Guest post by Bob Fernley-Jones by Bob Fernley-Jones AKA Bob_FJ
CAUTION: This is written in Anglo-Oz English.
Here is the diagram as extracted from their 2009 paper, it being an update of that in the IPCC report of 2007 (& also 2001):
The unusual aspect of this diagram is that instead of directly showing radiative Heat Transfer from the surface, it gives their depiction of the greenhouse effect in terms of radiation flux or Electro-Magnetic Radiation, (AKA; EMR and a number of other descriptions of conflict between applied scientists and physicists). EMR is a form of energy that is sometimes confused with HEAT. It will be explained later, that the 396 W/m^2 surface radiation depicted above has very different behaviour to HEAT. Furthermore, temperature change in matter can only take place when there is a HEAT transfer, regardless of how much EMR is whizzing around in the atmosphere.
A more popular schematic from various divisions around NASA and Wikipedia etc, is next, and it avoids the issue above:
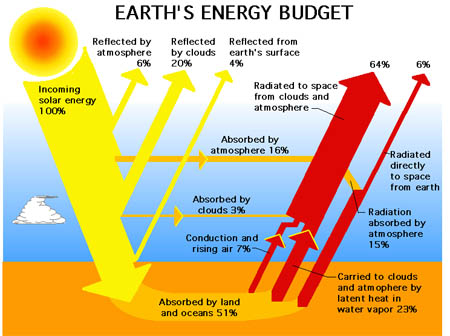
- Figure 2 NASA
Returning to the Trenberth et al paper, (link is in line 1 above), they give that the 396 W/m2 of EMR emitted from the surface in Fig.1 is calculated primarily by using the Stefan–Boltzmann law, and global year average conditions. Putting aside a few lesser but rather significant issues therein, it is useful to know that:
1) The Stefan-Boltzmann law (S-B) describes the total emission from a flat surface that is equally radiated in all directions, (is isotropic/hemispherical). Stefan found this via experimental measurement, and later his student Boltzmann derived it mathematically.
2) The validity of equally distributed hemispherical EMR is demonstrated quite well by observing the Sun. (with eye protection). It appears to be a flat disc of uniform brightness, but of course it is a sphere, and at its outer edge, the radiation towards Earth is tangential from its apparent surface, not vertical. It is not a perfect demonstration because of a phenomenon called limb darkening, due to the Sun not having a definable surface, but actually plasma with opacity effects. However, it is generally not apparent to the eye and the normally observed (shielded) eyeball observation is arguably adequate for purpose here.
3) Whilst reportedly the original Stefan lab test was for a small flat body radiating into a hemisphere, its conclusions can be extended to larger areas by simple addition of many small flat bodies of collectively flat configuration, because of the ability of EMR waves to pass through each other. This can be demonstrated by car driving at night, when approaching headlights do not change in brightness as a consequence of your own headlights opposing them. (not to be confused with any dazzling effects and fringe illumination)
4) My sketch below demonstrates how radiation is at its greatest concentration in the lateral directions. It applies to both the initial S-B hemispherical surface radiation and to subsequent spherical radiation from the atmosphere itself.
5) Expanding on the text in Figure 3: Air temperature decreases with altitude, (with lapse rate), but if we take any thin layer of air over a small region, and time interval, and with little turbulence, the temperature in the layer can be treated as constant. Yet, the most concentrated radiation within the layer is horizontal in all directions, but with a net heat transfer of zero. Where the radiation is not perfectly horizontal, adjacent layers will provide interception of it.
A more concise way of looking at it is with vectors, which put simply is a mathematical method for analysing parameters that  possess directional information. Figure 4, takes a random ray of EMR (C) at a modestly shallow angle, and analyses its vertical and horizontal vector components. The length of each vector is proportional to the power of the ray, in that direction, such that A + B = C. Of course this figure is only in 2D, and there are countless multi-directional rays in 3D, with the majority approaching the horizontal, through 360 planar degrees, where the vertical components also approach zero.
possess directional information. Figure 4, takes a random ray of EMR (C) at a modestly shallow angle, and analyses its vertical and horizontal vector components. The length of each vector is proportional to the power of the ray, in that direction, such that A + B = C. Of course this figure is only in 2D, and there are countless multi-directional rays in 3D, with the majority approaching the horizontal, through 360 planar degrees, where the vertical components also approach zero.
6) Trenberth’s figure 1 gives that 65% of the HEAT loss from the surface is via thermals and evapo-transpiration. What is not elaborated is that as a consequence of this upward HEAT transfer, additional infrared radiation takes place in the air column by virtue of it being warmed. This initially starts as spherical emission and absorption, but as the air progressively thins upwards, absorption slows, and that radiation ultimately escapes directly to space. Thus, the infrared radiation observable from space has complex sources from various altitudes, but has no labels to say where it came from, making some of the attributions “difficult”.
DISCUSSION; So what to make of this?
