Guest post by Bob Fernley-Jones by Bob Fernley-Jones AKA Bob_FJ
CAUTION: This is written in Anglo-Oz English.
Here is the diagram as extracted from their 2009 paper, it being an update of that in the IPCC report of 2007 (& also 2001):
The unusual aspect of this diagram is that instead of directly showing radiative Heat Transfer from the surface, it gives their depiction of the greenhouse effect in terms of radiation flux or Electro-Magnetic Radiation, (AKA; EMR and a number of other descriptions of conflict between applied scientists and physicists). EMR is a form of energy that is sometimes confused with HEAT. It will be explained later, that the 396 W/m^2 surface radiation depicted above has very different behaviour to HEAT. Furthermore, temperature change in matter can only take place when there is a HEAT transfer, regardless of how much EMR is whizzing around in the atmosphere.
A more popular schematic from various divisions around NASA and Wikipedia etc, is next, and it avoids the issue above:
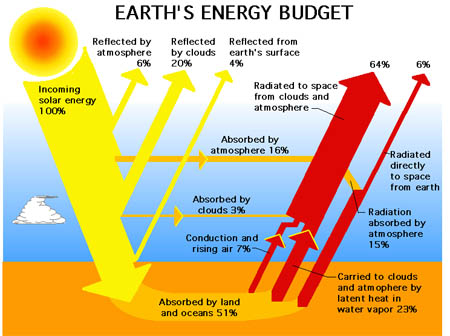
- Figure 2 NASA
Returning to the Trenberth et al paper, (link is in line 1 above), they give that the 396 W/m2 of EMR emitted from the surface in Fig.1 is calculated primarily by using the Stefan–Boltzmann law, and global year average conditions. Putting aside a few lesser but rather significant issues therein, it is useful to know that:
1) The Stefan-Boltzmann law (S-B) describes the total emission from a flat surface that is equally radiated in all directions, (is isotropic/hemispherical). Stefan found this via experimental measurement, and later his student Boltzmann derived it mathematically.
2) The validity of equally distributed hemispherical EMR is demonstrated quite well by observing the Sun. (with eye protection). It appears to be a flat disc of uniform brightness, but of course it is a sphere, and at its outer edge, the radiation towards Earth is tangential from its apparent surface, not vertical. It is not a perfect demonstration because of a phenomenon called limb darkening, due to the Sun not having a definable surface, but actually plasma with opacity effects. However, it is generally not apparent to the eye and the normally observed (shielded) eyeball observation is arguably adequate for purpose here.
3) Whilst reportedly the original Stefan lab test was for a small flat body radiating into a hemisphere, its conclusions can be extended to larger areas by simple addition of many small flat bodies of collectively flat configuration, because of the ability of EMR waves to pass through each other. This can be demonstrated by car driving at night, when approaching headlights do not change in brightness as a consequence of your own headlights opposing them. (not to be confused with any dazzling effects and fringe illumination)
4) My sketch below demonstrates how radiation is at its greatest concentration in the lateral directions. It applies to both the initial S-B hemispherical surface radiation and to subsequent spherical radiation from the atmosphere itself.
5) Expanding on the text in Figure 3: Air temperature decreases with altitude, (with lapse rate), but if we take any thin layer of air over a small region, and time interval, and with little turbulence, the temperature in the layer can be treated as constant. Yet, the most concentrated radiation within the layer is horizontal in all directions, but with a net heat transfer of zero. Where the radiation is not perfectly horizontal, adjacent layers will provide interception of it.
A more concise way of looking at it is with vectors, which put simply is a mathematical method for analysing parameters that  possess directional information. Figure 4, takes a random ray of EMR (C) at a modestly shallow angle, and analyses its vertical and horizontal vector components. The length of each vector is proportional to the power of the ray, in that direction, such that A + B = C. Of course this figure is only in 2D, and there are countless multi-directional rays in 3D, with the majority approaching the horizontal, through 360 planar degrees, where the vertical components also approach zero.
possess directional information. Figure 4, takes a random ray of EMR (C) at a modestly shallow angle, and analyses its vertical and horizontal vector components. The length of each vector is proportional to the power of the ray, in that direction, such that A + B = C. Of course this figure is only in 2D, and there are countless multi-directional rays in 3D, with the majority approaching the horizontal, through 360 planar degrees, where the vertical components also approach zero.
6) Trenberth’s figure 1 gives that 65% of the HEAT loss from the surface is via thermals and evapo-transpiration. What is not elaborated is that as a consequence of this upward HEAT transfer, additional infrared radiation takes place in the air column by virtue of it being warmed. This initially starts as spherical emission and absorption, but as the air progressively thins upwards, absorption slows, and that radiation ultimately escapes directly to space. Thus, the infrared radiation observable from space has complex sources from various altitudes, but has no labels to say where it came from, making some of the attributions “difficult”.
