
Science and media outlets claim ocean acidification is happening due to increased carbon dioxide in the atmosphere. But objective data show the ocean is far from acidic according to Dr. Caleb Rossiter, executive director of the CO2 Coalition and a statistician who has studied climate change closely.
Host Anthony Watts and Rossiter talk about how a pH of 7 is considered neutral, with anything below 7 considered acidic. Ocean pH averages 8.1, which is alkaline rather than acidic. Although climate models suggest the ocean’s surface pH may have dropped from pH 8.2 to 8.1 since 1750, that change was never actually measured.
The pH drop from 1850 is merely a modeled conjecture. The concept of pH was first introduced by in 1909, and agriculturalists first developed field instruments to measure pH in the 1930s.
Debasification?
Oceanographic science has certainly been debased by contamination with “climate science” ©.
“Science debasification”
+1000
You’re welcome.
Ineffably sad but true.
Consensus “climate science” is a corrosive agent eating away the core of real science.
Yeah, lets allow the students vote on the correct answers to the equations. What could go wrong?
Science by consensus of freshmen could hardly be worse than consensus of the Oreskes-Mann Axis of Weevil.
Why don’t they say “less caustic”?
No scary enough?
Less caustic sounds like a downright improvement!
I like it. Decaustification.
This subject always reminds me of Alice at the mad tea party. The Mad Hatter asks Alice, “would you like some more tea?” to which Alice responds, “well, I haven’t had ANY tea yet, so I couldn’t very well have MORE tea.”
The ocean hasn’t become acidic at all yet, so it couldn’t very well become MORE acidic, could it?
The ocean is not caustic at all, so it is wrong to say it is getting less so.
Caustic is not synonymous with basic.
Caustic refers to chemical action that causes burning and/or damage, commonly referring to damage or burns to living tissue or flesh. Caustic is more or less synonymous with corrosive, but most commonly refers to damage by solutions or chemicals with very high pH.
It helps to use a dictionary.
If warmistas used them, we would not have this nonsense where any lowering of pH is called acidification.
Acidification is defined in every dictionary as the process of making or becoming an acid.
While the terms caustic, alkaline, alkali or base are often used interchangeably, they’re not really synonymous. Caustic may refer to any strong base, especially alkalis. Whether seawater counts as truly caustic is debatable, but calling it that is more correct than using “acidification” instead of “neutralizing”. Making a solution less basic is neutralizing it, as has been repeatedly properly pointed out.
http://www.phadjustment.com/pH.html
The point is the terminology is incorrectly used.
Caustic does not simply mean very basic…it refers to something with the ability to burn or damage by chemical action (If you doubt this, find and cite a dictionary wherein the word “caustic” is a reference to a pH level alone, and makes no mention of corrosiveness or burning or damage).
Considering that the whole point of this discussion is regarding misused terminology, do you not find it jarring to give a nod to replacing one incorrectly employ word with another?
Somewhere in this thread a commenter tried to make the point that adding any amount of acid to any solution is correctly called “acidification”. This is plainly nonsense, and can be easily demonstrated to be the case that it is nonsense. If one were to add an amount of hydrochloric acid solution to a solution of sodium hydroxide, and did so in an amount that precisely matched the amount of hydroxide ions present, the resulting solution would be a neutral solution of saline water. There is no point of logical reference to describe that operation as “acidification”.
One could make all sorts of arguments to make this same point, some requiring long explanations of what an acid actually is, by way of pointing out that there are separate definitions of exactly what constitutes an acid or a base, and one of them does not mention or require hydrogen or hydronium ions. A Lewis acid denotes an electron pair acceptor.
But that is making the case more complicated than it needs to be or should be.
This is not complicated…it is very simple.
It is well known exactly when the term “ocean acidification” was coined and applied to the effect of increasing levels of atmospheric CO2. There have been several articles and long discussions on that point on this site over the past bunch of years.
The whole thing is about semantics and making up new definitions for words and alarmism.
My motivation is to point out when people say things that are false or wrong, and I will do so, no matter which side of a debate is making mistakes, whether purposefully or simply by not being rigorous and concise.
Rain water has always been acidic and it’s deadly. It’s 100,000% more acidic than sea water.
If one filled a bucket with rain water and fully kept one’s head immersed for only several minutes, death would surely follow.
Some managed to drink enough to kill herself without the aid of drugs (E)
Anyone with a MSc or PhD degree in any engineering or science field who claims that ocean acidification is happening should be stripped off his/her degree. It is basic (chemical) knowledge that any aqueous solution with a pH above 7 (like seawater) is not acid. Something that is not acid cannot become “more acid”.
So gentrification can’t occur unless a neighborhood is already gentrified in the first place, then? You might need to give up your BA degree.
Soil science and ag engineering folks know acidification is the lowering of pH. It isn’t necessarily the lowering of pH of something that is already acidic. But I’m sure you can set them straight and strip them “off” their degrees.
Michael, I studied soils and agricultural methodologies more than 30 years ago and it was always understood acidification of soils meant lowering PH below 7. Never did I hear anyone refer to reducing an alkaline soil PH but remaining above 7 as “acidification”. It was always referred to “becoming less base” – and that is technically the most correct.
In fact the term when referenced in dictionaries says “a process of making or becoming acidic”. I find no dictionary term that refers to acidification as remaining above base.
“acidification as remaining above base.”
correction – remaining base – not above base!
Exactly right. This is how the teachers in high school and then professors in first year in college told us what to do in some laboratory experiments. In order to do this or that for this experiment you have to “acidified”. Simply, yes add some acid in this solution!
For you this was more than 30 years ago. For me it was more than 60 years ago! But we both remember.
Jankowski
A poor analogy! I submit that a better one would be observing someone who was unrefined, and claim that they were becoming refined just because they did something that wasn’t as uncouth as usual. But, a worse instance would be claiming that the person was becoming more refined when they obviously were still unrefined.
