From the NORWEGIAN UNIVERSITY OF SCIENCE AND TECHNOLOGY comes this study where they actually did the experiment right – and dissed other studies where “lab tanks” were used to study the ocean.
Herring larvae could benefit from an acidifying ocean
A long-term field study in a Swedish fjord shows how rising CO2 levels can affect food webs and fish survival
One of the many downsides of too much carbon dioxide in the atmosphere is what happens when some of that CO2 is absorbed by the oceans. As atmospheric CO2 levels increase from burning fossil fuels, this carbon dioxide is soaked up by seawater and makes the oceans more acidic.
Increased acidity is bad news for coral reefs and creatures whose shells are made from calcium carbonate, but how does it affect the entire food web?
Using an unusual long-term research installation in the waters off the west coast of Sweden, a team of researchers from Germany, Sweden and Norway decided to find out. Their results have just been published in Nature Ecology and Evolution.
19-meter-long giant test tubes
Most studies on the effects of acidified seawater on fish species are done in the laboratory, says Fredrik Jutfelt, an associate professor at the Norwegian University of Science and Technology’s (NTNU) Department of Biology, who was one of the study’s authors. Some of those studies — of which there are relatively few –show reduced survival for fish in their early life stages, he said.
But studying fish in a tank don’t necessarily give researchers the chance to study the entire food web. For that you ideally need to be able to study fish in their natural habitat, the ocean. That’s where the experimental set up in Gullmarsfjord, Sweden comes in.

Here, researchers were able to use what they call “mesocosms”, or tall vertical floating “test tubes” filled with ocean water, 2.8 meters in diameter and 19 meters long, which were set out in Gullmarsfjord. Each mesocosm contained 50 m3 of ocean water.
The bags are long enough to contain an entire small ocean food web, from the tiny plankton that form the base of the food web, up to and including herring larvae.

CREDIT Rita Erven / GEOMAR
Five of the bags were filled with “normal” seawater to serve as a control, while the remaining five bags were filled with seawater that had been acidified with CO2. The researchers used predictions of the amount of CO2 that would be in the atmosphere at the end of this century to set the CO2 levels in these last five bags.
They then added fertilized herring eggs to all of the bags when the phytoplankton in the bags began to wake up and reproduce, in late April 2013. The herring larvae that hatched in both bags lived in and fed exclusively on the plankton in the bags until the experiment was ended on 28 June.
The researchers took regular samples of the physical, chemical and biological conditions in the mesocosms.
More food outweighs negative effects of acidity
The earliest days for a young herring are among the most critical in their survival, because they have to find enough of the food they like to eat.
What the researchers found was that the increased CO2 they introduced to the test bags stimulated the growth of plankton enough to improve the survival of these tiniest of young herring.
In the end, the researchers found that the young herring in the mesocosms exposed to elevated CO2 levels had a survival rate that was nearly 20 per cent higher than the herring in the control mesocosms. That’s in direct contrast to laboratory studies that have reported decreased larval fish survival.
“It appears that the herring will have an advantage over other more sensitive species in a future acidified ocean,” said Michael Sswat, a researcher from the GEOMAR Helmholtz Centre for Ocean Research Kiel, who was the lead author of the study.
Sswat and his colleagues also ran a parallel laboratory study where they raised siblings of the herring larvae in the mesocosms in the laboratory at comparable pH and CO2 levels.
“The surprising finding here was that while the herring were unaffected by CO2 in our simultaneous lab experiment, the herring in the natural ecosystem experiments in the mesocosms benefitted from high CO2,” said NTNU’s Jutfelt. “This finding was likely due to changes in the ecosystem, as that was the main difference between the lab and the field experiments. We also measured the ecosystem changes in the mesocosms and we saw that the extra CO2 stimulated growth of algae, which led to more zooplankton, meaning more food for fish.”
Catriona Clemmesen from GEOMAR, another study co-author, says that herring larvae may be naturally more able to adapt to ocean acidification.