DISCUSSION; So what to make of this?
The initial isotropic S-B surface emission, (Trenberth’s global 396 W/m2), would largely be absorbed by the greenhouse gases instantaneously near the surface. (ignoring some escaping directly to space through the so-called “atmospheric window”). However, a large proportion of the initial S-B 396 surface emission would be continuously lateral, at the Trenberth imposed constant conditions, without any heat transfer, and its horizontal vectors CANNOT be part of the alleged 396 vertical flux, because they are outside of the vertical field of view.
After the initial atmospheric absorptions, the S-B law, which applied initially to the surface, no longer applies to the air above. (although some clouds are sometimes considered to be not far-off from a black body). Most of the air’s initial absorption/emission is close to the surface, but the vertical distribution range is large, because of considerable variation in the photon free path lengths. These vary with many factors, a big one being the regional and more powerful GHG water vapour level range which varies globally between around ~0 to ~4%. (compared with CO2 at a somewhat constant ~0.04%). The total complexities in attempting to model/calculate what may be happening are huge and beyond the scope of this here, but the point is that every layer of air at ascending altitudes continuously possesses a great deal of lateral radiation that is partly driven by the S-B hemispherical 396, but cannot therefore be part of the vertical 396 claimed in Figure 1.
CONCLUSIONS:
The vertical radiative flux portrayed by Trenberth et al of 396 W/m^2 ascending from the surface to a high cloud level is not supported by first principle considerations. The S-B 396 W/m^2 is by definition isotropic as also is its ascending progeny, with always prevailing horizontal vector components that are not in the field of view of the vertical. The remaining vertical components of EMR from that source are thus less than 396 W/m^2.
It is apparent that HEAT loss from the surface via convective/evaporative processes must add to the real vertical EMR loss from the surface, and as observed from space. It may be that there is a resultant of similar order to 396 W/m^2, but that is NOT the S-B radiative process described by Trenberth.
~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~
ADDENDUM FOR AFICIONADOS
I Seek your advice
In figure 5 below, note that the NIMBUS 4 satellite data on the left must be for ALL sources of radiation as seen from space, in this case, at some point over the tropical Pacific. The total emissions, amount to the integrated area under the curve, which unfortunately is not given. However, for comparison purposes, a MODTRAN calculator, looking down from 100 Km gives some interesting information for the figure, which is further elaborated in the tables below. Unfortunately the calculator does not give global data or average cloud/sky conditions, so we have apples and pears to compare, not only with Nimbus, but also with Trenberth. However, they all seem to be of somewhat similar order, and see the additional tabulations.
| Compare MODTRAN & “Trenberth”, looking down from 2 altitudes, plus Surface Temperature | ||||
| Location | Kelvin | 10 metres | 100 Km. | (Centigrade) |
| Tropical Atmosphere | 300K | 419 W/m^2 | 288 W/m^2 | (27C) |
| Mid-latitude Summer | 294K | 391 W/m^2 | 280 W/m^2 | (21C) |
| Mid-latitude Winter | 272K | 291 W/m^2 | 228 W/m^2 | (-1C) |
| Sub-Arctic Winter | 257K | 235 W/m^2 | 196 W/m^2 | (-16C) |
| Trenberth Global | 288K ? | 396 W/m^2 | 239 W/m^2 | (15C ?) |
| Compare MODTRAN & “Trenberth”, looking UP from 4 altitudes: W/m^2 | ||||
| Location | From 10 m | From 2 Km | From 4Km | From 6Km |
| Tropical Atmosphere | 348 | 252 | 181 | 125 |
| Mid-latitude Summer | 310 | 232 | 168 | 118 |
| Mid-latitude Winter | 206 | 161 | 115 | 75 |
| Sub-Arctic Winter | 162 | 132 | 94 | 58 |
| Trenberth Global | 333 Shown as coming from high cloud area (= BS according to MODTRAN) | |||



Nasif Nahle, I was looking at the paper by Hottel http://dspace.mit.edu/handle/1721.1/42950 (direct link: http://dspace.mit.edu/bitstream/handle/1721.1/42950/02748698.pdf?sequence=1 ) for clues on how to interpret that table.
The data table that shows approximately the 0.2 emissivity for CO2 is table 1.4.1-2 on page 29.
