From NOT A LOT OF PEOPLE KNOW THAT
By Paul Homewood
h/t Philip Bratby/Paul Kolk
In BBC world, everything bad is due to climate change!

For Antarctic wildlife, exposure to the Sun’s damaging rays has increased in recent years, scientists say.
A hole in the ozone layer – the protective barrier of gas in the upper atmosphere – now lingers over the frozen continent for more of the year.
A major cause of ozone loss is believed to be the amount of smoke from unprecedented Australian wildfires, which were fuelled by climate change.
https://www.bbc.co.uk/news/science-environment-68906013
As we know, the 2019 drought was far from being unprecedented in SE Australia, the region worse affected, so climate change had no effect. Instead there is plenty of evidence that poor forestry management was the biggest factor.

In fact the paper itself makes no claims about “unprecedented Australian wildfires”, and the factors behind ozone loss are much more complex and nuanced than the BBC reports. In particular, La Nina, the polar vortex and the Hunga-Tongo volcano all play a role alongside Australian bushfires. The BBC report makes no mention of any of these other factors.


https://onlinelibrary.wiley.com/doi/epdf/10.1111/gcb.17283
As with all these sort of studies, the scientists are only looking at a few years worth of data, so they have absolutely no idea whether similar cycles of ozone loss occurred naturally and regularly in the past.
But that is of no concern to the BBC, who would rather persuade you that Antarctic animals are being sunburnt because of your consumption of fossil fuels.

Those who know me here will recall that I have long railed against the thought of thousands of penguins dying of skin cancer.
Certain areas below the cummerbund can be particularly sensitive.
Interesting chart 51 years are wetter than Norm and 72 years are dryer. Drier seems to be more the normal condition, at least for that particular 123 year chart span
Although it could be a 50 year dry cycle followed by a 50 year wet cycle then 20 years into the next 50 year dry cycle
Those penguins show absolutely zero sign of sunburn. !
Still as pure white as a Pommie in winter. !
https://www.nature.com/articles/s41467-023-42637-0 for a more nuanced view. Suggest GCMs aren’t working well in this regard.
The paper is a diabolical read, so I’ll summarise it for you: We, the authors, have absolutely no idea why the ozone hole is getting larger for longer.
True! If the CFCs are affecting the ozone layer – why only in Antarctica?
And what’s the big deal if the UV exposure there is roughly equivalent to San Diego?
(See the linked paper if you’re wondering what I’m going on about there’s actual measurements! So rare these days in science! It’s like we’re back in the days of Aristotle where no one bothers to test anything)
And why does a few weeks of round the clock sun in December close up the hole and bring down the UV dose?
Why are the northern hemisphere UV dosages basically unaffected?
Stop noticing! You’re disrupting The Narrative ™ and you’re gonna be sorry bub!
What is the highest that the sun appears in the Antarctic sky in December? Google Maps indicates that the northernmost tip of the Antarctic Peninsula is at 63°81’S,
Yesterday under brilliant blue skies, I cut my lawn for about an hour around noon. I’m a bit north of 41°N here in northern Connecticut. I am not sunburnt today. Had I done that in July without a hat and sunscreen, it would have been a different situation.
As September/October is the time of the Autumnal Equinox, I imagine that the penguins are happy to get a little sunshine after the June-July-August darkness!
Wouldn’t we call that their vernal equinox? We must acknowledge the sins of our hemisphericist past. Northern privilege and all that.
All those questions were answered years ago, basically temperature and the resulting noctilucent clouds (requires temperature below -78ºC).
You’re being very unclear Phil. Are you arguing that CFCs are in some way responsible for noctilucent clouds?
No that noctilucent clouds are agents in the destruction of ozone and because of the temperatures required they’re mainly formed over the Antarctic, hence the formation of the ‘Ozone hole’. The crystals formed allow a catalytic reaction which converts the inert form of chlorine in the CFC into reactive chlorine compounds which destroy O3 in a cyclical manner meaning that the chlorine compound is regenerated and continues to destroy the ozone. Also they remove nitrogen compounds which would reduce the chlorine destructive reactions.
See: https://csl.noaa.gov/assessments/ozone/2002/qandas9.pdf
Ok so you’re a believer in 80s pop science à la Algore. Is there any physical evidence that these angels dancing on the heads of pins actually exist in the real world up there in the stratosphere?
Perhaps you’d like to explain how your fantasy mechanism is consistent with the CFC curve that you posted showing a continuous decline in CFC concentration? That is, how it brought about a recent resurgence of the ozone ‘hole’ when the CFC concentration is even lower now than when the ozone had ‘recovered’.
Wait, wait, don’t tell me! You also buy into the latest fantasy that bushfire smoke is the cause?
Now I would offer up that other fairy tale that the ozone hole got worse because of illegal Chinese production of banned CFCs, except that that would be inconsistent with your chart showing a continuous decline.