“The tolerance of herring larvae to pH changes could be due to their life history strategy. “Herring spawn mostly near the ground, where naturally high CO2 levels prevail. They are therefore probably better adapted to ocean acidification than other fish species such as the cod that spawns near the surface,” Clemmesen said.
Not necessarily good news
Although the researchers found that the herring larvae did well in the high CO2 conditions, they caution that their findings are not a cause for celebration.
“Whereas herring larvae were shown to be tolerant to CO2 levels projected for the end of this century, larval development in other fish species, including the Atlantic cod (Gadus morhua), is negatively affected under projected ocean acidification scenarios,” the researchers wrote. “Also biodiversity of fish may be affected, as shown at natural volcanic vents, where changes in food availability and predation benefitted dominant fish species.”
###
References:
Sswat, M., M.H. Stiasny, J. Taucher, M. Alguero-Muñiz, L.T. Bach, F. Jutfelt, U. Riebesell, C. Clemmesen (2018): Food web changes under ocean acidification promote herring larvae survival. Nature Ecology & Evolution. http://dx.doi.org/10.1038/s41559-018-0514-6
Sswat, M., M.H. Stiasny, F. Jutfelt, U. Riebesell, C. Clemmesen (2018): Growth and survival of larval Atlantic herring, under the combined effects of elevated temperatures and CO2. PLOS ONE, https://doi.org/10.1371/journal.pone.0191947
‘Ocean acidification’ = red herring.
Nothing further, M’lud.
“and makes the oceans more acidic.”
Does the author know the difference between more acidic and less alkaline? One is correct and one is BS.
Makes the ocean slightly less caustic.
Don’t be fooled. This is bad news, since deep-ecologists are vegan and don’t eat fish. Many of them hate herring, except red herrings. They will say if you continue driving your suv, you’ll need to eat herring.
True is that. It would be easier to get new grants if the results were different.
In normal science the end discussion are possible interactions, further study questions, areas of further interest or inquiries into the impacts/implications of their study.
But here, in any thing associated with climate science that is not a CAGW theme, they MUST say: not to worry we are still doomed, we read this other paper that says its real bad, don’t take our IN-SITU proof out of context, back in the model-world things are more important than our paper, doom is still coming.
Grovel, snivel to the lords of CAGW while bowing and backing out of the room.
Timo –
1) This is a press release. You should expect the same things from a PR that you do from the original scientific publication.
2) Mentioning that not all fish species respond the same is a very important caveat, ensuring that people don’t make false assumptions. It need have nothing to do with the writer’s position on AGW.
You are very quick to condemn research when you presumably haven’t even seen the original publication. This is a consistent pattern, it seems, in many judgments of climate science. Surely by now the skeptic community should know better than to depend on the media for their science. Worse yet is condemnation for supposed omissions or methodological errors based solely on reading PRs and abstracts.
Wilding presumptuous of you K.S.
Didn’t condemn the research,
read the open access publication and it nicely and exactly closes with a scientific discussion.
However, the inaccessible article due to a paywall is the one supposedly quoted in the article with the ‘warnings’ to others.
You may think your caveat is important but it is a trivial bowing to CAGW.
Your “pattern”, is not our pattern but the pattern frequently found in published lit that goes against CAGW
where in order to publish they seemingly are forced to make statements like; “we all know …. because of GW, but…”
My first, exposure to this was 12 years ago a young researcher was publishing about a glacier in Greenland.
His abstract contained 3 sentences “bowing to CAGW” and then about the only real line in the abstract was about their study showed the opposite.
What I condemn I will make clear:
I condemn the PR releases and their required cowtowing,
I condemn your pompous attitudes of ‘your side’,
I condemn the condescending attitude diction of your ilk,
I condemn the failed to be released data, or code,
I condemn the likes of Lonnie Thompson, Michael Mann, and Jones,
I condemn people like Schmidt walking off stage and even refusing to be present with a fellow Ph.D scientist,
I condemn the virtue claims that the warmists raise as their ethical basis,
I condemn the models with decades of written code with real little evaluation required for such grand claims,
I condemn your holier than thou assumptions we don’t read the journal articles
And since we can assume you might know something about ph, answer this question about the above statement attributed:
“…effects of acidity.” How would you concisely explain exactly the correct usage of acidity. As you and your warmists seem to feel that ‘lower alkalinity’ should be called ‘acidification’ clarify for us this new use of acidity.