On page 24 it says:
“In order to check the consistency of the different independent experimenters and to check the agreement between the present computed emissivities and the experimental results it was necessary to reduce all data to a common basis of Pc = 0 and Pt = 1 atm.”
This normalization comment and tables such as table 4.2.2.5-1 on page 113 suggest that a single table (1.4.1-2) was constructed from values obtained at different atm. The table allows one to lookup path length easily.
I believe they use some theoretical procedure to make the conversion, so the experiment data is “corrupted” by this procedure, but it does suggest that what Tim Folkerts was saying is correct.
Let’s say CO2 in our atmosphere is at a partial pressure of .00038 atm. If we consider a height of say 1000000 cm (ie, 10 km) in our atmosphere, we multiply this by .00038 atm to get 380 cm*atm. We then look up 380 (cm*atm) on the table 1.4.1-2. The row with nearest PL has the entry 300 on the far left. Then on the next column over (300 Kelvin) we find .215. That value would be the emissivity for CO2 according to that table. That value represents a normalized result of numerous experiments calculating emissivity of CO2 at different temperatures, for different path lengths, at different atmospheres.
Nasif Nahle:
This paper “Effective Atmospheric Emissivity under Clear Skies” ( http://www.patarnott.com/atms411/pdf/StaleyJuricaEffectiveEmissivity.pdf ) is from March 1972 Journal of Applied Meteorology.
Fig 1 on page 352 (pg 4 of pdf) shows “effective atmosphere emissivity of CO2” at various pressures and for 3 mixing ratios. I think the mixing ratios (in g/kg) relate to fractional composition of CO2 in the atmosphere and would then appear to be in the ballpark of our current CO2 in the atmosphere (at about .04% in terms of number of molecules). All 3 curves suggest a 1013 mbar (1 atm) value of approximately between 0.17 and 0.2.
As a second example, Table 1 on pdf page 6 is for the 1013 mbar case and has CO2 at .19.
If these figures (and those from Hottel’s table 1.4.1-2) are in the ballpark (and if I am understanding properly from skimming the paper), they suggest the emissivity of CO2 in our atmosphere is on the order of tenths of 1 rather than thousandths of 1.
Jose_X @ur momisugly December 1, at 9:45 pm
I was going to ignore your comments, but it keeps coming back to me that it is very sad that you are so gullible and naïve as to believe for example the source that you quote:
You say that you want to study and learn, but when you are recommended to read FREE information, ….. Oh, I’d better stop there….. but will instead quote something that Wayne commented to you:
I could show more proof that the hockey-team and others are corrupt, but it is really up to you to study and learn, and it is off-topic.
Nasif Nahle, googling around for various things, I came across an article written by you, I’m assuming.
I will link to it here since it involves exactly the calculations and results you mentioned here for CO2 emissivity. http://jennifermarohasy.com/2011/03/total-emissivity-of-the-earth-and-atmospheric-carbon-dioxide/
I’ll try and go over it carefully tomorrow if possible (and maybe even look at the reference in the comments to Schwarzchild’s equations.)
Bob Fernley-Jones, your last comment didn’t make much sense to me. Is there a “free” link you want me to read? Is there a problem you found in Barry Bickmore’s write-up? Do you have a bias against Bickmore and automatically assume his write-up is garbage?
Nasif, I’m lost with the equation:
>> Ɛcd = [1 – (((a-1 * 1 –PE)/(a + b – (1 + PE)) * e^(-c (Log10 ((paL)m / paL)^2))] * (Ɛcd)0
I don’t know the meanings of:
1. a
2. b
3. PE
4. c
5. paL
6. m
7. (paL)m
8. (Ɛcd)0
Also, is the beginning:
1. a minus (1 times 1) minus PE
or
2. (a minus 1) times (1 minus PE)
Does “((paL)m / paL)” reduce to “m” or is (pal)m a distinct variable?
@ur momisugly Nasif Nahle
This is probably my last post too. You have mentioned a few calculations you have done to find emissivity.
You suggest the the emissivity of the CO2 in the atmosphere as a whole is ~ 0.002 — ie that well under 1% of the IR energy passing thru the CO in the atmosphere is absorbed
* The published reference I linked to disagrees
* The satellite data posted at the top of this page disagrees
* This simple experiment disagrees: http://www.youtube.com/watch?v=Ot5n9m4whaw
* The entire science behind global warming disagrees.
When such and array of data and theory are weighed against your calculations, I will believe these results and questions your calculations.