The initial isotropic S-B surface emission, (Trenberth’s global 396 W/m2), would largely be absorbed by the greenhouse gases instantaneously near the surface. (ignoring some escaping directly to space through the so-called “atmospheric window”). However, a large proportion of the initial S-B 396 surface emission would be continuously lateral, at the Trenberth imposed constant conditions, without any heat transfer, and its horizontal vectors CANNOT be part of the alleged 396 vertical flux, because they are outside of the vertical field of view.
After the initial atmospheric absorptions, the S-B law, which applied initially to the surface, no longer applies to the air above. (although some clouds are sometimes considered to be not far-off from a black body). Most of the air’s initial absorption/emission is close to the surface, but the vertical distribution range is large, because of considerable variation in the photon free path lengths. These vary with many factors, a big one being the regional and more powerful GHG water vapour level range which varies globally between around ~0 to ~4%. (compared with CO2 at a somewhat constant ~0.04%). The total complexities in attempting to model/calculate what may be happening are huge and beyond the scope of this here, but the point is that every layer of air at ascending altitudes continuously possesses a great deal of lateral radiation that is partly driven by the S-B hemispherical 396, but cannot therefore be part of the vertical 396 claimed in Figure 1.
CONCLUSIONS:
The vertical radiative flux portrayed by Trenberth et al of 396 W/m^2 ascending from the surface to a high cloud level is not supported by first principle considerations. The S-B 396 W/m^2 is by definition isotropic as also is its ascending progeny, with always prevailing horizontal vector components that are not in the field of view of the vertical. The remaining vertical components of EMR from that source are thus less than 396 W/m^2.
It is apparent that HEAT loss from the surface via convective/evaporative processes must add to the real vertical EMR loss from the surface, and as observed from space. It may be that there is a resultant of similar order to 396 W/m^2, but that is NOT the S-B radiative process described by Trenberth.
~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~
ADDENDUM FOR AFICIONADOS
I Seek your advice
In figure 5 below, note that the NIMBUS 4 satellite data on the left must be for ALL sources of radiation as seen from space, in this case, at some point over the tropical Pacific. The total emissions, amount to the integrated area under the curve, which unfortunately is not given. However, for comparison purposes, a MODTRAN calculator, looking down from 100 Km gives some interesting information for the figure, which is further elaborated in the tables below. Unfortunately the calculator does not give global data or average cloud/sky conditions, so we have apples and pears to compare, not only with Nimbus, but also with Trenberth. However, they all seem to be of somewhat similar order, and see the additional tabulations.
| Compare MODTRAN & “Trenberth”, looking down from 2 altitudes, plus Surface Temperature | ||||
| Location | Kelvin | 10 metres | 100 Km. | (Centigrade) |
| Tropical Atmosphere | 300K | 419 W/m^2 | 288 W/m^2 | (27C) |
| Mid-latitude Summer | 294K | 391 W/m^2 | 280 W/m^2 | (21C) |
| Mid-latitude Winter | 272K | 291 W/m^2 | 228 W/m^2 | (-1C) |
| Sub-Arctic Winter | 257K | 235 W/m^2 | 196 W/m^2 | (-16C) |
| Trenberth Global | 288K ? | 396 W/m^2 | 239 W/m^2 | (15C ?) |
| Compare MODTRAN & “Trenberth”, looking UP from 4 altitudes: W/m^2 | ||||
| Location | From 10 m | From 2 Km | From 4Km | From 6Km |
| Tropical Atmosphere | 348 | 252 | 181 | 125 |
| Mid-latitude Summer | 310 | 232 | 168 | 118 |
| Mid-latitude Winter | 206 | 161 | 115 | 75 |
| Sub-Arctic Winter | 162 | 132 | 94 | 58 |
| Trenberth Global | 333 Shown as coming from high cloud area (= BS according to MODTRAN) | |||



Bob,
“Yes, of course RW. Is it a trick question?”
No, it’s not a trick question. Truthfully, I’m still not quite sure I understand your fundamental objection here, so I’m attempting to get clarification.
Yes, I agree you are correct that the 396 W/m^2 LW flux emitted at the surface is not all emitted perpendicular to the surface.
Yes, I agree you are correct that most of the absorption occurs close the surface and subsequent LW emission in the atmosphere from that absorbed would be isotropic.
What am I missing?
Bob says: “For argument’s sake, let’s contemplate that the mean photon free path length from the surface is 10m.”
For the sake of argument, let’s also assume the atmosphere in those first 10 m is the same temperature as the ground. Ie the two are in thermodynamic equilibrium. And to be specific, we are considering a 1m x 1m x 10m tall rectangular volume of air right above the ground.
Bob says: “such photons cease to exist long before they could get there at only around 10m away from source. ”
Yes I agree. But we had assumed that the ground and air were the same temperature. As photons flow from the surface to the first 10 m of air, there is a flow of energy. But for two objects at the same temperature, there can be no net flow of energy (because that would contradict the thermodynamic equilibrium). Therefore, there must be exactly as much energy flowing down from the first 10 m of air to the ground in the form of IR photons as there is flowing up into the first 10 m of air.
But we also know the radiation from the air is isotropic. So the energy flowing up out of the top 1m x 1m is the same as that flowing down from the bottom 1m x 1m.