But can we take the credit for that?
https://ssrn.com/abstract=2669930
I think, it doesn’t deserve credit, because the authors (see PDF version) give the value of dissolved CO2 about 2.3 mmol/L, while in reality it is TOTAL INORGANIC CARBON (bicarbonate, carbonate and dissolved CO2), where dissolved CO2 is less than 1% of TIC.
Ocean acidification and not mentioning pH. Must have been that terribly acidic range of 8.1-8.2.
I saw that too, and so went to take a look.
The experiment went from natural conditions, CO2 = 450 ppm, pH =8.1, to horrid 22nd century conditions, CO2 = 900 ppm, pH = 7.8.
The ocean water stayed alkaline throughout. I don’t think any fish (or coral, or shelly invertebrate) would have any trouble living at pH 7.8.
There is just no way a pH 7.8 ocean can be called acidified; no way and be honest or competent, in any case.
+10 Pat Frank. Why not give the pH targets in the press release? Because any 6th grader with a pinch of earth science would laugh at the claim they used “acidified” conditions.
Pat,
If I were teaching a chemistry lab, which I used to do, and I instructed the students to acidify an alkaline solution, I would expect them to titrate the solution with an acid until the solution reached AND WENT PAST neutrality. It would then be acidic. Adding more acid would make it MORE ACIDIC. The alarmists are experts at obfuscation!
Pat Frank,
From the abstract: “The community was exposed to projected end-of-the-century CO2 conditions (~760 µatm pCO2)”
You: “I don’t think any fish (or coral, or shelly invertebrate) would have any trouble living at pH 7.8.”
You are free to think what you want, but that doesn’t make it true.
“There is just no way a pH 7.8 ocean can be called acidified; no way and be honest or competent, in any case.”
It doesn’t require honesty so much as an appreciation of what acidification (lowering of pH) means in the real-world biochemical reactions and equilibria. Those who have a problem with “acidification” meaning anything but turning pH from above 7 to below 7 are not in the field and haven’t the background (and therefore the competence) to judge.
Ms Silber==>you must have missed Rud Istvan’s comment, that the researchers depleted the buffering elements in the water, as doubling the amount of CO2 would not have ordinarily changed the pH that much in a buffered open system.
Finally somebody is doing actual science experiments. Reminds me of the CO2 fertilization experiments on vegetation and forests where real data is gathered.
Where is the experiment that verifies their default statement about the detrimental effects of CO2 on other species? This is actually more money wasted on a bigger, stupider unrealistic exercise by better funded idiots. The whole premise of climate science is that better funded idiots do better science. I don’t see the evidence.
So, John, it seems like you argue that science is a waste of money, then you say you don’t see the evidence. Do you want “evidence” or not? I don’t think so, or you would look for it. It’s much easier to deny science if one doesn’t know it’s out there.
Perhaps in an ideal world, those who are willing to contribute to ongoing science could be separated from those who aren’t, with the provision that those who don’t pay get none of the information coming from that science. This would be nice, but not practical. I have to pay for things that provide no direct benefit, and so do you. However, I accept this as part of living in a society and don’t whine about it.
“Where is the experiment that verifies their default statement about the detrimental effects of CO2 on other species?” Do you expect everything you want to be given to you so you don’t have to make an effort to inform yourself? Look it up.
Again Kristi you go holy on us. In response to your look it up. Let’s provide for you a little bit
Hint 1: Subarctic forest moths in Finnish Lapland studied for 3 decades.
Hint 2: More than 350K data points on more than 400 species.
Hint 3: 80+ dominant species studied.
Hint 4: 90% just fine or increasing abundance.
Overwhelming evidence of 3.5 degrees of warming improved their world.