I’m sure you have a compelling explanation, yet I remain puzzled that you choose to keep it under your hat.
When was the last time you ever explained any of your assertions?
Which assertions are you referring to Phil? Happy to defend any that you care to point out.
You are making the claim that CFCs are the cause of the inaptly named ozone ‘hole’. You presented a chart showing continuous declines in concentration for various CFCs over recent decades. It has been established or at least it had been asserted that the Montreal Protocol had resolved the ‘problem’, and then disappointingly the ‘hole’ came back. Do I need to justify these assertions or do you accept those as facts?
If CFC originally caused the problem when it was first observed in the 70s, and lower CFC concentration really was the cause of the ozone recovery in the early part of this century, then how to explain the back-sliding to a bigger ‘hole’ recently when CFC concentrations are apparently the lowest that they have been in 30 years or more? There must be an independent cause, much more powerful than CFCs that is to blame, n’est pas?
“ Do I need to justify these assertions or do you accept those as facts?”
I certainly don’t accept them as facts
No, I understand the science involved, there certainly is evidence for the existence of the species in the stratosphere, multiple measurements have been made of CFCs there. ClO measurements have been made there, From August onwards a strong increase in lower stratospheric ClO is observed, with a peak column amount usually occurring in early September. From mid-September onwards a strong decrease in ClO is observed which is exactly what you’d expect from the reaction mechanism destroying the ozone.
Here’s the data for O3 concentration, it shows no ‘recent resurgence’ that you are claiming.
And I thought the crazies said we solved the ozone hole problem by banning aerosol sprays.
Algore saved the world back in the 90s but you kept burning fossils and that led to forest fires and that wrecked the ozone again.
Recycling is a sacrament in the Church of Climastrology. This story is both recycling and a cover story at the same time.
Some of us predicted 30 years ago that eliminating CFCs would not eliminate seasonal reductions in ozone over Antarctica and that we were likely observing a cyclical pattern (much like Arctic sea ice extent is cyclical). Now we see evidence that this is the case. The CFCs are gone and the ozone is thinned out again.
Well that won’t do! Can we hide the decline or something?
What the Beeb settled on was recycling the ozone scare for a whole new generation of mind-numbed robots while simultaneously evoking the deus ex machina of Australian bushfires caused of course by Climate Change ™ to explain away the ‘inconvenient truth’ (to coin a phrase).
No the CFCs have not gone, they’re slowly decreasing:
https://www.fluorocarbons.org/wp-content/uploads/2020/07/Atmospheric-concentrations-04.svg
Ok, Phil, by your criteria, there probably are still a few atoms left in the atmosphere from atomic bomb testing in the 1950s.
When I said ‘the CFCs are gone’, I meant that we have stopped selling them for use in new refrigeration units and aerosol products.
Are you just being pedantic or are you arguing that the residual CFCs have caused a resurgence in the ozone ‘hole’?
So you made a false statement and are annoyed because someone took issue with it! As shown in the graph the CFC concentrations have dropped about 10% from their maximum and are still way above their levels in the 1980s when the Ozone hole was discovered and the O3 concentration was rapidly dropping to what it is now.
I’m hardly annoyed Phil. On the contrary, quite amused.
Are you now asserting that the ozone never recovered and has just been slowly improving in line with the slow decline in CFC concentration that you presented? Of course that would seem to be a counter-factual assertion, but is that your story? Measurement error perhaps?
Despite all of your blah blah woof woof misdirection quibbling around the edges, for some inexplicable reason you don’t seem to get around to explaining the recovery and resurgence of your ‘ozone hole’ in light of a continuous decline in CFC concentration and your hypothesis that CFCs catalyze ozone destruction.
Why is that?
Dobson himself observed ozone depletion above the Arctic during Winter 1936-37, and above the Antarctic twenty years later. Since CFC release only became detectable in the 1970’s it is unlikely they are responsible for the ozone hole.
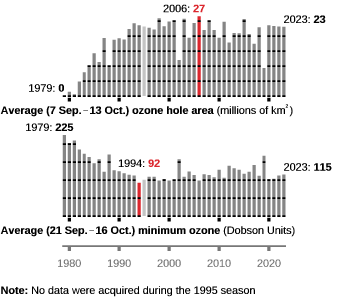
Yes Dobson detected an annual cycle in O3 concentration and data from 1924 indicated that the cycle was stable through the 1960s, since the 1980s the depletion in the minimum was dramatic, in the case of the Antarctic the minimum dropped from 225 in 1979 to 92 in 1994. It is that depletion beyond the normal annual cycle that is attributed to CFCs. Since the ending of CFC production the levels have stabilized but it’s a slow process because of the long lifetime of CFCs.
You accept that ozone depletion in Winter is a perfectly natural phenomenon?
