NASA Reveals New Results From Inside the Ozone Hole – Dec. 11, 2013

NASA scientists have revealed the inner workings of the ozone hole that forms annually over Antarctica and found that declining chlorine in the stratosphere has not yet caused a recovery of the ozone hole.
More than 20 years after the Montreal Protocol agreement limited human emissions of ozone-depleting substances, satellites have monitored the area of the annual ozone hole and watched it essentially stabilize, ceasing to grow substantially larger. However, two new studies show that signs of recovery are not yet present, and that temperature and winds are still driving any annual changes in ozone hole size.
“Ozone holes with smaller areas and a larger total amount of ozone are not necessarily evidence of recovery attributable to the expected chlorine decline,” said Susan Strahan of NASA’s Goddard Space Flight Center in Greenbelt, Md. “That assumption is like trying to understand what’s wrong with your car’s engine without lifting the hood.”
To find out what’s been happening under the ozone hole’s hood, Strahan and Natalya Kramarova, also of NASA Goddard, used satellite data to peer inside the hole. The research was presented Wednesday at the annual meeting of the American Geophysical Union in San Francisco.
Kramarova tackled the 2012 ozone hole, the second-smallest hole since the mid 1980s. To find out what caused the hole’s diminutive area, she turned to data from the NASA-NOAA Suomi National Polar-orbiting Partnership satellite, and gained the first look inside the hole with the satellite’s Ozone Mapper and Profiler Suite’s Limb Profiler. Next, data were converted into a map that shows how the amount of ozone differed with altitude throughout the stratosphere in the center of the hole during the 2012 season, from September through November.
The map revealed that the 2012 ozone hole was more complex than previously thought. Increases of ozone at upper altitudes in early October, carried there by winds, occurred above the ozone destruction in the lower stratosphere.
“Our work shows that the classic metrics based on the total ozone values have limitations – they don’t tell us the whole story,” Kramarova said.

The classic metrics create the impression that the ozone hole has improved as a result of the Montreal protocol. In reality, meteorology was responsible for the increased ozone and resulting smaller hole, as ozone-depleting substances that year were still elevated. The study has been submitted to the journal of Atmospheric Chemistry and Physics.
Separate research led by Strahan tackled the holes of 2006 and 2011 – two of the largest and deepest holes in the past decade. Despite their similar area, however, Strahan shows that they became that way for very different reasons.
Strahan used data from the NASA Aura satellite’s Microwave Limb Sounder to track the amount of nitrous oxide, a tracer gas inversely related to the amount of ozone depleting chlorine. The researchers were surprised to find that the holes of 2006 and 2011 contained different amounts of ozone-depleting chlorine. Given that fact, how could the two holes be equally severe?
The researchers next used a model to simulate the chemistry and winds of the atmosphere. Then they re-ran the simulation with the ozone-destroying reactions turned off to understand the role that the winds played in bringing ozone to the Antarctic. Results showed that in 2011, there was less ozone destruction than in 2006 because the winds transported less ozone to the Antarctic – so there was less ozone to lose. This was a meteorological, not chemical effect. In contrast, wind blew more ozone to the Antarctic in 2006 and thus there was more ozone destruction. The research has been submitted to the journal Geophysical Research Letters.
This work shows that the severity of the ozone hole as measured by the classic total column measurements does not reveal the significant year-to-year variations in the two factors that control ozone: the winds that bring ozone to the Antarctic and the chemical loss due to chlorine.
Until chlorine levels in the lower stratosphere decline below the early 1990s level – expected sometime after 2015 but likely by 2030 – temperature and winds will continue to dictate the variable area of the hole in any given year. Not until after the mid 2030s will the decline stratospheric chlorine be the primary factor in the decline of ozone hole area.
“We are still in the period where small changes in chlorine do not affect the area of the ozone hole, which is why it’s too soon to say the ozone hole is recovering,” Strahan said. “We’re going into a period of large variability and there will be bumps in the road before we can identify a clear recovery.”
Related Links
› NASA Goddard’s Ozone Hole Watch website
scarletmacaw says:
December 12, 2013 at 8:42 am
===============
Thanks for that reply, they seem rather skittish on “Lake Of The Woods” in Minnesota.
Home to the northern-most point in the contiguous United States, due to a badly written legal description that the surveyors had to follow 🙂
As I stated above, that was “An attempt by somebody to ‘debunk’ this aspect;” … I haven’t been able to nail or confirm everything as to ‘fact or fiction’, but, if R-12 was being manufactured in 1935 as claimed (and it would have to have been patented then or prior to 1935) THEN in 1955 that patent would have expired (20 some years after the 1935 date) … does that make sense? This is simple logic – patents ‘run’ (are effective, if paid up) for a period of time (IOW they have an ‘expiration’ date, if you will.)
