Editorial note: WUWT is publishing this paper as a contribution to open technical discussion, not as an endorsed or settled account of atmospheric physics. Readers should distinguish between the paper’s narrow laboratory claim—that IR-active gases can contribute measurable radiative effects under the conditions of this apparatus—and its much broader inferences about climate sensitivity, water-vapor feedback, Antarctica, and the relative role of CO2 in the real atmosphere. Those larger extrapolations remain contested, and this experiment by itself does not resolve them.- Anthony
Demonstrating the Reverse Greenhouse Effect in the Laboratory – Part 2 (Part 1 can be found here.)
1. Introduction
The ability of IR-active gases, the so-called Greenhouse(GH)-gases, to absorb thermal radiation is undeniable, as this has been demonstrated by countless infrared spectra. It is also widely agreed that Kirchhoff’s radiation law also applies to GH-gases at high altitudes, where the spectral absorption is equal to the spectral emission (here , here ). However, given the high collision rates of gas molecules at normal pressure, some authors assume that GH-gases in the lower atmosphere are not emitting and do not generate any counter- or back-radiation (here , here ). Accordingly, excited GH-gases would only contribute to heat by collisions through radiationless deactivation (thermalization). In order to test the thermalization hypothesis and, if necessary, in the sense of K. Popper to refute this, a special apparatus was developed that simulates the principle of the negative GH-effect (see Part 1).
In an earlier experimental set-up, a heated and a cooled plate were housed together in a thermally insulated container. Experiments with this apparatus demonstrated that GH-gases can increase the back radiation, so that with constant heating the temperature of the warm plate (representing the Earth) is further increasing [1]. Any influence of GH-gases on the air temperature could not be observed in these studies, as there were hardly any changes. The effects of water vapor could also not be investigated, as this gas had to be removed to prevent condensation on the cold plate.
These disadvantages are overcome by a two-chamber arrangement. The focus is no longer on the Earth’s temperature, but on the temperature of the atmosphere. In our actual experiments, the Earth’s atmosphere is represented by a layer of warm air above a cooled plate. The air and plate are separated by a polyethylene (PE) foil, which suppresses any mechanical heat flows. Furthermore, the effects of water vapor can now be investigated, as the PE film prevents direct contact with the cold plate. As a radiation receiver, the cold plate enables an energy flow that can be interpreted as a simulation of heat transport both from the atmosphere toward the cooler Earth’s surface and toward space.
The experiments show that even small amounts of a GH-gas are sufficient to significantly increase the thermal radiation of the air while simultaneously lowering its temperature. This demonstrates a negative GH-effect, which has the opposite effect to what is usually expected. We show on a laboratory scale and at an air pressure of the lower atmosphere that GH-gases can absorb the kinetic energy of their surrounding and convert it into thermal radiation through subsequent emission.
This detection is anything but simple, as a measurement with gases requires a cylinder as a container. And this is where the problems begin, because like all solid bodies, the cylinder emits thermal radiation. This so-called background radiation from the cylinder overlays the emissions of the GH-gases. As a result, only the tip of the gas radiation is visible, similar to an iceberg (see Part 1). This is also the reason why sophisticated, optimal experimental conditions are required to demonstrate this effect and why so many attempts have failed under these strict conditions. At first glance a horizontally positioned styrofoam box seems to be a convenient solution, as it is commercially available and easy to process. But as described in [2], such an experimental setup is unsuitable for detecting gas radiation, primarily because of its intense background radiation and also due to convection in the box.
2 . Experimental Set-Up
To examine how GH-gases affect the IR emission and temperature of an air parcel, all what you need is a heated cylinder and a cooled blackened plate PC. To prevent convection, the warm cylinder is placed vertically on top of the cold PC plate, creating a stable stratification (see Fig. 1).
The real challenge is reducing the cylinder’s background radiation. For this purpose, polished aluminum with a low emissivity of ε ~ 5% was used as the cylinder’s surface (Fig. 1c). This does not look much, but the cylinder’s inner surface is about ten times larger than the exit to the PC plate and thus, generates a background radiation of 78 W/m2 , while the radiance of CO2 is about ten times lower.
The emitted IR radiation, commonly referred to as heat flow Q to the PC plate, is detected by two independent detectors: a small blackened disc on an insulating layer (detector TD) and ten mini-Peltier elements (detector VP) located directly on the PC plate (Fig. 1d).
As described in Part 1, in the air-filled cylinder, approximately 94% of the heat is dissipated by radiation transfer I0 and 6% by mechanical heat conduction WL . For simplicity , this heat flow Q is further referred toas background radiation I0 , although it contains a smaller amount of heat conduction.
In contrast, the additional heat flow, when adding GH-gases, consists exclusively of infrared radiation from the GH-gas. This follows also from the temperature changes with the difference ΔT1 – ΔTC , which become negative when adding the GH-gases. A positive increase in this difference would be essential for an increase in mechanical heat conduction (Equation 1, Part 1), but this is not observed (see Table 2b ff.). The differential measurement of the heat flow before and after addition of the greenhouse gas thus measures the pure gas radiation IGas . However, this is attenuated by superposition and transmission losses before it reaches the PC plate (Chapter 4).
The temperatures T1 to T5 are determined using temperature data loggers (Elitech). Since their measuring tip is only 5 cm from the cylinder wall, a mixture of air and wall temperature is measured, which is referred to as cylinder temperature. The investigation is concerned with these cylinder temperatures and their changes. For this reason, in contrast to the previous apparatus [1], the cylinder is not heated electrically, but rather with thermostatted water at 51 ± 0.1 °C. For this purpose, the warm water TW is continuously passed through the PVC hose of the jacket heater. Without outgoing heat flow Q, the cylinder and heating water temperatures should be almost the same. However, due to the heat flow, differences arise between the cylinder temperatures and the heating water temperature TW (see Part 1). To enhance this effect, a 2 mm thin layer of Styrofoam insulation was placed between the PVC hose and the cylinder wall. This weakens the heat flow from the water to the cylinder. Using this measure, the gas radiation IGas can also be easily detected by the temperature decrease after the addition of GH-gases (see Fig. 4 ff.).
The PC plate consists of a blackened aluminum sheet bonded to a spirally bent copper pipe in a concrete bed. Thermostatically controlled water flows through this pipe at a temperature of 10 ± 0.1 °C, continuously dissipating the absorbed heat.
3. Preparatory Studies
Extensive preparation is required to ensure that the effects found are actually identified as radiation of the IR-active gases and not as other influences.
To detect greenhouse gas emissions, a steady state with constant temperatures is required. This is achieved by circulating heated and temperature-controlled water through the cylinder jacket several hours before the start of the experiment and waiting until a steady state is established. For evaluation, an average value is determined one hour before and one hour after addition of a sample gas.
When this gas is colder than the air in the cylinder, the temperature drops briefly, especially in the T1 range (see Fig. 4 ff.). Blind tests using normal air as sample gas show that thermal equilibrium with the original temperatures has established again after just 20 minutes [3].