Absolutely not, quite the opposite, ozone depletion occurs in the Spring as was observed by Dobson in his early work. He observed a seasonal variation with a minimum in the Spring, what changed in the 70s onwards was a significant depletion beyond the previously observed minimum. The graph I posted which you replied to showed the profile in the stratosphere at the time of the minimum with a peak O3 of about 12mPa, by the 80s that had been substantially depleted to about 3mPa, by 2001 there was no O3 between 14 and 20km! Now the bottom of the minimum is not as flat and some O3 remains.
How do you now this is not due to natural variability? There are years when the extent of the Ozone Hole shrinks almost to zero.
Not since 1979!
False. There was almost no ozone deficit in 2002.
https://www.copernicus.eu/en/news/news/observer-evolution-ozone-hole-1979-2021
What happens in the Earth’s atmosphere occasionally is a ‘Sudden Stratospheric Warming’ which causes substantial disruption to the polar vortex and substantial reduction in the Ozone hole. From the annual data I’ve shown you can see that it occurred in 1988, 2002 and 2019 (higher O3 and smaller Hole area). However the Hole did not shrink to “almost zero” as you asserted.

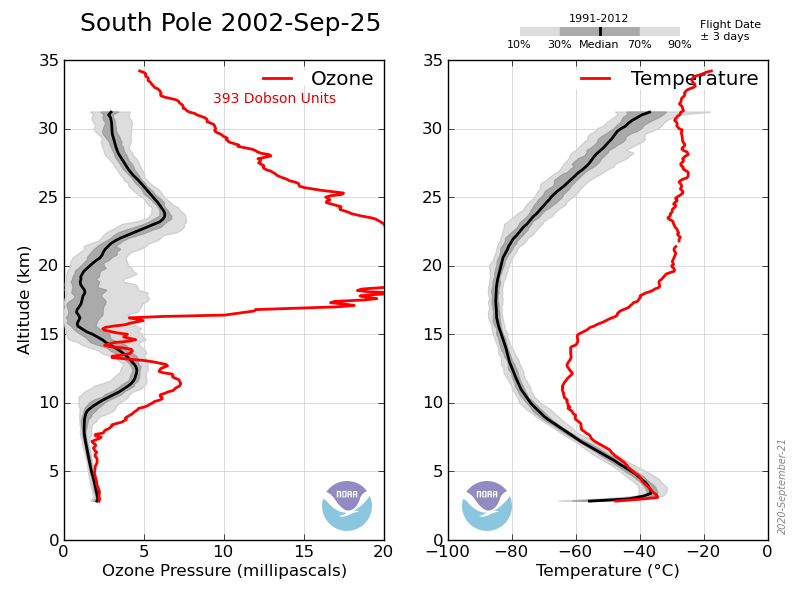
Here’s the sonde data for the South Pole on Sept 19, 2002, reasonably normal:
Here is the data for 6 days later, totally abnormal due to the SSW, temperatures in the Ozone depletion region 40ºC above normal and O3 levels through the roof!
By October 11 it was almost back to normal, returned again November, basically a stratospheric storm that occurs about every 20yrs.
Interesting graph. Why does the O3 increase at 10 km, where you have a higher concentration of CFC’s?
Because the temperature there is above -80ºC so the noctilucent clouds don’t form. From 14-20 km temperatures go below -80ºC thus catalyzing the depletion of the O3.
It’s absurd in the first place to argue that CFCs are catalyzing ozone depletion at part-per-trillion concentrations that they had back 30 years ago. But let’s stipulate that fantasy. So why/how did the ozone ‘recover’ and then reverse course based on your chart?
“So why/how did the ozone ‘recover’ and then reverse course based on your chart?
It didn’t happen, look at the data.
You’re absolutely correct. If the ozone hasn’t recovered since the Montreal Accord, perhaps CFC’s aren’t the cause.
What the Montreal Accord did was to stop the further release of CFCs into the atmosphere so the concentration reached a maximum and as I showed has started to slowly decrease but we still have ~90% of the maximum left so it will take a while to see a large difference in the O3 minimum. CFC-12 concentration peaked at 542 in 2002 and by 2018 had reached 508.
More importantly, cheap and efficient refrigerants were banned.
Yes. At a huge energy penalty.
Bring back 80’s big hair coiffed from Aqua Net.
Maybe we are seeing the start of a whole series of excuses for the ozone hole not having been fixed by stopping the use of CFCs.
….. and excuses for Nobel Prize winners not having to say sorry (and give their ill-gotten prizes back).
The same reason that was given years ago, the long lifetime of the CFCs.
https://www.fluorocarbons.org/wp-content/uploads/2020/07/Atmospheric-concentrations-04.svg
The long lifetimes of CFC’s should actually work against the hypothesis that they are responsible for ozone destruction. The dogma states that UV radiation dissociates a CFC into atomic chlorine and a free radical. The atomic chlorine is the actual catalytic species. The CFC molecule must therefore be decomposed before it can begin destroying ozone.