So the ground emits photons. Some of them are absorbed by the column of air. The column of air emits its own photons up thru the of the column that exactly matches the photons absorbed in the column of air. The spectrum up thru the top will be indistinguishable from the spectrum coming into the bottom of the column.
It is quite true that it is not the same photons 10 m up, but the energy and spectrum will be indistinguishable from the spectrum at the surface.
To disagree with this conclusion, you pretty much have to disagree either with radiation from the air being isotropic, or disagree with the definition of thermodynamic equilibrium.
Bob Fernley-Jones, I asked what you thought of those emails, if they disproved something from climate science and you didn’t state anything, so I assume you think they make certain scientists look bad/dishonest and perhaps invoke extra scrutiny over their other work. If you want to answer affirmative that more is at stake, go ahead.
Now, you may want to read this interpretation of those emails about hiding the decline and using the Nature trick: http://bbickmore.wordpress.com/2010/05/27/series-climate-conspiracy-theories-in-utah-part-1/ On first reading, you may want to skip to around the middle (where it first starts talking about “trick”).
Some of the main arguments there are (plus some other points I want to note):
(a) Current decades tree ring data appears not to represent temperature, and this is corroborated by satellite data. Experts have taken note of this as well.
(b) Further into the past tree ring data appears to do a good job and this is supported by other forms of temperature readings and proxy analysis. You may not want to accept tree ring data at all, but there is support for accepting it. The main divergence problems also appear to be in areas that have high urban populations suggesting acid rain or other causes might be affecting the tree growth of more recent years. Look at the graphs included there to see the effect plotted against satellite temp records.
(c) Mann removing this bad data makes sense and is usually considered good science. He seems to have reasons to believe older data might be accurate. In any case, it doesn’t suggest dishonesty as implied by many. [see next few points]
(d) The trick is likely as harmless as using known current temp data to either replace or compensate for the bad tree growth data that deviates more and more from the last few decades of temp readings. Additionally, the trick might include plotting as the main line (possibly showing error bars) a 50 year average with 25 of those years going into the future. This explains the reason the last 25 years prior to the publishing of the paper are not included — they couldn’t be by definition.
(e) The word “trick” has many innocent meanings and is used frequently as slang in technical fields by those who practice them. Surely, many students have referred to using the “math trick” or something related in their years of study to refer to various interesting results/processes/manipulation needed to solve math problems accurately. Surely, these math tricks don’t imply there is deception going on or that the math is anything but 100% sound.
(f) A formal review was done and found Mann clear on all the serious charges.
As I said before, I have reasons to believe (and will continue to do so for now) that most of this climate science is a sane attempt if not perfect. I am willing to hear arguments against it. Even the arguments just presented against Mann, the scientist, who does not represent climate science as a whole regardless, appear to come up far short of the apparent intentions by those leveraging the accusations. Those two links were opinion papers, I think with one expressing a belief data was removed. There was nothing but circumstantial evidence suggested (anchored in part by negative interpretations of the few words in perhaps a single email). In response, I provided a link to an alternative interpretation that actually draws upon a wider context and data set rather than merely guessing bad behavior. The alternative view draws a very different picture (a positive one) and not simply a slightly less negative one.
I asked you for help in identifying links because I cannot tell what you think is a good argument. I don’t think those two links were. If you have more, you can provide them, and I will try and read them (or at least skim). [I came across one link, for example, saying that random time series gave the same “hockey stick” effect. I would have to look closer at that. Do you want to make that your next link?]
RW @ur momisugly December 1, at 6:27 pm
I don’t know if I can explain it better than I did in the article, but perhaps the following comment from above may help:
http://wattsupwiththat.com/2011/10/26/does-the-trenberth-et-al-%e2%80%9cearth%e2%80%99s-energy-budget-diagram%e2%80%9d-contain-a-paradox/#comment-814677
Jose says: “Note, I did not clarify that by “massless” I meant zero rest mass only. E=mc^2 implies all energized particles have mass.”
Tim says: “Actually, you were closer to correct originally. Photons have no mass! I challenge anyone to find any authoritative reference that says photons have mass!
The full equation is not E = mc^2, but rather E^2 = (m^2 c^4) + (p^2 c^2). For particles with mass at rest, it becomes E = mc^2. For particles with no mass moving at the speed of light, it becomes E = pc. ”
Ok, if as you claim ρ²c² is massless (the kinetic term) then the rho (momentum) is zero and therefore energy is always zero. Is this really what you are saying Tim?
I think Jose_X was close here but should have used E=ρ²c² in his statement. If photons are not at rest and massless they have momentum therefore they have mass.
Oh Tim, you wanted a reference of a book… try “Gravity” (Misner,Thorne,Wheeler) p.580 on “Photon Reinterpretation of Geometric Optics” stating “Photons are particles of zero rest mass …” conversely implying “Photons are particles of non-zero mass when not at rest” or better non-zero momentum (four-momentum) demands non-zero mass.
