Guest post by Willis Eschenbach
There is a lot of misinformation floating around the web about how the greenhouse effect works. It is variously described as a “blanket” that keeps the Earth warm, a “mirror” that reflects part of the heat back to Earth, or “a pane of glass” that somehow keeps energy from escaping. It is none of these things.
A planetary “greenhouse” is a curiosity, a trick of nature. It works solely because although a sphere only has one side, a shell has two sides. The trick has nothing to do with greenhouse gases. It does not require an atmosphere. In fact, a planetary greenhouse can be built entirely of steel. A thought experiment shows how a steel greenhouse would work.

Before we start, however, a digression regarding temperature. The radiation emitted by a blackbody varies with the fourth power of the temperature. As a result, for a blackbody, we can measure the temperature in units of radiation, which are watts per square meter (W/m2). For objects with temperatures found on the Earth, this radiation is in the range called “longwave” or “infrared” radiation. See the Appendix for the formula that relates temperature to radiation.
This means that we can denote the temperature of a blackbody using W/m2 as well as the traditional measures (Fahrenheit, Celsius, Kelvin). The advantage is that while temperature (degrees) is not conserved, energy (W/m2) is conserved. So we can check to see if energy lost is equal to energy gained, since energy is neither being created nor destroyed by the climate.
For our thought experiment, imagine a planet the size of the Earth, a perfect blackbody, heated from the interior at 235 watts per square meter of surface area. How is it heated from the interior? Doesn’t matter, we’ll say “radioactive elements”, that sounds scientific.
The planet is in interstellar space, with no atmosphere and no nearby stars. The equilibrium surface temperature of this planet is, of course, 235 W/m2. To maintain the equilibrium temperature, it constantly radiates this amount of energy out to space. Coincidentally, this is the amount of solar radiation that makes it past the clouds to warm the Earth. If we convert 235 W/m2 to one of our normal temperature scales, it is -19 Celsius (C), or -3° Fahrenheit (F), or 254 Kelvins (K). It’s a possible temperature that the Earth might have if there were no greenhouse effect. That is to say … cold.
Now imagine that the planet gets completely surrounded by a thin black steel shell, located a few thousand meters above the surface, as shown in a cutaway view in the picture above, and in Figure 1 below. What happens to the surface temperature of the planet? (To simplify calculations, we can assume the shell has the same outer surface area as the surface. For an earth-sized planet with a shell two kilometers above the surface everywhere, the difference in area is only six-hundredths of one percent. This assumption makes no difference to the argument presented.)
In order to maintain its thermal equilibrium, including the new shell, the whole system must still radiate 235 W/m2 out to space. To do this, the steel shell must warm until it is radiating at 235 watts per square meter. Of course, since a shell has an inside and an outside, it will also radiate 235 watts inward to the planet. The planet is now being heated by 235 W/m2 of energy from the interior, and 235 W/m2 from the shell. This will warm the planetary surface until it reaches a temperature where it radiates at 470 watts per square meter. In vacuum conditions as described, this would be a perfect greenhouse, with no losses of any kind. Figure 1 shows how it works.

Figure 1. Building a steel greenhouse. (A) Planet without greenhouse. Surface temperature is 235 W/m2 heated from the interior. (B) Planet surrounded by steel greenhouse shell. Shell radiates the same amount to space as the planet without the shell, 235 W/m2. It radiates the same inward, which warms the planet to 470 W/m2 (29 C, 83°F, 302 K). [Clarification added] Note that the distance from the shell to the planet is greatly exaggerated. In reality, it is close enough to the planet that the difference in the areas can be neglected in practice.
The trick can be repeated again, by surrounding the planet and the shell with a second outer shell. In this two shell case, the planetary surface temperature (in W/m2) will be three times the initial surface temperature.
In nature, planets have atmospheric shells, of course, rather than steel shells. The trick works the same way, however. Rather than being warmed from the inside, the Earth receives 235 W/m2 from the sun. Solar radiation passes through the atmosphere. Outgoing longwave radiation is absorbed by the atmosphere, just as it is absorbed by the steel shell shown in Fig. 1.