Thermal conductivity of the gases in the cylinder can be excluded, as experiments with the noble gases argon and helium show [3].
By calibrating with an external radiation source of known intensity, a strictly linear relationship is obtained for the two sensors TD and VP with an identical correlation coefficient of R2 = 0.999 [3].
The respective radiation intensity can be calculated from the TD and VP values and the cross-section area A = 0.0855 m2, whereby the average of both values is used to increase accuracy.
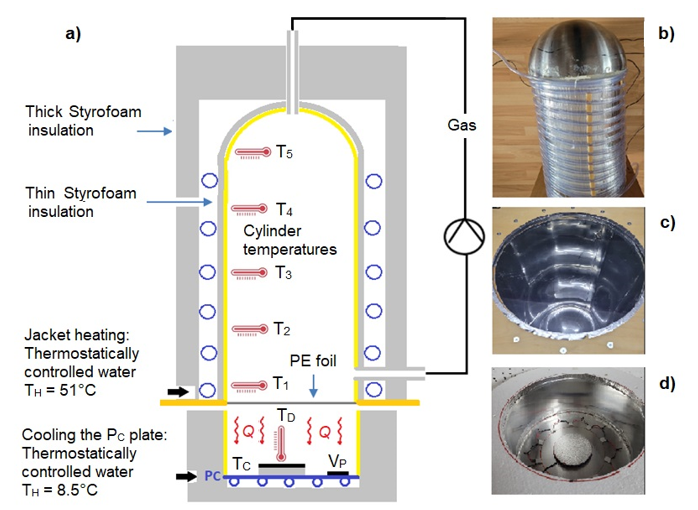
4. The Influence of Water Vapor on Background Radiation
To demonstrate the influence of water vapor, the cylinder temperatures T1 to T5 and the outgoing emission I0 are measured at threedifferent WV concentrations . The heating and cooling temperatures (TW and TC ) are the same as those used in later GH-gas measurements.
The different WV concentrations, except for the T1 measurement point, have minimal or no influence on the cylinder temperatures. This is due to the low WV concentration of only 1.9 vol.% compared to the other GH-gases (which were investigated at concentrations up to 8 vol.%) (see Part 1).
However, significant changes are seen in the background radiation I0 , which increases from 79.4 to 85.1 W/m2 with increasing humidity (Table 1).
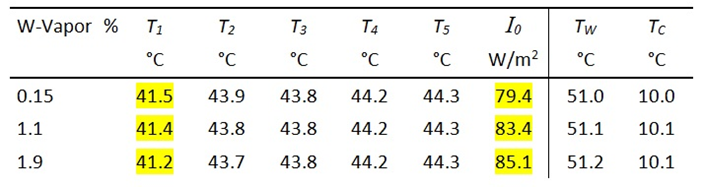
The measured values for this background radiation I0 can be very well reproduced by radiation transfer calculations, when the background radiation from the cylinder walls and transmission losses are taken into account (Fig. 2).
According to these calculations, 0.15 vol.% water vapor inside the cylinder generate an additional radiation intensity of 10.5 W/m2 . However, reaching the PC plate, this value drops to 2.0 W/m2 . In the case of 1.9% WV, only 7.7 W/m2 remain out of an expected WV intensity of 41.3 W/m2.
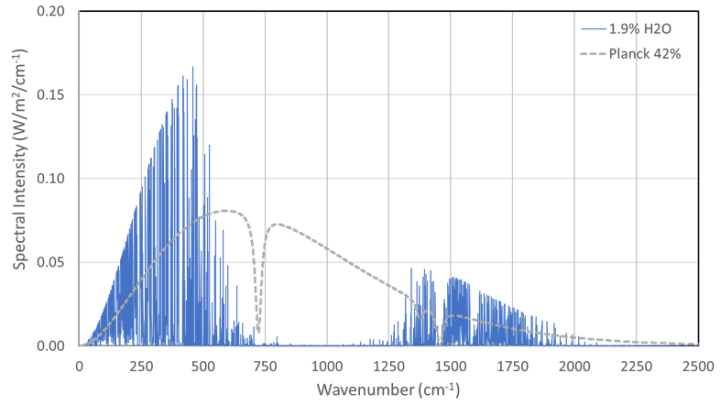
These losses arise from the superposition of the WV radiation with the infrared radiation from the cylinder walls, as well as from transmission losses to the PC plate. The strong attenuations reveal the difficulties in experimentally detecting GH-gas radiation. Figure 3 shows that the WV bands (Blue lines) occupy the same wavenumbers as those of the background radiation from the cylinder walls (Gray dashed line), including the absorption lines of the PE foil at 750 and 1500 cm-1. Due to these superpositions, only the WV intensities that exceed the gray line are effective.
The mutual superposition of the various greenhouse gases, especially with water vapor, occurs according to the same general principle:
The combined radiation of several greenhouse gases is always smaller than the sum of their individual contributions.
Within the atmosphere, there are further superpositions caused by aerosols and clouds. As Planck radiators with a continuous radiation spectrum, these solid and liquid particles can influence the emissions of all GH-gases. Therefore, the CO2 GH-effect is significantly weaker under high humidity and overcast skies than under clear skies and low humidity.
The superposition of WV radiation with that of the other atmospheric greenhouse gases leads to a significantly reduced water vapor feedback. The IPCC assumes a positive feedback by a factor of 2–3 to inflate the effects of CO2 , thus forming the backbone of climate alarmism (here). Without this hypothesis, a doubling of the CO2 concentration would only cause the Earth to warm by a harmless 0.6 to 1 °C (Harde 2013, 2014, 2017 [4 – 6]). Model calculations with a “water vapor feedback” assume so-called tropospheric hotspots, an increase in water vapor and temperature in the upper troposphere, between the equator and 30 degrees latitude. However, observational evidence for this hotspot has remained elusive, and subsequent analyses of radiosonde and satellite data have not confirmed the expected amplification in the tropical mid-troposphere, with some datasets showing trends inconsistent with model projections. See also the discussion in McKitrick & Christy (2018) [7]. Most of the warming since the 1970s has occurred near the surface (here).
5. The IR Emission of Greenhouse Gases
This section compiles the measurements and corresponding calculations for IR emission of the GH-gases CO2 , methane, nitrous oxide, and Freon 134a. All investigations are carried out at atmospheric pressure and constant initial temperatures. In addition to detecting IR radiation, the cooling of the gases, resulting from these emissions is also recorded.
The strongest cooling occurs at position T1 and gradually decreases towards the dome. This temperature gradient is consistent with radiation transfer according to the Schwarzschild equation and the layer model (Schwarzschild 1906 [8], Harde 2013 [4]). The T1 gas layer, located just 5 cm from the PE foil, can transfer its infrared radiation to the PC plate almost unhindered and cools down most strongly. All more distant layers must transport the energy through absorption and re-emission, resulting in attenuation with increasing distance from the PE foil. This effect is particularly evident with nitrous oxide and Freon, as these gases influence all five measuring points T1 – T5 (Subsections 5.1 and 5.4).
