Guest Post by Willis Eschenbach
Pushed by a commenter on another thread, I thought I’d discuss the R. W. Wood experiment, done in 1909. Many people hold that this experiment shows that CO2 absorption and/or back-radiation doesn’t exist, or at least that the poorly named “greenhouse effect” is trivially small. I say it doesn’t show anything at all. Let me show you the manifold problems with the experiment.
To start with, let me give a curious example of the greenhouse effect, that of the Steel Greenhouse. Imagine a planet in the vacuum of space. A residue of nuclear material reacting in the core warms it to where it is radiating at say 235 watts per square metre (W/m2). Figure 1 shows the situation.
 Figure 1. Planet in outer space, heated from the interior. Drawing show equilibrium situation
Figure 1. Planet in outer space, heated from the interior. Drawing show equilibrium situation
This planet is at equilibrium. The natural reactor in the core of the planet is generating power that at the planet’s surface amounts to 235 W/m2. It is radiating the same amount, so it is neither warming nor cooling.
Now, imagine that without changing anything else, we put a steel shell around the planet. Figure 2 shows that situation, with one side of the shell temporarily removed so we can look inside.
 Figure 2. As in Figure 1, but with a solid steel shell surrounding the planet. Near side of the shell temporarily removed to view interior. Vertical distance of the shell from the surface is greatly exaggerated for clarity—in reality the shell and the shell have nearly the same surface area. (A shell 6 miles (10 km) above the Earth has an exterior area only 0.3% larger than the Earth’s surface area.)
Figure 2. As in Figure 1, but with a solid steel shell surrounding the planet. Near side of the shell temporarily removed to view interior. Vertical distance of the shell from the surface is greatly exaggerated for clarity—in reality the shell and the shell have nearly the same surface area. (A shell 6 miles (10 km) above the Earth has an exterior area only 0.3% larger than the Earth’s surface area.)
[UPDATE: Misunderstandings revealed in the comments demonstrated that I lacked clarity. To expand, let me note that because the difference in exterior surface area of the shell and the surface is only 0.3%, I am making the simplifying assumption that they are equal. This clarifies the situation greatly. Yes, it introduces a whopping error of 0.3% in the calculations, which people have jumped all over in the comments as if it meant something … really, folks, 0.3%? If you like, you can do the calculations in total watts, which comes to the same answer. I am also making the simplifying assumption that both the planet and shell are “blackbodies”, meaning they absorb all of the infrared that hits them.]
Now, note what happens when we add a shell around the planet. The shell warms up and it begins to radiate as well … but it radiates the same amount inwards and outwards. The inwards radiation warms the surface of the planet, until it is radiating at 470 W/m2. At that point the system is back in equilibrium. The planet is receiving 235 W/m2 from the interior, plus 235 W/m2 from the shell, and it is radiating the total amount, 470 W/m2. The shell is receiving 470 W/m2 from the planet, and it is radiating the same amount, half inwards back to the planet and half outwards to outer space. Note also that despite the fact that the planetary surface ends up much warmer (radiating 470 W/m2), energy is conserved. The same 235 W/m2 of power is emitted to space as in Figure 1.
And that is all that there is to the poorly named greenhouse effect. It does not require CO2 or an atmosphere, it can be built out of steel. It depends entirely on the fact that a shell has two sides and a solid body only has one side.
Now, this magical system works because there is a vacuum between the planet and the shell. As a result, the planet and the shell can take up very different temperatures. If they could not do so, if for example the shell were held up by huge thick pillars that efficiently conducted the heat from the surface to the shell, then the two would always be at the same temperature, and that temperature would be such that the system radiated at 235 W/m2. There would be no differential heating of the surface, and there would be no greenhouse effect.
Another way to lower the efficiency of the system is to introduce an atmosphere. Each watt of power lost by atmospheric convection of heat from the surface to the shell reduces the radiation temperature of the surface by the same amount. If the atmosphere can conduct the surface temperature effectively enough to the shell, the surface ends up only slightly warmer than the shell.
Let me summarize. In order for the greenhouse effect to function, the shell has to be thermally isolated from the surface so that the temperatures of the two can differ substantially. If the atmosphere or other means efficiently transfers surface heat to the shell there will be very little difference in temperature between the two.