Nasif, I found the book Modest, Michael F. Radiative Heat Transfer-Second Edition. 2003. Elsevier Science, USA and Academic Press, UK. on googlebooks: http://books.google.com/books?id=lLT-aKLTxkQC&pg=PA826&lpg=PA826&dq=Modest,+Michael+F.+Radiative+Heat+Transfer-Second+Edition.+2003.+Elsevier+Science,+USA+and+Academic+Press,+UK.&source=bl&ots=7k4Sxqi87s&sig=DqLHm-TPB9S-a3ICO2YSyvX-Yko&hl=en&ei=nsfaTvGiFZDAgQf154GpBg&sa=X&oi=book_result&ct=result&resnum=3&ved=0CC0Q6AEwAg#v=onepage&q=paL&f=false
This is a book you referenced in the http://jennifermarohasy.com/2011/03/total-emissivity-of-the-earth-and-atmospheric-carbon-dioxide/ article for the formula you used [the one having a, b, c, PE, and some other values].
On page 342 there is a formula that looks sort of like what you provided, but the formula is set equal to a ratio and is used as an adjustment where you look up the base emissivity and then apply the formula if you are working under non 1 atm environment. The book also provides a table of the meanings of the various constants/variables (many of which look like what you provided).
The book also leverages the Hottel data (and the Leckner fixes, which are similar) and mentions that some of his extrapolated data is a bit off, but I think the deviations are on the order of some percentage points and mostly for the higher temp cases.
I applied the formula 10.145 (although I didn’t have to for 1 atm environment, as the calculations below will show) using values of .00038 bar for CO2 partial pressure and 300 Kelvin and total pressure of 1 bar. The result of the ratio was very close to 1 (just like in the book example 10.11 that uses different scenario). Because the ratio is about 1, the emissivity of CO2 would be the reference one, which basically can be estimated from Hottel’s work as given on figure 10-25 on page 342 of the google book. Those values are shown as curves and resemble the Hottel table Tim Folkerts linked to. Each curve corresponds to a row in the table. The “y” value of the curve is the emissivity and the “x” values would be the temperature. These curves in figure 10-25 seem to almost overlap the table in emissivity values.
Notes:
formula 10.145 with values already substituted:
emiss /(emiss base) = 1 + ((.000057 / 1.8001) * exp((-1.47) * (log(.6)^2))) = 1.00002945;
using t=.3, a=1.57, b=.23, c=1.47, (PaL)m / (PaL)0=.6, PE=1.0001 (see table 10.5 on page 344 of google book).
Recap: We can look up the values for CO2 emissivity for a given depth * partial pressure at some temp using the Hottel experimental results done at about 1 atm. The ratio formula (10.145) is used as a correction factor if we were calculating for a pressure other than 1 atm. [so that formula was not useful for the earth atmosphere climate case; it would be useful for lab experiments and any system under different enough pressure from 1 atm.] Conclusion, the CO2 emissivity can be taken (more or less) from the table on page 29 of the 1972 Hottel paper linked to earlier. That is what the Modest, Michael F. Radiative Heat Transfer-Second Edition appears to be doing around page 342+.
BTW, this and the last prior comment I made resourcing Modest, Michael F. Radiative Heat Transfer-Second Edition and performing a calculation (equal to 1) with a formula that looks a fair amount like the formula used by Nasif when deriving .002 emissivity of CO2, and where that book appears to reference Hottel’s and Leckner’s work, will probably be my last comment (following Tim and maybe Nasif’s lead) debating the emissivity value of CO2. The result appears to be pretty much what Tim Folkerts had concluded numerous comments earlier upon first(?) referencing Hottel’s 1972 work on CO2 and H2O emissivity. Going several kilometers up the atmosphere, the CO2 emissivity appears to be near 0.2.
Oh… Now, I see. The formula in the book is supposed to be the one Nasif stated.
To answer a prior comment…
First, the formula is wrong in that the parenthesis are off (probably due to copy/paste formatting slip-up).
As per Radiative Heat Transfer-Second Edition:
Ɛcd = [1 – [(a-1)*(1-PE)/(a + b – 1 + PE)] * e (-c (Log10 (p_CO2_L_m / p_CO2_L_0) )^2) ] * Ɛcd_0
.. and not //// XXXX Ɛcd = [1 – (((a-1 * 1 –PE)/(a + b – (1 + PE)) * e (-c (Log10 ((paL)m / paL)^2))] * (Ɛcd)0
Ɛcd = emissivity. The formula is useful mainly when the total pressure under consideration is not 1 bar (aka, 1 atm) since otherwise we likely get approximately Ɛcd = Ɛcd_0.
Ɛcd_0 = emissivity value at 1 bar. For CO2, we can look up from the Hottel table.
T = temperature in kelvin; eg, 300 K.