The intensity of the IR radiation reaching the PC plate is determined by the temperature or voltage increases of the TD or VP sensors and due to their calibration converted to W/m2 [3].
I0 is the background radiation of the experimental setup before, and IG the total radiation intensity after addition of a GH-gas. By measuring the difference with and without GH-gases, the radiation component of the gas, ΔIGas, can be determined.
For control purposes, the changes in heating and cooling temperatures are also recorded as ΔTW or ΔTC.These values should be approximately zero to demonstrate that emissions and cylinder temperatures are not caused by external factors. This is entirely true for CO2 and methane. For the stronger GH-gases nitrous oxide and especially Freon 134a, however, only the heating ΔTW is approximately zero, while the cooling temperature TC shows a significant increase of up to 0.7 °C, caused by the strong increase in gas radiation.
5.1. Freon 134a Radiation
Tetrafluoroethane (CF2CF3), also known as HFC-134a or Freon 134a, has been used for many years as a chlorine-free (not a CFC) efficient refrigerant. According to EU regulation, it is to be gradually phased out by 2030 on the basis of its measured GWP₁₀₀ of approximately 1,430 – meaning its radiative forcing per molecule is substantially higher than CO₂ over a 100-year horizon. The present experiments confirm that this reflects real IR emission properties, not merely a theoretical construct.
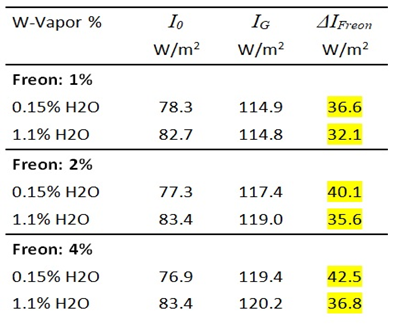
Due to its strong GH-effect, Freon 134a is ideal for demonstrating gas radiation. A concentration of just 1 vol.% leads to an impressive increase in IR emission of 36 W/m² and a temperature reduction of the cylinder air of up to 2.8°C (see Fig. 4 and Table 2).
In view of these clear data, the thesis that greenhouse gases at normal pressure are only absorbers and do not emit infrared radiation or generate counter-radiation is clearly refuted.
Despite its strong effect, the effectiveness of Freon 134a also depends on the WV concentration. At a WV concentration of 1.1 vol.%, the radiation intensity is only about 88% of that of dried air (Table 2b).
Since Freon 134a is not listed as a line-by-line dataset in the HITRAN database, verification by radiation transfer calculations is not possible at this point.
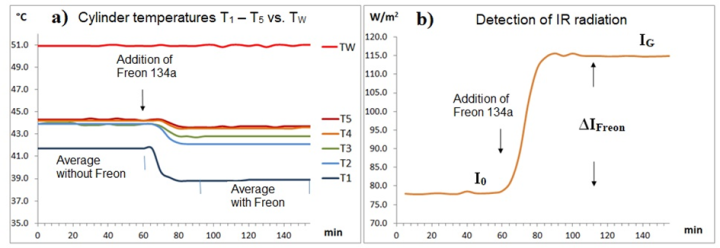
Table 2a: Influence of Freon 134a and WV on the temperatures T 1 – T 5 .
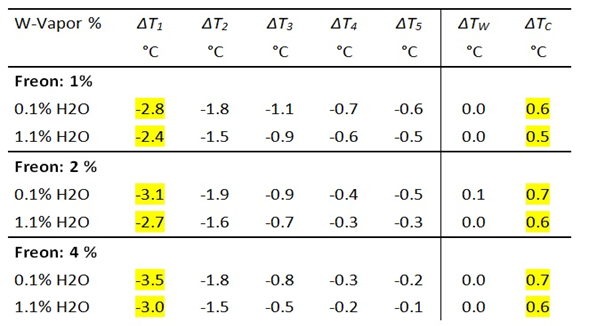
Table 2b: Measurement of IR intensities before and after addition of Freon 134a. DI Freon = Change of Intensity due to Freon radiation.
5.2. CO2 Radiation
The effectiveness of CO2 is much weaker than that of Freon. Therefore, as with the following GH-gases, higher concentrations of 2, 4, and 8 vol.% are used. At these concentrations, the addition of CO2 leads to a noticeable cooling of the T1 and T2 temperatures with a simultaneous increase in the IR radiation intensity ΔICO2 (Fig. 5).

Note: Due to the significantly lower effect compared to Freon 134a , the diagrams for CO2, methane and nitrous oxide are each shown at 8 vol.%.
Table 3a: Influence of CO 2 and WV on temperatures T 1 – T 5 .
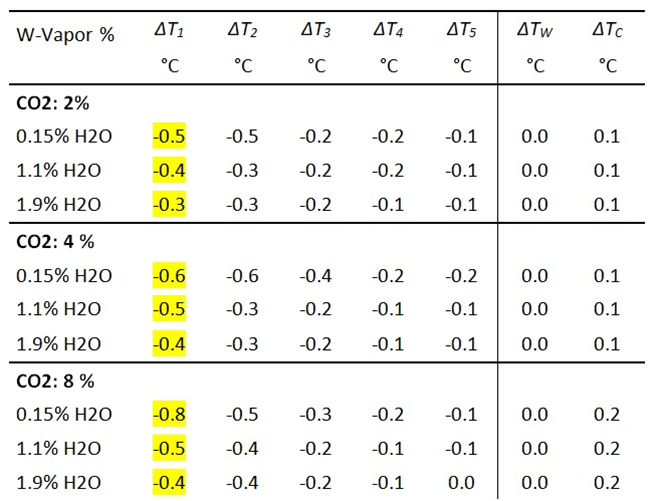
In the case of CO2 , the influence of water vapor is examined at three concentrations (0.15%, 1.1%, and 1.9 vol%) (Tables 3a and 3b). With increasing WV concentration, it is shown that the background radiation continues to increase, leading to a weakening of the CO 2 radiation component. This weakening is particularly noticeable at lower concentrations of WV and CO2 , as shown by the different gradients after the addition of CO2. For example, an H2O concentration of 0.15 % results in an increase of 7.7 W/m2, whereas for 1.9% WV the increase is only 5.7 W/m2 (see Table 3b, last column). This is only 74% of the original intensity. The weakening is due to an increase in the background and thus an increased saturation of the CO2 flanks.
Table 3b: Measurement of IR intensities before and after addition of CO2 .

For verification, the measured data for the total intensity IG are compared with radiative transfer calculations for CO2 and H2O (Fig. 6, Mea = Measured, Cal = Calculated). Details of these calculations, which are also available for methane and nitrous oxide, can be found in [3].