Now, remember that I started out to discuss the R. W. Wood experiment. Here is the report of that experiment, from the author. I have highlighted the experimental setup.
Note on the Theory of the Greenhouse
By Professor R. W. Wood (Communicated by the Author)
THERE appears to be a widespread belief that the comparatively high temperature produced within a closed space covered with glass, and exposed to solar radiation, results from a transformation of wave-length, that is, that the heat waves from the sun, which are able to penetrate the glass, fall upon the walls of the enclosure and raise its temperature: the heat energy is re-emitted by the walls in the form of much longer waves, which are unable to penetrate the glass, the greenhouse acting as a radiation trap.
I have always felt some doubt as to whether this action played any very large part in the elevation of temperature. It appeared much more probable that the part played by the glass was the prevention of the escape of the warm air heated by the ground within the enclosure. If we open the doors of a greenhouse on a cold and windy day, the trapping of radiation appears to lose much of its efficacy. As a matter of fact I am of the opinion that a greenhouse made of a glass transparent to waves of every possible length would show a temperature nearly, if not quite, as high as that observed in a glass house. The transparent screen allows the solar radiation to warm the ground, and the ground in turn warms the air, but only the limited amount within the enclosure. In the “open,” the ground is continually brought into contact with cold air by convection currents.
To test the matter I constructed two enclosures of dead black cardboard, one covered with a glass plate, the other with a plate of rock-salt of equal thickness. The bulb of a thermometer was inserted in each enclosure and the whole packed in cotton, with the exception of the transparent plates which were exposed. When exposed to sunlight the temperature rose gradually to 65 oC., the enclosure covered with the salt plate keeping a little ahead of the other, owing to the fact that it transmitted the longer waves from the sun, which were stopped by the glass. In order to eliminate this action the sunlight was first passed through a glass plate.
There was now scarcely a difference of one degree between the temperatures of the two enclosures. The maximum temperature reached was about 55 oC. From what we know about the distribution of energy in the spectrum of the radiation emitted by a body at 55 o, it is clear that the rock-salt plate is capable of transmitting practically all of it, while the glass plate stops it entirely. This shows us that the loss of temperature of the ground by radiation is very small in comparison to the loss by convection, in other words that we gain very little from the circumstance that the radiation is trapped.
Is it therefore necessary to pay attention to trapped radiation in deducing the temperature of a planet as affected by its atmosphere? The solar rays penetrate the atmosphere, warm the ground which in turn warms the atmosphere by contact and by convection currents. The heat received is thus stored up in the atmosphere, remaining there on account of the very low radiating power of a gas. It seems to me very doubtful if the atmosphere is warmed to any great extent by absorbing the radiation from the ground, even under the most favourable conditions.
I do not pretend to have gone very deeply into the matter, and publish this note merely to draw attention to the fact that trapped radiation appears to play but a very small part in the actual cases with which we are familiar.
Here would be my interpretation of his experimental setup:
 Figure 3. Cross section of the R. W. Wood experiment. The two cardboard boxes are painted black. One is covered with glass, which absorbs and re-emits infrared. The other is covered with rock salt, which is transparent to infrared. They are packed in cotton wool. Thermometers not shown.
Figure 3. Cross section of the R. W. Wood experiment. The two cardboard boxes are painted black. One is covered with glass, which absorbs and re-emits infrared. The other is covered with rock salt, which is transparent to infrared. They are packed in cotton wool. Thermometers not shown.
Bearing in mind the discussion of the steel greenhouse above, I leave it as an exercise for the interested reader to work out why this is not a valid test of infrared back-radiation on a planetary scale … please consider the presence of the air in the boxes, the efficiency of the convective heat transfer through that air from the box to the cover plates, the vertical temperature profile of that air, the transfer of power from the “surface” to the “shell” through the walls of the box, and the relative temperatures of the air, the box, and the transparent cover.
Seems to me like with a few small changes it could indeed be a valid test, however.
Best regards,
w.