You’ll also notice, per Wiki’s own words: “The critical DuPont manufacturing patent ” which is a patent titled “Process for fluorinating halohydro-carbons“, and NOT a patent for R12 or Freon (Freon appears to be a trademark, a different entity) per se. Does that make sense? IOW it’s a patent on how a particular process is performed, to wit, fluorinating halohydro-carbons – a manufacturing process that is covered by patent acting to protect Du Pont’s intellectual property as a manufacturer, as it says.
Wiki, BTW, needs cross-checking just as any other source up to the original documents (like the patent cite.)
Are things completely cleared up? I don’t think so, not yet … certain dates need to be verified yet.
.
Gary Pearse says:
December 12, 2013 at 9:33 am
ferd berple, you have come closest to recognizing an element of my hypothesis (my post of 603 am):
==========
Gary you may well be onto something. When I first read you post it didn’t register at all. How can air be magnetic? It isn’t something you learn in school.
But one of things that has really bugged me about arctic warming is the rapid shift in the earth’s north magnetic pole. It is easy to dismiss this as coincidence, but on what basis?
I had thought it might be an interaction of the magnetic field with the solar wind, but now you have given us another previously unrecognize mechansim. A paramagnetic material (O2) moving in a magnetic field may well separate the O2.
Werner Brozek says:
December 12, 2013 at 1:15 pm
I see no way that gases in the atmosphere will be affected by any magnetic field as low as Earth’s.
===========
my fridge magnets have a stronger magnetic field locally, but they don’t deflect compasses all over the earth.
re: Khwarizmi says December 12, 2013 at 4:49 pm
I think this is the patent you seek (note: in the following CCl2Fl2* represents Freon R-12):
– – – – – – – – –
Title: Manufacture of halo-fluoro derivative of aliphatic hydrocarbons, US 2007208 A
Publication date . . Jul 9, 1935
Filing date . . . . . Feb 24, 1931
Priority date . . . . Feb 24, 1931
Inventors . . . . . . . Henne Albert L, Midgley Jr Thomas, Reed Mcnary Robert
Original Assignee . . Gen Motors Corp
https://www.google.com/patents/US2007208
.
.
* CCl_sub2_Fl_sub2
ferd berple says:
December 12, 2013 at 5:31 pm
my fridge magnets have a stronger magnetic field locally, but they don’t deflect compasses all over the earth.
True, but if you held your fridge magnet near a compass on your kitchen table, the compass would move according to your fridge magnet. So if Earth’s magnetic field is expected to attract O2 molecules, the fridge magnet should do so much more. But if the fridge magnet cannot do so locally (unless you have proof to the contrary) then why should Earth be expected to do so globally?
Thanks _Jim,
I’ll have a look at that later today. It was the method of manufacture, not the product, that was subject to the patent.
Meanwhile, here’s a brief summary of Sagan’s weird and interesting version of the history:
http://wattsupwiththat.com/2013/07/20/the-handsomest-fox-in-the-henhouse/#comment-1367758
Cheers.
Just to tie a ribbon on it, the announcement of dichlorodifiuoromethane (R-12) in a white paper in 1930:
“Organic Fluorides as Refrigerants”
Thomas Midgley Jr., Albert L. Henne
Ind. Eng. Chem., 1930, 22 (5), pp 542–545
http://pubs.acs.org/doi/abs/10.1021/ie50245a031?journalCode=iechad
The more or less ‘formal’ incorporation of dichlorodifiuoromethane as a constituent part of an ‘invention'” via this patent:
– – – – – – – – – – – – – –
Title: “Heat transfer” US 1833847 A
Publication number US1833847 A
Publication type . . . Grant
Publication date . . . Nov 24, 1931
Filing date . . . . . . . Feb 8, 1930
Priority date . . . . . . Feb 8, 1930
Inventors . . . . . . . . Henne Albert L, Mcnary Robert R, Midgley Jr Thomas
Original Assignee . . Frigidaire Corp
– – – – – – – – – – – – – –
We find the ‘meat of the nut’ in the claims section:
I got there in a roundabout way using these works:
1. http://www.fluoride-history.de/p-freon.htm
2. http://www.scs.illinois.edu/~mainzv/HIST/bulletin_open_access/v31-2/v31-2%20p66-74.pdf
Number 2 contains a lot of background and history and corrects some of the ‘myths’ surrounding the reason and development of the first “Freon”.