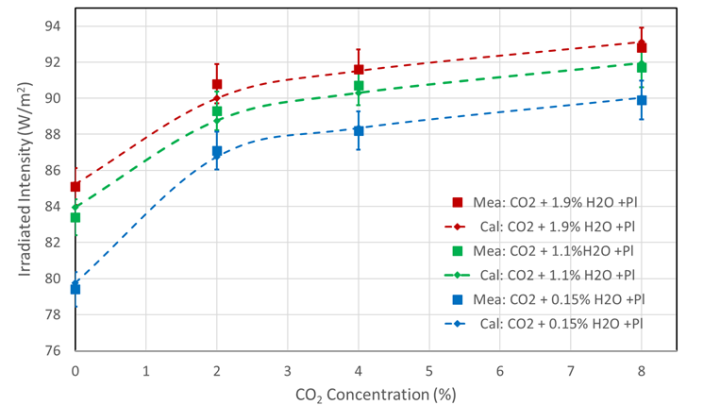
However, due to the long propagation paths in the atmosphere and the 30 – 40 times higher WV concentration compared to CO2,the weak overlap of the spectra around 670 cm-1 (part 1, Fig. 2) leads to a significant limitation of the CO2 climate sensitivity and also to a reduced water vapor feedback (Harde 2014 [5], Harde 2017 [6]).
The different increase in the CO2 radiation intensity at concentrations below and above 2% is striking, changing from an almost linear to a logarithmic curve and reflecting the clear saturation of the absorption and emission processes in the main band around 670 cm-1. For concentrations above 2%, the further increase in intensity is primarily determined by the unsaturated wings and weaker bands.
This phenomenon of a “kink” in the increase in radiation intensity can be observed for all greenhouse gases. This change in sensitivity is the reason why the so-called “global warming potential,” the comparison of a greenhouse gas at very low concentrations with CO2, is a comparison of apples and oranges (see methane).
5.3 Methane radiation
Fig. 7 displays a measurement for 8 vol.% of CH4 in dry air. Methane is considered as a particularly dangerous greenhouse gas because, depending on the timescale, it is said to have a global warming potential 25 to 84 times higher than CO2. This classification paradoxically arises from its low atmospheric concentration of approximately 2 ppm. In this range, the optical density is still very low compared to CO2, so there is a linear increase in the potential. CO2, on the other hand, at 420 ppm, is already in the logarithmic range (see Chapter 5.2). The completely different spectral overlaps with water vapor are another reason for the accusation of “apples and oranges” (see Part 1, Fig. 2).
Global warming potential is often mistakenly confused with effectiveness. In fact, based on equal concentrations, methane is a weaker greenhouse gas than CO2 (Table 4b vs. Table 3b). Theoretical calculations (Part 1, Chapter 2) have already shown that the IR radiation of methane is lower than that of CO2. This has now been confirmed by experiments. Methane is oxidized to CO2 in the atmosphere under the influence of ozone and UV light and therefore has a relatively short residence time of approximately 9 to 12 years.
Table 4a: Influence of CH4 and WV on the gas temperatures T 1 – T 5 .
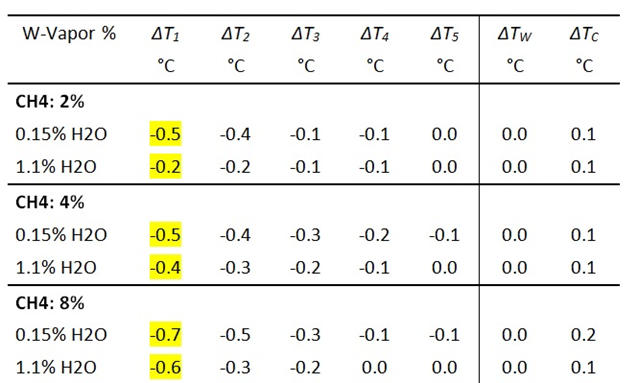
Table 4b: Measurement of IR intensities before and after addition of CH4. DICH4 = intensity change due to CH4 radiation.
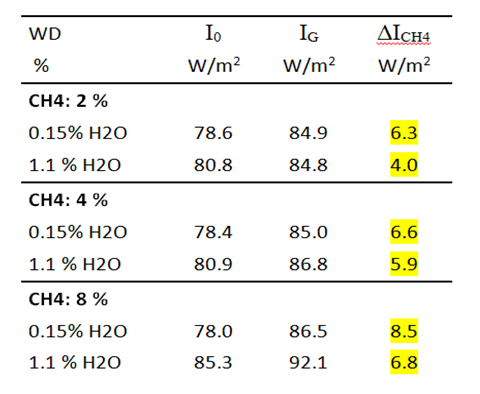
As with CO2, methane radiation also depends on the water vapor concentration. The greatest methane effect is found at a H2O concentration of 0.15 vol.% and is only 78% of the original effect at 1.1% water vapor (Table 4b).
5.4 Nitrous Oxide Radiation
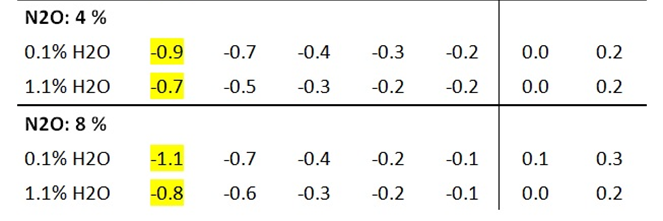
Nitrous oxide (N2O, laughing gas), the third most important long-lived greenhouse gas, is thought to contribute significantly to global warming due to its long atmospheric residence time and its greenhouse potential, which is approximately 300 times higher than that of CO2. In radiation experiments, the effect of N2O is noticeably greater than that of CO2, but only by a factor of 1.5 at the same concentration (Table 5b vs. Table 3b). Even in theoretical calculations, the IR radiation of nitrous oxide is only slightly higher than that of CO2 (Part 1, Chapter 2).
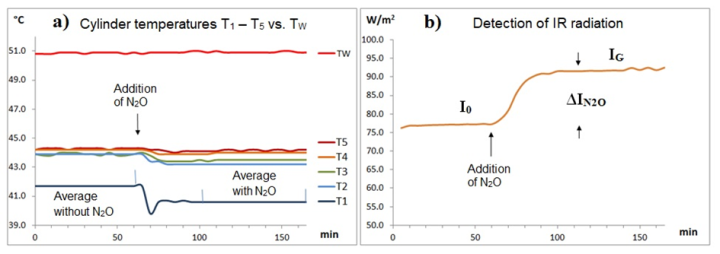
Table 5a: Influence of N2O and WV on the gas temperatures T 1 – T 5 .