A suburb hasn’t become more gentrified if it’s still a slum. The process isn’t one where the end suburb is one where the unemployed might flock to from an even bigger hell hole, but where the upper middle class would like to live. The process actually requires some streets attracting them.
Never will the oceans drop below 7 because of emissions.
Michael Jankowski: You’re utterly wrong, as well as being pompous. Chemists use the term “acidification” to mean lowering the pH below 7. Changing the pH from 8.2 to 8.1 (if that’s the case, which I doubt) would be called “a barely detectable lowering of the pH”.
BTW, I am a chemist.
No they dont. They use the term less basic. Its all about the power of the hydrogen Ion. Alkalinity is how the ocean is measured. not pH. In alkaline water the H+ has less power.
What on Earth are you talking about? In alkaline solutions (pH >7) the concentration of H+ is less than 10e-7 moles per litre. Nothing to do with the “power” of H+.
The definition of pH is very simple and well known to anyone who has studied chemistry or looked it up…or at least it ought to be.
pH is minus the log of the hydrogen ion concentration of a solution.
Very simple.
Mick, what you said is a confused mess.
The scientific term used in chemistry is less basic. either way. Not more acidic.
Besides, alkalinity is a better measure of ocean health
The question is a simple one…what does the word “acidification” mean?
One can check any sort of dictionary and the definition is the same, whether a regular dictionary, a scientific one, a legal dictionary…whatever.
Acidification is the process of making or becoming an acid.
Nowhere does it mean making a basic solution somewhat less basic, or even making a basic solution neutral, except in the BS made-up jargon of climate alarmists.
dealkalinization?
The pH level is changing in the direction of acidity.
Agreed, Jack Dale. But the changes are small and there is no evidence that it is harmful.
Given that the interior of living organisms is more acidic than the ocean, it is more likely that the changes are net beneficial.
“In the 200-plus years since the industrial revolution began, the concentration of carbon dioxide (CO2) in the atmosphere has increased due to human actions. During this time, the pH of surface ocean waters has fallen by 0.1 pH units. This might not sound like much, but the pH scale is logarithmic, so this change represents approximately a 30 percent increase in acidity.”
The shells and skeletons are affected.
https://www.noaa.gov/education/resource-collections/ocean-coasts/ocean-acidification
Human stomach acid has a pH level of 1.5 to 3.5 which enough to damage our skin which has ideal pH level is just below 5.
The only problem with the claim that ocean “acidification” is affecting shells and skeletons, is that the shells and skeletons show no signs of being affected.
The oft quoted 30 percent increase in acidity is incorrect. Let me explain.
The hydronium ion concentration might be 30% higher at the lower pH, but it has a concentration of < 10E-7 M. That is not acidic at all. Acidity is the ability to titrate a base, typically something like an alkali metal hydroxide. In the case of sea water, the dominant acidic species that can titrate a base is boric acid. Bicarbonate can also, except that at sea water pH it is essentially buffered and gets back titrated by the buffering capacity of sea water.
People generally don't have a good understanding of pH and what it means. At higher pH, the hydronium ion concentration is so low that it is of little chemical consequence. Just as the term "ocean acidification" was invented for its "shock value," the claim of a 30% increase in acidity is similarly deceitful and wrong.
I haven't studied such things in detail since my last graduate course in electrochemistry, but as a PhD chemist, I have used these principles practically for decades.
Nail on head.
The Green Movement is incredibly dishonest in its use of numbers. A friend of mine was incredulous when I pointed out that an increase of atmospheric CO2 concentration from 300 to 400 ppm equated to one extra molecule of CO2 in 10 000.
Scissor, exactly the right rebuttal!
Acidic solutions (pH 7.0) are considered to have insignificant amount of H+ ions in comparison to oxidizing agents, such as OH- ions.
“pH is the negative log of hydrogen ion concentration in a water-based solution. The term “pH” was first described by Danish biochemist Søren Peter Lauritz Sørensen in 1909. pH is an abbreviation for “power of hydrogen” where “p” is short for the German word for power, potenz, and H is the element symbol for hydrogen.”—source: https://www.thoughtco.com/what-does-ph-stand-for-608888#:~:text=pH%20is%20the%20negative%20log,the%20element%20symbol%20for%20hydrogen.
Applying logic based on the above definition of an acidic solution (i.e., considering the need for significant quantities of H+ ions or equivalent reducing ions to be present for a solution be consider as “acidic”), it is INCORRECT to associate changes in pH levels above 7.0 with becoming more or less acidic.
OOOPS . . . something happened to my second paragraph’s original text during posting. The text should read:
“Acidic solutions (pH 7.0) have insignificant amounts of H+ ions in comparison to oxidizing agents, such as OH- ions.”
I cannot believe World Press or whatever bot is handling WUWT postings did it again (mangling my second paragraph) . . . here’s my last ditch attempt at correcting this issue (removing the “less than” and “greater than” symbols:
“Acidic solutions (pH less than 7.0) have significant amounts of H+ ions, which actually appear as “protonated” water molecules or the hydronium ions (H3O+), a reducing agent, in comparison to basic solutions (pH greater than 7.0) which contain oxidizing agents, such as OH- ions, and an essential absence of reducing agents.”
Scissor,
The obfuscation at the root of this aspect of the discussion is that the hydrogen ion/hydronium ion concentration in a solution is synonymous with the word “acidity”.
We use the pH scale and the actual molar concentration of that ion to describe this.
The word “acid” has a very simple definition when used in describing aqueous solutions…it is a solution with a pH below 7.
Warmistas are simply using the usual leftist tactic of employing linguistic switcheroos when they confuse the subject thusly.
To restate it again, redundantly: Hydrogen ion concentration is not another word for “acidity”.
“Jack Dale June 11, 2020 at 4:03 pm
…the pH of surface ocean waters has fallen by 0.1 pH units.”
Which is an estimate because no-one has actually measured ocean pH.