So that’s the trick of the greenhouse. It has nothing to do with blankets, mirrors, or greenhouse gases. It works even when it is built out of steel. It depends on the fact that a shell radiates in two directions, inwards and outwards. This radiation keeps the planet warmer than it would be without the shell.
Now, it is tempting to think that we could model the Earth in this manner, as a sphere surrounded by a single shell. This is called a “two-layer” model, with the two layers being the surface and the atmospheric shell. In fact, a number of simplified climate models have been built in this way. Unnoticed by their programmers, however, is that is not physically possible to model the Earth as a two-layer system. Figure 2 shows why. Note that in all cases, the system has to remain in thermal equilibrium. This means that the surface must radiate as much as it absorbs, and the shell must also radiate as much as it absorbs. In addition, radiation from the shell to space (upwelling longwave radiation or ULR) must equal radiation from the shell to the Earth (downwelling longwave radiation, or DLR)
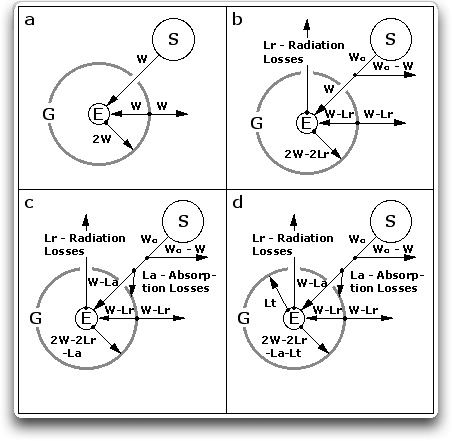
Figure 2. Single-shell (“two-layer”) greenhouse system, including various losses. S is the sun, E is the Earth, and G is the atmospheric greenhouse shell around the Earth. The height of the shell is greatly exaggerated; in reality the shell is so close to the Earth that they have about the same area, and thus the small difference in area can be neglected. Fig. 2(a) shows a perfect greenhouse. W is the total watts/m2 available to the greenhouse system after albedo. Fig. 2(b) is the same as Fig. 2(a) plus radiation losses Lr which pass through the atmosphere. Fig. 2(c) is the same as Fig. 2(b), plus the effect of absorption losses La. Fig. 2(d) is the same as Fig. 2(c), plus the effect of thermal losses Lt.
Figure 2(a) shows the same situation as Figure 1(B), which is a perfect planetary greenhouse. In this case, however, it is heated by an amount of energy “W”, which is coming from the sun. The planet receives solar radiation in the amount of “W” from the sun, and longwave radiation “W” from the atmospheric shell. The surface temperature is thus 2W. All energy flows are in Watts/square metre (W/m2).
Figure 2(b) adds two losses. The first is the reflection from the Earth’s albedo (Wo – W). This is energy which never enters the system and is reflected back into space. We are still getting the same energy entering the system (W). The second loss Lr is from radiation which goes from the surface to space through the “atmospheric window”. Because of the second loss, the surface of the Earth does not get as warm as in a perfect system. In a perfect system, the temperature of the surface is 2W. But including the radiation loss Lr, the surface temperature drops to 2W – 2Lr.
Figure 2(c) adds another loss La, which is the solar radiation which is absorbed by the shell. This cuts the radiation hitting the surface down to W – La. Including this loss, the surface temperature is 2W – 2Lr – La.
Figure 2(d) adds the final loss. This is Lt, the thermal loss. It is the sensible energy (energy you can feel) and latent energy (evaporation) which is transported from the surface to the shell by convection. Including all of these losses, the surface temperature of the Earth is 2W – 2Lr – La – Lt.
Now, why can’t the Earth be modeled in this manner? A look at the size of the various losses shows why. Here is the canonical global energy budget, called the “Kiehl/Trenberth” budget, or the K/T budget.