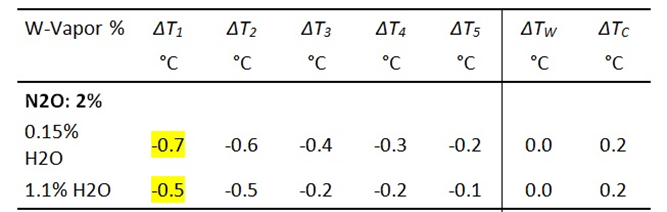
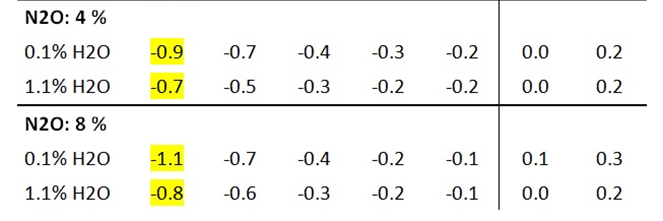
Table 5b: Measurement of IR intensities before and after addition of N2O. DIN2O = intensity change due to N2O radiation
With 0.3 ppm, the concentration of N2O in the atmosphere is 1400 times lower than that of CO2 , raising questions about its effectiveness.
As with CO2 and CH4, nitrous oxide radiation also depends on the water vapor concentration. The greatest effect is again achieved at an H2O concentration of 0.15 vol.% and is reduced to 86% of the original effect at 1.1% water vapor (Table 5b).
6. Summary
This study was performed to demonstrate, on the one hand, the emission properties of greenhouse gases under conditions similar to those in the lower atmosphere and, on the other hand, to demonstrate the existence of the negative GH-effect on a model scale.
For this purpose, a two-chamber vertical set-up was developed. The upper part of this apparatus is a heated gas cylinder, and the lower part is a thermally insulated cylinder with a cooled plate containing thermal radiation detectors.
CO2, methane, or nitrous oxide are added to the upper cylinder and dependent on their concentration, a decrease in air temperatures and a simultaneous increase in IR radiation are observed. This means that these GH-gases can absorb the kinetic energy of their surroundings through inelastic collisions with other air molecules and convert this energy into thermal radiation through subsequent emission.
A cooling of the air accompanied by an increase in thermal radiation is called the “negative GH-effect,” since it is usually the other way around. Such reversals are also part of the Earth’s climate system in inversion weather conditions, the nighttime cooling of the air near the ground, or the air currents toward the winter poles.
The theory that a GH-effect is fundamentally impossible, since collision processes supposedly lead to radiationless deactivation and thus no back radiation, is refuted by these experiments. In the case of only thermalization, the effects of GH-gases would have to be exactly the opposite, because then the air would have to warm up, and attenuating the IR emission, which was not observed.
Although the water vapor concentration can only be increased to a limited extent due to its tendency to condense, a significant attenuation of the effects of the above-mentioned GH-gases was observed in the presence of water vapor.
The often erroneously cited theory that gas radiation violates the second law of thermodynamics does not hold in these studies, as they only consider the heat flow from warm to cold. This eliminates a fundamental argument of the skeptics.
A plausibility check using radiation transfer calculations shows good agreement with the measured data when unavoidable losses are taken into account.
Detecting the gas radiation is anything but simple, as the background radiation from the upper cylinder masks the radiation from the GH-gases, making only a fraction of their radiation visible. Detecting these attenuated effects requires sophisticated technology. In addition to mirrored surfaces, a vertical experimental setup that prevents convection is a minimum requirement.
References
- H. Harde, M. Schnell, 2022: Verification of the Greenhouse Effect in the Laboratory , Science of Climate Change, Vol. 2.1, 1- 33. https://doi.org/10.53234/scc202203/10
- M. Schnell, H. Harde, 2025: The Negative Greenhouse Effect Part I: Experimental Studies with a Common Laboratory Set-Up , Science of Climate Change, Vol. 5.3., pp. 1-9, https://doi.org/10.53234/scc202510/ 02 .
- H. Harde, M. Schnell 2025: The Negative Greenhouse Effect Part II: Studies of Infrared Gas Emission with an Advanced Experimental Set-Up , Science of Climate Change, Vol. 5.3., pp. 10-34, https://doi.org/10.53234/scc202510/03 .
- H. Harde, 2013: Radiation and Heat Transfer in the Atmosphere: A Comprehensive Approach on a Molecular Basis , International Journal of Atmospheric Sciences (Open Access), vol. 2013, http://dx.doi.org/10.1155/2013/503727
- H. Harde, 2014: Advanced Two-Layer Climate Model for the Assessment of Global Warming by CO2, Open Journal of Atmospheric and Climate Change, Vol. 1.3, November 2014, https://web.archive.org/web/20160429061756/http://www.scipublish.com/journals/ACC/papers/download/3001-846.pdf .
- H. Harde, 2017: Radiation Transfer Calculations and Assessment of Global Warming by CO2,International Journal of Atmospheric Sciences, Volume 2017, Article ID 9251034, pp. 1-30, https://doi.org/10.1155/2017/9251034 .
- K. Schwarzschild, 1906: Über das Gleichgewicht der Sonnenatmosphäre. In: Nachrichten von der Königlichen Gesellschaft der Wissenschaften zu Göttingen, Mathematisch-Physikalische Klasse, 1906, Heft 1, pp. 41–53 (13. January 1906).
- R. McKitrick, J. Christy, 2018: A Test of the Tropical 200- to 300-hPa Warming Rate in Climate Models, Earth and Space Science, 5, 529–536, https://doi.org/10.1029/2018EA000401
A plausibility check
This is old paradigm thinking. Isn’t avoiding any real bona fide validation – save for climate models; averaged – the key to “the science” of the climate? 2100 has long been their go to year and they know full well that a person born today would be in their mid 70s in 2100.
Could this situation ultimately be influenced by those other famous users of massive supercomputers: hyper-intelligent pan-dimensional beings (presenting as mice). After all, Deep Thought took some 7.5 million years to arrive at an answer of 42.
That required an even bigger computer….
“a person born today would be in their mid 70s in 2100”
I’ll be 150. 🙂
I wonder what the contemporary scares will be in 2100?
I’ll only be 139!
And yet using an “open-system” this controlled lab experiment, this one produced data proved that water vapor constituted ~95% of the natural greenhouse gases, and that CO2 at its current atmospheric concentration (0.042%) had no measurable effect on the absorption temperature. This is directly inconsistent with the 2007 IPCC assessment report which predicted that if the natural CO2 concentration were doubled to 842 ppmv, it would increase the atmospheric temperature by 5.4˚F based on mathematical modeling. In this laboratory test, a 15-fold increase only raised the absorption temperature by 1.2˚F. This means the IPCC’s mathematical model is off by a factor of 31 times.
This data also shows the inconsistency with the CO2-water vapour control knob theory.
https://www.scirp.org/journal/paperinformation?paperid=145896
Reply canceled, was a general reply…
Reproduced here, as it is not a reply to Macha:
As a repeat of a similar objection in part 1:
The earlier experiment used an electrical heating element to heat up the atmosphere in the upper part of the cylinder.
That gives a constant input of energy Q(in) to the inside air, regardless of its temperature.
That experiment didn’t show much “cooling” effect of GHGs at near ground pressure and temperature.
This experiment uses a water jacket at a fixed temperature, which gives a different input Q(in), depending of the temperature difference between water and inside air.
