On February 11th, NBC News published a story by writers Kenzi Abou-Sabe, Yasmine Salam, and Cynthia McFadden titled “Can giving the ocean an antacid help curb climate change?,” describing ways to artificially induce the world’s oceans to capture and store more carbon dioxide.
Whether artificially altering the oceans chemistry is a worthwhile or valuable goal to fight climate change is unclear, what is fairly clear, it is impractical due to scale, and could possibly produce unintended negative consequences.
The NBC News story is perhaps best summed up in the first few paragraphs of the story:
Just over a year ago, Canadian oceanographer Will Burt was in Fairbanks, Alaska, teaching college students about the effects of global warming on marine life when a former colleague approached him about a startup seeking to use the ocean to remove carbon from the atmosphere.
“I didn’t have to think about it,” Burt said.
…
Burt works for Planetary Technologies, a Canadian startup that’s attempting to harness and accelerate that potential by adding antacid powder to the ocean.
The theory goes that by altering seawater chemistry, the ocean’s surface could absorb far more atmospheric carbon than it does naturally.
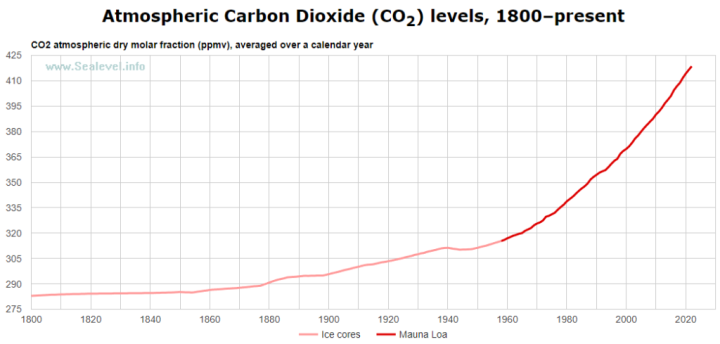
The premise is that with human intervention the world’s oceans could absorb more carbon dioxide (CO2), to be dissolved in them, in order to slow or reverse the growth of CO2 in the atmosphere. However, Burt and other researchers seem to misunderstand basic chemistry, which tell us that as the temperature of the oceans increase, CO2 will outgas from it. This has been known for decades, as seen in the figure below.
So, just like soda pop fizzes out more carbonation when it is warm vs. cold, the oceans outgas more CO2 as they warm. The Earth is modestly warming, and has been since about 1850. As water temperature increases with a warmer planet, CO2 solubility becomes less, and the oceans outgas more CO2. As seen in Figure 2, CO2 has been increasing in the atmosphere, partly due to human contributions, but also due to outgassing of CO2 from the oceans as the Earth warmed since the end of the Little Ice Age, which ended around1850.
Adding antacids won’t have any effect on the temperature dependent chemical process in the oceans, and these researchers will soon discover that their efforts would be miniscule in the big picture.
Of course, when Burt said “I didn’t have to think about it.” he was correct, and any logical person can see the folly of his quest just on the impossibly large scale alone, never mind the uphill battle against decreasing CO2 solubility and ocean outgassing as temperature increases.
And, have oceanographers like Burt given any thought whatsoever to dumping huge amounts of an antacid compound into the ocean? On one hand, we are told the oceans are fragile, on the other hand we are told by Burt and others they should be used for an experiment in industrial-scale antacid tests. The article says, “Planetary intends to recycle mine waste from a defunct asbestos mine in Quebec to produce pure magnesium hydroxide, which the company believes would help accelerate the ocean’s carbon uptake ability in the areas where it’s used.”
Dump asbestos mine waste in the ocean? Surely there must already be an environmental law against such a thing.
The idea that we ought to attempt to intentionally alter global ocean chemistry by dumping refined asbestos mine waste into them is wrong-headed. Even if it made sense, with no unintended consequences, the scale needed to appreciably reduce atmospheric CO2 is likely impossible.

Anthony Watts is a senior fellow for environment and climate at The Heartland Institute. Watts has been in the weather business both in front of, and behind the camera as an on-air television meteorologist since 1978, and currently does daily radio forecasts. He has created weather graphics presentation systems for television, specialized weather instrumentation, as well as co-authored peer-reviewed papers on climate issues. He operates the most viewed website in the world on climate, the award-winning website wattsupwiththat.com.