.
re: Khwarizmi says December 12, 2013 at 7:14 pm
This might be of interest, too, given your last:
THOMAS MIDGLEY, JR., AND THE INVENTION OF CHLOROFLUOROCARBON
REFRIGERANTS: IT AIN’T NECESSARILY SO
By Carmen J. Giunta, Le Moyne College
http://www.scs.illinois.edu/~mainzv/HIST/bulletin_open_access/v31-2/v31-2%20p66-74.pdf
This generally applicable paragraph buried towards the end of the above document particularly caught my eye:
The above cited doc also has a rich ‘references and notes’ section.
.
Brian says: mumble, mumble, mumble. crap, mumble mumble,……
December 12, 2013 at 8:50 am
Just wondering how in Brian’s mind that critiquing bad science equates to conspiracy theory ideation.
(This is getting old .. second submission of this particular post since the first ‘bounced’)
re: Khwarizmi says December 12, 2013 at 7:14 pm
This might be of interest, too, given your last:
THOMAS MIDGLEY, JR., AND THE INVENTION OF CHLORO FLUORO CARBON
REFRIG ER ANTS: IT AIN’T NECESSARILY SO
By Carmen J. Giunta, Le Moyne College
http://www.scs.illinois.edu/~mainzv/HIST/bulletin_open_access/v31-2/v31-2%20p66-74.pdf
This generally applicable paragraph buried towards the end of the above document particularly caught my eye:
The above cited doc also has a rich ‘references and notes’ section.
.
(Try a 2nd post on this one too as the 1st disappeared right promptly into lala-land)
Just to tie a ribbon on it, the announcement of di chloro di fluoro methane (R-12) in a white paper in 1930:
“Organic Fluorides as Refrig erants”
Thomas Mi dgley Jr., Albert L. Henne
Ind. Eng. Chem., 1930, 22 (5), pp 542–545
http://pubs.acs.org/doi/abs/10.1021/ie50245a031?journalCode=iechad
The more or less ‘formal’ incorporation of di chloro di fluoro methane as a constituent part of an ‘invention'” via this patent:
– – – – – – – – – – – – – –
Title: “Heat transfer” US 1833847 A
Publication number US 1833847 A
Publication type . . . Grant
Publication date . . . Nov 24, 1931
Filing date . . . . . . . Feb 8, 1930
Priority date . . . . . . Feb 8, 1930
Inventors . . . . . . . . Henne Albert L, Mcnary Robert R, Midgley Jr Thomas
Original Assignee . . Frigidaire Corp
– – – – – – – – – – – – – –
We find the ‘meat of the nut’ in the claims section:
I got there in a roundabout way using these works:
1. http://www.fluoride-history.de/p-freon.htm
2. http://www.scs.illinois.edu/~mainzv/HIST/bulletin_open_access/v31-2/v31-2%20p66-74.pdf
Number 2 contains a lot of background and history and corrects some of the ‘myths’ surrounding the reason and development of the first “Freon”.
.
Oh – thanks guys / m o d s! _Jim
john robertson says:
December 11, 2013 at 6:57 pm
They do sound bewildered and frustrated, as their methods worked so well in the ozone scare, perhaps they do not understand why the CO2 scare is failing so miserably.
There is something that might explain it. Civilization can get along without CFCs, but not without fossil fuels.
ROM says:
December 12, 2013 at 3:59 am
The corruption of the natural sciences was preceded and foreshadowed by the corruption of economics. We used to think that, short of outright dictatorship, the physical sciences were immune to such manipulation. We were wrong.
u.k.(us) says:
Good fences make good neighbors.” (Robert Frost)
Frost only quoted this saying, and not with approval. Read the poem.
Since Svensmark published his evidence for cosmic ray chemistry in the atmosphere, i’ve wonered if the chloroflurocarbons have been given a bad rap. …. maybe it should be revisited in light of the
new knowledge of the muon produced high energy electrons in the troposphere …
Werner Brozek says:
December 12, 2013 at 1:15 pm
Gary Pearse says:
December 12, 2013 at 11:46 am
http://en.wikipedia.org/wiki/Earth's_magnetic_field
“Earth’s surface ranges from 25 to 65 micro Tesla”
“In your Table 2, the range is from 25 to 750,000 milliT. So the lowest number here is about 1000 times stronger than Earth’s magnetic field. Is that right? And the ”
Werner, It is not a strong field, but please lets agree that there is an effect. Happily I found a demonstration on You Tube by a Harvard prof. The only thing is the prof doesn’t appear to understand the diamagnetic aspect. N2 is not just unattracted to the magnet, it is REPELLED by the magnet. So if you have molecules attracted to the magnet AND molecules repulsed by a magnet, there will be some degree of separation.