As GHGs do absorb and emit IR at any temperature, the increase of GHGs anyway will give more downwelling IR that is detected by the cooling of the atmosphere and the instruments at the bottom, which give Q(out).
The point is that without measuring or calculating the input Q(in), one can’t say that the real atmosphere is gaining or loosing energy, as that depends of the sign of Q(in) – Q(out).
That doesn’t mean that there is no effect at all, as indeed some days and at some parts of the globe, the atmosphere is warmer than the surface, but this experiment doesn’t quantify that.
From post:”As GHGs do absorb and emit IR at any temperature,…”
Gases are not black bodies and do not emit based on temperature. CO2 has three emission frequencies about 2.5 micro, 4.7 micro, and 15 micro. The atmospheric window shows this.
Sorry, I should have been more detailed: GHGs absorb and emit in specific wavelengths, independent of their own temperature…
“Gases are not black bodies”.
I get your point, that gases are not cavity radiators. But the earth’s atmosphere is black at wavelengths of sufficient optical depth. Black (thermal) radiation comes from matter of radiative absorption high enough that matter and photons are (nearly) in thermal equilibrium. The earth’s atmosphere is black at 15 micron as can be seen from measurements, and not in the atmospheric window as you correctly observed.
Black bodies also don’t have specific heat capacities, Earth does.
Demonstrating the reverse….
More drilling in North Sea ‘not the answer’ for UK energy security, say former military leaders
“…the UK needed an energy strategy that was “focused on greater energy efficiency to reduce demand”. – Retired R Adm Neil Morisetti, a professor of climate and resource security at University College London – Guess who
To coin a phrase, wheeling out the big guns. A growing economy will by definition require more energy, not less. Given that the UK now has no military it makes perfect sense that idle Rear Admirals etc become a professor of climate of one sort or another. That and not national defence is where the money is.
When you have more admirals than warships…
You mean all these people whose main aim is stars on shoulder pads and spout the latest war-monger propaganda
I suppose all this stuff is of some intellectual academic interest, but the thing we should really worry about is not the effects of CO2 on the climate. Its the consequences of the lunatic efforts to move everything to electricity, and at the same time move electricity generation to wind and solar, while having no solution to intermittency. It cannot be done, and its main result will be blackouts and rationing in the few countries that seriously try it.
This is the important practical issue. Arguments about the effects or lack of them of CO2, well, whatever they are they are going to happen, because without any solution to intermittency no-one outside a few English speaking countries is seriously trying, or going to try, to reduce emissions.
That’s the argument to have. Whether you are right or wrong about the climate, all you are going to do is produce blackouts in London, without having any effect on the climate.
So please just stop it!
‘I suppose all this stuff is of some intellectual academic interest…’
More than that, unfortunately. Accepting the phenomenological physics of radiative transfer theory is equivalent to accepting the alarmist narrative that CO2 is the ‘control knob’ of the Earth’s climate. One hand washes the other, which the Left has taken full advantage of.
Frankly,……
It is amazing that engineers go to jail if they screw up, while climate scientists get ever bigger budgets to prognosticate on settled science.
The paleo record shows cause (co2) follows effect (warming), reversing the arrow of time.
Yes they (mostly) do, with one important exception – the PETM.
The Earth’s carbon cycle naturally works with temp leading.
IE: warming temps cause greater vegetative decay and SST’s to warm, both of which cause a rising atmospheric CO2 concentration.
The current situation, and that of the PETM is that of an injection (not meaning of a short time-scale) of a large quantity of CO2, which overwhelms the CC, such that it cannot be fully absorbed by the biosphere, and over time it accumulates in the atmosphere.
The PETM:
“The Paleocene-Eocene Thermal Maximum (PETM) was a rapid global warming event ~56 million years ago, where temperatures rose by 5–6°C over a few thousand years. Driven by massive carbon releases (likely volcanic), it caused severe ocean acidification, major ecosystem shifts, and benthic extinction. It is a key geological analogue for modern climate change”
stupid question from a person with minimal physical chemistry background.
wouldn’t the higher pressures near sea level push CO2 molecules closer together and favor pushing LWIR back into space?
just asking.
Many times, I have hoped in public for more experiments of this type.
However, despite 50 years of scientific study myself, I cannot comprehend the essence of this paper by Harde and Schnell. In general, I appreciate the difficulty of writing complicated concepts when English is not the primary language of authors and I lament their consequent failure to communicate what might be significant scientific measurements.
I suggest that a capable scientific author collaborates with the present authors to create a version in more readable English, which is a major language for such purposes.
Specifically, it is difficult to use part of the brain to try to translate while another part runs below optimum when trying to comprehend the science. Geoff S
@ur momisugly sherro01
He writes: ‘I suggest that a capable scientific author collaborates with the present authors to create a version in more readable English, which is a major language for such purposes.’
Thank you for the very constructive suggestion. Perhaps with your 50 years of scientific experience, you are ready to collaborates with us to create a version in better understandable English?
In any way, despite our terrible English, I am surprised that an expert who has followed the climate debate for so many years, cannot recognize what these studies should demonstrate.
Since Wood’s experiments in 1909 and the continuous discussion about the existence or nonexistence of the GHE (see also this blog, e.g., Anthony Watts, Kevin Kilty, Siim & Olsen or the German blog EIKE here and here) sceptics argue, such effect would violate the 2nd law of thermodynamics; or radiation under conditions as found in the lower troposphere can only be absorbed and thermalized but not reradiated by greenhouse gases due to the dominating collisions.
Even with bad English it should be clear that the graphs show a distinct increase in the detected IR radiation with increasing GHG concentration in the cylinder and simultaneously a decrease in the temperature of the gas mixture. To our knowledge this is the only experiment, which shows such a correlation. Readers are free to draw their own conclusions from this.
What we tried with these publications was to contribute to real science based on reproduceable observations and to limit the endless speculations. But of course, this does not preclude any criticism of our approach and presentation.
Also a short reply to Ferdinand Enelbeen’s comment: ‘The point is that without measuring or calculating the input Q(in), one can’t say that the real atmosphere is gaining or losing energy, as that depends of the sign of Q(in) – Q(out)’.
For the real atmosphere the incident energy flux Q(in) can only be estimated by a total energy and radiation balance of the Earth-atmosphere system. The uncertainties involved with such balance are illustrated by the current paper of Cohler et al. In our set-up a well-controlled temperature of the inner cylinder jacket is ensured by the water heating. This supplies the jacket with a constant influx to compensate for any losses. Without GHGs we measure typical radiation losses at the sensors of 77 W/m2, caused by the polished inner walls. With GHGs this increases, with 2 % Freon even by 40 W/m2, and at the same time the gas temperature reduces. At new equilibrium the additionally radiated and detected flux can only be supplied by the incident heat flux , which is increasing accordingly. Finally it holds Q(in) – Q(out).
I would be delighted to help improve the English readability, but it needs you authors at the same table as the translator for several days face-to-face to convey your deeper intent accurately.