The California school system gave me an aversion to math, but any dumping of antacids in the ocean would be orders of magnitude too small.
Rather like the odds of reaching escape velocity by putting your head between your legs and spitting.
The problem is there is no problem with water chemistry; it works very well. The problem resides in their heads.
Yeah, Planetary Technologies fails to recognize at least a couple of facts.
The oceans already are basic in pH, not acidic, and whatever they do is just a fart in the wind. Of course, they are happy to fart in wind so long as they get paid for it.
Seeing as how their goal is to collect a few $million via the “Donate” button on their website so they can perform tugboat scale ocean experiments, it is quite possible they will be a successful start-up…..results, nah….
Worth of your attention?
This is, one might say ‘unprecedented’. We have a new historical data record (going back to 1979) for the month of February (after the all time minimum was equalled just 6 weeks ago) for the SSW (sudden stratospheric warming), making it 50 degree C difference for the six weeks period.
The SSW spike may be an early warning of another very cold Arctic blast heading for Canad, USA and possibly NW Europe.
Do you mean the “cold” already on the move, or do you imply continuing cold in March?
This is current state at 10hP
https://www.ventusky.com/?p=53;-44;1&l=wind-10hpa
(You can blame Russians and Putin the Grozny)
It might take few days for change to propagate further down. I suspect polar vortex is breaking down and losing control of the jet stream, which will turn from mainly zonal to pronounced meridional circulation, with cold Arctic air masses moving into southern Canada and the USA.
Why is this happening now.
Number of years I formulated hypothesis that explosive volcanic eruptions in Kamchatka push a dome of warm air into stratosphere which at 56 deg latitude can be as low as 8 km (5 miles).
Schiveluch (height aprox 3.3km/2 miles) erupted explosively for nearly a week 10 days ago.
see report from Smithsonian
https://volcano.si.edu/volcano.cfm?vn=300270
Some of the readers not too familiar with the above, might find it surprising that stratospheric temperature (10hP- 30km altitude) over the N. Pole (-22C) is more than 30C (almost 90F) degrees warmer than over the Central USA (-55C)
https://www.ventusky.com/?p=53;-124;1&l=temperature-10hpa
What is to follow:
I expect that large loop over N. Canada to narrow down a bit and to centre just N.W of the Hudson Bay and the Russian one widen and move further east over Central Siberia north of lake Baikal.
If that happens instead one strong polar vortex we will have two weak ones, and jet stream will loop to much lower altitudes.
If so this interactive NOAA’s forecast for a week from now might look very wrong with the hindsight (select date by clicking along the box below current date).
https://www.netweather.tv/charts-and-data/global-jetstream#2023/02/26/1500Z/jetstream/surface/level/overlay=jetstream/orthographic=-87.87,44.70,356
Is is the Arctic blast of cold air on the way?
All signs are there, I rate probability of more than 50-50.
Four days ago I sugested that polar vortes is about to split (https://wattsupwiththat.com/2023/02/18/no-nbc-news-giving-the-ocean-antacids-will-not-help-curb-climate-change/#comment-3682889
back date Ventusky to 18. Feb)
According to Venusky that has happened already.
The SSW is almost over due to Kamchatka explosive eruption of two weeks ago, now electrostatic charge and geomagnetic field are in charge.
For time being MetOffice (30 day forecast) is not too certain:
“Temperatures will most likely start off around average, but there is increasing likelihood of colder conditions (relative to average). Cold nights are likely in places throughout the period.”
Nevermind the destruction to the sea life that would happen. Truly these people are mad.
Plastic straws kill everything in the ocean, but changing its fundamental chemistry? Where do we sign up for that?
One thing you can say for the Roman Empire is that when it finally did collapse it didn’t take the planet with it.
“The premise is that with human intervention..”
Isn’t it always? Polar bears, penguins ad nauseam will not survive [whatever] without human assistance of some sort.
Isn’t that a bit of an insult to nature? I think it is.
I just read the 1991 Jacques Cousteau UNESCO interview in which he blames humans for creating the Sahara desert. Apparently Homo habilis had terraforming capability.
The ocean already does this. Calcium carbonate (Limestone eventually) precipitates out of WARM shallow seas. CO2 is tied up with bicarbonate ions and dissolved alkaline earth minerals when the water is very cold. The soluble bicarbonates break down to CO2 and Ca or Mg carbonates when the water warms (or some living creature exspells it. There is about 1.7 g/l of Mg and Ca (combined) in the ocean wheras the concentration of CO2+bicarbonate is closer to ~0.1g/l in its various forms. The solution is not more alkaline earth, its heat in shallow parts of the ocean.