Tim Folkerts @ur momisugly December 1, at 6:47 pm
Sorry Tim but you must have been a bit hasty or perhaps over-imbibed with some good vino; let me guess; Cabernet-Merlot?
Why? Does it ever happen over the ground? (OR at sea?)
• No, according to the great prophet Trenberth, the surface (land and sea) is a black body. (emissivity =1). However, the atmosphere is not even a body, let alone a black body. The back-radiation is not a full Plancky, but follows the emission lines/bands of the emissive gases. (probably within the embrace of a partly definitive blackbody Plancky). The energy output is also reduced.
• Also, according to Trenberth, the surface emission is 396, whereas the backradiation is 333. According to you, previously, the 333 is from the total atmosphere above, whereas you now seem to be claiming that 396 is backradiated to the surface from a lesser source only 10m high. If that were the case, there would be no net heat transfer upwards.
• You have also cited MODTRAN as being definitive. Check-out my table 2 under Fig. 5, and perhaps elaborate more if you do not see a trend that is inconvenient to your claims therein
Tim, let me correct a typo before you surely catch it, that should have read E²=ρ²c², not E=ρ²c², but that’s par for me.☺
RW, further my recent post to you above, please also see the last two paragraphs in:
http://wattsupwiththat.com/2011/10/26/does-the-trenberth-et-al-%e2%80%9cearth%e2%80%99s-energy-budget-diagram%e2%80%9d-contain-a-paradox/#comment-813371
Wayne, I don’t have a copy of that book handy. Is it actually printed in the book that “Photons are particles of non-zero mass when not at rest”?
I would be surprised to see such an actual statement.
While the term “relativistic mass” has been used for “mass when not at rest”, the more typical current interpretation in special relativity follows more on the lines of wikipedia: “The term mass in special relativity usually refers to the rest mass of the object, which is the Newtonian mass as measured by an observer moving along with the object. The invariant mass is another name for the rest mass”
I was a little surprised to learn that in general relativity, the term “mass” cannot always be uniquely defined!
TIM>>For the sake of argument, let’s also assume the atmosphere in those
TIM>>first 10 m is the same temperature as the ground.
BOB>Why? Does it ever happen over the ground? (OR at sea?)
Clearly the air is sometimes warmer than the ground, and sometimes cooler. It is pretty clear that sometimes it must be the same temperature. My assumption was made to simplify the discussion so there was one less variable floating around.
TIM>> The spectrum up thru the top will be indistinguishable from the
TIM>> spectrum coming into the bottom of the column.
BOB>No … The energy output is also reduced.
Sorry, but I have to object to your objection. First, note that I am talking about the energy UP out of the top of the 10 m column (analogous to the 396 W/m^2), not DOWN into the column (analogous to the 333 W/m^2).
You seem to be suggesting that because the ground is a BB (and hence a good radiator) and the air column is not a black body (and hence a poor radiator), that the ground will send more energy into the air column than the air column sends into the ground — even when they are the same temperature (call it T(1)).
But then then there must be some temperature T(2) > T(1) that I could warm the air to such that there was no net energy flow.
But then there must be some temperature T(3) between T(1) and T(2) where there is still a net flow from the ground to the air.
Alas, we have now violated the 2nd law of thermodynamics –> there is a net flow of heat from the cooler ground (T(1)), to the warmer air (T(3))
Therefore at T(1), there must be identical flows off energy (ie identical absorption of IR photons) from the air to the ground as from the ground to the air independent of their emissivities.
And since the IR radiation from the air is isotropic, the photons the 10m tall air column emits upward must exactly match the photons it emits downward, which we just established exactly match the photons absorbed from below.
~~~~~~~~~~~~~~~~~~~~~~~~
That said, there is indeed some drop in temperature as the altitude increases. In 10 m = 0.01 km, that would be about 0.06 K cooler at the top of the column (at a typical lapse rate of 6 K/km). This will lead to a slight drop in IR radiation from the top, but a quick estimate suggest that the intensity at the top of the 10 m tall column will still be at least 99.9% as strong as at the ground in the absorption bands (and 100% in the “windows”). Of course, as you keep going up, the air keeps getting cooler and the amount out thru the top will get a bigger and bigger “bite” taken out of it.
@Jose_X…
A good book to start on is Quantum Thermodynamics, by Jochen Gemmer and M. Michel, 2004.
@Tim O'Donovan Folkers…
The equation C = Cp (δ) (r) is to calculate the capacity of energy storage (C) of any thermodynamic system. Cp is heat capacity, (δ is density of the system, and r is radius of the system or length of a parcel to be analyzed. It is a very simple formula that has been systematically ignored.
The results of multiplying J/kg K by kg/m^3 by m gives those square meters you are asking for.
The following formula is to calculate the time a molecule of a given substance takes to have all its excited electrons in ground state:
t =√(ΔE*(1/((m^2 kg) )) ) = √(1.345 x 10^(-20) (kg m^2/s^2) * (1/((m^2 kg) )) ) = 1.16 x 10^(-10) s
The term 1/(m^2 kg) is a correction factor derived from moment of inertia. The original formula would be:
t =√(ΔE /(m^2 kg))
In this case moment of inertia is taken as the unit 1 because inertial tensors are symmetrical.