Dale
http://wattsupwiththat.com/2015/09/15/are-the-oceans-becoming-more-acidic/
Calcifiers that live in zones of upwelling protect against potential damage by covering their carbonate-based shells with mucous and chitin. Some even allow other organisms to colonize their chitin-covered shells as an additional layer of protection. That way, they don’t have to expend extra energy to repair damage from transient excursions of pH to levels low enough to affect their shells.
Care to explain how bivalves with calcium carbonate shells live perfectly happily in neutral or even acidic river water?
**represents approximately a 30 percent increase in acidity.”**
FALSE. It cannot be acidic until it drops below 7.0.
Therefore it cannot become MORE acidic when it is NOT acidic.
try again.
**represents approximately a 30 percent increase in acidity.”**
Although from made-up non-data, and is scientific nonsense because it isn’t acid to start with
Stating it this way means there has to be further approx 2000% change in H+ concentration to even become neutral
Anyone using such a statement has to be a complete scientifically ignorant jackass.
Only a complete jackass looks to NOAA for education on science.
It is sematic nonsense to refer to the acidity of a basic solution.
An acidic solution is one having a pH below 7.
In referring to solutions, the words acid and base are binary…a solution is either one or the other, not both at once.
“The pH level is changing in the direction of acidity”
Except it isn’t.
Dehydrogenization?
Lots of ways to express what allegedly is happening. The one fact upon which all can agree is that “acidification” doesn’t describe the supposed process.
At best, “neutralization” could be defended, since change in the direction of neutral, yet far from acidification.
Consensus “climate science” © has corrupted even chemistry.
Neutralization is, in fact, the right term. But it is as far from scary as one can imagine, and thus not allowed in the narrative.
Yes, that’s true except that neutralization as implies that the solution is taken to neutrality, which it is not because sea water is basic.
As a practicing chemist, I would just say lowers the pH, and to be more quantitative explain by how much.
The term “ocean acidification” was created deceitfully and is used deceitfully.
Right. At best “neutralizing”, not neutraliztion or neutralification.
I agree with you the term “ocean acidification” is used deceitfully.
But the term “acidification” itself was used by our teachers in high school and professors in first year in college to tell us that adding some small among of acid was needed to initiate an experiment on something.
We were told so many times “acidifies” and the term “acidification” was a so very common way to tell students what they needed to do.
It was never used, as far a s I know, to reduce the pH below 7.0. It was simply used to reduced the pH to get something going. Sometimes we were at a pH below 7.0, sometime above 7.0. It did not mater, it did not matter if the pH to be reduced was above or below 7.0, we were told “acidified is needed”.
rd50
I can remember experiments where we were instructed to titrate an alkaline solution with acid until it was neutral (as evidenced by change of color of the indicator) and then add a slight excess of acid to acidify it. The important point of the difference between an acid solution and an alkaline one is that there is an excess of hydronium ions that are free to react with a metal or anions to drive a reaction. That is, the ratio of hydronium ions to anions is greater than one (1).
RD you are wrong.
I am also a chemist by training, and your explanation is flat out wrong.
Simply adding some small amount of acid to a solution is not called acidification.
The oceans will never become acidic. It is impossible.
So referring to a slight alteration of pH as ocean acidification is simply wrong.
Period.
rd:
It means one of two things. Your instructor did not understand and gave you an incomplete lesson, or you did not understand the process.
You were not working in the ocean where it is impossible to make it acidic by adding a few drops of acid.
“…adding some small among of acid was needed to initiate an experiment on something.”
This is flat out nonsense.
As well as being demonstrably wrong.
Either you just made it up, or you have a bad memory, or you had awful and mixed up instructors.
It makes no sense at all.
Chemical reactions can be initiated by any number of means, including simply combining two or more substances and waiting, adding heat, adding water, exposing to sunlight, allowing something to dry or evaporate, etc.
Chemical reactions proceed due to any number of causative factors. And there are all sorts of reactions and “experiments” that have nothing whatsoever to do with acids, bases, or even aqueous solutions.
Maybe you are just leaving out a large part of whatever it is you are trying to say or explain, but the fact is, as stated, your comment makes no sense and is wrong.
Even outside of the world of science, if one is trying to explain something, it is necessary to be concise and correct if one wants to be able to communicate effectively.
There is no generic explanation of what constitutes and “experiment” that requires the addition of an acid to something.
Clyde has it right…it sounds like you are trying to describe the titration of a basic solution…to be charitable. But even regarding titration of a solution in acid base chemistry…one could be titrating an acidic solution, in which case one would slowly add base to gradually neutralize the acid.
As far as beginner level schooling…that is when it is most important to start at the beginning, leave out no details, and get everything right. Otherwise, one is not educating students, but miseducating them at best.
Michael Kelly is exactly right.
Adding an acid to a base (or vice versa) is called neutralization.
Hell, even Wikipedia says so.
When combined, acids and bases neutralize each other.
The situation with seater and CO2 is much more complicated, but the terminology of chemistry is plain enough, and has a log history.
Science is not meant to be a process of obfuscation, no matter what warmista jackasses say.
“Just as we describe an increase in temperature from -40°F to -20°F as warming, even though neither the starting nor the ending temperature is “warm,” the term “acidification” describes a direction of change (i.e. increase) in the level of acidity in the global oceans, not an absolute end point. When CO2 is added to seawater, it reacts with water to form carbonic acid (H2CO3); hence acid is being added to seawater, thereby acidifying it. Similarly, in the example about human blood, a drop in pH is referred to as acidosis, even though the point where acidemia begins (7.35) is still above 7.”
https://pmel.noaa.gov/co2/story/A+primer+on+pH
Your first answer was a lot better 😀 [dealkalinization? The pH level is changing in the direction of acidity.
] and right.
Maybe “increasing temperature” is the right description of that phenomen ? 😀
”The pH drop from 1850 is merely a modeled conjecture.” Another words the oceans pH have been studied for only around a hundred years. Maybe you may like to hazard a guess as to the variance of pH in the oceans within the life of corals, which is millions of years. My guess is the levels been a lot worse and the corals are still here..