Yes, the chemistry shows that the UV dissociates a CFC molecule producing 2 or 3 chlorine radicals each of which can remove 100,000 O3 molecules.
You really believe those chlorine atoms are going to survive decades in the stratosphere?
It’s not the Cl atoms that survive, those that are generated from CFCs in the Spring and are removed in the process of destroying the O3. However ~90% of the CFCs survive and are able to generate more Cl in the following Spring, the current decrease in CFC-12 is about 1%/year.
How does the production of atomic chlorine from CFC’s (annual global emission ca.18 000 tonnes) compare with that from marine algal chloromethane (ca. 4 million tonnes per annum)?
Chloromethane is a natural chlorine source, it’s tropospheric lifetime is about 1yr due to OH reaction, reaction in soil and ocean uptake. Its level has been very stable and is only about 10% above its level between 1700 and 1900 CE.
Check this out:
 ?2023
?2023
I don’t understand this graph. Why is the Y axis labelled “Reactive halogen” and not simply parts per trillion?
Because it takes account of the number of reactive halogen atoms per molecule: CCl4 contains 4, CFC-12 contains 2 Cl, CFC-11 contains 3 Cl etc.
Chloromethane has a far shorter half life than CFC’s and is produced in vastly higher amounts (4mT versus 18kT per annum). Therefore, the former must contribute far more to the production of atomic chlorine than CFC’s.
Do you really believe there is something especially bad about atomic chlorine produced from chloromethane compared with that from CFC’s?
Only if it makes it to the stratosphere, the Chloromethane is reacting in the troposphere unlike the CFCs.
How do you know that chloromethane does not make it to the stratosphere?
It takes 2-5 years to reach the stratosphere from near the surface, you said its lifetime is ~1year, work it out!
If 90% of CFC molecules survive a year in the stratosphere, that means that at most only 10% will produce atomic chlorine. The longer the half life, the less chlorine will be produced per unit time.
Actually ~99% survive a year in the stratosphere that’s why the problem will take such a long time to clear. That’s enough to destroy all the O3 between 14 and 20 km each spring.
You seem to think that CFC’s are responsible for destroying ozone when it is the atomic chlorine from the decomposition of the CFC’s which does this. CFC’s can only damage the ozone layer if they are destroyed first.
Would there be any ozone depletion if CFC’s were 100% stable? You seem to think so.
You don’t appear to understand plain English, I’ve pointed out several times that it is the chlorine atoms produced in the breakdown of CFCs that destroy the O3, and that each one is regenerated multiple times and is capable of destroying 10s of thousands of O3 molecules. As I pointed out the 1% of CFCs destroyed each spring is enough to destroy all the O3 between 14 and 20km.
I know perfectly well that the catalytic species is atomic chlorine.
Do you really believe a chlorine atom has a lifetime of years in the stratosphere? How do you know that chloromethane is not responsible for the majority of ozone destruction?
How do you know ozone depletion did not occur before the introduction of CFC’s?
“Do you really believe a chlorine atom has a lifetime of years in the stratosphere?”
No, as I explained to you before!
https://wattsupwiththat.com/2024/04/27/ozone-hole-why-antarctic-wildlife-is-being-sunburnt/#comment-3902853
“How do you know that chloromethane is not responsible for the majority of ozone destruction?”
Amongst other things the data shown in this graph, which you admitted you didn’t understand!
https://wattsupwiththat.com/2024/04/27/ozone-hole-why-antarctic-wildlife-is-being-sunburnt/#comment-3902941
“How do you know ozone depletion did not occur before the introduction of CFC’s?”
The data:
Your last graph: Correlation is not causation. CFC’s were already in use in the 1950’s, yet the decline only really started around 1980. Why is that? How do you now there is not a long-term cycle or natural variability? Dobson observed seasonal ozone thinning in 1935-6. Was this due to CFC’s?
You still haven’t explained why the atomic chlorine produced by decomposition of 1% of CFC’s destroys more ozone than that from chloromethane (4mT/year emitted). Nor have you explained how Cl atoms survive years in the stratosphere.
“Your last graph: Correlation is not causation. CFC’s were already in use in the 1950’s, yet the decline only really started around 1980. Why is that?”
Because the production rate was relatively low and it takes about 5 years to get from the N hemisphere to the S Pole.
“How do you now there is not a long-term cycle or natural variability? Dobson observed seasonal ozone thinning in 1935-6. Was this due to CFC’s?”
Already explained that, the seasonal cycle observed by Dobson and others from the 20’s onwards was smaller than the present and very stable, what happened in the 80’s was a major increase in the annual depletion.
“You still haven’t explained why the atomic chlorine produced by decomposition of 1% of CFC’s destroys more ozone than that from chloromethane (4mT/year emitted).”
Already answered, can’t you read?
“Nor have you explained how Cl atoms survive years in the stratosphere.”
No I explained that they don’t, clearly you don’t read.