T_0 = 1000 K.
t = T / T_0; eg, 0.3 .
a = 1 + ((.1) / (t^1.45)) for CO2; eg, 1.57 .
b = .23 for CO2.
c = 1.47 for CO2.
p = total pressure in bar (1 bar is about 1 atm); eg, 1 bar.
p_CO2 = partial pressure of CO2 in bar; eg, .00038 bar is about partial pressure of CO2 in atmosphere.
PE = (p + .28 * p_CO2) / (1 bar) for CO2; eg, 1.0001
p_CO2_L_m / p_CO2_L_0 = 0.054 / t^2 for CO2 and t<0.7 according to the book table, but this may need adjustment using the actual definition of p_CO2_L_m and with P_CO2_L_0 = 1 bar cm.
Jose_X @ur momisugly December 3, at 4:38 pm
YAWN; I REPEAT part of what I said with added bold: I could show more proof that the hockey-team and others are corrupt but it is really up to you to study and learn, and it is off-topic.
As you have repeatedly stated that you want to study and learn, and you do appear to be quite an intelligent chappie with much divergent thinking, it would be appropriate if you were to follow your declared principles, and read and digest the stuff recommended to you. (although I find it distressing that you cannot afford the valuable journalistic research of the IPCC corruptness in the downloadable Laframboise digital book at $4.99)
Oh BTW, did you ever hear about the Amman and Wahl fiasco involving the IPCC, Mann, McIntyre, journal rejections, editorial staff changes, and different versions and dates of their papers used for various naughty purposes, and substantial other whatnots. Here follows a nice lucid and pleasantly readable account, and it’s FREE:
http://bishophill.squarespace.com/blog/2008/8/11/caspar-and-the-jesus-paper.html
You won’t like it of course, because it does not fit with your dogma. Nevertheless, you might benefit from the high quality of the English prose.
If it weren’t so serious, the A & W fiasco would be funny.
Jose, BTW, please read my Andrew Montford (Bishop Hill) reference just above, carefully and with an open mind. I don’t know if language might be an issue here but I guess that your native tongue might be Spanish or Portuguese. If that is the case, I sincerely complement you in your good English, as a second tongue, but wonder if you need to be more cautious in what you say.
Perhaps draft what you want to say in a word processor, and then come back to it later, preferably the next day, and examine it for ambiguities. This was something I as an Englishman by birth commonly did in the past in industry, especially with tricky issues.
With your divergent thinking you may be able to contribute here, but please be more careful.
Jose_X says:
December 3, 2011 at 8:20 am
Myrhh, where do you draw the boundary between electromagnetic radiation that can impart kinetic energy and that which cannot? Why do you draw the boundary there?
I draw the boundary here because traditional practical real world tried and tested and well understood physics has already categorised it, it is the difference between Heat and Light, the science fields of optics and thermodynamics. Since the AGWSF manipulation of the education system this distinction has been so blurred that older papers can’t be understood properly by most now and typically we have all the above confusion as seen in these arguments, for example Tim who makes out that heat is either something esoteric that can’t be understood or it’s all a matter of semantics so use the word as you want. Tim is typical here in AGWSF influence in denying that heat is as traditional physics describes it, as he has done a few posts up, and, typical in denying that thermal infrared direct from the Sun heats the land and oceans, going with the science fiction KT97 ilk produced by AGW pushers to dumb down the population, that visible light heats land and oceans, in the real world this is impossible. Visible light is Light, not Heat. OK, let’s start with traditional understanding of Heat.
So, lots of confusion created by AGWSF to better sell its propaganda, it has reduced the physical world to a one dimensional reality, nothing in it has any properties or characteristics; not as here in the meme ‘all electromagnetic energy is the same’ and ‘all absorption of energy heats’, nor in the properties of molecules where ‘oxygen, nitrogen and carbon dioxide are ideal gases’. You really need to appreciate what I’m saying here, in traditional physics you have to put those properties, and so the differences between them, back into the picture. Molecules have weight and volume and attraction and electromagnetic waves have size and so on.
These are simple, basic differences, don’t be distracted into making it more complicated than it is, even a primary school child should be able to understand the following, and certainly by secondary school level. It is now longer easy to find traditional physics taught, you have to go into the applied science world where it you’ll find it because real physics is a description of the real world and they learn how real things work.. For advanced take dscott’s advice here: http://wattsupwiththat.com/2011/09/29/1-k-or-not-1-k-that-is-the-question-2/#comment-755585
For basics, solid understanding of basics without which you cannot build anything and understanding of which will stop you from going off into dead end muddled tangents as Tim et al do, and, which will enable you to see where Tim and ilk confuse read this:
Italics as used in the piece.