I posted, about 5 years ago, a link to a study from Pacific Northwest ocean waters (I think it was in PNAS, which once had credibility). The intra-day variation in pH was in the one whole unit range.
phil
Which is not unusual for water along coasts that experience upwelling. That is one of the reasons that the Monterey Bay Aquarium monitors the pH of the water they bring in for their tanks.
In other words, the hydronium ion concentration varies by a factor of TEN within a day! And yet marine organisms manage this perfectly happily.
-40 to -20 is going from effing cold to dam cold. Had that experience more often then I would like to remember. Once after experiencing temps no warmer than -22 F for 72 hours when the cold started to break I unzip my coat snow-blowing the driveway at -19, it felt rather balmy at -19.
In my experience working in northern canada below -35c your body can’t tell the difference, it’s just damn cold.
I worked in -55c once, my one boot broke from the cold.
I laugh when greens and politicians blather about the north and hydrocarbon use, up there during cold snaps plugging your vehicle in doesn’t help, they just leave them run all night.
Without diesel all those remote settlements would cease to exist in 24 hours
Give it up, Jack. The highest NATURAL source of CO2 is the ocean. Carbonic acid is just about the weakest acid known, and 0.04% of the atmosphere is not sufficient to “acidify” anything. Try it yourself, fill a glass with distilled water, leave it outside overnight and measure the PH in the morning. Report back with any significant change, we’re all eager to hear the results of the experiment.
The ocean is a net sink.
When did you wake up to sense?
The Baltic Sea is a net source
😀
RHS, I think you need to reconsider your statement that the “the highest natural source of CO2 is the ocean”. But it is likely true that “the highest natural sink of CO2 is the ocean”.
The world’s oceans are very stable, relatively-strongly buffered solutions with an average pH of 8.1-8.2. When CO2 from the atmosphere dissolves into such basic seawater, it first forms carbonic acid (H2CO3), which in itself is unstable. Carbonic acid in basic seawater quickly dissociates to form bicarbonate and carbonate ions. An insignificant amount of CO2 remains as a gas in aqueous solution (see Figure 1, the Bjerrum plot, at https://www.soest.hawaii.edu/oceanography/faculty/zeebe_files/Publications/ZeebeWolfEnclp07.pdf ) The bicarbonate and carbonate ions create the buffering capacity inherent in seawater; that is, seawater can resist drastic pH changes even after the addition of weak bases and acids. This is the primary reason that the massive amounts of rain water (with pH of 5.0-5.5) that fall into the world’s oceans each and every day do little to change the pH of the world’s oceans.
Moreover, the governing chemical equations are:
CO2 + H2O —> H2CO3
which forms bicarbonate:
H2CO3 –> (HCO3-) + (H+)
which in terms forms carbonate:
(HCO3-) —> (CO3–) + (H+)
In a solution with a pH at or below 7.0 (i.e., acidic solutions), the released H+ ions would not exist as free ionized hydrogen atoms but would instead form “protonated” water, aka hydronium, (H3O+). However, in a basic solution any hydronium ion would have essentially zero residence time, quickly reacting with excesses quantities of (OH-) ions, or other oxidizing ions, resulting in the availability of (H+) becoming insignificant. What this means fundamentally is that the above chemical equations are not reversible as long as ocean water pH remains on the basic side.
Therefore, it is incorrect to infer that CO2 solubility in seawater follows Henry’s Law of partial pressure (a statement about equilibrium conditions, by the way, that neglects chemical reactions between the gas and solvent) and this fundamentally means that the solution of atmospheric CO2 into seawater is, to first order, NOT REVERSIBLE . . . the world’s ocean would have a very hard time outgassing gaseous CO2 independent of atmospheric CO2 concentrations, as long as they remain as basic solutions.
And, no, warming ocean waters will not release significant quantities of “dissolved CO2” back into Earth’s atmosphere for the same fundamental reason.
Ever wonder why all carbonated beverages are acidic?
Gordon,
You got a lot of details correct here, and obviously know some chemistry, or at least took the time to look some things up.
But your comment seems to overlook that only a tiny traction of the CO2 in water is in the form of any of those species you mention…the vast majority exists as a dissolved gas in solution, and plays no part in any of those equilibrium reactions…it is just dissolved gas in solution.
To figure out how much, you need to look at the hydration equilibrium constant, which for CO2 in water at 25 C is 1.7 x 10^-3 in pure water and about 1.2 x 10^-3 in seawater.
What that tells us is that the amount which is converted into carbonic acid to begin with, and from there is in equilibrium with bicarbonate and carbonate, is only about 1 or 2 one-thousandths of the amount of CO2 in the water. About a tenth of one percent of it.
CO2 is constantly entering and leaving the ocean, depending on the temperature of the water at a given location, how much CO2 is in a given parcel or surface water, and the exact amount of CO2 in the air. CO2 tends to enter the ocean in Winter and/or at high latitudes where the water is cold, and tends to exit the ocean in the tropics or in Summer or wherever the water is very warm.
In keeping with the format of a closing question, ever wonder why the ice core CO2 data shows a delayed (by something like ~800 years last time I checked) correlation with temperature?
The accepted reason for this is due to outgassing or absorption by the oceans as the cycles of glacial advances and retreats alternated.
Oops…traction = fraction.
Sorries.
BTW…carbonated beverages typically have phosphoric acid added.
Nicholas, thank you for your comments.
As to your assertion “. . . only a tiny traction of the CO2 in water is in the form of any of those species you mention…the vast majority exists as a dissolved gas in solution . . .”, if you carefully re-read my previous post you will see that I specifically stated the following:
“An insignificant amount of CO2 remains as a gas in aqueous solution (see Figure 1, the Bjerrum plot, at https://www.soest.hawaii.edu/oceanography/faculty/zeebe_files/Publications/ZeebeWolfEnclp07.pdf )
If you had consulted the referenced Bjerrum plot you would have seen that it actually plots the equilibrium concentration of CO2 that exists as a DISSOLVED gaseous component in seawater as a function of pH. You will see that between a pH of about 4.5 and a pH a bit above 8.0, the dissolved CO2 content (in mole/kg) DECREASES by approximately 10^(2.3), or a factor of about 200.