Isn’t that ozone hole largest in winter? Is it not so that in Antarctica the sun does not shine in winter? Hence the penguins get a sunburn from a sun that does not shine or raises barely over the horizon.
What type of BBC moron writes that stuff?
Is there more than one type of BBC moron?
LOL and an excellent question!
I’m sure with that many morons they probably can be classified and sorted.
Point taken.
The maximum hole area is Sept/Oct and still over 50% in early December, so it peaks in Spring and is still significant in early Summer. The Adelie penguins breed between October and February, in October on the coastline where they breed there’s about 15 hrs/day of sunlight, beginning of December it’s 24 hrs/day.
Again with the ambiguous comment. Adelie penguins breed while there’s a lower concentration of ozone overhead. Does that matter? Is that known to be different from the situation five centuries ago?
The sun may be shining for the whole day but never higher than 25 degrees above the horizon.
But you said “Isn’t that ozone hole largest in winter? Is it not so that in Antarctica the sun does not shine in winter? ” which is patently false. That’s what I addressed.
Yes, yes, you ‘address’ — most would say nitpick — many tangential points where perhaps others have not used precise language or have even made factual errors on essentially irrelevant points.
What you fail to address is the main point. If CFCs are the cause of ozone depletion and CFC concentration has been slowly decreasing, how do you explain the ozone levels recovered and then resumed dropping while CFC concentration was on a steady decline?
I suppose I have asked this three or four different ways. It cannot be that you missed the question.
Not ‘nitpicking’ to point out that the poster had mistaken spring/summer for winter!
Make that about 45º where the penguins are breeding.
You can’t even get the geography correct. With the exception of the Peninsula, ALL of Antarctica is South of the Antarctic Circle (63 Deg South).
Here’s a picture of where Emperor Penguins breed:
https://www.researchgate.net/figure/Location-of-the-54-emperor-penguin-breeding-colonies-around-the-Antarctic-continent-The_fig3_312870660
70ºS-23ºS=47º
In response to: “The sun may be shining for the whole day but never higher than 25 degrees above the horizon.”
Yes I was unaware that the Antarctic Circle had moved from 66.5ºS, thank you for updating that!
If you look at Wikioedia you can see that between 1960 and 1983 the area burnt in Australia was about 3 times that burnt this century.
That raises the question why smoke affected the Ozone Layer now and not in the 1970s and 80s?
How would anyone know if it did?
We weren’t looking back then.
The 2019 bushfires in Australia were caused mainly by 2 rather dry warm years, which followed some normal years. Massive amounts of fuel build-up occurred in those previous years, and nothing was done about it.
The actual area burnt was nowhere near a historic maximum,, but the areas burnt just happened to be near towns.
There was nothing “unprecedented” about the 2019 Australian bushfires.
If it was “climate change” why hasn’t there been a strong fire season since then…
Even the ranted and yelled “hottest year ever” in 2023 ….. and nada, zip !!
Although, fuel is starting to build up in many areas again, so another bad year will probably happen in a few years.
It is Australia. !! That is what happens.
I helped my SIL clear some brush in WA today and I get the impression that fire is pretty much expected here, it’s kind of interesting to learn that even a few birds (black kites) get into the arson business.
You do get around a bit Scissor.
Hope you studiously track your “carbon footprint” 🙂
Anyone actuality make ground-level measurements of UV light in the arctic where the sun is always at an angle (not overhead like the tropics) and MORE of the atmosphere is in the ‘path’ of the incoming UV?
The world panicked when the ozone hole was first seen and (without wondering if it had always been like that) made all sorts of changes to refrigerants at great cost. It now appears to be a normal and variable aspect of our climate and it changes with many different inputs. YES, we need to study it but the effect is already known not to be human-related in the way once feared. But why isn’t this stressed sufficiently by the media?
We can study it and that might be interesting, but I am not sure that we need to study it.
We’re not responsible for it and it doesn’t impact a single person, so why do we need to study it?
Are there not researchers of mitochondrial DNA anomalies in African fruit flies that struggle to get proper funding? And of course the MoSW has been chronically underfunded vis à vis the MoD since Cleese et al. exposed the scandal on MPFC.
The ozone hole solution has been characterized as the poster child for climate change. To admit they got it wrong would destroy the narrative and likely lead to many more people questioning climate change.
The story served it purpose back then. The attack against western societies has moved on since. There is no reason to distract from the current focus by bothering to explain away discrepancies.
And exactly how do the sun’s rays bend through the ozone hole to burn anything?
“And exactly how do the sun’s rays bend through the ozone hole to burn anything?”
Lee, don’t you know nuffing,
1st, the science … 97% of all CO2 rushes down south to fill the hole, forming a concentrating lens (like a CO2 laser cutter); that’s why Antarctica is splitting (nothing to do with tectonic plate movement) & the seas are boiling, ask Al Gore if you don’t believe me.