What a silly example. Prior to the metal shell being put around the sphere, the temperature of the sphere would be just sufficient to radiate the wattage of the internal heat source to the vacuum of space. If the temperature was too low, the sphere would continue to heat until the temperature was just high enough to radiate away the energy. Adding the additional thermal resistance of a steel shell in series with the thermal resistance to the vacuum of space will actually reduce the temperature of the sphere as the emitting area is increased. Leaving a space, presumably containing a vacuum between the sphere and the shell would do nothing since this would have the same thermal resistance as from the sphere to space.
This would be the same as adding another heatsink on top of the existing heatsink on your cpu. The thermal resistance between the heatsink and the atmosphere is so high that adding additional metal actually helps cooling by increasing the surface area for emitting heat while adding only very slightly to the series thermal resistance between the chip and atmosphere.
For another example, adding a 1″ length of 20′ diameter pipe to a mile long length of 3/4″ hose will not appreciably increase the flow through the hose.
Also, note that your soup doesn’t get hotter when you put it into a Thermos in spite of the reflected IR radiation from the silvered sides of the Dewar flask.
Problem #4
The radiative ghe changes the observed temperature of the earth as seen from space by exactly zero. With no ghe the earth as seen from space would be 255K and the temperature at surface would be 255K. As seen from space with a ghe, the temperature of the earth IS 255K and the temperature of the surface IS 288K, However, the average height at which 255K occurs is 14 km instead of at surface.
As this experiment only measured equilibrium temperature, and only at one elevation in each box, it does not and cannot be used as a simulation of the radiative ghe.
Problem #5
The radiative ghe is distributed from earth surface to TOA. This experiment puts a sheet of glass in to represent the ghe of the atmosphere, but does so at what would be the TOA, but with conductance and convection still active, which doesn’t exist at the TOA.
Greg, of course it has been proven, many times over. It’s in every college thermo textbook.
>>>>>>>>>>>>>>>>
Not to mention that verifying the thermo laws via experimentation is part of the curriculum in many universities. A quick search for SB Law alone gets:
http://uregina.ca/~szymanss/uglabs/p242/Experiments/EXPT1.pdf
http://www.fiziks.net/lifesciences/exp54.htm
http://www3.wooster.edu/physics/jris/Files/Carter.pdf
http://personal.tcu.edu/zerda/manual/lab22.htm
http://wanda.fiu.edu/teaching/courses/Modern_lab_manual/stefan_boltzmann.html
http://sky.campus.mcgill.ca/Exp/Manuals/en0049.pdf
Is that enough or should I copy and paste the entire 177,000 results that searching “stefan-boltzmann law experiment” produced?
Not to mention that when a bunch of engineers design, oh, I don’t know, let’s go with a nuclear reactor…they figure out exactly how much power the fission process will produce, how much of it they can capture and use to run generators, how much will turn into waste heat, how much that waste heat will raise the temperature of everything from the boilers to the turbines, exactly how much water at a given temperature and flow rate will be required to keep the system cooled to a given operating temperature, and they nail it to fractions of a percent before the construction even starts, and they do so using the precise same laws of physics that we’re discussing right now.
Who out there thinks the laws of physics don’t work like the thermo texts say they do and the engineers just nailed their design by shear luck? A few hundred nuclear reactors in a row? C’mon, hands up. Greg and who else?
Michael Moon
I’m not saying Willis is right but I don’t think he’s wrong because of what you say.
“The steel would radiate to space, but transfer ZERO heat back to the planet, as we all know that the Second Law is true”
If by 2nd law you mean the movement of a system to maximum entropy this will not invalidate the above model. Remember this is a conceptual model? Why would it not radiate back to the planet, if the shell is cooler then the net transfer would be outward from the planet but there would be a some transfer of energy to the planet from the shell (radiative transfer is emitted from and toward bodies irrespective of current state (it doesn’t care about energy state); maximum entropy only requires that the net flow will be toward the cooler body). Eventually, if the planet were to run out of its internal heat then both would eventually become the same temperature (all other things being equal).
Your point about the steel being cooler. Yes to begin with, but with constant supply of energy it will warm, and yes it can’t get warmer than the emitter but it can get warmer and will return some energy back to the surface with the result that the surface of the planet will warm.