Scientific conclusion: some mystery mechanism must be protecting them from harm.
In your words: “Go look it up.”
But since scholar.google.com might be difficult for you; https://onlinelibrary.wiley.com/doi/abs/10.1111/gcb.12529?utm_campaign=283df1d873-DAILY_BRIEFING&utm_medium=email&utm_source=Daily+Carbon+Briefing&utm_term=0_876aab4fd7-283df1d873-
You want the negative effects? Read the article and discover “…in models…”, “model predict…” but the
IN-SITU data, research and analysis says nothing of the sort.
I know red herring have been exploding.
They tested herring- and found out that herring actually thrived(20% better) under enhanced co2 due to increased plant growth increaseing the amount of food available .
now they need to expand the test to other cold water species, like cod etc.
it is my belief that enhanced co2= more plankton= more food fish eat= more fish mature= more fish that eat fish mature due to more fish that eat plankton/zooplankton maturing, etc…its the great circle of life…
more of low end food ULTIMATELY = more food for top predators= more live
… and the ultimate follow up studies.
“Higher survival rate of herring fry will allow poor genes to proliferate”.
“Ocean acidification helps otherwise inferior herring fry out compete their healthy brothers and sisters”
“Ocean food chain disruption similar to, and caused by the same culprit as climate disruption”
There’s got to be a lot more here … help me out Nick (you’re really good at this kind of thing).
Exactly. Why scientists haven’t tried geoengineering parts of the open ocean by seeding it with iron oxide which would naturally speed up phytoplankton growth—the foundation for everything that lives in the oceans—is beyond me. Such experiments would allow CO2 to be removed from the atmosphere and returned to the deep oceans as well as provide much-needed sources of food for otherwise nutrient-deprived fisheries. Claiming “we don’t know the ultimate outcome” of an experiment is ridiculously narrow-minded, given the brouhaha being foisted on a gullible public about ocean acidification causing depleted fisheries.
bang on the money len. they cod will be negatively affected. this goes against historical fish stocks. cod in the north east atlantic areas where herring are their main prey fish have always had higher spawning stock biomass when herring stocks have been high.
the two are inextricably linked, with cod eggs,larvae and fry being part of the herring diet at various stages.
Now, if they came out of the ocean as pickled herring, then they would gain my attention. 🙂
Acidic? You mean slightly less alkaline?
Yes, that’s my first reaction to this “ocean acidification” BS. MAJOR pet peeve. You can’t “make the oceans MORE acidic,” because the oceans AREN’T “acidic,” they are ALKALINE. You can’t have “INCREASED acidity” in something that ISN’T “acidic” to begin with. This is just intelligence insulting word games to make a minuscule ph change (which probably has a range of error as big or bigger than the supposed amount of “change”) sound “scary.”
If the “ph is lower,” just say that. OR, state it accurately, as daveburton suggests – say the oceans are “becoming less caustic.” So, we’re moving the oceans further away from, you know, LYE. Which should be a boon to ocean life. Can’t have those oceans becoming too caustic, after all…
“MAJOR pet peeve. You can’t “make the oceans MORE acidic,” because the oceans AREN’T “acidic,” they are ALKALINE.”
One of my pet peeves, too. The way they talk, you would think we are dealing with an ocean of sulfuric acid.
The growth of plants on the Earth’s surface has increased about 14 percent, so it has also probably increased the growth of plants in the oceans by quite a bit, too. More CO2 looks like just what the doctor ordered. 🙂
Ocean acidification is a distortion of the truth meant to scare uninformed people.
Re: “…makes the oceans more acidic.”
No, it only makes the oceans slightly less caustic.
Re: “Increased acidity is bad news for coral reefs and creatures whose shells are made from calcium carbonate,”
Increased CO2 levels dramatically increase growth of calcifying coccolithophores, thereby removing CO2 from the oceans. This effect seems to be much greater than expected:
https://hub.jhu.edu/2015/11/26/rapid-plankton-growth-could-signal-climate-change/
http://www.techtimes.com/articles/111246/20151127/rapid-growth-of-plankton-caused-by-increased-carbon-levels-in-the-ocean.htm
Remember the white cliffs of Dover. All built by coccolithores living and thriving in those terrible warm, acidic Cretaceous seas.