Regarding the impossibility for a molecule of a given species to absorb the energy emitted by molecules of its own species resides in the fact of specificity of absorption spectral bands. The molecule of carbon dioxide emits quanta/waves with wavelengths pertaining to radio, such quanta/waves are not thermal radiation; consequently, they cannot be transferred as heat.
The unusable potential energy transformed in a molecule of carbon dioxide or any other molecules corresponds to sinks from where it cannot be taken to do work.
Something worth to be mentioned here is that the absorptivity of carbon dioxide at its stronger band of absorption (14 μm) is only 0.002, which is also its emissivity potential.
Jose_X, would you say that the wikipedia chart applies to quite or very moist air and that my 70% to 90% pertains too dry air?
‘Looking at this rough picture at the top section http://en.wikipedia.org/wiki/File:Atmospheric_Transmission.png, it looks like the area that is blue is much closer to 40% than to 70-90% range’.
Jose_X, my calculations came up with the following:
Total emissive power of CO2 bands = 79.8W/m^2
Emissive power entire spectrum @ur momisugly 15 deg = 392W/m^2
>> A good book to start on is Quantum Thermodynamics,
Thanks for the book reference Nasif, but I am interested in your argument here and I am very unlikely to buy that book now, especially if I can get the information from elsewhere or derive it with some help. I have loads of physics books I have hardly looked at from many years back. Where the physics may have changed (I doubt it), I have google and can read up on select papers or discussions that I might find online.
>> The equation C = Cp (δ) (r) is to calculate the capacity of energy storage
OK.
It’s not clear to me what 609.7 J/m^2 K represents or how you got it.
(a) Together with the 120 picoseconds remark (which I’ll currently assume is a reasonable approximation to the length of time CO2 holds an excited state — I’m sure this value can be looked up) and
(b) noting that the units are 1/m^2,
are you saying that you figured out how much each square meter column of atmosphere on earth has of CO2 and multiplied this by how much each such CO2 can store in extra heat (as Joules) for each Kelvin degree in temperature.. and you got that 609 value?
Am I close?
If I am close, then how did you leverage this information to deduce the effect a doubling of CO2 would have on surface temperatures?
I don’t see any reference to the nonlinear time-dependent partial differential equations that must be solved on our complex earth system in order to try to try to deduce future temperature values. [This is the sort of calculation I think is done by climate models using supercomputers if necessary to perform many iterations using boundary conditions of current and past temp and other recorded values.]
>> The following formula is to calculate the time a molecule of a given substance takes to have all its excited electrons in ground state:
OK, I have a few more questions on this.
I was hoping you’d say which equations/operators lead to that very simple expression. Generally, I’d think that computing the decay times for arbitrary molecules is not something easy to do.
Also, what does delta E represent if you have multiple electrons in various excited states?
And does this equation consider LTE effects among other gas molecules? We don’t just have to deal with what CO2 can hold. CO2 will interact with other gas molecules. I heard (as mentioned already) that LTE time constants are much shorter than decay time constants so the CO2 serves as a conduit for imparting kinetic energy onto N2 and other gases.
And as mentioned above with the differential equations remark, it’s not just how much energy CO2 can hold or share with other gas molecules because excited CO2 and other ghg molecules do decay, with some of the energy going back down in the direction of the earth+oceans (which have a lot more mass). If possible, we should try to leverage as much of this information as possible to compute accurate future values.
[I think anyone wanting to check up on the predictions of these models should be taking a serious look at open source computer models, eg, GISS’ model E. We could use every extra eyeball there to make sure the models aren’t accidentally using sophisticated curve fitting that actually produces a result consistent with the past but also with pre-desired future value ranges. I think peer review probably has this covered, but a skeptical individual should really consider this path.]
Regardless of this equation, t=sqrt(delta E), or any other, I think these values have been measured and/or derived by many people already.
So my main question is still, how are you using this result to prove your claims? ..especially if it appears you are not using (generally, time-dependent) PDEs. The earth is a very complex system. We need heavy-duty calculations to get answers.
>> The molecule of carbon dioxide emits quanta/waves with wavelengths pertaining to radio,
Can you offer calculations to support this or experiments? I think much of the physics scientific community has concluded this is not an accurate statement.
>> Something worth to be mentioned here is that the absorptivity of carbon dioxide at its stronger band of absorption (14 μm) is only 0.002, which is also its emissivity potential.
I think we covered this above and Tim Folkerts presented an old paper by Hottel showing calculated tables dependent on distance-atmospheres. If we wanted to take that path to calculating some values for CO2, we need to know how much CO2 we are talking about. A huge tank of CO2 absorbs more than a little glass. The whole atmosphere absorbs much more still.
OK, I look forward to corrections and clarifications by anyone (Nasif or anyone else). This is a learning opportunity for me.
And no, I haven’t looked at the details of the climate computer models. Again, I think anyone seriously wanting to challenge details of current accepted theories needs to look at those carefully to make sure the wider community is not overlooking things and perhaps even doing elaborate curve fitting.