The Bjerrum plot gives the absolute CO2 equilibrium concentration for pH=8.1-8.2 seawater at the stated T=15 C, P=1 atm conditions to be about 5.0E-5 mol/kg, which is equivalent to 2.2 ppm wt. As I previously indicated, this is insignificant compared to the current atmospheric CO2 concentration of 410 ppmv (equivalent to about 623 ppm wt.).
Also, please not that the title text to Figure 1 indicates ΣCO2 = 2000 μmol kg−1, meaning that the total concentration of CO2 that contributed all of the chemical species noted in the plot (ions as well as dissolved CO2, but excluding B(OH)3 and B(OH)4-) is equivalent to about 88 ppm wt. This means that only 2.2/88 = 2.5% wt. of the CO2 actually entering seawater (not already having CO2 content) will remain as (gaseous) CO2 dissolved in seawater with pH in the range 8.1-8.2 . . . the other 97.5 % breaks down into the ion species per the chemical formulas I provided. And those chemical reactions are NOT reversible, for the reason I stated.
Finally, the reason carbonated beverages typically have phosphoric acid added is specifically to give the solution a pH of 4 or lower for the very reason that such acidic conditions are needed to enable gaseous CO2 to saturate/supersaturate the drink to any appreciable degree.
Dale
Warming refers to a process, whereas, “hot” refers to a state of being. Only a person who was daft or dishonest would refer to a change in temperatures, both of which are cold enough to cause frost bite quickly to exposed skin, as getting hotter. That is a better analogy to becoming “more acidic.”
NOAA is infected with political correctness.
So called “correctness”
In reality it’s not correct at all
😀
Jack:
Wrong. I do not use warming. I stress less cold, similarly i do nor use acidification.
Gerald
Typically, at those temperatures, I would say, “It is not as cold today as yesterday.”
I’ve posted this before on here years ago, but this was an easy one for the people with CVSD (Chronic Virtue-Signaling Disorder) thanks to people, i.e. chemists like me. Although I’m a bit wiser now, although not wise enough to know why 1,000s of people marched through my neighborhood last night wanting to defund the Oakland Police Department (I kid you not), it never occurred to me that if I was in the lab. and taking something from pH 12 say, to pH 8 with hydrochloric acid I might say I was acidifying the solution and this word would be hijacked by the leftard loonies and 12th-rate “scientists”. I suspect the people who came up with the words liberal, progressive and gay are having second thoughts about their nomenclature actions too.
…. and add “defund” to that list too. I think it might mean “more money for us” although I’ll wait for that one to play out.
Defund climate change research!
Might I respectfully disagree:
Start up a program of funding climate change research, after defunding every last vestige of phony climate change research.
Defund Climate “Science”. Fund Climatology.
Thanks Phil,
I now know where they conjured up the word COVID from.
COnstant VIrtue Disorder. The number 19 was added to convey the average age of the unaffected yet vocal activists.
It all makes sense now, those climate alarmists are cunning folk.
I think you’ll find the 19 refers to IQ
I am very confident the use of the word “acidification” is due in large part to the scare-tactic impacts it has on laypeople and utter morons. But it had been a widely-accepted and defined term for lowering pH in soil science well below any ocean acidification debate.
Jankowski
You claimed, “But it had been a widely-accepted and defined term for lowering pH in soil science …” I find that difficult to believe. Can you provide a citation to support it?
Everyone should read Hofman et al 2011,“High Frequency Dynamics of Ocean Ph etc.”
Professor Gretchen Hofman and a team of scientists checked Ph in oceans around the world and found considerable variations from the 8.1/ 8.2 figure currently identified as the norm by month, week, day and even by the hour.
In one instance the Ph was currently at figures presented as “ dangerous”for marine life by centuries end!
“Ocean acidification” is a confected non issue.
I think I posted this before, too good to ignore. Enough with the acid business —https://doi.org/10.1016/j.jembe.2014.07.011 “We examined seasonal differences in invertebrate community structure between two distinct pH zones: control (pH 8.1) and acidified (pH 7.8) and show that many groups of invertebrate taxa were robust to acidification effects.” and “The survival pH level for this nemertean ranged from 5.00 to 9.20” https://doi.org/10.1016/j.jembe.2005.07.003 First paper do your homework, second interesting.
Everybody calling pH7+ acid should be required to put their fist in a fresh shark’s stomach. That’s acid, I did it. Measuring pH with precision when I was in college long ago was difficult.
Forgot these, real acid
Perry, C.M. Chronic effects of low pH on length and weight of Atlantic salmon, Salmo salar . Environ Biol Fish 27, 153–155 (1990). https://doi.org/10.1007/BF00001945
The Cortland salmon were significantly different from the other four sources in respect to their weight over time at neutral pH. At pH 5.2, there were no significant differences between any of the five sources.
Orsatti, S.D., Colgan, P.W. Effects of sulphuric acid exposure on the behaviour of largemouth bass,Micropterus salmoides . Environ Biol Fish 19, 119–129 (1987). https://doi.org/10.1007/BF00001882
“(pH levels 7.2, 6.1, 4.8, and 3.7)”
Look at some waters in Amazone rainforests.
Most fishes from there need water below pH 7, these fishes are nice ones.
Discus, Scalares, Neon..,
“Science and media outlets claim ocean acidification is happening due to increased carbon dioxide in the atmosphere. But objective data show the ocean is far from acidic”
To be fair, there is nothing wrong with the use of the term “acidification” to mean “lowering of pH”. However, there are some serious unresolved issues in the ocean acidification claims of climate science including statistical flaws in the claim that the observed rise in atmosCO2 is caused by fossil fuel emissions and that it can therefore be attenuated with climate action in the form of reducing fossil fuel emissions.