I didn’t see your more complete explanation when I posted mine. Good job.
Interesting point about temperature as a catalyst although I’m not 100% clear if you are saying warming would accelerate precipitation? It does seem you are saying that by relative dissolved weight there is plenty of Mg and Ca already available.
Of course, what is missing in the story (and Anthony’s retort) is these folks are not talking about dissolving more CO2 in the ocean (other than incidentally) but about precipitate mineralization. e.g. here is a paper about where to get the MagnesiumHyrdoxide that explains the effort is for mineralization not for higher dissolved concentrations of CO2, i.e., regardless of water temperature, if you mineralize dissolved CO2 then there is less in solution and more can be dissolved, rinse and repeat.
While I’m skeptical that climate change and CO2 are a particularly significant problem and engaging this on a massive scale isn’t justified by present knowledge, I respect people who are looking at sequestration as a method for reducing CO2 if that were truly to prove desirable and some good thinking is applied to unintended consequences. Although 10 demerits to this post for tryng to take advantage of the fact that this magnesium hydroxide would be sourced from asbestos mine waste. This is playing on fears the same way that climate catastrophists do and completely unnecessary. Asbestos, arsenic, etc. are naturally occuring. Concentrations can be impacted by failing to sequester tailings safely, but the point in the paper I linked is that production of Magensium Hydroxide can be carbon intensive so existing offal might be turned into something useful. Of course it has to handled to minimize other contaminants of concern but there is already plenty of natural transport of Asbestos into the ocean so some kind of holier than though standard of perfection is not necessarily called for.
Regardless of source, as NBC did manage to report, introduction of magnesium hydroxide is intended to mimic natural processes where the substance is naturally washed from weathered minerals into rivers and thence to the ocean where the bulk of the remineralization reaction can then take place. (they just forgot to describe what the reaction is but they did not even use the word “dissolve”, they used the word “Absorb” which metaphorically describes what’s going on but leaves out the chemistry involved.)
I can recall Ian Plimer’s impressive presentations at several Heartland Conferences that first got me thinking of mineralization of dissolved CO2 as something to consider not on an eons long timescale but in real time. He was speaking largely to natural processes and not encouraging intervention that I recall, but where a great deal of CO2 is anthropogenic, I hardly think it is offbase at least to consider whether enhancing/accelerating natural processes that mineralize it could be useful. As @Sean2828 has pointed out, one consideration might be that lack of akaline minerals might not be the chokepoint for this reaction.
Of course there are other approaches attempting to optimize various forms of ocean surface plantlife to digest CO2 and in turn produce fuel. The folks looking at these options at least are not engaged in a war on energy, and get plenty of hostility from environmental purists (who, as Patrick Michaels recently noted are nonetheless willing to ignore possible negative impacts of ‘sustainable’ offshore wind). I don’t think we need to pile on too.
Carbonate mineral formation has always been a sink for CO2 and it forms in warm shallow seas. That’s because the calcium and magnesium bicarbonate are thermally unstable and break down to mineral carbonate and CO2 in warm water. If that happens over deep water, the mineral precipitate redissolves in the cold depths. That’s why coral atolls form on the shallow western side of the oceans where water is warmer. Certainly a lot of micro organisms help in this.
Ocean circulation allows CO2 to be picked up in cold polar regions and converted to bicarbonate then carried to the tropics warmed up and sequestered as carbonate mineral. Every bit of this is natural process. What would it take to enhance precipitation in tropical regions? There are likely biological methods and chemical methods that could be employed. Remember, 97% of CO2 emission is natural as was its re-absorption so you don’t need much enhancement to bring balance. And to your question about necessity, that’s not clear at all.
Yep, I stayed awake forty-five years ago in chemistry class on the day Mr. Gau taught Le Chatelier’s Principle, you know, The Equilibrium Law. The more of a reactant added to a reaction, the more the reaction responds to reduce the concentration of said reactant. H2CO3 + Ca++ => 2H+ + CaCO3. Addition of CO2 to ocean water BINDS Calcium in limestone, sea shells, and coral. You can test this by sealing some bits of limestone, sea shells, and coral in a bottle of seltzer water. Better pick a different acid to FUBAR the oceans.