Further:
So. What we feel is heat created by the mechanical energy in rubbing our hands together, and, heat is thermal energy in transfer. There’s no confusion here. Why not? Because it’s one and the same thing.
The only confusion is in Tim et al (sorry Tim, just using you as example here because you’ve posted), confusing the situation because they don’t understand this. Because they don’t understand it they make up their own claims about what it is, and then they extrapolate from them to claim, to them it appears logical, that visible light from the Sun heats land and oceans, but get stumped when asked to prove it, because they can’t. All they can do is obfuscate more. As dscott put it so well: “What we have here is the classic false proof, where the assumption at the outset of logical thought process is incorrect. In typical fashion you scientific types run with the flawed assumptions, apply flawless logic and then end up with a false conclusion.”
So remember, it’s not semantics, it’s a knowledgeable use of the word heat by people who understand what it is and can tell by context when others understand what it is, and can see when they don’t:
“Thermal energy and heat are often confused. Rightly so because they are physically the same thing. Heat is always the thermal energy of some system. Using the word heat helps physicists to make a distinction relative to the system they are talking about.”
So, the great thermal energy of the Sun is heat in the Sun; it is heat in radiative transfer to us as it flows from the Sun in straight lines and reaches us in around eight minutes; it is this heat we feel when our bodies absorb this thermal energy transfered to us by radiation, (one of the three ways of heat transfer) as it heats the water in our bodies; the heat from the Sun in transfer by radiation is heat; this the invisible thermal infrared, which is heat, which is heat energy, which is thermal energy in transfer by radiation, which is why it is called thermal.
It is not visible light, visible light is not thermal. Near infrared is not thermal.
Recall the kid’s page from NASA – the difference in size between the not hot near infrared and thermal infrared, near infrared microscopic, thermal infrared, the invisible heat we can feel, the size of a pin head.
So back to: “where do you draw the boundary between electromagnetic radiation that can impart kinetic energy and that which cannot”
The link I gave from the wiki page on transparency and translucency has a section which gives the basic basics of the difference, where the line is drawn between thermal and non-thermal infrared is that near is not thermal, it is reflective, and from mid infrared on it is thermal, absorptive. Near classified with Light not Heat, think cameras. Just as an ordinary camera captures the light reflected from objects so does the near infrared, it is what is reflected by our bodies, not as thermal infrared cameras capture, the heat energy radiating out from our bodies. Near infrared can penetrate deeper than visible before being reflected out, visible can penetrate deeper than uv, uv doesn’t make it past the first of the three layers of the epidermis. Highly energetic = powerful? Put a shirt on to block it..
From the wiki page:
That’s the cut off point, the boundary.
Now, read on from there, UV-Vis: Electronic transitions, bearing in mind that this will also apply to near infrared which is not hot, which is reflective, think cameras, as are visible and uv (there are plants deeper in the ocean than visible red can reach which use near infrared light for photosynthesis, they’re a sort of violet colour, the colour they don’t absorb at those deeper levels reflected out as the green reflected out generally by plants).
There are four bullet points listed: When photons (individual packets of light energy) come in contact with the valence electrons of atom, one of several things can and will occur:
NB, can and will occur.
The second is how the difference between reflective and absorptive comes about in the general difference between Light and Heat, light being reflective, heat being absorptive. Here light is absorptive in a light context, not as a comparison in heat and light differences which is reflective visible, absorptive on atomic/molecular level thermal.
Remember, visible light is even smaller than near infrared, light works on an electronic transition level, and in reflection/scattering it hits the electron and its energy briefly absorbed and is bounced back out as bullet 2 explains. This is what is happening in the atmosphere, the blue sky we have is because blue light is even more energetic than red and so is more easily bounced around by the electrons of the molecules of oxygen and nitrogen, think pin ball machine.
The atmosphere, therefore, is wrongly claimed by AGWSF to be transparent to visible light, as in the ‘greenhouse’ descriptions, but it isn’t, because it is absorbed on an electron level. Bear this in mind, because the next point is a description of a real transparent medium to visible light.
The third bullet point describes this: An electron cannot absorb the energy of the photon and the photon continues on its path. This results in transmission
Water is a real transparent medium to visible light, it is not absorbed by the electrons but transmitted through.
AGWScience Fiction claims that visible light heats water. How? It takes moving the whole atom and molecule into vibration to heat matter which is not in the level of capability of visible light, which are in Electronic Transitions not Vibrational (kinetic) modes of operation.