Three links below.
https://tambonthongchai.com/2020/06/10/a-monte-carlo-simulation-of-the-carbon-cycle/
https://tambonthongchai.com/2020/01/26/ocean-acidification-the-evil-twin-of-climate-change/
https://tambonthongchai.com/2020/02/27/a-co2-nightmare-hydrothermal-vents/
Semantically true, practically, no, even in low salinity estuaries where I sometimes worked it was difficult to lower it because carbon dioxide forms a temporary very weak acid, quickly changed from the, in this case highly diluted alkalinity with rich organic matter, so the term is incorrectly used. It was a new expression, as the talk said, deliberate.
Chaamjamal
You said, “To be fair, there is nothing wrong with the use of the term ‘acidification’ to mean ‘lowering of pH.”’ I would disagree. If one started with an alkaline pH and reduced it below 7, then acidification would be grammatically correct. However, if the intent and outcome was to move along the spectrum of alkaline pH values from a high pH to a lower alkaline pH, then the process is most succinctly described as lowering the pH because the characteristics and chemical behavior of an alkaline solution is distinctly different from an acidic solution.
The answer is far more retardedly simple.
The old tests for ocean acidity involved transporting CORK PLUGGED PHIALS back to Europe from around the world… and that cork would reduce the acidity of the water inside – well known problem now.
Back then it simply made the world’s oceans more base by experimental bias.
And the scientists who claim the ocean is “more acid” are retards. They slow down and damage the progress of science. That’s what the invective means, that the person accused is dragging down the whole group by a failure of preparation, attention, cooperation or incompetence. They are the applied brake.
NOAA is wrong. Whether they are being intentionally deceitful like Ken Caldeira, I do not know.
If I were a little closer to retirement, I would correct them, but good things do not happen to those who don’t tow the party line and since some of my funding is from NOAA, I must tread cautiously.
If they wanted to forecast a really positive, hoped for kind of trend, could someone please try to arrange for a ‘pH DD’, i.e., a ‘PhD Deficit’?
The role of pH buffering in the oceans is never discussed by the Lunatics who push the AO scam. The buffering capacity is vast. If you’ve ever done simple pH-buffer “experiments” in the lab you’ll know, that while the in a strongly buffered solution, the pH initially drops downwards a bit, the bufferin action releases counter anions to hold the pH steady after that initial perturbation.
That is essentially how the oceans have responded to this step increase in dissolved CO2-carbonic acid from anthropogenic emissions of the last 70 years. There was an initial drop of about 0.10 to 0.18 pH units. Getting the oceans to drop further below 8.1 though will require vastly more CO2 addition than there are fossil fuels to reasonably burn in the next few centuries.
The AO scam has always been a climate charlatan’s hoax on the naive public.
I had a “Joe Biden moment”: I meant to write OA, not AO. (as in Ocean Acidification).
(P.S. A Joe Biden memory moment is not to be confused with Joe Biden female-groping moment though.)
Your AO is AZ, correct? Or WUWT.
I am always struck by the lack of understanding of ocean geology. The longest mountain chain in the world is the great spreading center which passes through the Atlantic Ocean, Indian Ocean circling the Antarctic Continent, then running up north through the eastern Pacific Ocean. Basaltic material is exposed to sea water along the spreading center and is serpentinized. This is a basic chemical reaction. Until this process is taken into consideration, any talk of ocean water becoming less basic is faulty.
Made that similar comment many times here at WUWT. Basalt is basic salts containing lots of magnesium and calcium. Acts as a pH buffer in the deep ocean. The continental margins around the world are also ringed with calcium carbonate deposits. Again a buffering action.
The OA scam depends on people not understanding the physical chemistry of acid-base and hiw buffering systems work to stabilize pH.
Q: Do you know what part of the ocean has its pH affected by dissolved anthropogenic CO2?
A: The surface waters. The effect is confined mainly to surface waters.
Q: Do you know what part of the ocean has the highest pH (i.e., is most alkaline)?
A: The surface waters.
Here’s a graph:
●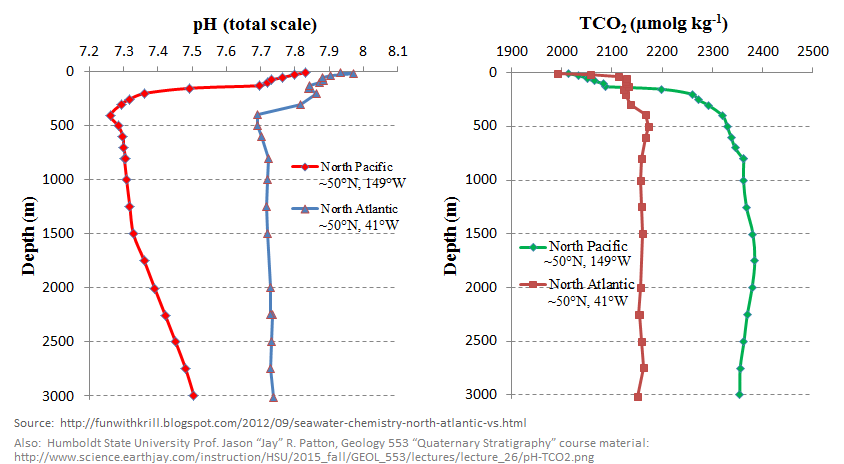
Several things are obvious in that graph:
● There’s a large difference in pH profiles between the North Atlantic and North Pacific
● In both basins, there’s a large difference in pH with depth
● The most caustic part of both oceans is the surface waters.
Since the surface waters are also where so-called “ocean acidification” happens, that means the reduction in ocean pH is confined almost entirely to the most caustic part of the ocean.
Another way of stating it is that the effect of dissolved eCO2 on pH is to slightly is to slightly reduce the variation in alkalinity with depth.