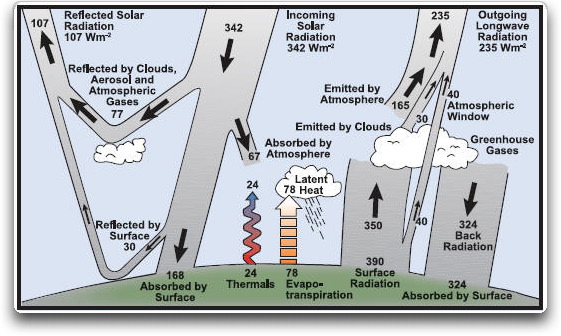
Figure 3. The Kiehl/Trenberth Global Energy Budget. This is a “two layer” representation, with the surface and the atmosphere being the two layers. Lr, the radiation loss, is the 40 W/m2 labeled “Atmospheric Window”. La, the absorption loss, is the 67 W/m2 labelled “Absorbed by Atmosphere”. Lt, the thermal loss, is 102 W/m2. This is the sum of the 24 W/m2 labelled “Thermals” and the 78 W/m2 labelled “Evapo-transpiration”. W, the energy from the sun, is the incoming solar radiation of 342 W/m2 less the 107 W/m2 that is reflected by the surface and the clouds. This means that W is 235 W/m2. SOURCE
What’s wrong with this picture? Note that the temperature of the Earth is 390 W/m2, labeled as “Surface Radiation”. This is 15 C, or 59°F. But from Fig. 2(d), we know that the surface temperature of a greenhouse system with a single shell, as shown in the drawing, is 2W (470) – 2Lr (80) – La (67) – Lt (102) = 221 W/m2. This is far below the known surface temperature of the Earth. In other words, a single shell greenhouse system simply isn’t efficient enough to give a surface temperature which is warm enough to allow for the known losses.
So where is the problem with the K/T budget diagram? The hidden fault is that the upward radiation from the atmospheric layer does not equal the downward radiation. There is 195 W/m2 going to space from the atmospheric shell, and 324 W/m2 going down to the surface.
In order to get enough energy to allow for the known losses, the simplest model requires two atmospheric shells. A perfect greenhouse with two shells would give a surface temperature of 3W, or 705 W/m2. This is enough to allow for the known losses, and still give a surface temperature which matches that of the Earth. Figure 4 shows one such possible system.
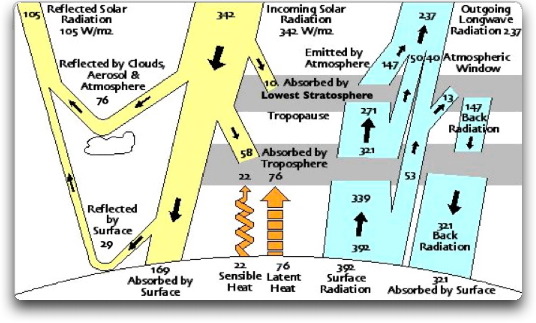
Figure 4. Simplest greenhouse global energy budget capable of representing the Earth. Note that all major flows in the K/T energy budget have been maintained. There are two shells, which must be physically separate. These are the lower troposphere and the lowest stratosphere. They are separated by the tropopause.
This budget fulfills all of the requirements for thermal equilibrium. The same amount is radiated upwards and downwards by each layer. The amount absorbed by each part of the system equals the amount radiated. Further verification of this energy budget is the 147 W/m2 emission from just above the tropopause. This converts to a Celsius temperature of about -50 C, which is a typical temperature for the lowest part of the stratosphere.
I have written a simplified Excel radiation/evaporation/convection model which encapsulates the above system. It is available here. Click on the “File” menu on the webpage and select “Download”.
I invite people to play with the model. It is what I term a “Tinkertoy” model, which is what I call the simplest possible model that can represent a particular reality. One of the interesting results from the model is that there can be very little thermal loss between the inner and the outer atmospheric layers. If there is more than a trivial amount of leakage, the surface cools significantly. Among the other insights yielded by the model is that a change equivalent to a doubling of CO2 (an increase of 3.7 W/m2 downwelling radiation at the top of the atmosphere) can be canceled by a 1% increase in the upper and lower cloud reflections.
Experiment with the values, it is an interesting insight into the energy flows in the simplest possible climate model that can represent the Earth’s greenhouse system.