So be aware, AGWSF misuses the word ‘absorption’ as it does the word ‘heat’; that oceans are described as ‘absorbing different colours to different depths’ does not mean visible light is heating the water, it can’t, it can’t even get to the electrons of water as it does in the atmosphere because it is transmitted through transparent mediums. Again, don’t get yourself distracted into tangents that try to obscure this basic difference in well known tried and tested real world physics fact..
If you can get these simple basics clear, then you’ll be able to spot more easily where AGWSF takes liberties with real physics elsewhere; by taking laws out of context, by giving the property of one thing to another, as here giving the properties of thermal infrared to visible light, by stipping all properties to hide the differences and so the actual effects, as in ‘carbon dioxide well mixed in the atmosphere and able to accumulate’ because ‘the atmosphere is empty space with all the molecules zipping around at tremendous speeds’ and not the real world fluid gaseous heavy volume pressing down on us a ton/sq ft, where we have sound.., and so on.
I am convinced the confusing manipulations of terms is deliberate, because the more I explored this the more I saw it takes someone with a real grounding of physics to be able to create the confusion by these fiction sound bites, and produce the so called ‘proofs’ which on examination turn out to be more sleight of hand, as in the ‘heating experiments’.
Remember the real boundary in these arguments:
As dscott put it so well: “What we have here is the classic false proof, where the assumption at the outset of logical thought process is incorrect. In typical fashion you scientific types run with the flawed assumptions, apply flawless logic and then end up with a false conclusion.”
p.s. sorry, for clarity:
“The link I gave from the wiki page on transparency and translucency has a section which gives the basic basics of the difference, where the line is drawn”
From “where the line is drawn” is separate point to the info on the link, best put in brackets.
Jose_X @ur momisugly December 3, at 8:28 am
Well for a start the maths would be a bit tricky. How do you subtract a downward facing hemisphere from an upward facing hemisphere? OK, Trenberth does it as 396 – 333 = 63. Is this net heat transfer of 63 a hemisphere, full sphere, a weird shape, or vertical? Oh, and the horizontal stuff does not have a relative sign BTW.
Another bit of fun for you is that surface radiation can only be hemispherical, whereas the 333 comes from the atmosphere which gives spherical radiation. Thus there is another 333 “upwards”. So, that means that there is a total “upwards” of 396 + 333…….
Bob Fernley-Jones >> you want to study and learn
Yes, but I have not the time nor desire to follow and absorb every link someone posts. I have to be judicious in how I spend my time. As is, I have a lot more things I would like to be doing unrelated directly to posting comments related to climate science.
You provided a link loaded with links. I read over that page to get an idea, but I am not going to follow all the links of every link someone gives. I am not going to read hundreds of comments on an article. If you have done the reading yourself, you should be able to point out the more important points and lay out your argument in a bit more detail. [Obviously, if I were really into this or saw things from your perspective more, I would not need much prodding at all.]
>> although I find it distressing that you cannot afford the valuable journalistic research of the IPCC corruptness in the downloadable Laframboise digital book at $4.99
I can’t control your level of distress, duh. I was honest about some of my views towards paying for literature because why kid you? There are many great pieces of fiction and non-fiction, and, in another lifetime, I might be able to read them. I don’t make a lot of money and have IMO much more important things — free reading — I still have not found time to read. I haven’t even read the free IPCC WG 1 report (only bits and pieces) or anything but a super-duper, teeny-weeny, tiny fraction of formal papers on this subject [and don’t intend to read except as necessary). I am certainly not caught up on the physics (which I do find much more interesting than office politics or other people’s dirty laundry). I am not passionate about bringing down the IPCC.
To repeat, I am no one’s keeper. These are not my heroes. If any specific individual is proven a fraud, that will lead to a bit of re-evaluation, but it should not be that much. Even if they were honest, the science should speak for itself (though obviously there is much you or I can’t read, understand, or confirm). I don’t work in this industry and am not affected very much at all no matter which way the coin falls. I am affected as any other human might be if the news is bad. I certainly don’t think humans are a negligible force on this planet. I do care about the integrity of science generally. This is one reason why I both want to help improve it if I can and avoid having it be tarnished unnecessarily. Science works by bringing in something that works better.
OK, I will post this comment and then go and read the “Caspar and the Jesus paper”.
[FWIW, I catch some language mistakes after I post. Yes, I review what I write but maybe not as much as I should. And my understanding of (English) grammar only goes so far. The biggest reason my skills fall far short of those of Shakespeare has little to do with English not being my native language. You become skillful in areas where you spend time. I put in enough time so that I can be understood generally and at times even entertain a bit, but I don’t go too far beyond that. This is not a literature forum. A great poem or story is not as important here IMO as a fair dissection of some science related point. Although it might be true that my expectations are misplaced.]