The main practical effect seems to be to stimulate the growth of calcifying coccolithophores, which remove carbon from the upper ocean, sequestering it in carbonates. Here are some references:
● https://academic.oup.com/icesjms/article/73/3/529/2459146
● https://hub.jhu.edu/2015/11/26/rapid-plankton-growth-could-signal-climate-change/
● https://dx.doi.org/10.1073%2Fpnas.1117508109
● http://science.sciencemag.org/content/350/6267/1533
“Ocean acidification”[sic] is minuscule, and harmless, and dwarfed by natural spacial and temporal variations in ocean pH.
Sea water is not caustic.
Nicholas
Yes, “caustic” implies extreme alkalinity or very high pH.
Long ago, when I took Chemistry, when describing aqueous solutions, the adjectives “caustic,” “alkaline,” and “basic” were synonyms, which all meant “has pH > 7.00”.
The adjective “acidic” meant “has pH < 7.00"; "sour" was occasionally used as a synonym.
The adjective "neutral" meant "has pH = 7.00".
Solutions with a pH only slightly above 7.00 were said to be "weakly alkaline," "weakly caustic," or "weakly basic."
(Of course, in other contexts, the meanings aren't necessarily opposite. For instance, the same person could be said to have a "caustic sense of humor" or an "acid wit.")
Perhaps terminology has drifted. I've noticed that most young whippersnappers say "Celsius" instead of centigrade, and "ml" instead of "cc," these days.
(But nobody says “spacial.” I meant spatial. I don’t know how I typed that.)
Look at the etymology, if in doubt.
There are overlaps in these various terms, but I for one have never seen caustic used to describe any solution that is even slightly basic, i.e. has a pH above 7.
The root is originally from the Greek, either kaustos ‘combustible’, or from kaiein ‘to burn’…or via Latin…according to Merriam-Webster: “Latin causticus, from Greek kaustikos, from kaiein to burn”.
Personally, I find it “better” to look words up in numerous sources and have a close look if anything is contradictory or unclear.
Memory is a notoriously bad way to try to settle anything factual. After all, in addition to simply misremembering something, one might be recalling an instance of incorrect usage by one or more individuals, or else context might be missing from the recollection.
Anthony, you need to look at two things and two things only:
CO2+H2O converts to alkali products bicarbonate+carbonate.
90% of our oceans alkali products is a buffer which will always remain at 90%, of bicarbonate which has the pH varying around….. 8.1-8.2 depending on temperature and salinity.
Oddgeir
“acidification” is technically correct; one might equally well say the arctic is warming. The denotation of the word is “trending toward acidity,” which is correct.
But the connotation of “acidification” is that the ocean is becoming acidic, which is misleading. So scrupulous debaters would not employ the word in a way that would mislead a layman.
LOOK OUT!!! Here comes SCIENCE!!
The pH scale is log so every whole number is a power/factor of ten.
By definition pH is the negative exponent of the hydrogen ion concentration.
For instance, pH 9 is 10^-9 or 1 part per billion, 0.000000001.
pH 8 is 10^-8 or 10 parts per billion, 0.000000010.
To go from pH 9 to pH 8 is factor of 10 or 1,000%!!!! Makes 26% look trivial.
Ocean “acidification” of pH 8.2 to pH 8.1 is an alkalinity change in H ions of 1 ppb.
I’m fairly certain the ocean flora and fauna don’t even notice.
Nick, I believe your statement “For instance, pH 9 is 10^-9 or 1 part per billion, 0.000000001” is misleading, if not outright incorrect.
“The pH scale is traceable to a set of standard solutions whose pH is established by international agreement. Primary pH standard values are determined using a concentration cell with transference, by measuring the potential difference between a hydrogen electrode and a standard electrode such as the silver chloride electrode . . .
“A solution of a strong acid, such as hydrochloric acid, at concentration 1 mol dm−3 has a pH of 0. A solution of a strong alkali, such as sodium hydroxide, at concentration 1 mol dm−3, has a pH of 14. Thus, measured pH values will lie mostly in the range 0 to 14, though negative pH values and values above 14 are entirely possible.” — source: https://en.wikipedia.org/wiki/PH
Please take note of the fact that negative pH values and values above 14 are possible, whereby “negative pH” contradicts your statement “By definition pH is the negative exponent of the hydrogen ion concentration”.
HCl at an aqueous concentration level of 1 mole/dm^3 = 36.5 gm HCl per 1000 gm H20. Since the pH scale is base-10 logarithmic, a pH of 9 would be equivalent to hydrochloric acid at a concentration of (1E-9)* (36.5 gm HCl per 1000 gm H20, or 0.04 parts per billion by weight. It would also translate to 0.02 parts per billion by molar volume. Acids at different molecular weights would, of course, give different ratios.
Alternatively, NaOH at an aqueous concentration level of 1 mole/dm^3 = 40 gm NaOH per 1000 gm H20. Therefore, a pH of 9 would be equivalent to dissolved sodium hydroxide at a concentration of (1E-5)* (40 gm NaOH per 1000 gm H20, or 400 parts per billion by weight. And likewise, different bases of different molecular weights would give different ratios.
It’s all relative.
On one hand we are told that the oceans are taking up CO2, contributing to acidification, on the other we are told that the warming water is out-gassing the stuff which is a positive climate feedback. Often you get both claims in the same sentence. What kind of magic could this be?
You need to exercise a little scepticism Eric the half halibut. You’re “told” that the CO2 must be from warmer oceans out-gassing… you’ve been gaslighted if you believe that. The ocean is a net CO2 sink, no magic required.
Of course it’s nonsense, I don’t fall for sophistry either. I recall a recent visit to Heron Island on the Great Barrier Reef where my wife and I were shown through the UQ reef research facility. A bright shiny-eyed young graduate told us all about pH and CO2, how everything is doomed and how they had proven it by subjecting little fishes and sea cucumbers to various conditions in PVC and glass tanks. It took a dig in the ribs from my wife to stop me walking out. As a UQ science graduate herself, she knows full well that a degree is much harder to get if you don’t pay lip service to the lecturers’ line of thinking.