APPENDIX
The formula that relates the temperature to radiation is called the “Stefan-Bolzmann” equation:
R = sigma * epsilon * T^4
where r = radiation (W/m2)
sigma = the Stefan-Boltzmann constant, 5.67 * 10^-8
epsilon = the emissivity of the body, which for a blackbody = 1
T^4 = absolute temperature in Kelvins (which is Celsius plus 273.15) raised to the fourth power
yonason (18:55:21) :
You aren’t the first person to bring this up; it’s been addressed above. Willis was lazy and neglected the difference in surface area, just so he could keep using fluxes. Not a big deal. Assuming the shell is close to the earth, the error is small. If you really want, redo the math with a different radius. Keep in mind that this cartoon works no matter how close the shell is to the earth. So put the shell 1 centimeter above the earth’s surface. The difference in area is trivial.
Joel Shore,
All recent measurements of ocean heat content show a rate of increase of about half of Hansen’s 0.85 W/m2 at worst and probably more like 0.25 W/m2 over the last two decades. Since 2003, OHC has been increasing even more slowly, possibly not at all. Maybe there’s some loss to the deep ocean, but the usual estimate of the proportion of heat transfer I’ve seen is ~90% to the upper ocean. Hansen’s estimate has not been upheld by measurement.
Mr. Eschenbach,
I have a question but I think it “might” be related in the explaination of “different energies”. I state different in that there’s “sensible temperature” and “radiant temperature”. I’m going to use my Wood/Coal addon in my House as an example.
I burn wood mainly because I don’t have to pay for it. I love wood heat because it’s a “Bone Warming” heat and I was raised with it on the Farm. I hope you’ve heard that phrase before. Now should I decide to go buy some coal and “mix” it or use it by itself, the feel of the “Bone Warming” is different, it actually changes in the “feel” of the Heat in my living space and of course around my Wood/Coal Stove. My question is “Why does it feel different? Is is because of different IR emitting against the Heat Exchanger? I think this might be the case because of
two “different” types of fuel.
I’ve been a HVAC Service Technician for over 15 years and when I walk into a Home in winter, I will know with most certain accuracy what kind of Heat is being produced.
You input would be most kind if you would. I think it would also explain the differences in “energy” being used in the Complex Climate Change System that we all endure.
Regards,
Denny
Willis Eschenbach says:
Take a look at the link when things get back, I can’t access it either at the moment. From memory, Hansen et. al used a ten year period during which the model was a close fit to the ocean temperature observations to make his claims. However, I took a look at the earlier ocean temperature observations from the same source he used, and the model Hansen used does a very poor job of replicating those. It is out on the order of ±6 W/m2. Given this size of error, both the size and the precision of his claimed imbalance (0.85 W/m2) are simply not credible.
Always good to hear from you,
w.
Denny (19:50:35) :
Radiation is emitted by everything. The only difference in the radiation is the frequency/wavelength, which changes with the temperature. Hot steel glows red, but if your hand was that hot it would glow the same color red as well …
So there is no difference in the type of radiation. But there may be a difference in how much energy is emitted as radiation versus hot gas. This is because as wood burns, typically there are a lot of flames. Also, the wood ash tends to flake off and expose new surfaces, that burst into flame. Coal, on the other hand, tends to end up with a shell of ash and clinker surrounding it, and with much less visible flame. So that’s a possible explanation.
My guess would be, though, that your sense of smell is what tips you off. Houses heated by coal have a distinctive smell to me, which I can smell even if the stove is not lit.
That’s my guesses, for what they’re worth.
yonason (18:55:21) :
Indeed it is confusing. yonason, again I can’t get to whatever is the nature of your complaint, or the source of your misunderstanding. Let me suggest again that you look at Fig. 1 and ask some pointed, short questions that show exactly what you find wrong about that Figure.
Willis Eschenbach (23:47:52) :
par5 (21:22:15) :
Willis- with this reverse greenhouse (heat from within) will there still be changes in the atmosphere ie cool and warm phases? Will the steel green house provide a constant temp worldwide? Will there be polar ice caps?
Since my thought experiment has no atmosphere, none of them are possible.
Willis- If you raise the temp of a black body in space, with a core temp as you decribe, surrounded by a shell as you descibe, then the surface temp will rise high enough to melt and evaporate water. Even if no water is on your planet, there will still be outgassing. You will have an atmosphere- like it or not. The temp of your planet can not be determined without the gas tables, and knowing the mass and pressure. I concede your physics because I see your POV, and agree. Will you concede that your experiment must attain an atmosphere?