Yes, it’s amazing how ANY metric they can drum up to show SOMETHING has CHANGED (as if the Earth has never seen any of THAT before WE came along – LMFAO) is immediate cause for panic, because the ASSUMPTIONS (and that is ALL they are) are that WE are at “fault” for the “change,” and that if WE don’t “DO something” (that the Climate Fascists demand), the “trend” (from whatever cherry-picked time frame to maximize its extent) will continue infinitely until [fill in catastrophe of choice, in this example until metal boats dissolve in the oceans or something].
CO2 is THE BASIS OF ALL LIFE ON THIS PLANET. MORE is BETTER! FFS!
Acid, n. — a molecule or other entity that can donate a proton or accept an electron pair in reactions.
Is sea water an acid, no. Can it become more of something that it is not, no. Much in the same way that putting on lipstick does not make a male any more female, despite some being confused on the issue.
THERE’s your answer – it doesn’t matter what the oceans’ ph is – they “identify” as alkaline, so that’s what they are. /sarc
Well, it is “fluid,” after all. 😉
“The herring larvae that hatched in both bags lived in and fed exclusively on the plankton in the bags until the experiment was ended on 28 June…”
at which point the researchers had a big smorgasbord of herring.
No kidding … I would have loved to have been invited to that end-of-experiment celebration. Norwegians do a heck of a job making those tiny fish tasty in all manner of preparation.
God, now I’m salivating …..
I wasent dere but I been herring it wass real goot! Yah!
Supplying more of the essential nutrient CO2 to the plants (in this case, plankton) increases plant production, providing more food for herbivore animals like herring. Why is this ‘surprising’?
Breaking News: Researchers discover ‘Green Herrings’! Details to follow…. video at eleven.
I wonder too. Look at a map of primary production in the ocean (=the amount of phytoplankton):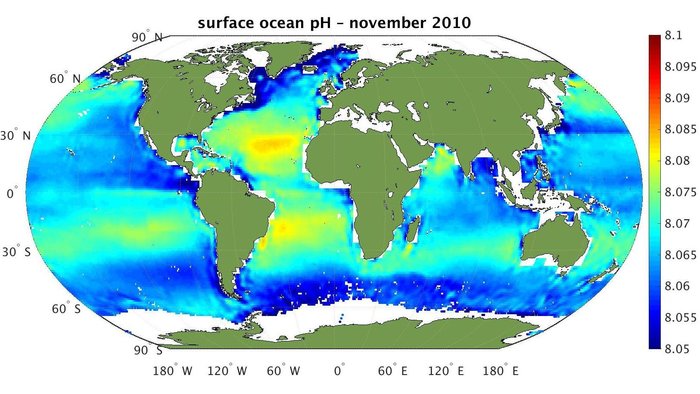
http://old.grida.no/graphic.aspx?f=series/rr-in-dead-water/Figure05.jpg
then look at ocean pH (=the amount of CO2):
and the main fishing-grounds:
http://old.grida.no/images/series/rr-in-dead-water/Figure06.jpg
Notice a pattern?
Interesting – I think you’re on to something there, tty!
Slightly lower pH seems to correspond with higher phytoplankton production and fish catch production.
Of course! And it is well known to fishermen (and should be to scientists). Areas with high CO2-water/low pH have high primary production and are also the prime fishing-grounds.
I don’t think correlation has any causation here. I think the real causation is sea surface temperature. Fish like cool water. It just so happens that cool water absorbs more CO2, and becomes somewhat less alkaline.
The most alkaline regions are the surface, but the surface layer is nutrient poor due to sinking. Upwelling zones bring more nutrients as well as more DIC and thus lower pH water into the sunlit waters where phytoplankton can flourish and thus so can fish.