The silver lining here is that antacids for oceans should be difficult to beat for unmitigated craziness. Unfortunately it’s a new marker – someone is bound to try.
Geoengineering in all of its forms is without merit.
Some things have merit.
At the very least, if we want the current greening of the earth to continue, then we should continue to combust fossil fuels for energy. Also, combustion of fossil fuels likely will help prevent or forestall the next glaciation.
It’s a bit unfair to call it geoengineering – to real engineers.
why is that. isn’t agriculture geoengineering? I kind of like that.
it certainly has had an incredible impact on the biosphere. I don’t know how it logically follows that unintended consequences are fine, but if you intend to make changes that is wrong headed.
of course we can be hubristic and once unintended consequences become intended consequences it is possible to miss other unintended consequences. But these notions should be entertained skeptically which is to say rationally.
Are they aware that magnesium hydroxide is a laxative? And I can testify that it works very well in that manner.
Well, for those that have sh*t for brains, it’d leave room for more air-head ideas?
(Maybe AOC could make a few extra bucks if they her face on every bottle?)
“Whether artificially altering the oceans chemistry is a worthwhile or valuable goal to fight climate change is unclear,…”
No and no. As already pointed out. What a dipstick. Planetary Technologies, a Canadian startup, and any potential customers/investors should start it down again if they have any morals.
Scientifically, the most interesting thing is what is NOT in the Engineering Toolbox diagram of CO2 solubility with water. It stops at zero Celsius.
Well duhh… you might say. That’s when liquid water freezes. But not until about -50 Celsius in supercooled clouds in the atmosphere it doesn’t. Extrapolate that graph backwards from zero and one might begin to understand how the carbon-cycle models ignore some physical fundamentals.
Just another grifter scam intended to have the rubes part with their money to fund a illogical non-science based non-solution. PT Barnum must be so proud of these con artists in whatever afterlife he finds himself.
I’m trying to think of some idiotic scheme I can come up with to reap the monetary benefits which seem to abound in the world these days. Unfortunately, I can’t lie very well, as my wife constantly reminds me of that as one of my shortcomings and the reason I make her laugh a lot. Oh well.
Yes. Lying doesn’t come easily to many of us. Seems like you found a good wife.
Sorry, Anthony. I saw the headline and immediately said aloud, “Plop, plop, fizz, fizz. Oh, what a relief it is.”
Also, if the C in NBC stands for climate, what do the N and B stand for?
Regards,
Bob
N nonsense B bulls*** (or as my father would say baloney) C climate
As a group, we should all STOP using the word carbon (anything)….call it the element of life…the fraudsters want everyone to think of its as dirty black substance…instead of a colorless beneficial life-giving gas.
Like the backlit photos of factory smokesatcks belching …….steam!
No Bodys Climate
or National Broadcasting Corporation
“Whether artificially altering the oceans chemistry is a worthwhile or valuable goal to fight climate change is unclear”
Attempting to “fight climate change” is not only not worthwhile, it holds no value whatsoever. Oh, and it’s impossible to win such a battle.
“Believing CO2 controls the climate is pretty close to believing in magic.”
Claimed by: Richard Lindzen
The pH of seawater varies based on season and location, but on average, it is greater than 8. Seawater is a basic, not an acidic solution. The entire conversation about ocean acidification is based on a perversion of science. According to Halevy and Bachan, the pH of seawater has increased (become more basic) from 6.5 (slightly acidic) to today’s value greater than 8. Their work states that this occurred over many hundreds of million years.
When people use the term “ocean acidification” they fully expect those to whom they are communicating to infer that the oceans have already become acidic and are becoming more acidic. This is simply not true. Seawater is a mild base and is very definitely not acidic. Seawater is not even close to neutral pH (7.0). I have grown weary of those who pervert science for shock value.
Yes, but now we know that the oceans are full of that hazardous substance, dihydrogen monoxide, which can be quite deadly.
Conflation is a very useful tool when addressing the multitudes.
You spelled “manipulating” wrong.
The graph of solubility of CO2 in water ends at a freezing point. That means it describes fresh water, not sea water.
Second, “the oceans outgas more CO2 as they warm” – does not follow from the graph. Oceans are not saturated with CO2, far from it. A little science would help the authors a lot.
Yes, the solubility of CO2 in ocean water is much higher because ocean water in basic in pH. Further, it’s a buffered solution, so that bicarbonate is essentially constant at ocean pH.