Denny 19:50:35
On the difference of “feel” of heat coming from coal or from wood. Coal burns at a much higher temperature than wood, therefore its radiation curve is different, with higher frequencies. This will be felt next to the stove burning the coal/wood.
Now for the house, it is not clear.
If it is heated air circulating then the same holds as above, the convected air has the spectrum of the air next to the stove.
If it is water radiators it must be a mind thing: because one has felt the difference next to the stove the mind assumes it carries out in the house. Else it is the smell difference between coal and wood, as Willis has suggested.
“In order to maintain its thermal equilibrium, the
whole system must still radiate 235 W/m2 out to space…”
– from paragraph just above fig.1
In other words, by definition, the steel shell is arbitrarily
assigned the same temperature as the earth?
No. The Earth rises to 470 W/m2, due to the ‘insulating’ (re-radiating) properties of having the shell around it – just as a naked flam will turn into an oven if you place a shall around it.
That is the whole point of the experiment. And, as I see it, the ultimate point of this exercise, is to demonstrate that a single layer system would not warm the real Earth sufficiently, and that we need two layers in the atmosphere.
The experiment is a VERY simple model to demonstrate this hypothesis by Willis. Whether he is right in his TWO LAYER system, I do not know, but the thought experiment to demonstrate this IS DESIGNED TO BE VERY SIMPLE AND CLEAR TO UNDERSTAND.
I fail to see why so many people are having problems with it. But it does demonstrate the problems we have in explaining scientific points to politicians, who have to decide where to place the nation’s money. It must be as easy to bamboozle politicians as it is to bamboozle the contributors to this blog. Hence the problem we have with AGW taking over the political world.
.
>>>At thermal equilibrium, (T1^4-T2^4) = 0.
>>There will be no net heat exchange, BY DEFINITION
>>not assertion.
Yes, NET heat exchange – but that does not mean that there is no heat exchange. Place two white-hot suns next to each other and there will be LOTS of heat exchange going on – but no net exchange.
.
A “thought experiment” is an experiment wherein we can specify conditions in order to simplify and understand a system. I specified that there is no atmosphere. Why? I don’t know. Perhaps the planet is made out of solid Cantobtainium with a radioactive core. In any case, 470 W/m2 (the eventual steady-state temperature of the planet with a shell) is 28C, which is far too cool to melt much of anything.
But in any case, no, there is no atmosphere.
Cantobtainium? Are you no longer patient? Thought experiments have no ramifications?
“The planet is in interstellar space, with no atmosphere and no nearby stars.”
I’m pretty sure this was before you built your shell, yes? Perhaps it doesn’t matter?
Willis you said :
“My apologies, Tom, but I find your writing impenetrable. Please clarify and simplify what your objections are, and I’ll be happy to answer.”
.
Fair enough even if I think that I have been too long only because I tried to be as simple as possible and to demonstrate every step of the argument mathematically .
My basic assumption is that I assume that you are familiar with the derivation of the SB law . If not , then you will benefit of looking it up .
So that means that the SB law is just a consequence of some MORE basic law and this more basic law is the Planck’s law .
OK , you’re with me ?
1)
Now the Planck’s law gives the spectral radiance I (units W/m²/sr/Hz) .
If you ask what power will be emitted by some surface into some solid angle you will integrate the Planck’s law for all frequencies and within some solid angle and obtain the SB law .
The particular case used here is the case where the Planck’s law is integrated for solid angles in a “half sphere” e.g the plane surface sees only a “half space” .
That means concretely that the SB constant is not an universal constant but is only valid for plane surfaces emitting in a half space . For instance a ponctual source or a double faced shell emit in the whole space and the SB law for them has an SB constant which is DOUBLE of the one you used .
I went farther by saying that the constant you are using is only valid for convex bodies . No problem for your model because you have indeed chosen convex bodies but it would be wrong to think that the SB constant you use is valid for any body . Especially bodies with cavities are tricky .
2)
W/m² (or irradiances) generally do NOT conserve .