Robert S, again, it’s not a big deal here IMO since we aren’t doing much with the value anyway, but I was replying to what you said specifically:
[Robert Stevenson]>> This range is called ‘open radiation window’, because it is here that the least amount of absorption by water vapour and CO2 takes place. This window allows 70 to 90% of the radiation from Earth to escape into space. It includes the peak emission.
Maybe you got confused, but your calculation of CO2, which very well may be very accurate, is not the calculation we need to address the question of radiation leaving into space unimpeded. We would need to calculate similar figures for water vapor (which you even mentioned by name) and other ghgs.
The wikipedia link I think was over the total absorbed and passed through. I didn’t do any calculations, but presumably others have (or have good measurements). I think that picture addressed your question to cover “all” ghgs. That was what you seemed to be “asking” literally, even if perhaps half of your mind was thinking only of CO2 coverage (as CO2 is the main discussion gas).
This article is related to a link offered recently. It’s a fresh guest write-up by B Bickmore.
http://bycommonconsent.com/2011/12/02/global-warming-and-the-mobs
Nasif,
I am still confused about some things you say, and I disagree with others.
>absorptivity of carbon dioxide at its stronger band of absorption (14 μm) is only 0.002
Of course, absorption depends on the amount of CO2 involved. I would believe 0.2% gets absorbed in a few cm or so of CO2 in the air. Bob suggests that close to 100% is absorbed in the absorption bands within 10 m. I suggested a number more like 100 m earlier in the thread. But it is meaningless to give a number for absorption without a reference to distance & concentration.
>The molecule of carbon dioxide emits quanta/waves with wavelengths pertaining to radio …
CO2 emits strongly at ~15 um, as well as ~4 um and 2.7 um. These are definitely IR photons, not radio photons. The emission of thermal radio waves by CO2 would be insignificantly small.
>… such quanta/waves are not thermal radiation
Whether or not it is “thermal radiation” depends on the source, not the wavelength. The cosmic microwave background is thermal radiation. Microwave ovens are not thermal radiation. Visible light from an incandescent bulb is thermal; visible light from a fluorescent bulb in not thermal.
>C = Cp (δ) (r)
>Cp is heat capacity, δ is density of the system, and r is radius of the system
>For atmospheric carbon dioxide in current Earth’s atmosphere, the capacity
>of carbon dioxide to store energy during 120 picoseconds
>(1.2 × 10 ^ (-10) seconds) is 609.7 J/m^2 K.
This still greatly confuses me. I *think* you were using this equation to find the “capacity to store energy” for CO2. What values of Cp, δ, and r did you use for CO2 to calculate 609.7 J/m^2 K?
>The original formula would be: t =√(ΔE /(m^2 kg))
That doesn’t even have proper units, so it cannot possibly be correct!
Energy is in [J] = [kg*m^2/s^2]. If we divide by [kg*m^2] we get s^(-2). After taking the square root, this might be an equation for 1/t, but it is definitely not an equation for t.
Tim Folkerts,
Whether or not it is “thermal radiation” depends on the source, not the wavelength.
I think Nasif Nahle was referring to the absorption spectrum for CO2 and how that was not radio waves (which allegedly would be emitted by other CO2).
I think this is just another case of using words that may mean different things to different people at different times but generally conveys the right idea. I’d guess “thermal” meaning radiation overlapping significantly in the “short” IR range such as were CO2 absorbs.
>> After taking the square root, this might be an equation for 1/t, but it is definitely not an equation for t.
I didn’t look carefully, but yes it does appear that eqn is 1/s. Maybe wordpress blogging messed up his submission. Maybe the “1” in the numerator (of the correction factor that has moment of inertia term in the denominator) comes with units that were left out for one reason or another.
I forgot to quote above.
It’s:
****
>> Whether or not it is “thermal radiation” depends on the source, not the wavelength.
I think Nasif Nahle was referring to the absorption spectrum …
****
Also, I haven’t checked any calculations. I presume they can be done in time or looked up as necessary (especially since I don’t think the values were used).
Ah, I forgot to say one more thing [Nasif]
Looking around a bit with google, I get the impression that perhaps decay rates for excited molecules tend to be exponential, so I think that unlike the doubt expressed earlier, a simple calculation (whatever be the formula) may be reasonable.
@Tim O'Donovan Folkers…
“But it is meaningless to give a number for absorption without a reference to distance & concentration.”
Atmospheric carbon dioxide current partial pressure of 0.00038 atm and a column of length 7000 m.
“CO2 emits strongly at ~15 um, as well as ~4 um and 2.7 um. These are definitely IR photons, not radio photons. The emission of thermal radio waves by CO2 would be insignificantly small.”
The carbon dioxide molecule emits quantum/waves with energy E = 5.4 x 10^(-26) J, which corresponds to wavelength (λ) = 3.75 m. those are radio waves, not thermal radiation. An excited molecule of carbon dioxide has not available microstates, so it cannot absorb more energy; consequently, it only is able to emit quanta/waves with longer wavelength and lower frequency:
Serway, Raymond A., Moses, Clement J., Moyer, Curt A. Modern Physics-3rd Edition. Brooks Cole. 2005.