Bob Fernley-Jones, don’t interpret what I just wrote to mean that I think I am always understood, but in the end what someone might understand someone else won’t. There is no science. I think I have shown I am eager to understand and be understood, even if it takes numerous comment exchanges.
Bob Fernley-Jones, ..and I do care about abuse and exploitation, but an unjustified attack on the science is what would really be an abuse and exploitation. I want to make sure we get the science right.
Jose,
Don’t get me wrong, I wish my Italian and nearly forgotten Spanish was half as good as your English.
BTW, congratulations for posting comment #600
Bob Fernley-Jones, that was a neat article. I did not read the whole thing. What I got out of it during this run was that some scientists very possibly overplayed the value of their paper and they probably had some help from people high up in facilitating a path for that paper to make cut offs that should then not have been met. Both of these things (to the extent true) obviously have some repercussions, but in the big picture it “probably” isn’t much of a blow at all to the science (although if it is, then so be it) nor to most people. We mostly all gain from more accurate science.
I liked the statistics related coverage because I want to learn more about some of those basics covered in the article (so I will go back to read with more care and do some cross reading).
I like the fact that a paper that perhaps likely should be put in its place will be so put in time if not immediately and that predictions will probably be adjusted to reflect what we currently know and based on our statistics/probabilities. But in the end little may change if the statistics of that one experiment were a little short of meeting some cut-off. [Maybe my statistics ignorance is showing here.] It appears this is a case where the potential costs to individual careers and/or pride is much higher than to the science. I am not a fan of punishing individuals (you can invoke here the “cast the first stone” and “take the plank out of your eye first” mentality), but I do care about restricting the damage individuals can cause.
Sorry, if there were more serious issues I missed. I did read fast in order to get a taste of what was there. Have thoughts you want to share on this preliminary “summary” I just wrote?
Bob Fernley-Jones, I have not given much of an opinion on the 333; however, a simple diagram can convey useful information while still having many limitations. There is no need to think that a given diagram must present material in a way where certain complex or precise calculations must be achievable.
And it can even have “insider” meaning to the extent it might specify the value of mathematical variables of some model. The values would be useful to someone who knows the details of such a model. [I am not a climatologist and don’t “get” this inside angle that may exist.]
I do like the theme of this last reply of yours. I may think a bit about the math of hemi-planes and may also think more about that 333. Actually, I am still trying to understand atmosphere physics, so the 333 will have to wait. The 396 doesn’t seem so problematic to me at this point in my understanding.
>> Don’t get me wrong, I wish my Italian and nearly forgotten Spanish was half as good as your English.
Don’t be too impressed. I essentially grew up in the US… um, please don’t take that as an attack on the US educational system. 😀
>> BTW, congratulations for posting comment #600
It’s hard to miss key numbers like that when I post 80 comments in a row!
OK, not quite 80.
Speaking of cherry-picking, did I post #500 and #400?? How about #444 or #555????
Bob Fernley-Jones, reading over http://wattsupwiththat.com/2011/10/26/does-the-trenberth-et-al-%e2%80%9cearth%e2%80%99s-energy-budget-diagram%e2%80%9d-contain-a-paradox/#comment-818852 , I could find problems with how I conveyed my thoughts.
For one, the tone might be taken to be aggressive even though I was trying to be more “factual” than complaining in tone (maybe I was a little defensive but mostly just trying to clarify) and was not angry at all.
>> and have IMO much more important things — free reading — I still have not found time to read.
More accurately:
and have IMO much more important things I still have not found time to read (or do), including a great lot of reading which happens to be free.
Finding and fixing the other problems with that comment (if any) will be left as an exercise for the reader.
[ .. have I hit #666 yet?]
Tim Folkerts @ur momisugly December 2, at 6:14 am
Tim, sorry for the delay.
One of the fundamental oversights in your various hypotheses’ which I’m not going to grind through, is that if we accept the numbers of the great prophet of cartoons, in fig 1, then there are four basic heat transfer happenings from the surface. They are, in his terminology and W/m^2;
Thermals = 17; Evapotranspiration = 80; Surface radiation absorbed by atmosphere = 23; & surface radiation direct to space = 40. (Total = 160)
Thus the portion of thermal activity that you were discussing was only ~14 % of the total. However, you cannot sensibly ignore the other processes which happen interactively.
>> and was not angry at all
Well, a little bit of frustration that goes along with writing almost any “defensive” comment was surely there and an inkling of anger might necessarily follow frustration.
But I can’t remember.