and regeneration later on of photons or by molecules coming into contact with other molecules.. leading to the eventual photon radiation that simply leaves the atmosphere). A hypothetical measurement of this extra time would be seen by measuring the time constants of the earth temp dropping towards 0 K if we turn off the sun when the earth is at current temps.
Remember that the sun provides a constant addition of energy. If it takes longer to radiate into space any given addition of energy, then we have on average now a greater amount of energy on the earth’s atmosphere at any new point in time. By definition, I think, temperature of gas represents the average kinetic energy of each particle. More energy in the atmosphere with the same number of particles, volume, etc means a higher energy. — Sure, the volume might increase… that is something to consider. — And with higher temp, we get a more rapid rate of radiation, which may eventually lead to greater or equal radiation outward as we receive from the sun (but at that higher temperature). When the rate out is equal, we should have stable global average temps (assuming no other chemical or other reaction disturbs this balance). When rate out is greater, we will now start to cool as more energy will be leaving then entering.
We can look at temp as a measurement of the amount of energy (eg, kinetic energy) in place in the (eg) atmosphere at any given point in time. An analogy would be with water level (temp) in a tub where we have a source of water into the tub and a drain.
To clarify on the comment I made recently:
1: “On earth, most radiators would wind down in energy if not constantly heated by the sun.”
In other words: Most objects on earth would lose “all” (aka, “most of”) their energy in a noticeable time period if it weren’t for the continuous energy the planet receives from the sun. Technically, many objects on earth pretty much “get their energy from” other objects directly (but ultimately from the sun if we follow the chain of energy acquisitions).
This might be a silly clarification to make, normally, but I’ll do it now in the context of the current discussion of, “what is a photon?”
I may have misspoke any number of times (and will in the future) because I frequently wear the hat of a layperson and enough times don’t use technically correct jargon. Ask away if anything I say sounds odd. We are all on this same boat anyway.
To sum up that that first part (to mkelly’s comment), basically I’m saying that power does add up, in the sense that energy adds up, but, to know what happens to temperature, we’d have to know enough details to know the power/energy values to add AND to subtract, before then applying S-B or equivalent to get the temperature. So I think mkelly’s comment was too vague to conclude that the final temperature is simply the temp of “the highest radiator” (not sure what that means). Also, I can see how we might achieve here on earth a temp higher than the sun’s temp even though ultimately the energy probably came from the sun at some point in the past.
2: “More energy in the atmosphere with the same number of particles, volume, etc means a higher energy.”
The last word should be “temperature” and not “energy”.
The main point in that section is that temperature of the earth system can have many internally different temperatures (eg, average temp at TOA vs. close to the ground vs. in the oceans vs. in volcanoes, etc). Think of power going into an oven and power radiating out. A well insulated oven might have an internal high temperature for a given measured amount of power in and out (in/out in equilibrium), while a less quality oven might have a lower internal temperature for the same given amount of power in/out. The earth might receive some power and radiate some power yet have built up furnaces of high temp deeper below the TOA.
So I think its rather natural to accept the possibility of “greenhouse effect” within our atmosphere. We may disagree over the details of which gas (if any) has how much greenhouse effect, but the mere idea that energy would be retained to enable a higher temperature (and power radiation) in one part of the earth system internally vs. at its boundary with space should not be too hard to accept. We see many related examples all around us. If conduction and convection (which are what specifically?) can be kept at gradients via judicious use of barriers in the context of given “energy sources”, why can’t radiation also be a part of that equation and possibly apply within the atmosphere?
@Jose_X…
“In short, if you want a response form me in order to get a clue about my background and biases and lapses,”
Forget about it… Professional background, and biases and lapses do not matter in science when they are not related with the information that is being examined.
“…a photon is a massless entity with a fixed amount of energy.”
In physics and biology, entity is an abstraction of the real world. If we add the word “analytical”, for example “physics analytical entity, we are referring to an abstraction created with the purpose of describing and analyzing physical processes. If we classify photons as physical analytical entities, we would be creating an abstraction which would permit us to describe photons and processes related to photons. Therefore, the label “entity” does not fit with the definition of photon; at least, it is not the concept of photon handled in quantum physics.