The world oceans are all nicely alkaline and likely to remain that way! I have actually been testing oceans around the world (unlike Scrips) and have never gotten a reading below pH 8. Maybe if they would climb out of their basement lab full of large aquariums and go outside to test the nearby Pacific, they would find similar conditions.
The famous geochemist Konrad Krauskopff stated that even in the most extreme of conditions in the oceans, anoxic sulfur dioxide-rich waters, it would be improbable for the waters to even reach neutrality let alone become acidic.
Oceans are buffered. In those pH 7.8 tubes the buffer must have been used up. Doubling CO2 given actual seawater buffering cannot lower ocean pH from 8.1 to below about 7.95. Chemistry Details with references in essay Shell Games.
So even tho in a fjord, those mesocosms are still just very big artificial acquariums.
Good point.
Would Al Gore fit in one of these bags?
I’m sure his carbon footprint is too large. What about just his cod dead hands?
Only if he ditched his ego.
This is not entirely fair, but only took me a short time, enough to suggest considerable evaluation for revision and probably shortening, as also checking carefully any criticism. It takes a long time to do this properly. I worked on larval fishes once and found this an interesting mesocosm study. The stage from yolk to predation has long been known to be a most critical stage, which they acknowledge, when mortality rates are as bad as it gets, and 5 day sampling is a long time in their life.
From the herring paper—-“Ocean warming has been suggested to increase total energy expenditure for metabolism [82], while ocean acidification has been hypothesized to mainly increase energy expenditure for osmoregulation [75].” See below
From the abstract (last line)—-“ These findings emphasize the need to include biotic factors such as energy supply via prey availability in future studies on interactive effects of multiple stressors.” This is so generalized and well known as to be unnecessary to be included.
From citation number 75 for cod paper–
https://link.springer.com/article/10.1007%2Fs00360-015-0923-7
From the introduction —“Depending on emission scenario atmospheric PCO2 levels are projected to reach between 420 and 940 µatm by the year 2100, consistent with an average decrease in surface ocean pH by 0.13–0.42 units.” That is quite a range, not something I would be proud of having any significance.
From the abstract—-“Overall, the gill as a key ion regulation organ seems to be highly effective in supporting the resilience of cod to effects of ocean warming and acidification.” Seems a bit more positive than the citation above, “highly effective…resilience” more important than “mainly increase energy expenditure.” Their glass is too often half empty (or more).
The Dutch love herring. Every year, the first day with ‘New Herring’ is celebrated, nation wide. In the Netherlands, the result of this research must definitely switch the balance in favour of CO2! ?zoom=1.5&resize=297%2C170
?zoom=1.5&resize=297%2C170
Thanks for this. I will add this to my Earth Day talk on ocean acidification
It is at sea as on land. When pCO2 increases, photosynthesis increases, and when photosynthesis increases at sea the number of phytoplankton increases and subsequently those that graze on them. Pending on the fish species, some fish larvae start on phytoplankton before they gradually switch to to zooplankton during the very first phase of their life. Those that start directly on zooplankton will also benefit from more larval zooplankton forms around. Fish larvae have a extremely bad odds and the mortality rates are as highest during first feeding. Vision is limited as is swimming capacity – down to some body lengths for some species. Thus survival is closely connected to the number of and often also the right number of food items nearby. The number of food items is far more important than having some extra energy allocated to what models claim is an extra energy cost. Mesocosmos studies have sampling restrictions: To improve data resolution you increase the risk of depleting the animal at study too fast. Anyway, the key message is that more CO2 is good for fish stock recruitment. The Peruvians should be thrilled by this study as their anchovy has algae as part of their food even as adult fish. The same should be the case for all commercial fishermen and game fishermen catching fish species with small larvae at hatching cause many of them need algae too to get going.
The exuberant Cambrian explosion during which multicellular life triumphantly burst onto the planet out of single-celled ancestors, took place under atmospheric concentration of CO2 of 10,000 – 50,000 ppm.