For spheres they definitely don’t (they do for INFINITE parallel plans) .
The irradiance of your shell is P.r²/R² where P is the irradiance of the inner sphere . As P.r²/R² < P follows that :
a) the shell has a lower temperature than the inner sphere
b) the shell and the inner sphere are not in thermal equilibrium (e.g there is a net flux going from the inner sphere to the shell)
c) The difference of temperature between the shell and the inner sphere (so the net flux) increases as R increases .
.
I could have written only very shortly the following synthesis but then you could have rightly asked me where is the proof .
So consider that if you find what follows not clear , then what is above is the proof .
.
========================================
1) SB constant is not universal and depends on the solid angle over which the Planck's law is integrated .
.
2) The apparent paradox between the shell emitting "two times as much" as the inner sphere is due to the fact that the double faced shell has an SB constant which is 2 times the one of the inner sphere .
.
3) The shell and the inner sphere are not in a thermal equilibrium and have different temperatures .
.
4) W/m² (irradiances) do not conserve . The W/m² of the shell are r²/R² times smaller than the W/m² of the inner sphere .
========================================
.
I find your remark condescending (cantobtainium) because of my chemistry background. I conceded your point on the physics, yet you refuse to see the ramifications of your own thought experiment. I’m calling Shenanigans.
lgl (10:37:03)
it still makes little sense. if 256 re-radiate back to the source and 256 exits the barrier, its still 256 one way and 256 the other that can’t be added together. (Addidion is the unified magnitude of several units) . translating it to temperatures. if an object radiates to an energy measured at 20C to a shell for a sufficent time to heat it to that temperature, then the shell isn’t going to be 40C because radiation leaves it bi-directionally, 20C each way. Temperatures don’t add up mathematically like that. If they did you’d have a litre of water at 10C and a litre at 30C combined to give 2 litres at 40C
blow up a balloon
TomVonk (04:22:04) :
1. What is conserved
Several people have become upset that he has conserved fluxes (W/m^2), not conserved flows (W). It’s been addressed a few times. If the shell is thin and near the planet, the error is trivial. If you don’t like it, redo it using actual areas and flows. You’ll see nothing much changes, so long as the shell is near the planet.
2. Stefan-Boltzmann
Your point about Stefan-Boltzmann is an absolute red herring.
Yes, the derivation starts with Planck’s law. You assume the surface is a diffuse emitter (intensity has no directional dependence), so integrating over the hemisphere of vision is trivial (it just adds a factor of Pi), then you integrate over wavelength and you get the Stefan-Boltzmann law. So what?
a. Curvature of the emitting surface
Stefan-Boltzmann gives the total power emitted by a surface. You think it should only be used for plane surfaces? Fine, then break up the earth or the shell into tiny differential areas (each of equal area); each differential area dA is essentially planar. Apply Stefan-Boltzmann to each of those areas dA to get the power emitted at each, dP. Now, add it all up. the differential areas dA all add up to the surface area of the sphere or shell, 4*pi*r^2. Adding up all the dP will just get you…exactly what you’d get by applying Stefan-Boltzmann to the entire curved body, because dP is the same for every area dA. So the fact that the body is slightly curved is completely inconsequential, so long as you know its surface area.
b. Solid Angle
Are you then worried about the solid angle, then? Why? Stefan-boltzmann gives the total power emitted by the surface, in all directions. And that is exactly what is needed here. Every last bit of power emitted by the planet will reach the inner surface of the shell. Every last bit of power emitted by the inner surface of the shell will reach the planet.
Willis-
Here is a web-based calculator for your two-layer model:
http://home.comcast.net/~stevehaker/GlobalWarmingCalculator-II/
I put your figure in there for reference. I hope you don’t mind. I will take it out if you prefer.
I had fun making it. Playing around with the numbers is interesting.