Photon is a perturbation of the electromagnetic field which manifests itself as a particle with inertial mass and a wave with wavelength and frequency according to the energy that it has available which is usable on other fields.
Given that wavelengths and frequencies of quantum/waves depend on the amount of energy that they transport, it is evident that the energy of quantum/waves is not fixed.
On the other hand, matter particles also exhibit wave’s nature.
The great question is about the definition of photons as massless particles; we cannot say that photons are massless particles because they have inertial mass, and in a given moment it can be interpreted as gravitational mass. The latter was evidenced when researchers measured the redshift of quantum/waves due to gravity. Gravity makes a photon to shorten its wavelength and to increase its frequency:
Δf = f ((gH)/c^2)
Consequently, a quantum/wave originated in the Sun carries a higher amount of energy which diminishes as the condensation goes farther from the source and increases as the quantum/wave goes approaching to any interfering system with gravitational mass which is capable of absorbing it.
C = Cp (δ) (r)
For atmospheric carbon dioxide in current Earth’s atmosphere, the capacity of carbon dioxide to store energy during 120 picoseconds (1.2 × 10 ^ (-10) seconds) is 609.7 J/m^2 K.
t =√(ΔE*(1/((m^2 kg) )) )= √(1.345 x 10^(-20) (kg m^2/s^2) * (1/((m^2 kg) )) ) = 1.16 x 10^(-10) s
As way of comparison, the whole atmosphere retains 2000 times more energy than carbon dioxide during 138 picoseconds.
It is preferable to think about a distribution of energy more than about “heat retained”.
The major mistake that I have read from some articles published in the Internet involves the amount of quantum energy absorbed by a system. No system in the known universe absorbs the whole load of energy transported by a quantum/wave. It is erroneous to suppose that the load of energy carried by absorbed photons is transformed into stationary energy. There is a retrograde movement of the absorbent molecules when a quantum/wave hits upon them. Additionally, there is an amount of dynamic energy that is transformed into unusable potential energy; consequently, the absorbed energy by a molecule of carbon dioxide is not the same as the energy carried by the absorbed photon.
Regarding the question about the annihilation of a quantum/wave as soon as it is absorbed by a system, it is true; the quantum/wave emitted is not the same quantum/wave that was absorbed. Emitted photons have longer wavelengths and lower frequencies, so emitted photons are less energetic than absorbed photons. This characteristic avoids that molecules of same species absorb photons emitted by molecules of their own species.
There is not physical basis in support of global warming caused by carbon dioxide.
Nasif
@ur momisugly Nasif
Very clear as usual. I do have a question for you along the lines of the atmosphere’s shear mass in relation to what Miskolczi’s work tends to imply on tau, but I will defer that for later, not to jostle this thread here.
@ur momisugly Jose_X
I’ll deem this as a free lesson/tutorial the minute either you or I actually do learn something new to our respective understanding of these topics.
The x/6’ths seemed to throw you above. I could have placed that in spherical coordinates with equal ease but as I told you before, I like to say things here on WUWT as simply as possible for the many different backgrounds of those taking the time to read these comment, many of them never comment at all.
You see Jose_X, if you are so intelligent in these topics you should be able to mentally transform my simple x,y,z Cartesian example into the more complex proper physics, principle by principle, and agreedwith most of what I was explaining. IR photographs do not show that low-level whole-atmosphere IR radiation because that portion a CCD registers is zeroed out to give you a good photograph. Those ‘events’ do occu. It does not mean the quantum/waves are not present (at an event level).
Read it again later. (and yes, those energy transfers are ‘after-the-fact’ absorptions of the quantum/waves in an explicit direction and at an exact time since it was separated into vectors and accumulated to give a rate.) Personally, I will continue to take the time to word things here in a manner that any person should understand with a little thought, regardless of their background or nomenclature.
If I were you I would not assume people here are as clueless as you apparently assume. Your smart, translate, fill in, assume correctness, read between the lines. You keep saying your willing to learn but if you continue to splice out partial thoughts just to tear them into pieces, I for one will stop feeding you. You say you want to learn, climb off that horse.
Nasif Nahle, OK, what you wrote did not land smoothly upon my eyes. Some of my objection might be reasonable. But surely this is an opportunity for me to learn and review some physics.