I live in Australia and you do not. At my age of 84, health issues prevent air travel.
I am not being critical of your experiment. I simply cannot follow your thought processes well enough to comprehend. I am not expressing any “sceptical” attitude to send me to your naughty corner. As I said, I appreciate more of this type of lab experiment and am simply suggesting that your work could be made more readable and so more valuable. Geoff S
“Even with bad English it should be clear that the graphs show a distinct increase in the detected IR radiation with increasing GHG concentration in the cylinder and simultaneously a decrease in the temperature of the gas mixture.”
This does *NOT* mean an increase in the temperature of the source of the increase of radiative flux, i.e. the Earth. Temperature is driven by heat (joules), not flux (joules/sec). If the increase in IR radiation is merely the return of heat (joules) already emitted by the source it will not raise the temperature of the source beyond what it was when the radiation was actually emitted by the source. If the reflected IR is only a portion of that emitted by the source and not a complete reflection then the temperature of the source will decrease, it will continue cooling although perhaps at a slower rate.
Heat (joules) is a time function, especially in a non-blackbody that has thermal inertia. You can’t use instantaneous measurement in incoming radiative flux to project temperature of the source. The temperature of the source is dependent on what happened before the measurement, not what is happening in the moment of measurement.
Reply to Tim Gotman
He writes: ‘This does *NOT* mean an increase in the temperature of the source of the increase of radiative flux, i.e. the Earth. Temperature is driven by heat (joules), not flux (joules/sec) …’
It’s not clear what this comment is actually referring to. In a preceding reply I stated that with GHGs the measured intensity at the detectors further increases – compared to the background radiation caused by the cylinder walls – and at the same time the temperature of the gas decreases. I didn’t talk about a temperature increase at the source of the increased radiation flux, just opposite.
To make this clear: With the detectors below the PE-foil we measure the IR-radiation escaping from the upper cylinder. In addition to mechanical heat fluxes (conduction, sensible and latent heat) in the standard literature this radiation is also known as heat flux and the radiation intensity specified in W/m2.
The source of the additional radiation is the GHG, which due to this continuous energy loss is cooling down. Without new excitation by collisions with the buffer gas, and thus without heat exchange, the radiation would come to an end. But with an increasing temperature difference to the cylinder jacket also the heat flux from the walls to the gas volume is increasing and so ensures a further continuous radiation level, which is measured as intensity.
As the inner wall (polished aluminum foil) is separated from the outer thermostatically controlled jacket by a 2 mm thin insulation layer, the temperature gradient between the outer PVC hose and the inner wall – thus also the heat flux to the gas volume – is essentially determined by this isolation layer. Therefore, also the inner wall is slightly cooling down, at least at the bottom of the cylinder (temperature T1). This and the reduced temperature of the gas demonstrates radiative cooling by GHGs, how this occurs in the upper atmosphere as radiation to space and also under inversion conditions (increasing temperature with altitude) in the lower troposphere.
Tim Gorman writes: ‘The temperature of the source is dependent on what happened before the measurement, not what is happening in the moment of measurement.’ He should look at our measurement procedure (Section 3, Preparatory Studies). He will see that our measurements represent equilibrium conditions, which typically establish within 20 minutes, but the specified data are average values derived one hour before and one hour after addition of a sample gas. This holds for the diverse temperatures in and outside the cylinder and also for the radiation with Q(in) = Q(out).
It seems to me that your experiments address certain questions I have had about the ongoing arguments about ‘back-radiation’ and the collision thermalization of radiation, as you actually mention in your paper.
The collision thermalization argument makes sense, because the collision rate is much faster than the spontaneous emission time. But this would mean (correct me if I’m wrong) that the radiative cooling can only happen in the upper atmosphere where the collision rate is much smaller.
However, we know that the atmosphere, at ground level, radiates under inversion conditions. Observations performed in the 1950s of the clear, night sky, and in the arctic, measured the atmosphere radiating at ground level. I never see this mentioned in the articles about back-radiation and thermalization.
It seems to me that the micro physics argument on the collision thermalization might need to be amended. The inelastic collisions that produce the population inversion cannot radiate by spontaneous emission because its time is too long. But there is a thermal radiation ‘gas’ existing with the molecules, and could we expect induced emission to complete the reradiation process?
Reply to apsteffe:
He writes: ‘The collision thermalization argument makes sense, because the collision rate is much faster than the spontaneous emission time. But this would mean (correct me if I’m wrong) that the radiative cooling can only happen in the upper atmosphere where the collision rate is much smaller.’
Exactly this conclusion is often found. However, such interpretation overlooks that the typical collision rates of several GHz, as they are observed in the lower atmosphere, are only reducing by a factor of 4 – 5 at an altitude of about 11 km and therefore are still some 100 million times larger than the spontaneous transition rate on the CO2 bending mode (~1 Hz). When such interpretation would be true, there would also be absolutely no emission in the higher atmosphere.
Instead, what can be observed is just the opposite. Our measurements clearly demonstrate that also at normal pressure GHGs are emitting. This was one of the reasons for performing these experiments which show full agreement with theory. At the tropopause at 11 km altitude the radiated intensity of CO2 is just 12% of the intensity observed in a 100 m thick gas layer close to the ground. Next to a reduced gas density the main reason for this lower emission rate is the lower temperature. For a detailed explanation of spontaneous emission in the presence of collisions and its temperature dependence, see: Harde 2013, subsec. 2.5, also Harde & Schnell 2022, subsec. 3.2, shortened version, subsec. 4.2.
Our measurements are also supported by Kirchhoff’s law (see, e.g., Goody & Yung, 1989 Radiation: Theoretical Basis; and Harde 2013, eq.(89)). According to Conrad Ziefle that is first year thermodynamics (see this blog). Therefore, different to the introductory editorial notes of WUWT to our articles we certainly see our studies as an endorsed and settled account of atmospheric physics.
You write: ‘I never see this mentioned in the articles about back-radiation and thermalization.’
See Part 1, section 6 and the original paper Harde & Schnell 2025, subsec. 4.4.
You ask: ‘But there is a thermal radiation ‘gas’ existing with the molecules, and could we expect induced emission to complete the reradiation process?’
The thermally radiating gas is the GHG which determines the background radiation, for which Einstein concluded that a radiation field, interacting with the molecules at thermal and radiation equilibrium, just had to be of the type of a Planckian radiator. As shown in Harde 2013, subsec. 2.5, the origin of this thermal background radiation is just the spontaneous emission of the molecules. Because of its origin this radiation only exists on discrete frequencies, given by the transition frequencies and the linewidths of the transitions. But on these frequencies and over longer paths the radiation strength is the same as that of a blackbody radiator, and at thermal equilibrium it is only controlled by the gas temperature TG.