Also, you may or may not be aware of a phenomenon known as a ferro fluid:
“In a gradient field the whole fluid responds as a homogeneous magnetic liquid which moves to the region of highest flux. This means that ferrofluids can be precisely positioned and controlled by an external magnetic field. The forces holding the magnetic fluid in place are proportional to the gradient of the external field and the magnetization value of the fluid. This means that the retention force of a ferrofluid can be adjusted by changing either the magnetization of the fluid or the magnetic field in the region.”
https://www.ferrotec.com/technology/ferrofluid/
It causes the particles (read oxygen) to align and they too become a magnetic body that further repels its diamagnetic friends. I found this effect occurring when trying to separate fine ferromagnetic grains from very fine grained paramagnetic rare earth mineral grains in a liquid. The ferromagnetic grains immediately magnetized the liquid and a captured its neighboring weaker paramagnetic grains and removed them from the suspension with them, even though the rare earth minerals were too weakly magnetic to be attracted to the outside field applied.
re: Gary Pearse says December 13, 2013 at 5:54 am
I got no ‘bite’ on this comment linked below the first time (5 days ago?) regarding gas ‘reaction’ to magnetic fields, so I point back to it now:
. . . http://wattsupwiththat.com/2013/12/07/ipccs-report-on-climate-change-myths-realities/#comment-1495479
In particular, the paper below which used a really strong magnetic field to affect the sublimation of CO2 from a block of dry ice:
. . Effects of Gradient Magnetic Fields on CO2 Sublimation in Dry Ice
. . http://iopscience.iop.org/1742-6596/156/1/012029/pdf/1742-6596_156_1_012029.pdf
Also note the other paper was looking at the mixing ratio between O2 and N2 and at heights above 100km where some ‘separation’ or settling out (stratification) may occur as opposed to lower altitudes where turbulent vertical mixing keeps the two well mixed:
. . http://acmg.seas.harvard.edu/people/faculty/djj/book/bookchap2.html
_Jim says:
December 13, 2013 at 6:57 am
re: Gary Pearse says December 13, 2013 at 5:54 am
“I got no ‘bite’ on this comment linked below the first time (5 days ago?) regarding gas ‘reaction’ to magnetic fields, so I point back to it now:”
Thanks Jim and sorry I seem to have missed it. Yes, I accept that the idea should be fleshed out with other data and, indeed, some calculations. Although I have used ferro, para and diamagnetic phenomena in the mineral processing field, I am someway out of my depth to present calculations of this effect in the atmosphere – a medium that seems to be even giving physicists a lot of trouble. I proposed a long time ago that given uranium’s significant paramagnetism, it could be cleaned out of affected water using a high gradient “mesh” or steel wool magnetic filter. I was told the idea should be tried and possibly patented – but like the atmospheric situation, I didn’t have the resources to do much about it, except to mention on as thread somewhere when Fukushima reactors were breached that the water could be pumped from the site through a magnetic filter.
Regarding data, the first round should be devoted to see if there is difference in gas species content around the south pole, the temperate and equatorial regions. I didn’t know that NO is also paramagnetic before some small research I did in response to questions. Maybe it is more abundant at the South Pole, too. Ive mentioned confounding aspects or weather and biology in the equatorial zone wrt CO2 and O2 but one should find elevated N2 and noble gases there and a hole for all atmospheric gases except O2 and NO at the south polar region. Once again, I would be obliged to defer such research to someone else. It would be nice if someone reading this could put there hands on whatever data there is. After that, I would also have to rely on a physicist or engineer familiar with the details of magnetic behavior in such a setting to get a “measure” of the effect – tiny or significant.
Thanks for your interest and links.
The ozone hole over Antarctica has always had too large seasonal changes for it to be anything other than mainly natural cycle. What, if any human affect on it by CFC’s, is too small to even notice.
http://www.cpc.ncep.noaa.gov/products/stratosphere/polar/gif_files/ozone_hole_plot.png
I’m betting there has always been a ozone hole (area of weak concentration) and we cant distinguish any natural changes with unnatural.
scarletmacaw says:
December 12, 2013 at 9:00 am
Mike Tremblay says:
December 12, 2013 at 1:14 am
You present a very good summary.
The correlation between CFC production and the decrease in ozone levels at the south pole do provide evidence that CFCs are the cause. But we don’t have any measurements before CFCs were introduced into the atmosphere, so there’s no definitive proof.