Willis wrote (re multiple light bulbs in close proximity);
”I say both of them will run hotter in that situation, because A warms B, and B warms A. If you disagree, I urge you to buy a thermometer and bulbs and sockets and do the test. Me, I don’t need to do it, the physics of the situation is quite clear to me.”
Please note that in this example you have TWO energy SOURCES.
Please replicate this thought experiment with two steel shells in close proximity (sans additional heat sources), do you speculate that steel shell A heats steel shell B (and vice versa) and the temperature of both rise ?
If that’s the case I could just replace my electric baseboard heaters with steel slabs, boy that would sure be a lot cheaper.
Cheers, Kevin.
@ur momisugly Kev-in-Uk: Actually “Greenhouse Effect” is also an incorrect term/expression, as greenhouses rely on physical barriers, not just varying amounts of gasses, to do what they do. You are correct that while the planet does NATURALLY warm and cool, anyone who suggests there is any measurable ANTHROPOGENIC warming is only fooling themselves.
Willis Eschenbach says:
February 6, 2013 at 5:01 pm
“…60-watt bulb. Measure the temperature, we’ll say it’s 150°C. Try a 100-watt bulb, it’s hotter, say 200°C.”
“Now, mount another socket right next to the first one. Insert a 100-watt bulb in one and a 60-watt bulb in the other. What will the final temperatures of the bulbs be?”
The temperature will not exceed 160 watts.
cd says:
“Your point about the steel being cooler. Yes to begin with, but with constant supply of energy it will warm, and yes it can’t get warmer than the emitter but it can get warmer and will return some energy back to the surface with the result that the surface of the planet will warm.”
Not true. Thermodynamic entropy is defined in units of energy per degree of temperature. Once temperatures are matched (delta T = 0), then no energy is transferred between bodies.
Willis:
I don’t know how many times I’ve seen your steel shell-game, but I still ain’t buyin’ it. You are creating energy from nothing there, IMHO. I’m certainly not spending any time disputing this nonsense.
But my main interest is in the Wood experiments, and despite reading your post several times, I still haven’t figured out just how the shell-game is relevant. I just want to know why the IR backradiation from the glass does not make the inside of the glass greenhouse any warmer than the air in the “salt” greenhouse, which cannot have any such backradiation. And how the atmosphere can cause warming (or slow down cooling), if even the glass cannot do it.
Then you end the post with a puzzle for us:
“Bearing in mind the discussion of the steel greenhouse above, I leave it as an exercise for the interested reader to work out why this is not a valid test of infrared back-radiation on a planetary scale … please consider the presence of the air in the boxes, the efficiency of the convective heat transfer through that air from the box to the cover plates, the vertical temperature profile of that air, the transfer of energy from the “surface” to the “shell” through the walls of the box, and the relative temperatures of the air, the box, and the transparent cover.”
After all those words, you are leaving it as an exercise to the reader….?? WOW, this seems like a cop-out! Aren’t YOU the one who is supposed to be demonstrating why this experiment does not tell us anything about how backradiation helps heat (or prevents heat loss, or whatever)??
KevinK
Please replicate this thought experiment with two steel shells in close proximity (sans additional heat sources)
>>>>>>>>>>>
You’ve constructed a thought experiment which is in thermal equilibrium. By definition, net energy flux is zero. This tells you precisely nothing about a system which is not in thermal equilibrium (such as the baseboard heaters in your house).
Sparks says:
“The temperature will not exceed 160 watts.”
Temperature is not measured in watts. You have the right idea though. The temperature measured in any body will be the equivalent of having absorbed 160Watts of energy, which is defined by the heat capacity of the body defined in terms such as J / (kg K).
Who out there thinks the laws of physics don’t work like the thermo texts say they do and the engineers just nailed their design by shear luck? A few hundred nuclear reactors in a row? C’mon, hands up. Greg and who else?
>>>>>>>>>>>>>>>>
OK two hands. Greg and jae. Anyone else?
davidmhoffer says: “Greg, of course it [that A warms B and then B warms A back] has been proven, many times over. It’s in every college thermo textbook.“.