2nd, the solution … The UN should send a task force to supply penguins with factor 50 sun-cream & straw sun hats (crowns for the emperor penguins ).
Finally, a pragmatic approach.
Although you neglected the most important solutions—stop burning fossil fuels and more socialism, less freedom.
Oh boy. The Law of Unintended Consequences is huge when it comes to the last Global Rush To Judgement: The Ozone CFC Treaty.
True story: I was a consulting electrical engineer for ~30 years. I was the senior power engineer at the largest semiconductor wafer fab plant ever built in California (i.e. computer chips). One day, c.1995, Marc, the senior mechanical engineer (and the best I’ve ever known) came up to me and said “we are going to convert the (giant 4160V 1200 ton) chillers from R11 to R134 15 years before the Treaty deadline, so the company can show how green we are. My first Q: What will this do to their efficiencies? He said “about a 5% loss of efficiency.” So I did a quick study, that showed that over then 15 years the loss in efficiency would equal about 50,000 barrels of oil (I like to put things in terms like that, so management can more easily understand). Then I emailed the boss. He killed the project.
(A few years later, he converted one machine on the sly, then told me. I measured it’s amps. Sure enough, used 5% more power.)
The non-CFC refrigerants are very inefficient compared to the old CFC Freons. This is a loss in the refrigerants’ thermodynamic efficiency, so it comes off the top, and remains even when machine design improves.
Now, in professional engineering, we do lots of studies, only we rarely publish them – they are usually for clients, often in book form. But, knowing that mechanical refrigeration was one of the hugest uses of energy in the USA (far bigger than lights), I decided to do a quick study on the national implications of the CFC Treaty vs. the CO2 scare. Using US Govt DOE data and Marc’s 5% loss (I searched the literature; his 5% may even be conservative – the range I found was 3% to 15% loss), I calculated, conservatively I believe, that the annual penalty, in terms of coal, was equal to about a 1000 mile long coal train! That’s a lot of CO2.
Did the media EVER tell America about this huge penalty of their last global rush tp judgement? (If you can find where, please let me know.)
Excellent post sir.
Only about one quarter of the CFCs were used for refrigeration the rest were used for things like aerosol cans and sprayed into the atmosphere.
Well, if that’s true, then the CFC treaty banning Freons from refrigeration machines looks even more irrational. Because they could have eliminated 75% of the “ozone depleting” CFC without the huge CO2 and “forever” energy penalty caused by using the replacement refrigerants. AND the irony is that those replacement refrigerants were, what (I forget the exact data), 2000X worse in (highly questionable, since CO2 appears saturated anyhow) “global warming potential” than CO2?
Even if the CO2 scare turns out to be 100% irrational, as I expect, the CFC treaty is ensuring that we are still wasting vast amounts of precious fossil fuels.
So, the moral may be, GLOBAL RUSHES TO JUDGEMENT are really stupid!
Oh boy^2.
They wouldn’t have “eliminated 75% of the “ozone depleting” CFC” they would have merely slowed it’s growth.
Restoration of the Ozone Layer Is Back on Track, Scientists SayRogue emissions from China of ozone-depleting chemicals had threatened to delay recovery by a decade. But the emissions were stopped.
https://www.nytimes.com/2023/01/09/climate/ozone-hole-restoration-montreal-protocol.html
Even the NYT goes for chemicals…
The article says that the ‘rogue’ CFC11 emissions were under control by 2019 and that they only delayed ozone recovery by one year. But recent years have seen much larger ozone holes. All we need is the ozone hole charted against CFC concentration over Antarctica, then we can see for ourselves whether the ozone hole recovery is on track and whether it relates to CFCs. I wonder why they never show such a chart.
YES!!!
I good friend of mine, who is a well known geochemist, has always suggested that the output of Mt. Erebus volcano is a major, even the dominant, contributor to the Antarctic ozone hole, and its variability.
Well he’s wrong.
Why do you keep posting ambiguous comments without explaining what you think is the right answer?
Phil says he’s wrong… so the geochemist is almost certainly not wrong..
The so-called ‘ozone hole’ doesn’t really start to form until after the Winter circumpolar vortex is in place. One of the consequences is that the stratospheric ozone created in the tropics, and moved pole-wards by the Brewer-Dobson Circulation, creates anomalously-high ozone concentrations outside the vortex. Ozone is effectively metastable, having a short half-life, and can also be photo-catalytically decomposed. This is what happens in the Spring in Antarctica. When the sun comes up over the horizon, halides adsorbed onto ice crystals result in rapid destruction of the ozone that has survived through the darkness of Winter. However, during warm Springs, the vortex may break up early and the hole is minimal in size. In any event, when the vortex does break up, the anomalously high concentration of ozone outside the vortex is able to move in and replace the missing ozone, ending the growth of the ‘ozone hole.’ Low temperature plays an important role in the photo-catalytic destruction of ozone by creating the ice crystals necessary as carriers for the halides. IIRC, Erebus has an unusual amount of fluorine in its emissions. That can exacerbate the problem, but the ice crystals are essential.