There is one other comment I’d like to make. As far as I know, no one has ever actually detected CFCs in the stratosphere. I remember reading that a satellite was launched to measure CFCs in the stratosphere, but got a null result and the results were then suppressed. I don’t know whether or not that story is true.
CFCs are routinely measured in the stratosphere, here’s a graph.
http://www.epa.gov/ozone/science/images/FIG-FAQ02.JPG
This paper describes measurements made as early as 1975!
http://link.springer.com/article/10.1007%2FBF01636907#page-1
re: Phil. says December 13, 2013 at 10:45 am
Post was kinda thin on specifics there Phil .. one resource is sitting behind a paywall.
.
“That assumption is like trying to understand what’s wrong with your car’s engine without lifting the hood.”
Sorry, I just can’t leave that go Susan! This must be magic, eh?
Welcome to the 90’s ↘
Thanks for the links. I’m curious as to what they actually say since the first link is an undescribed graph and the second is just an abstract. How were the measurements made in 1975?
The first link appears too smooth to be a graph of actual measurements, so I suspect it is a model.
Gary Pearse says: December 12, 2013 at 9:33 am
ferd berple says: December 12, 2013 at 6:46 am
Yes and yes. Here is helpful image that shows how the Northern Polar Vortex formed and created a Northern “Ozone Hole” in 2011:
http://ozonewatch.gsfc.nasa.gov/facts/images/2011-02-22_LNH.png
For reference the images above are:
And this:
[caption id="" align="alignnone" width="578"]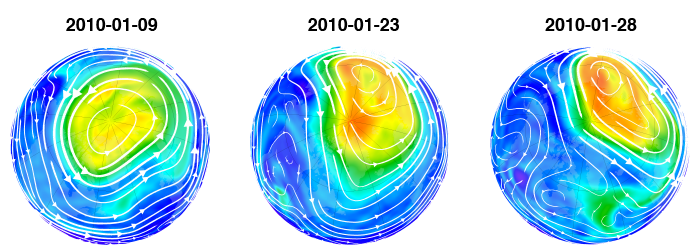 NOAA – National Aeronautics and Space Administration (NASA) Goddard Space Flight Center (GSFC) – Click the pic to view at source[/caption]
NOAA – National Aeronautics and Space Administration (NASA) Goddard Space Flight Center (GSFC) – Click the pic to view at source[/caption] NOAA – National Aeronautics and Space Administration (NASA) Goddard Space Flight Center (GSFC) – Click the pic to view at source[/caption]
NOAA – National Aeronautics and Space Administration (NASA) Goddard Space Flight Center (GSFC) – Click the pic to view at source[/caption]
The source of this images, this NASA page on Polar Vortices;
[caption id="" align="alignnone" width="578"]
is very informative, however it is confounding that they demonstrate the mechanics of the natural formation of “Ozone Holes” within Polar Vortices and then claim on this page;
http://ozonewatch.gsfc.nasa.gov/facts/NH.html
that:
If there is a physical process that adequately explains the existence of Ozone Holes, why do we need a supplemental CFC based chemical process? How big would the ozone hole be if CFCs didn’t exist? It would be the same size, as ozone hold size depends on how big the Polar Vortex is and how deep it penetrates into the atmosphere.
There is also another important physical process in play within the Polar Vortex, is that
Air towards the top of the stratosphere has lower concentrations of ozone;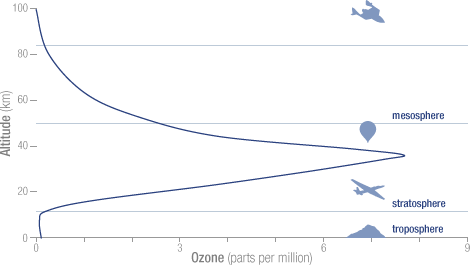 NOAA – National Aeronautics and Space Administration (NASA) Goddard Space Flight Center (GSFC) – Click the pic to view at source[/caption]
NOAA – National Aeronautics and Space Administration (NASA) Goddard Space Flight Center (GSFC) – Click the pic to view at source[/caption]
[caption id="" align="alignnone" width="578"]
As such, when this “air from very high altitudes descends vertically through the center of the vortex” it displaces the air below it, decreasing the concentration of ozone within the Polar Vortex. The combination of the low pressure area formed by the centrifugal force of the Polar Vortex and the air from very high altitudes with lower concentrations of ozone that descends vertically through the center of the vortex, creates the “Ozone Hole”.