>> massless
Note, I did not clarify that by “massless” I meant zero rest mass only. E=mc^2 implies all energized particles have mass.
>> If we classify photons as physical analytical entities, we would be creating an abstraction which would permit us to describe photons and processes related to photons. Therefore, the label “entity” does not fit with the definition of photon; at least, it is not the concept of photon handled in quantum physics.
I did not understand what distinction you were trying to make with my use of the word “entity”. I chose what I thought was a simple dictionary word that is rather general in ability to describe something we believe exists. I did not realize there was a specific special meaning for that word in physics and biology. Can you provide a reference where I may be able to read up on the use of that word in these fields of study?
I am not a philosophy major by a long shot, but, at least in science, the only meaning that really exists is that which forms a part of some model or other. I don’t see what distinction you would create between an actual object and a model of that object.
>> Photon is a perturbation of the electromagnetic field..
I will note that electromagnetic field (and everything else in physics) is just a model.
Anyway, this appears to be a philosophy diversion. Feel free not to clarify this point (I’m not sure why you brought it up), and I will just move on regardless. I generally don’t want to get hung up with words.
And like I said, I will try to work with whatever definition you find suitable (which will depend on what tools/models you use to analyze the problem).
>> ..which manifests itself as a particle with inertial mass and a wave with wavelength and frequency according to the energy that it has available which is usable on other fields.
To the extent I cut short my education and likely will have to read up on this, are you referring to classical E&M field theory?
>> Given that wavelengths and frequencies of quantum/waves depend on the amount of energy that they transport, it is evident that the energy of quantum/waves is not fixed.
I don’t follow. Do you mean anything beyond the Heisenberg Uncertainty Principle?
>> we cannot say that photons are massless particles because they have inertial mass
Agreed. I meant zero rest mass.
>> Consequently, a quantum/wave originated in the Sun carries a higher amount of energy which diminishes as the condensation goes farther from the source and increases as the quantum/wave goes approaching to any interfering system with gravitational mass which is capable of absorbing it.
Can you specify the mathematical form of this wave or at least specify if you are referring to a single photon or a group of photons (what is the quantum system you refer to)?
I have very little experience with wave equations, but I will try to follow. Or you can use some other description (eg, dump the wave function approach for the sake of this discussion/analysis).
>> For atmospheric carbon dioxide in current Earth’s atmosphere, the capacity of carbon dioxide to store energy during 120 picoseconds (1.2 × 10 ^ (-10) seconds) is 609.7 J/m^2 K.
>> t =√(ΔE*(1/((m^2 kg) )) )= √(1.345 x 10^(-20) (kg m^2/s^2) * (1/((m^2 kg) )) ) = 1.16 x 10^(-10) s
Do you mean the capacity to store energy for a time interval of at least 120 picoseconds or is 120 the average?
Can you specify from what you derived this (or name of this formula). Is this a calculation of some sort of time expectation on a wavefunction? Can you give details. I cannot follow your logic without more clues.
I’ll repeat I am not practiced here, so any derivations I may have to perform may take time, but you can help by pointing to the name of the equations you started with or something.
>> C = Cp (δ) (r)
This may be obvious to you, but can you specify the name of these quantities or the formula name?
I am willing to do research, but since I don’t use these formulas and may never have seen them, it helps to at least specify what these variables represent.
>> No system in the known universe absorbs the whole load of energy transported by a quantum/wave. It is erroneous to suppose that the load of energy carried by absorbed photons is transformed into stationary energy.
Wave functions refer to probabilities, so I am not sure what you mean here by total energy.
[I find this very interesting because these are calculations I have not done.]
..If the wave is of many photons, then of course no molecule is going to absorb all the energy. I am not sure why you would look at the wavefunction of many photons yet then talk about a single molecule, but if that is what you are doing, I’m all ears.
>> Additionally, there is an amount of dynamic energy that is transformed into unusable potential energy; consequently, the absorbed energy by a molecule of carbon dioxide is not the same as the energy carried by the absorbed photon.
..If you are talking of a single photon, can you reference which experiments support the view that only a fraction of the energy of a photon is absorbed by a molecule?