I have found very few references to actual Antarctic ground-level UV measurements, almost always suggesting that our concern about biological effects should be focused on ozone as a proxy for UV. However, focusing on the proxy ignores the facts that when the sun is low on the horizon, it’s rays have a longer slant-range than when the sun is overhead, which it never is at those latitudes. Basically, the ozone alarmists are either ignorant of the solar geometry, or choose to ignore it. Either behavior does not speak well for those claiming to be scientists. Additionally, sometimes the solar rays pass through the high concentration of ozone outside the vortex. The situation is more complex than is usually acknowledged.
Many years ago, I wrote a computer model to simulate the ground-level UV concentration based on the solar geometry and ozone measurements from the Total Ozone Mapping Spectrometer. What it showed was that there appeared to be a slight upward drift in surface UV-flux during the times of the year when sunlight (and UV) was weak; however, after the break-up of the vortex, when the sun was higher in the sky and sunlight was stronger, the UV flux was not increasing. That is, when animals might need protection from UV, the UV flux was probably the same as what they had evolved with.
Interesting post. Thanks.
Another factor is the emission of halogenated compounds by marine algae. It is thought that around 4 million tonnes of chloromethane is produced annually.
A chart of ozone hole against Mt Erebus output could settle the argument?
Stratosphere water vapour injection of Hunga Tonga
Hans,
That’s pretty impressive. Curious question, there also appears to be a largish injection in 2012/2013 (end of first graph/beginning of second). Does that correlate with anything that happened around that time (I can’t think of anything off the top of my head)/
According to NASA, the water ejected from Tonga depleted the ozone layer by between 5% and 45%.
Water is not a CFC.
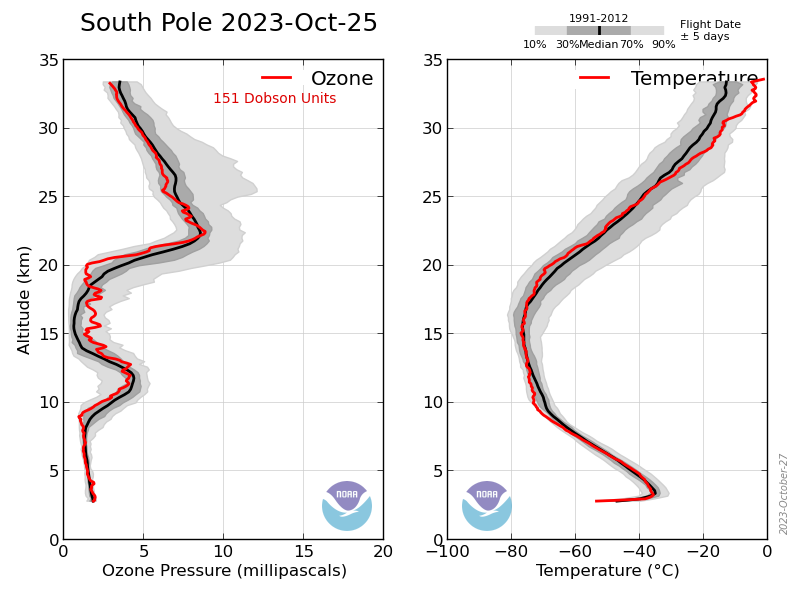
Got a reference for that? It could lead to more noctilucent clouds which would lead to increased O3 depletion. No sign that the 2023 season depletion was out of the ordinary.
“A major cause of ozone loss is believed to be the amount of smoke from unprecedented Australian wildfires, which were fuelled by climate change.”
Did the aborigines do much landscape burning- purposefully or by accident?
Well, to pick up on the “largely normal” theme from a couple of days ago, the BBC header is actually largely correct:
For Antarctic wildlife, exposure to the Sun’s damaging rays has increased in recent years, scientists say.
They only got one word wrong, see:
For Antarctic wildlife, exposure to the Sun’s damaging rays has increased in recent years, idiots say.
I know I’m getting old, but I can remember way back in ancient history when the ozone hole science was settled.
Until it wasn’t. There appeared to be an improvement until about the last 4 years, and since then it has been similar to what it was in the 1990s.
I do hope a way is found to plug this hole. I’m busy assembling a business plan to open a chain of Penguin Tanning Salons across Antarctica for when it is.
So it is clear CFCs we’re not the cause of the ozone hole. It is time for government and academics to stop lying to us. Enough is enough.
In general I thought the ozone thinning (not a “hole”) occurs in the Southern winter. Due to sun angle,even in Southern summer, the sun is not intense over Antarctica, and far less so moving to winter. I remember when the ozone “hole” was a big deal some years back, we in North America could pretty much eliminate the extra UV from our lives by moving 10 miles or so north!