Of the twenty-something phyla (basic designs) of living animals today, nearly all appeared during this extraordinary “dream-time”, about 541 million years ago, evolving over a period of thousands to a few million years. Thus the term “explosion”. Nearly all the significant evolution of basic body plans took place during this intense crucible of life-forms. Evolution in the following half billion years has amounted to little more than minor tweaking on established designs.
Among all the phyla emerging at this time were several which developed calcified parts – the shelled Mollusca, the bony Chordates, the sessile corals and sponges, the armour plated arthropods and others.
These calcified forms evolved from single celled and then microscopic multicelled organisms. If ever there was a time when the biosphere was vulnerable to CO2 acidification of the sea compromising
calcium carbonate/phosphate precipitation in biological tissue, then this was it! Frail and tiny life-forms living in seawater experimented with calcifying their tissues – could they have done this in an acidic ocean with the pH of a bottle of coke?
And yet they calcified. Somehow not getting the memo about CO2 acidification, they proliferated millions upon millions of varying calcified body variations, upon which natural selection fashioned the eventual animal forms and phyla that have – through constantly changing climate and CO2 levels, survived to this day.
The “ocean acidification” scare is a testament to just how spectacularly wrong a supposedly scientific investigation can be when it goes down a reductionist inductive route, ignoring direct deductive inference from the salient facts and building and inductive structure of assumption upon assumption upon poorly representative lab water tank experiments, like a rickety pile of child’s play-bricks. The scientific community needs to learn from this failure.
Technology will never over-rule epistemology.
Had two good friends that did very large mesocosm research in Western Canada coastal region. Much large mesocosm then were used here. What I don’t understand is how these Norwegian research jumped to conclusions about cod. My “guess” would be that cod larvae would have improved survival and growth just as the herrings did. Adult cod are demersal fish but their larvae are pelagic.
A quick internet search found that Black Smokers (hydrothermal vents) have a pH of around 2-5 in the immediate vicinity of the fluids coming out. However, there is no shortage of adapted life living in the hot transition zone.
Clyde, I have used deep sea hydrothermal vents since their discovery in 1977 as an example of just how little we know about the Earth, especially the oceans,75% of the Earth’s surface. After years where some claimed they were rare and unusual now it seems each time someone tries to estimate how many there are the estimates go up dramatically. From one in every 200 kilometers to one in every 20 kilometers along the mid-ocean ridges. Somewhere I had an article about several being studied off the southern tip of South American that flowed more water through than than the Amazon. With water temperature approaching 700 degrees F that is a lot of hot acidic water.
“Life” has a tremendous diversity of species, able to survive under nearly all circumstances: from the hottest to the coldest circumstances, from very low pressure high in the mountains to a pressure of hundreds of atmospheres down in the deep sea where species are surviving without any light at all. At every level, some species thrive, others just survive. As soon as circumstances change, other species will thrive, while the previously dominant species will diminish.
When ocean water cools as it flows to the poles, other species will do well. We do find corals at the poles but we don’t find tropical coral fishes over there.
In AlgaeBase different species of algae are collected. On the website is written: “About 200,000 records have been entered, but the task is still very incomplete, particularly for non-marine algae.” For Europe I did a search: 28359 Found Species.
There is some more information about the number of species of bacteria. In the Abstract of ‘Species Numbers in Bacteria’ I found the following numbers: “From this I estimate there are a million species of bacteria in 30 grams of rich forest topsoil and propose that there will be at least a billion species worldwide.”
Life will always survive. It adapts to changing circumstances. As it does do when warm surface water flows to the poles. Which happens 24/7/365, proving that ‘life’ adapts, every day of the year.
Info:
Corals at the poles: https://soundwaves.usgs.gov/2010/11/meetings.html
Algaebase: http://www.algaebase.org/search/distribution/
Bacteria: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3160642/
Do they mention how much the pH changed? Could it be that the pH is irrelevant and that increased planckton grows is the reason the larvae did well?
Sorry, growth.
I wonder reading this, have they done similar experiments with cods or do they just assume that cod will be suffering by increased acidification of their habitat…?