Unfortunately, as I read them, all the links you provided were only tests of Stefan Boltzmann Law, not of “A warms B and then B warms A back”.
I don’t doubt that “A warms B and then B warms A back” but it seems that a test of that specific hypothesis is needed to settle the matter. Roy Spencer’s set-up is definitely testable.
KevinK says:
February 6, 2013 at 5:31 pm
Willis wrote (re multiple light bulbs in close proximity);
Please note that in this example you have TWO energy SOURCES.
Your argument, as I understand it, is that a cooler object cannot radiate energy to a warmer object.
If that is correct, then the fact that they are transforming energy from electricity to heat is immaterial.
w.
jae says:
February 6, 2013 at 6:17 pm
I can’t tell you how I wish your final sentence were actually true … and no matter how many times you’ve seen it, you still haven’t found an error in it, or if you have you’re keeping it secret.
w.
Willis, pretty picture but,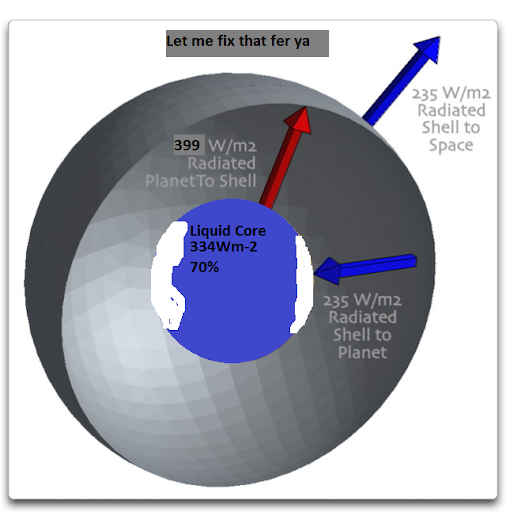
Let me fix that for ya.
KevinK @ur momisugly 4:50 – During the winter there is generally less humidity, so that would cause the air to feel cooler than in the summer with the same temperature; but, in line with your point, the folks around here with brick houses require less wood to keep their homes heated once the bricks absorb the heat. I place a pot of water on my wood stove to increase the percentage of water vapor in the house to help with the sensation of heat.
Chad Jessup
the reason that houses with warm bricks require less ‘heat’ to maintain temperature is that the bricks are no longer absorbing heat, and their emissivity/conductivity is such that the rate of heat transfer from the exterior of the wall is very low. Hence the heating system only needs to keep up with the rate the bricks carry heat to the outside world .
All the units and quantities are irrelevant because the implication here is that a photon leaves the planet surface, interacts with an electron in the shell then somehow one photon is emitted back and another one into space.
Doesn’t seem right.
Mike Jonas;
Unfortunately, as I read them, all the links you provided were only tests of Stefan Boltzmann Law, not of “A warms B and then B warms A back”.
>>>>>>>>>>>>>>>>
My point was that the laws of thermodynamics are found in every university level text book and are validated through experimentation, of which SB Law is a single example. You really want me to go through the list of all the laws and post links to experiments validating each and every one of them?
On the other hand, your complaint that SB Law has nothing to do with a warms b suggests that you don’t have a clue what SB Law is or how to apply it. Otherwise you wouldn’t have said such a thing.
Gino says:
February 6, 2013 at 6:21 pm
Sparks says:
“The temperature will not exceed 160 watts.”
Temperature is not measured in watts. You have the right idea though. The temperature measured in any body will be the equivalent of having absorbed 160Watts of energy, which is defined by the heat capacity of the body defined in terms such as J / (kg K).
The temperature will not exceed 160 watts, convert it to joules to watts and watts to temperature. My point is valid.
Doing a simple calculation with the Stephan-Boltzman equation suggests that with black body radiation within a vacuum, the ratio of the planet surface temperature to the steel shell temperature is 2^.25 or (1.189). So if the surface of the planet was 100 degrees C, shell would have to be 40 degrees C.
joletaxi in English so all can understand the exchange:
Willis responds:
Sparks:
Watts do not convert to temperature without the heat capacity of the material. The point being that two items can have the same temperature but vastly different heat quantities.