Think I’ll rush out and buy all the packs of Raberprazole & Lanzaprazole!
The oceans already take antacids. Overwhelmingly river waters carry bicarbonates to discharge into the oceans. The real problem to solve is with so much bicarbonate entering the world’s oceans, why don’t they become increasingly alkaline with time? As alkaline as, say, the Great Salt Lake?
Sea monkeys will take over earth.
Because of all the calcite that settles to the floor of the oceans and becomes limestone — like the White Cliffs of Dover.
Simple answer. Salt is alkaline(the opposite of acid). in fact the thought of heaven forbid the oceans becoming acidic is ridiculous. pH of the ocean is around 8.4, Seven being neutral. Monkeying with the pH of the ocean would be harmful if at all possible. making the oceans more alkaline as this suggest is simply idiotic. but then again the scientists who are all knowing wanted to paint the arctic black so the sun would melt more ice and stop global cooling back in the 70’s. Here’s the bottom line: These people will do and say anything to get a government grant. And the people giving out the grants, well lets face it, aren’t too bright.
But, But it worked. The soot from coal fired power plants, especially those from China and the USSR DID cause ice to melt at a marginally higher rate and so THAT caused CAGW.
Do I need the s/off?
so when I look at pictures of the arctic and Antarctic everything looks white, Oh I get it! the camera people must have used the film negatives.
my point was the earth is too big and we are too small to affect it much. Besides none of the warmists predictions have come true except after the fact.
“Don’t attribute to malice that which can be explained by simple stupidity.” A fair aphorism. Except when looking at government. There, it is “Don’t attribute to simple stupidity that which can be explained by massive greed.”
Every one of those grants handed out includes graft for the grant evaluators – and kickbacks through handing out inflated contracts to their cronies.
The level of CO2 doesn’t matter
It’s going to cost us $20 million to dispose of that mine waste? Damn those enviro-wackjobs!
Wait! I’ve got an idea. Let’s get the loons to pay us subsidy money to dump it in the ocean to solve Climate Change!
But sir, there’s no way it will have any significant impact.
Of course not, foolish lackey.
But are they really that stupid?
No, but they do a convincing impression after the Big Guy gets his 10%!
Sounds like a plan to get rid of fluoride by putting it in drinking water.
First you get dentists involved.
Then you get politicians involved.
Then you sell the waste product for a profit instead of having to spend a bundle to safely dispose of it.
No need for such measures, the toxic black cloud from Ohio will soon spread over the ocean blocking the sun, and the resultant drop in temperature will see more CO2 absorbed.
I remember the old Alka-Seltzer commercials!
A glass of water with some dialog. The dialog might have something to do with what they ate or the mother-in-law was coming to visit, etc.
Then a couple of tablets being dropped into the glass.
I always wanted to see one with a nagging wife for the audio and tablet after tablet after tablet after … being dropped into the glass.
Sounds like that’s what these guys wants to do.
No “what a relief it is!” would be the result.
It’s absolutely amazing innit, how someone, anyone, in this day & age could get so many things so completely wrong and come up with an answer that is frankly surreal
That they really think that digging up the Earth’s land surface and throwing it into the water will fix the weather.
In the final endgame, when all the land has been dumped into the water, yes it would but there’d be, by definition, no-one around to benefit from all that effort.
You know me, “Doing the exact opposite *would* fix the climate”
Pulling silt and mud off the continental shelves, then spreading it on farmland and deserts would do wonders for simply everything, everybody, everycritter, everyplant everywhere
iow: Reverse Soil Erosion
And when there was still a trace doubt whether they believed their own “precautionary principle” crap, they remove it!
The oceans are a complex buffer system and dissolved carbon dioxide simply cannot alter that. NOAA has also admitted that they are not seeing ocean acidification. Ocean pH varies quite widely on a sunny day, with bays and inlets becoming more alkaline and then acidifying at night. Water passing through a coral reef is acidified by the organic acids given off by the life in the reef. Oceans are much more dynamic than the idiot activists understand. Ignorance is bliss and then they run with it.
As noted before, we need to bring back medieval punishments for anyone intentionally experimenting with the atmosphere.
The rack is one idea
But I think what the English did to William Wallace should be the template.
These people have to potential to be the greatest mass murderers in history.
It has to be made clear in advance what the consequences will be of catastrophe.
We will find you.