You can likely get away with many models that in the end come to the same conclusion (we would have to know the details of the theory); however, I have not delved deeply into this area and the simple models I remember have each photon energy be imparted entirely or not at all, iirc. The classical view works differently, but that is because it describes many photons and a reduction of that set gives the appearance that a fraction of the energy of the whole was imparted.
Again, if you could be clear about your wavefunction or the theories/experiments you are invoking, I could better follow your argument.
I do recognize that a “collision” between any given particles may produce some of the same particles (with different energy levels, etc) as we started out with, so perhaps this is what you mean. [Can you clarify and provide experiment or math?]
>> Emitted photons have longer wavelengths and lower frequencies, so emitted photons are less energetic than absorbed photons.
Well, this is an extension of your point.. which I don’t agree with, generally, from what I remember of basic quantum mechanics (and I simply may not remember my physics from many years back). However, I can certainly change my mind if I have some basis (eg, experimental basis or a mathematical analysis that requires the same type of particle exist afterward but with different values).
>> This characteristic avoids that molecules of same species absorb photons emitted by molecules of their own species.
This is a conclusion I cannot accept since I don’t currently buy your earlier points.
I do get the feeling that this is not standard accepted physics.
Also, it doesn’t readily follow from you claims earlier since molecules of the same specie can absorb different amounts of energy (within some “band” range of values), so it would be quite possible to have one CO2 molecule emit a less energized photon (on this argument you provide) that would still be acceptable to a different CO2 molecule (ie, would still fall within the overall absorption band range).
I suppose it’s also possible (and I noted this confusion on my part in an earlier comment.. I think to wayne, iirc) that the photon energy can be used to affect the electrons in the molecules and also the whole molecule’s average kinetic energy. I have to believe this is very possible, but I have not thought about this very much or remember explicitly reading this (which may be a very basic common fact). In any case, this would not support your conclusion that CO2 cannot absorb photons emitted from other CO2 since we’d have a clear mechanism for the same molecule to accept a very wide range of values .. In fact, I really think this is true; however, a very high energy photon probably is more likely than not (QM wavefunction probability analysis) to simply pass over a CO2 molecule rather than to impart its energy and ionize the molecule.
>> There is not physical basis in support of global warming caused by carbon dioxide.
I disagree and suppose you deduce this from the view you present that CO2 cannot pass photons to other CO2.
Honestly, this sounds rather strange to me: that no molecule can ever pass energy to another of the same type via a photon seems rather absurd.
wayne, why are you going back to that x/6? There is no fundamental rule or definition that creates that and says “up” is associated with it (and if you believe their is, please reference that definition). The coordinate system is immaterial. Your “6” is an arbitrary division of the space you decided to create because you wanted to define “up” in that context.
You mentioned Nasif was “very clear as usual”, in which case, every question I posed Nasif I pose to you as well because I found a number of things that weren’t very clear to me about that comment.
Also, based on your earlier replies, I find it suspect you would claim Nasif’s comment was clear. [BTW, you never addressed what part of that radio astronomy webpage you referenced stated something that was inconsistent with what I have been saying. Supposedly much on that page was, so it would be useful to me if you could be specific.]
>> Personally, I will continue to take the time to word things here in a manner that any person should understand with a little thought, regardless of their background or nomenclature.
I agree that is generally a good idea for everyone to follow, but many conversations cannot procede except with math (it depends on the nature of the particular sub-discussion).
>> If I were you I would not assume people here are as clueless as you apparently assume. Your smart, translate, fill in, assume correctness, read between the lines.
I am all ears for healthy discussion. There is little need for assumption of what others know or don’t. Simply put, let’s argue/discuss. I’ll try to follow as much as I can and state my views, of course.
>> You keep saying your willing to learn but if you continue to splice out partial thoughts just to tear them into pieces
I don’t think I have been closed minded. In fact, I just asked Nasif (and you) a load of questions so I can better understand what models he(?) is using.
Jose_X @ur momisugly December 1, at 7:22 am
[1] It shouldn’t have taken you long to find it. See item 38: http://wattsupwiththat.com/2011/11/28/a-response-from-jeff-severinghaus-on-why-the-trees-dont-make-good-thermometers-after-1950-i-did-indeed-feel-at-the-time-that-mike-mann-had-not-given-me-a-straight-answer/
WUWT item 52 is also relevant, and takes you to Steve McIntyre’s new article:
Hide-the-Decline Plus: http://climateaudit.org/2011/12/01/hide-the-decline-plus/
Remember “Hide the Decline” in one of Phil Jones’ emails in climategate I?