“Whether or not it is “thermal radiation” depends on the source, not the wavelength. The cosmic microwave background is thermal radiation. Microwave ovens are not thermal radiation. Visible light from an incandescent bulb is thermal; visible light from a fluorescent bulb in not thermal.
Thermal Radiation is the fraction of the electromagnetic spectrum between wavelengths 0.1 μm and 100 μm.
Thermal Radiation includes visible spectrum, almost the whole ultraviolet spectrum (Vacuum UV, Far UV, C-UV, Middle UV, B-UV, Near UV, and A-UV. Low UV is excluded), and almost the whole infrared (IR) spectrum, except the portion of the spectrum corresponding to wavelengths of Far-IR, from 101 μm to 1000 μm:
Pitts, Donald and Sissom, Leighton. Heat Transfer. 1998. McGraw-Hill. Pp. 289-311.
“This still greatly confuses me. I *think* you were using this equation to find the “capacity to store energy” for CO2. What values of Cp, δ, and r did you use for CO2 to calculate 609.7 J/m^2 K?”
The values found at 300 K, p of 1 atm, 1000 m and density 0.00069 kg/m^3.
It is a very known and simple formula applied in heat transfer science, everytime and everywhere; however, it has been strangely dismissed by most authors. Liquid water is very efficient on storing energy. Let’s take 10 m depth of clean water:
C = 4181.3 J / kg K * 998.2 kg m ^3 * 10 m =
41,737,736.6 J/m^2 K
And C of water vapor at 373.15 K, in a proportion of 3%, and a length of 10 m is 917.7 J/m^2 K
At height of 1000 m, for comparison with atmospheric carbon dioxide, the capacityof water vapor to store energy is 91,774.5 J/m^2 K.
Those small simple things forgotten by biased science… 😉
“>The original formula would be: t =√(ΔE /(m^2 kg))
That doesn’t even have proper units, so it cannot possibly be correct!”
Units of moment of inertia are m^2 kg.
Nasif,
>absorptivity of carbon dioxide at its stronger band of absorption (14 μm) is only 0.002 ..
> (at partial pressure of 0.00038 atm and a column of length 7000 m)
Check this reference page 144: http://dspace.mit.edu/handle/1721.1/42950
For 0.00038 atm and a column of length 7000 m = 700,000 cm, ie 266 atm*cm, we are off the top of the chart and the emissivity is above 0.2, so 20% of ALL wavelengths are absorbed. Very close to 100% is absorbed in the CO2 absorption bands. The chart might be off slightly, but your number (0.002) is simply WAY off from any possibly correct value (~1).
>Thermal Radiation is the fraction of the electromagnetic spectrum
>between wavelengths 0.1 μm and 100 μm.
I have seen “thermal IR” defined as ~ 3-15 um. But I have never seen “0.1 – 100 um” called “thermal radiation”. A more typical definition is
“Thermal radiation is electromagnetic radiation generated by the thermal motion of charged particles in matter. All matter with a temperature greater than absolute zero emits thermal radiation.” wikipedia
That is one source — Google gives many similar definitions. Thermal radiation is usually considered any radiation due to thermal energy on the early Google hits..
>C = Cp (δ) (r)
Thanks for explaining. The equation makes sense. Mostly. With somewhat odd notation.
Let Cp = specific heat capacity (at constant pressure) = (heat capacity / mass). Density is (δ) (although I usually see density as “ρ” and specific heat capacity with a lower case “c”)
.
The heat capacity, which I will call C* is then C* = Cp * m. But m = δ * V = δ * (l*w*h) = δ * (A * h) , so C* = Cp * δ * (A * h), or finally
C = (C* / A) = Cp * δ * (h)
>it has been strangely dismissed by most authors
I would instead say that most authors choose to write the equation in the more transparent form
(heat capacity) = (specific heat capacity) * (mass)
C = c * m
rather than your version which is
(heat capacity per unit area) = (specific heat) * (density) * (height)
(C/A) = c * ρ * h
>Those small simple things forgotten by biased science… 😉
I really don’t think science of any stripe has forgotten the meaning or importance of “heat capacity”. I think science was just confused by your notation.
>Units of moment of inertia are m^2 kg.
You missed the point. The left side of your equation has units of seconds. The right side is 1/seconds. There is no way such an equation can be correct.
Bob,
“Clearly, the 396, by definition, comes from the surface, but does not make-it to space. (or even to the high clouds as shown in the cartoon). The 396 and its progeny of new-generation emissions contain horizontal vector components which cannot be seen in the normal view. Thus, the vertical components must be less than the total S-B emission.”
I agree the full 396 W/m^2 does not make it to the high clouds, and in this sense Trenberth’s depiction is not accurate. But all of the 396 W/m^2 of S-B emission in the LW IR has an upward component to its direction, some of which is more horizontally or diagonally upward but it’s upward toward and in the direction of space none the less.