The SB may give total power emitted by surface, although other calibation equipment gives better measurements of power emitted form a surface. at 100w/m2 uniformly over a flat surface, is enough energy to produce 24C. 300-500w/m2 is enough energy to cook food from frozen in oil (see citiations above)
Why is this equation used to describe anything but incandescent/hot or superheated metals? If a black box were the absorbing heat at maximum potential, then it theoretically emits as much as it receives, but only if it is out of equilibrium. If it is in equilibrium then the rate for this theortical plane is 235w/m2 no more, no less between the two spheres. (if you thing the SB constant is a valid measure of energy).
this is why AGW theory is all fraud. If earth receives this amount of solar energy, there is no reason why it should emit as much, or even more. if it is in a fair equilibrium with the incoming solar that hits the surface then earth gives off very little radiation. Repeat: When two objects are at the same temperature in the same vicinity and are in thermal equilibrium, energy is not being radiated from these objects, as objects only give off radiation when they are getting colder relative to their surroundings. Bring the temperature down then the said objects cool and give off radiation until they reach the new equilibrium
P Wilson: You aren’t just arguing against AGW, you’re arguing against physics that has been successfully used by a lot of people for a lot of things for a long, long time. When that is the case, you must consider the strong likelihood that the physics is just fine, but you misunderstand it.
“The SB may give total power emitted by surface,”
Stefan-Boltzmann gives the maximum possible power – that emitted by a blackbody.
“although other calibation equipment gives better measurements of power emitted form a surface. ”
To the extent that something is not actually acting as a perfect blackbody, yes, some measurements are needed.
“at 100w/m2 uniformly over a flat surface, is enough energy to produce 24C.”
“300-500w/m2 is enough energy to cook food from frozen in oil”
By itself, you can’t say any of these things. You need to describe the entire system.
“Why is this equation used to describe anything but incandescent/hot or superheated metals?”
Because it works. For some reason, you are having a difficulty with infrared radiation. I am not sure why.
“If earth receives this amount of solar energy, there is no reason why it should emit as much, or even more.”
If the temperature of a body is stable, that means that energy is coming to that body at the same rate it is leaving.
“as objects only give off radiation when they are getting colder relative to their surroundings.”
Absolutely incorrect. All matter gives off radiation, proportional to T^4. Any body is always radiating, and receiving the radiation from surrounding bodies. The NET exchange between the bodies will be from the higher temperature body to the lower temperature body.
P Wilson, all of these things have been described before, above. All of them are easily learned in a physics text. All of them are used in the design of useful things. All of them can be observed. If you’re clever enough, it can even be derived from first principles. One really should pause before declaring that some fundamental principle of physics is invalid.
Denny (19:50:35):
Excellent question! I think you are noticing the phenomenon that different wavelengths of IR radiation penetrate different depths into the human body (as you instinctively suspect when you talk about “bone-warming heat”). This was something my heat transfer prof loved to demonstrate in the lab, because he knew it would really “get” us. I still remember the demonstration 30 years later!
So if the coal burns significantly hotter than the wood, as Anna V claims, then yes, you could “feel” the difference if you are attuned to it.
This lets me segue into another issue on this thread — radiative power from cooler bodies to warmer bodies. (Yes, it does occur, even though John A will never concede the point, but of course, it is less than the radiative power from the warmer body to the cooler body.)
One underemphasized benefit of having good insulation inside your walls is that it makes the room surface of the wall warmer so it radiates more heat back to the people in the room. This lets the people have an equivalent comfort level with the air temperature of the room several degrees below what they would want with cold walls.
If you are attuned to it, you can “feel” this difference too when you walk into a room.
Also, there is a significant difference in “feel” between radiative heat sources, where the surfaces are heating the air, and forced-air heat sources, where the air is heating the surfaces.
carrot eater (10:03:18)
The SB works well for IR radiation such as the sun, and heated metals in engines or but not normal temperature, or LW surfaces in equilibrium. If a black box in your room was next to a white box, according to SB they should be different temperatures and the black should be emitting more radiation than the white box, meaning they ought to have different temperatures. However, if they are both in thermal equilibrium at that room temperature then they’re the same temperature. Normal temperature matter just doesn’t don’t give off so much radiation that the constant gives it. (if its in equilibrium)
Willis,
Sorry to be late to the party but it has been difficult to find the time to read the 400 comments to get to this point. Obviously you have generated great interest.