In the atmosphere this has been observed as upwelling radiation by satellites (Data Courtesy of D. Tobin, 1986: Space Science and Engineering Center, University of Wisconsin-Madison) and as downwelling radiation at ground-stations (Arnott 2008: University of Nevada, Reno, ATMS 749; Feldmann et al. 2015). Comparison with line-by-line radiation transfer calculations show excellent agreement (see Harde 2013, Figs 21 and 22).
On the other hand, induced transition rates do not matter. They are only about 3 – 4 % of the spontaneous rates.
“ He will see that our measurements represent equilibrium conditions”
This raises the question of how do your measurements at equilibrium relate to the real world? Radiation out and radiation in cannot balance at any moment in time in the real world, e.g. equilibrium, because of the different time intervals involved over the diurnal cycle. Equilibrium in the real world for radiative flux would mean the earth is losing far more heat than it could possibly gain from the sun.
It appears to me that all your experiment really shows is that adding radiative molecules to a parcel of air causes an increase in the radiative flux from that parcel of air. It does not prove that a reflective object (i.e. CO2 in the atmosphere) can raise the temperature of the source object beyond the beginning temperature the source object. By retarding the cooling rate of the source object, it actually causes an increase in the actual heat loss from source object because it remains at a higher temperature for longer. The heat loss is the integral of the temperature profile, an exponential decay function. As the slope of the temperature curve increases toward zero (i.e. less negative) the greater the integral will be, i.e. more area under the curve.
This is part of what Planck spoke of as “compensation”. I’m not seeing where your experiment invalidates the concept of compensation.
Reply to Tim Gorman
He writes: ‘This raises the question of how do your measurements at equilibrium relate to the real world? Radiation out and radiation in cannot balance at any moment in time in the real world, e.g. equilibrium, because of the different time intervals involved over the diurnal cycle. Equilibrium in the real world for radiative flux would mean the earth is losing far more heat than it could possibly gain from the sun.’
Why people are performing laboratory experiments? Often, they do this to study processes under well controlled and reproduceable conditions, which in reality are superimposed by perturbations or are running so fast that any details cannot be observed or characterized accurately enough (see example below).
Also, everyone knows that the signal-to-noise ratio of a measurement is improved by reducing the bandwidth and integrating over longer times. This comes at the cost of reduced time resolution.
When there would be available highly sensitive and fast detectors in the mid-IR, comparable to photon-counting with photomultipliers in the visible and UV, we hadn’t to integrate with temperature sensors or piezoelectric elements over longer times. But anyway, to show the difference without and with GHGs, before starting a measurement we have to wait, till quasi-equilibrium has established. Otherwise it cannot be clearly verified the emission of GHGs under regular pressure conditions and at the same time the cooling of the gas.
Then he writes: ‘It appears to me that all your experiment really shows is that adding radiative molecules to a parcel of air causes an increase in the radiative flux from that parcel of air. It does not prove that a reflective object (i.e. CO2 in the atmosphere) can raise the temperature of the source object beyond the beginning temperature the source object.’
At least, from the first sentence I see that he agrees, GHGs are emitters also at regular pressure. The second sentence doesn’t make too much sense to me. Everyone knows that GHGs are not reflecting, there can be Rayleigh scattering of solar radiation or absorption and emission – the latter sometimes also called resonant scattering, but no reflection; and in these two WUWT-papers we didn’t claim that an ‘object’ could raise the temperature beyond the beginning temperature of the source. Here we discuss just the opposite, reverse GHE. But when he wants to have an example, how to prove the regular GHE, he should look to Harde & Schnell 2022.
A special example for extremely fast transients and their measurement is shown below: Femtosecond THz-pulses are propagating through methyl chloride vapor. (details see Harde & Grischkowsky 2015).
Such a pump-probe measurement takes about 1 hour. Nevertheless it samples with femtosecond resolution the electric field of the incident pulses (upper graph), which are reshaping over time and propagation length due to dispersion and absorption in the vapor (lower graph); and they are followed by a series of coherent transients, which appear every 40 ps as commensurate echoes on the free induction decay. After excitation, the CH3Cl molecules are freely oscillating on individual rotation frequencies with the consequence that the individual oscillators are periodically dephasing and rephasing. This appears with a beat frequency of 25 GHz, given by the frequency spacing between adjacent rotational lines and results in the formation of a macroscopic polarization and the reemission of the periodic echoes. The relatively fast decay of the echoes is a consequence of line broadening due to the molecular collisions.
Even with fastest high-speed camera techniques such observations wouldn’t be possible. But with respective integration for each time-delay the S/N-ratio is better 1000:1.
“The second sentence doesn’t make too much sense to me. Everyone knows that GHGs are not reflecting, there can be Rayleigh scattering of solar radiation or absorption and emission”
Planck doesn’t require “reflectors” to be mirrors, only that they return energy originating at a source back to the source. That is exactly what CO2 in the atmosphere does.
Planck: “For example, if we let the rays emitted by the body fall back on it, say by suitable reflection, the body, while again absorbing these rays, will necessarily be at the same time emitting new rays, and this is the compensation required by the second principle.”
I will raise one additional point. If a parcel of air contains sufficient CO2 molecules to be considered an isotropic radiator, then that parcel will return an amount of radiation back to the earth that diminishes with altitude. This involves at least two processes. 1. the angle the earth subtends on the isotropic radiator (i.e. a parcel of CO2) decreases and, 2. a significant portion of the radiation in the subtended angle will be intercepted by CO2 at lower elevation and converted to kinetic energy. It’s kind of like a Plinko board, lots of bins at the bottom but a ball (packet of radiated energy) dropped at the top doesn’t always fall into the bin directly below it.
Perhaps I’ve missed it somewhere but I’ve never seen an attempt to quantify exactly how much source-energy from the earth actually gets returned back to the earth. Lots of assumed amounts – but guesses are guesses.
Yeah, ε=α. We’re taught that is first year thermodynamics. How could it be ignored so long? So the water vapor (clouds) at high altitude radiate heat to space, with very little interference.
Anthony wrote:
“The often erroneously cited theory that [atmospheric] gas radiation [power to the surface] violates the second law of thermodynamics”
Hmm. What do you think the Second Law actually says, Anthony? And what do you think “radiation” is? Because energy transfer from a colder object to a warmer one does violate the Second Law.
“does not hold in these studies”
The Second Law holds in every study, including this one, of course.
“as they only consider the heat flow from warm to cold.”
That is the only direction in which heat can flow. And, of course, the only direction in which anyone has ever seen it flow. (Technically, heat doesn’t “flow” in the first place, but we’ll let that pass for now, as a colloquialism…)
“This eliminates a fundamental argument of the skeptics [of radiant greenhouse effect theory].”
It does no such thing, of course.
Better late than never.
Earth is cooler w atmos/water vapor/30% albedo not warmer.
Ubiquitous GHE balance graphics don’t + violate GAAP & LoT.
Kinetic heat transfer processes of contiguous atmos molecules render “extra” GHE energy from a BB surface impossible.
GHE = bogus & CAGW = scam.
Y’all be bickering over the mechanism behind a process that does not exist.