“Loydo June 11, 2020 at 9:43 pm
You’re “told” that the CO2…”
We’re “told” CO2 causes climate change, and a tax on energy will fix it! The “science” is in!! Apparently!
Loydo wrote, “You’re ‘told’ that the CO2 must be from warmer oceans out-gassing… you’ve been gaslighted if you believe that. The ocean is a net CO2 sink…”
Exactly correct, Loydo.
Mankind is currently adding about 5 ppmv of CO2 (about 11 PgC) to the atmosphere each year, but the atmospheric CO2 level is only rising at about 2.5 ppmv per year. The difference is the rate at which natural negative feedbacks (mainly terrestrial greening and absorption by the oceans) remove CO2 from the air: currently about 2.5 ppmv per year. Someone who claims nature is raising the atmospheric CO2 level must be incapable of subtracting 2.5 from 5.
However, the solubility of gases like CO2 (or CH4) in water does decrease as the water gets warmer (per the temperature dependence of Henry’s law), so as the oceans warm they would outgas CO2, if nothing else changed. The capacity of the water to hold dissolved CO2 decreases by about 3% per 1°C by which the water warms.
So, when the oceans are absorbing CO2, as is currently the case in most places other than the tropics, if the water warms then the oceans absorb CO2 slightly more slowly.
The CO2, in turn, works as a GHG to cause warming, which is surely one of the reasons that atmospheric CO2 levels swing up & down by about 90 ppmv over glaciation/deglaciation cycles. (There are almost certainly also biological [2] and/or ice sheet burial mechanisms at work, which increase the magnitude of glacial-interglacial CO2 swings.)
This is a slight positive feedback mechanism.
That positive feedback loop is undoubtedly one of the causes for the apparent hysteresis [2] in the temperature and CO2 records: Over the last million years, the Earth’s climate has tended to be either mild, as in our current interglacial (the Holocene), or, more of the time, heavily glaciated and cold, with relatively brief, unstable transitions between. (But see also: Deglaciation / Volcanism / CO2 Feedback.)
In paleoclimate reconstructions from ice cores, CO2 level changes generally lag temperature changes by hundreds of years, which is consistent with the fact that higher CO2 levels not only cause higher temperatures, but are also caused by higher ocean temperatures, and ocean temperature is slow to respond to air temperature changes.
Note that the rate at which the ocean absorbs CO2 from the air is proportional to CO2’s partial pressure in the air. That’s intuitively obvious when you remember that the concentration of CO2 in the air determines the rate at which CO2 molecules collide with and are absorbed by the surface of the ocean, and falling raindrops. So the measly 3% per °C, by which CO2 solubility in water decreases as the water warms, is dwarfed by the 48% by which solubility increased as atmospheric CO2 concentration rose by 48% (from 280 ppmv to 414 ppmv), and as atmospheric CO2 level continues to rise, the rate at which the oceans remove CO2 from the air will continue to accelerate.
Well put Dave. The wilful delusion that the 45% increase in CO2 is not human-caused, is in my opinion infantile.
Loydo
Reference please!
5 – 2.5 = 2.5
Starting with an example that the pH of the ocean is typically between 8 and 8.2. Then if the pH drops to 7.95 I would suggest that the ocean is in a state of acidosis, and if the pH rises to 8.3 then it is in a state of alkalosis. This is based on terms used in medicine for the state of blood, https://en.wikipedia.org/wiki/Acidosis.
That’s a faulty analogy. There are places in the depths of the ocean where vents release very acidic solutions. There are other places where liquid CO2 pools. There are places that are at the opposite extreme where processes make the water more basic than typical.
Diurnally, at certain locations, pH varies by one unit or more, for instance around many estuaries. In addition, like plant growth on land affecting local CO2 concentrations, ocean plankton do the same.
Yes, except for this: “There are other places where liquid CO2 pools.”
I don’t think so, on Earth.
https://www.pnas.org/content/103/38/14164.full
Microbial community in a sediment-hosted CO2 lake of the southern Okinawa Trough hydrothermal system
It happens where temperature is low and pressure is high enough.
Ridiculous analogy.
The range of blood pH which a human being can have and survive is very narrow.
There is no such narrow range which applies to ocean life in general.
The range of pH values that occur in the oceans is enormous.
The pH changes with depth, it changes with latitude, it changes with temperature, it changes diurnally, it changes from one ocean basin to another, it changes where any of a huge number of processes alter the chemical and physical makeup of the sea, such as where rivers enter the ocean, where rain falls, or where some geologic process such as volcanic or tectonic activity changes the chemical composition of the water. Biological activity can and does also alter pH by a significant amount.
All of these various changes exceed the small changes caused by the changing amount of CO2 in the air and then dissolved into the water from the air.
It wasn’t meant as an analogy, but rather an example of terminology used when the pH falls outside the accepted limits, in this case mammalian blood.
You are claiming the situations are analogous, hence you were attempting an analogy.
John
So, you are suggesting that oceanographers and chemists should adopt a terminology for oceans that is guided by what physicians use for human blood?
I apologise for contributing I am just an unintelligent ignoramus. I won’t bother again.
seeing this article today is amazing – just this week I was showing and explaining to my teenage science students who have to complete a dodgy assessment task on climate change – how I googled – ‘ alkaline oceans’ …………. dots indicate a few more words and then ‘alkaline’ again’at the end of my search (I did not type in the word acid or acidification) . I got from all the URL’s that the google search engine generated was information about acidification (alkaline is the opposite of this). These hits included – nature (sci journal) sites with edu in the URL. AND FINALLY one on the 5th page of google – had sceptical in the URL – and it was the only one that did not use the words acid or acidifcation of the oceans. I told my students – ‘”no wonder a fellow science teacher assumed and believed the oceans were acidic and I told them that this misinformation breaks my heart’) – it’s unbelievable sometimes – just how much misinformation is available on the internet but not surprising that people believe it because most of the time – this is all that is available via google – is misinformation
‘alkaline sea water’ search on https://duckduckgo.com/ gives good results.
Weak beer is stronger tea!