Your model appears correct for a solely radiative heat transfer process occurring in a vacuum. Unfortunately radiative heat transfer plays a relatively small role in the heat transfer processes occurring between a sphere and a relatively dense, gaseous atmosphere under the influence of a gravitational field.
Heat transfer flux for all three modes of heat transfer (conduction, convection and radiation) is basically a function of the temperature differential between the source and receptor and the heat transfer coefficient. In your model, the sphere heats up due to the reduced net heat transfer output flux from the sphere that results when the ∆T between the sphere and shell decreases (the input flux to the sphere (Sun) remains constant). In a planet with an atmosphere all three modes of heat transfer will operate in the same manner. If the atmosphere is colder than the surface, heat transfer will occur from the surface to the atmosphere until and if the surface and atmosphere reach the same temperature. Since the total heat transfer is the sum of the three heat transfer modes, the heat transfer coefficients for each process determine which modes will predominate.
Since the heat transfer coefficient for conduction in gases is very small, conduction plays only a minor role (primarily to initiate convection from the surface).
Radiative heat transfer will behave quite differently in a gaseous system in contrast to your model where all bodies are solids. First, radiative heat transfer will only occur with the approximately 2% of the gaseous molecules that are IR active. Second, in the lower and mid troposphere, the IR active molecules will experience approximately 3000 – 10,000 collisions with other molecules during the relaxation time between absorbance and emission. Thus the transferred heat will be thermalized among the non IR active molecules and the temperature of the total gas mixture will rise. Very little emissivity will occur. In other words, in the lower and mid troposphere, the heat transfer coefficient for radiation heat transfer in a gas is very low (especially at or near ambient conditions). Heat flux absorbed by the gas from radiation will be converted to convective heat transfer. (In the upper troposphere, where molecular density and the chance for molecular collisions is much lower, emissivity will play a much larger role and is a factor in the total OLR).
Convection and the gravitational field are the primary players in tropospheric thermodynamics. The heat transfer coefficient for convection is large (and becomes larger as turbulence increases). You illustrated part of the convection role very well in your previous article on the thermostat hypothesis (this was a very good article, by the way). But you did not explain the other part of convection which is subsidence. Meteorologists frequently use the term “convective overturning” which illustrates the convection/subsidence cycle that is true for a convection process. “What goes up must come down”.
The gravitational field is important in that as mass is convected to a higher altitude via expansion and work on its surroundings, thermal energy is traded off for potential energy. As a result, the temperature of the mass parcel decreases due to adiabatic expansion and the dry adiabatic lapse rate is created. But the total energy content remains constant. At higher altitudes radiation heat transfer becomes more significant and OLR is emitted and the parcel cools. This is one reason why the greybody temperature of the earth is lower than the surface temperature. Subsidence then occurs and the mass parcel descends and trades off potential energy for thermal energy and the parcel increases in temperature due to adiabatic compression. This is why the surface temperature is higher than the greybody temperature. This subsidence conversion of potential energy to thermal energy represents the bulk of the 324 W/m2 down arrow in the K&T cartoon. Very little is due to “back radiation”.
I tried to keep this as brief as possible and a lot of detail is missing. But if you or anyone else would like to pursue this approach, I would be glad to elaborate more fully. But I am late to the table and I’m not sure if many are still plugged into this thread. But either way, thanks for listening.
par5 (04:19:34) :
“Cantobtainium” is a humorous name for an element which we can’t obtain, which I sometimes use in thought experiments to indicate a substance with certain properties which real substances might not have. I could just as well have said “Steel”. Neither steel nor cantobtainium vaporize at 470W/m2.
Remember, this is a thought experiment, designed to simplify the issues so that the main principles become clear. The substances used are immaterial.
And no, I haven’t lost my patience, although some posters tempt me to temporarily mislay it …
If you take the human basal metabolic rate as a standard of comparison – it is the calories required against calories burned, converted into w/m2, which for most people is 58w/m2, increasing on average to 100w/m2. In the following experiment radiation is measured fromleaving individuals.
in google enter:
Description of a human direct calorimeter
and it takes straight to the cambridge journals which conducted the experiment.
It stands to reason that if a human optimum emites 100w/m2 of radiation through a duration, that earth is emitting much less per square metre.