Somewhere above in all that waffle, you say that you are willing to study and learn. You should apply this to what I’ve recommended that you read.
[2] It is yet more proof that Mann and other hockey-stick creators, e.g. Jones, Briffa, etc, omitted tree ring data from the mid 1900’s without explanation, because it showed an inconvenient downturn against the published instrumental data. Esper 2002 seems to be the only Dendro’ to openly discuss it, but he was Swiss, not part of the US/UK “team”, and was unpopular with the IPCC. He subsequently removed that paper from his publication list
If you study this you will see that the team were far from honest in their dealings… see Climategate I & II emails
“Hide the decline” is also known as “The Divergence Problem”
Oh, BTW; Oz =Australia(n) in this context.
Jose says: “Note, I did not clarify that by “massless” I meant zero rest mass only. E=mc^2 implies all energized particles have mass.”
Actually, you were closer to correct originally. Photons have no mass! I challenge anyone to find any authoritative reference that says photons have mass!
The full equation is not E = mc^2, but rather E^2 = (m^2 c^4) + (p^2 c^2). For particles with mass at rest, it becomes E = mc^2. For particles with no mass moving at the speed of light, it becomes E = pc.
So photons have energy. Photons have momentum. Photons are even affected by gravity. But they have no mass.
Jose says: “Anyway, this [the definition of “entity”] appears to be a philosophy diversion. ”
I agree. I have never seen “entity” used as a specifically defined term in physics. I think your use was understandable and reasonable.
Bob,
“Clearly, the 396, by definition, comes from the surface, but does not make-it to space. (or even to the high clouds as shown in the cartoon). The 396 and its progeny of new-generation emissions contain horizontal vector components which cannot be seen in the normal view. Thus, the vertical components must be less than the total S-B emission.”
Do you agree that the required emitted flux from the atmosphere that eventually passes into space at the TOA is not all from vertical emission from the atmosphere? By ‘vertical’ I mean emission perpedicular to the surface?
@ur momisugly Nasif
I am reading a bit between the lines but when you said:
—> This characteristic avoids that molecules of same species absorb photons emitted by molecules of their own species.
didn’t you mean “This characteristic avoids that molecules of same species absorb photons emitted by molecules of their own species at the same frequency of the quantum/wave that was originally absorbed?
To me, and you from what I have gathered from you, that the re-emitted radiation will always be of lower frequency, lower energy, multiple photons, due to both the momentum transfer/loss and that all thermal absorption is of ro-vibrational in nature, never strictly vibrational. (that is unless a path existed where some other event could separately add more energy to that same molecule.) Close?
Or are you saying due to the above characteristics that the frequency is basically always shifted so much lower and the energy split to prevent basically any like-species cross radiative transfer.
I can see both sides, but which is closer to reality? Would love a clue.
RW @ur momisugly December 1, at 5:01 pm
Yes, of course RW. Is it a trick question?
Nasif says:
Did you mean …
“Consequently, a photon originated in the Sun carries a higher amount of energy which diminishes as the photon goes farther from the source and increases as the photon goes approaching to any .” ?
I would agree with that — it sounds like a standard gravitational redshift.
What is this equation? What do the symbols stand for?
Where does this come from? Why is 120 picoseconds significant for energy storage? What is the square meter factor?
1.345 x 10^(-20) (kg m^2/s^2) seems to be the energy of a ~15 um photon. What is the significance of 1/(m^2 kg)? What is this equation?
I can’t tell quite what your objection is. Yes, there will be some energy that goes into KE of the CO2 molecule as it recoils. So, yes, the energy is not transformed entirely into “stationary energy”.
But what sort of “unusable potential energy” does the atom gain?
Molecules are generally quite good at absorbing the same photons that the same species of molecule emits. Between Doppler broadening and the natural line width related to the Heisenberg Uncertainty Principle, there are a variety of energies of photons that can be emitted and absorbed by a particular molecule.
But all this is rather off-topic from the original topic….