This is really not the biggest ‘problem’ with Trenberth’s diagram. Most importantly his depiction does not show gross net energy flux in any discernable way, and he incorrectly mixes non-radiative and radiative energy fluxes in a way that doesn’t account for the non-radiative energy returned to the surface as the temperature component of precipitation, wind, weather, etc.
RW @ur momisugly December 2, at 9:29 pm
Thanks for your continued rational interest RW, and concerning your comments on part of what I’ve said.
So why does the 396 not make it up there?
Let’s approach this from a different direction, whilst simplistically ignoring the interacting thermals and evapotranspiration and stuff:
If the atmosphere were transparent, we would NOT be holding this discussion. In that imaginary situation, all the isotropic components of surface radiation would make it to space, and thus with a wide angle sensor in space, the 396 would be seen as if it were a vertical total. However, because we have an absorbent atmosphere, the critical wavelength photons from the surface are annihilated in quite short distances. When they are annihilated, they can no longer be observed.
See particularly my item 4 including Fig 3 in the article. Do you disagree that most radiation is sideways? Can you not see that the critical wavelengths are annihilated before escape to space is possible? Then please note that the horizontal vector components are persistently there as part of the isotropic emissions. (They do not go away). Are you hypothesising that when those spawning photons from the surface are absorbed that somehow their progeny after random collisional thermalization with N2 and O2 and whatnot are rotated through 90 degrees to the vertical?
@Tim O'Donovan Folkerts…
“For 0.00038 atm and a column of length 7000 m = 700,000 cm, ie 266 atm*cm we are off the top of the chart and the emissivity is above 0.2, so 20% of ALL wavelengths are absorbed. Very close to 100% is absorbed in the CO2 absorption bands. The chart might be off slightly, but your number (0.002) is simply WAY off from any possibly correct value (~1).”
Calculate it by yourself. The formula is as follows:
Ɛcd = [1 – (((a-1 * 1 –PE)/(a + b – (1 + PE)) * e^(-c (Log10 ((paL)m / paL)^2))] * (Ɛcd)0
A very important procedure concerns to the term e^(-c (Log10 ((paL)m / paL)^2))] and the term (Ɛcd)0.
I have the articles by Hottel, Sarofim, Leckner and Lapp and used them as references.
We cannot simply multiply partial pressure by length because it gives erroneous results, like those 266 atm*cm that you obtained as the partial pressure of the gas which is 266 times the total atmospheric pressure at sea level.
The correct procedure is to obtain the pressure that carbon dioxide would exert if it was alone occupying the whole volume of the Earth’s atmosphere; once you have obtained the pressure of the gas in each meter, you must convert atm-m to atm-cm. It is unphysical that the carbon dioxide could exhibits a pressure of 266 atm in each one of the centimeters of a column of air.
http://en.wikipedia.org/wiki/Partial_pressure
“That is one source — Google gives many similar definitions. Thermal radiation is usually considered any radiation due to thermal energy on the early Google hits..”
From Wikipedia:
“Thermal radiation is electromagnetic radiation generated by the thermal motion of charged particles in matter. All matter with a temperature greater than absolute zero emits thermal radiation.
Examples of thermal radiation include visible light emitted by an incandescent light bulb, infrared radiation emitted by animals and detectable with an infrared camera, and the cosmic microwave background radiation. Thermal radiation is different from thermal convection and thermal conduction–a person near a raging bonfire feels radiant heating from the fire, even if the surrounding air is very cold.
Source:
http://en.wikipedia.org/wiki/Thermal_radiation
Googled:
http://casswww.ucsd.edu/archive/public/tutorial/Planck.html
http://www.ptbmagazine.com/features/2009/feat1_0909.html (please, notice the big arrow which says “Thermal Radiation”. It is similar to graphs you can find in books of heat transfer.
https://docs.google.com/viewer?a=v&q=cache:J4yUwp8bBH8J:www.handbook.ifrf.net/handbook/dl.html/index.pdf?id%3D67%26type%3Dpdf+&hl=en&pid=bl&srcid=ADGEEShC7qK-yhnK8gb-xxiChSPGtN9ZnlypaeSCnvOkSX_U2Z8MgTLtLPq86vbtR-Raj3PLFAPqBnNKnoe22xOCJHZD6bTpgbO2Ck5RqUDTj2YpKSDLOOk5qeA0AmXduf5Z0LjtxUDg&sig=AHIEtbRWjWyjBUPMDrvCSNVwgOFDkMl-wA&pli=1
Etcetera, etcetera…
“rather than your version which is (heat capacity per unit area) = (specific heat) * (density) * (height) (C/A) = c * ρ * h”
Not my version… You can find it almost in all books of heat transfer.
“I think science was just confused by your notation.
I will dismiss this argument. It’s off topic and unproductive regarding science.
“You missed the point. The left side of your equation has units of seconds. The right side is 1/seconds. There is no way such an equation can be correct.”
I thought the same when I read the formula from two books of Modern Physics, but the formula was written exactly as I wrote it. Perhaps a (s^4) was dropped by the authors.