Incorrect, the hole forms in the Austral spring not the winter.
sunburn is related to UV and thats related to ozone layer, which was level 600 in 1950 and now 120 in a lot of places not just the poles, skincancer is now in top 3 deadly diseases, and why? Cause airliners were allowed to fly in jetstream which steers gulfstream. That more UV also harms fythoplankton which has no protection like landplants, and thus absorbs less CO2 and gives back less O2…it’s all measured, and what on airliners in climate accords ? NOPE NOTHING
In 2019 max level UV was 4 no in april 8 which is claimed “unhealthy”, in Pacific there are places with levels 13 and up…
It appears that you are mixing ozone Dobson units and relative meteorological UV Index units.
The surface UV flux varies with the season, latitude, cloud cover, and aerosols/dust. Any UVC that makes it through the stratosphere will create ozone in the upper troposphere, and in so doing will be absorbed. Plants have defenses against UV, such as waxy coatings, hairs, and the ability to change their orientation during the day.
The so called dangers of UV radiation are greatly exaggerated. Only naked human skin and the noses of palominos are in danger of carcinogenesis. Hair, feathers or scales protects everything else…The unprotected protista have an evolutionary advantage from UV exposure, having an increased mutation rate, therefore an increased chance of beneficial mutations.
Also, water is a strong absorber of UV, so all the plankton have to do is hang out a little deeper in the water.
Fortunately for the Penguins, the sunlight they receive standing on the Antartica continent, has nothing to do with atmospheric chemical conditions directly overhead.
Take a globe. Put the proper sized ozone hole over one or the other pole (it matter not which).
No put the sun it its proper position. Draw a vector from the sun to the ozone hole.
Tell me how much sunlight directly hits the penguins.
Simple geometry belies this entirely.
Bear in mind that the penguins are at the shoreline, ~70ºS.
Where the cosine(70) = 0.34, which is the relative intensity of sunlight spread out over a larger footprint at that latitude.
However, the important point is that any solar rays that might impact the penguins have to actually travel entirely (or mostly) through the ozone-depleted column of air, with a boundary perpendicular to the surface of the Earth. Generally speaking, if the penguins are at 70 deg latitude, the sunlight (UV) reaching them will have encountered the atmosphere at a lower latitude than where the penguins are, and have a longer path length than at the Equator. That is, there is a good chance that UV will be absorbed in the anomalously ozone-rich areas outside the circumpolar vortex, and if not, will still experience absorption along a longer path length.
The ozone depleted layer is between 14 and 20 km altitude so at a 45º angle it will only be encountering the atmosphere 14-29 km further north with a ~40% longer path length.
I haven’t studied this UV issue much, but I do know that the reason that the sun is red in the morning is because its rays go through much more of the atmosphere, and the shorter wavelengths, like blues, violets, and presumably UV, are absorbed, leaving longer wavelengths like reds, IR, etc.
So, since the angle of incidence at the poles (I live in low UV Alaska) is so close to the horizon, why would more than a very minor amount of UV make it to the antarctic surface?
Those colors are not absorbed, they’re scattered at wider angles, particularly in humid atmospheres when there are small water droplets in the air, smoke particles also have a big effect.
Bear in mind the ozone is 100 miles up.
Actually the depleted layer is between 14 and 20 km
Per NASA, between 10 and 50 km with the thickest being at 32 km.
Your graph shows the greatest Dobson Units at about 22 km, which is offered by other sites.
Such is the fallacy of going by memory rather than verification.
Yeah in the 60s before the hole formation the max used to be about 16km at ~13mPa.
I went back and checked. The area of Antarctica equates to a perimeter at 70 S. You are correct.
I also double checked with NASA. The ozone hole is not empty of ozone, it has fallen below a measurable threshold. The measurement is made on a vector from the satellite to the center of the earth.
Also, not 100 miles. The range is 10 to 50 km, with the maximum at 32 km.
Doing the geometry, the area of the ozone hole has a perimeter equivalent to 65 S latitude so there is an overlap.
I did not do atmospheric thickness, but there is more air to go through relative to the sun. That, too, is geometry.
Smoke from Australian wildfires is supposed to cause an ozone hole over Antarctica?
All of the Australian continent (except for the island of Tasmania) is north of 40 degrees south latitude. The closest approach of the Antarctic continent to Australia is at 66 degrees south latitude.
So we’re supposed to believe that the smoke from the Australian wildfires traveled over 26 degrees of latitude (about 2900 km or 1800 miles) due south over the ocean to break up the ozone over Antarctica?
There is very little land between 40 and 66 degrees south latitude, except for the south island of New Zealand, and the southern part of Argentina and Chile, which is about 115 degrees of longitude east of the east coast of Australia. The remainder of that “ring around the world” is ocean, dominated by prevailing westerly winds. Any smoke from the Australian wildfires that drifted south would have been blown over New Zealand and the southern Pacific Ocean, and widely dispersed before it could affect the ozone over Antarctica.