Guest post by Bob Fernley-Jones by Bob Fernley-Jones AKA Bob_FJ
CAUTION: This is written in Anglo-Oz English.
Here is the diagram as extracted from their 2009 paper, it being an update of that in the IPCC report of 2007 (& also 2001):
The unusual aspect of this diagram is that instead of directly showing radiative Heat Transfer from the surface, it gives their depiction of the greenhouse effect in terms of radiation flux or Electro-Magnetic Radiation, (AKA; EMR and a number of other descriptions of conflict between applied scientists and physicists). EMR is a form of energy that is sometimes confused with HEAT. It will be explained later, that the 396 W/m^2 surface radiation depicted above has very different behaviour to HEAT. Furthermore, temperature change in matter can only take place when there is a HEAT transfer, regardless of how much EMR is whizzing around in the atmosphere.
A more popular schematic from various divisions around NASA and Wikipedia etc, is next, and it avoids the issue above:
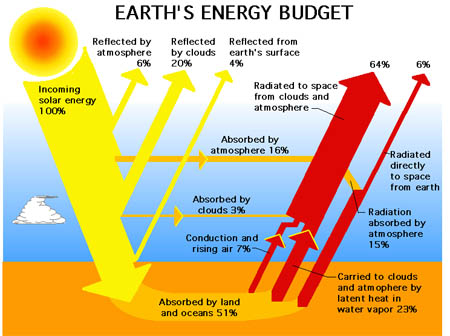
- Figure 2 NASA
Returning to the Trenberth et al paper, (link is in line 1 above), they give that the 396 W/m2 of EMR emitted from the surface in Fig.1 is calculated primarily by using the Stefan–Boltzmann law, and global year average conditions. Putting aside a few lesser but rather significant issues therein, it is useful to know that:
1) The Stefan-Boltzmann law (S-B) describes the total emission from a flat surface that is equally radiated in all directions, (is isotropic/hemispherical). Stefan found this via experimental measurement, and later his student Boltzmann derived it mathematically.
2) The validity of equally distributed hemispherical EMR is demonstrated quite well by observing the Sun. (with eye protection). It appears to be a flat disc of uniform brightness, but of course it is a sphere, and at its outer edge, the radiation towards Earth is tangential from its apparent surface, not vertical. It is not a perfect demonstration because of a phenomenon called limb darkening, due to the Sun not having a definable surface, but actually plasma with opacity effects. However, it is generally not apparent to the eye and the normally observed (shielded) eyeball observation is arguably adequate for purpose here.
3) Whilst reportedly the original Stefan lab test was for a small flat body radiating into a hemisphere, its conclusions can be extended to larger areas by simple addition of many small flat bodies of collectively flat configuration, because of the ability of EMR waves to pass through each other. This can be demonstrated by car driving at night, when approaching headlights do not change in brightness as a consequence of your own headlights opposing them. (not to be confused with any dazzling effects and fringe illumination)
4) My sketch below demonstrates how radiation is at its greatest concentration in the lateral directions. It applies to both the initial S-B hemispherical surface radiation and to subsequent spherical radiation from the atmosphere itself.
5) Expanding on the text in Figure 3: Air temperature decreases with altitude, (with lapse rate), but if we take any thin layer of air over a small region, and time interval, and with little turbulence, the temperature in the layer can be treated as constant. Yet, the most concentrated radiation within the layer is horizontal in all directions, but with a net heat transfer of zero. Where the radiation is not perfectly horizontal, adjacent layers will provide interception of it.
A more concise way of looking at it is with vectors, which put simply is a mathematical method for analysing parameters that  possess directional information. Figure 4, takes a random ray of EMR (C) at a modestly shallow angle, and analyses its vertical and horizontal vector components. The length of each vector is proportional to the power of the ray, in that direction, such that A + B = C. Of course this figure is only in 2D, and there are countless multi-directional rays in 3D, with the majority approaching the horizontal, through 360 planar degrees, where the vertical components also approach zero.
possess directional information. Figure 4, takes a random ray of EMR (C) at a modestly shallow angle, and analyses its vertical and horizontal vector components. The length of each vector is proportional to the power of the ray, in that direction, such that A + B = C. Of course this figure is only in 2D, and there are countless multi-directional rays in 3D, with the majority approaching the horizontal, through 360 planar degrees, where the vertical components also approach zero.
6) Trenberth’s figure 1 gives that 65% of the HEAT loss from the surface is via thermals and evapo-transpiration. What is not elaborated is that as a consequence of this upward HEAT transfer, additional infrared radiation takes place in the air column by virtue of it being warmed. This initially starts as spherical emission and absorption, but as the air progressively thins upwards, absorption slows, and that radiation ultimately escapes directly to space. Thus, the infrared radiation observable from space has complex sources from various altitudes, but has no labels to say where it came from, making some of the attributions “difficult”.
DISCUSSION; So what to make of this?
The initial isotropic S-B surface emission, (Trenberth’s global 396 W/m2), would largely be absorbed by the greenhouse gases instantaneously near the surface. (ignoring some escaping directly to space through the so-called “atmospheric window”). However, a large proportion of the initial S-B 396 surface emission would be continuously lateral, at the Trenberth imposed constant conditions, without any heat transfer, and its horizontal vectors CANNOT be part of the alleged 396 vertical flux, because they are outside of the vertical field of view.
After the initial atmospheric absorptions, the S-B law, which applied initially to the surface, no longer applies to the air above. (although some clouds are sometimes considered to be not far-off from a black body). Most of the air’s initial absorption/emission is close to the surface, but the vertical distribution range is large, because of considerable variation in the photon free path lengths. These vary with many factors, a big one being the regional and more powerful GHG water vapour level range which varies globally between around ~0 to ~4%. (compared with CO2 at a somewhat constant ~0.04%). The total complexities in attempting to model/calculate what may be happening are huge and beyond the scope of this here, but the point is that every layer of air at ascending altitudes continuously possesses a great deal of lateral radiation that is partly driven by the S-B hemispherical 396, but cannot therefore be part of the vertical 396 claimed in Figure 1.
CONCLUSIONS:
The vertical radiative flux portrayed by Trenberth et al of 396 W/m^2 ascending from the surface to a high cloud level is not supported by first principle considerations. The S-B 396 W/m^2 is by definition isotropic as also is its ascending progeny, with always prevailing horizontal vector components that are not in the field of view of the vertical. The remaining vertical components of EMR from that source are thus less than 396 W/m^2.
It is apparent that HEAT loss from the surface via convective/evaporative processes must add to the real vertical EMR loss from the surface, and as observed from space. It may be that there is a resultant of similar order to 396 W/m^2, but that is NOT the S-B radiative process described by Trenberth.
~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~
ADDENDUM FOR AFICIONADOS
I Seek your advice
In figure 5 below, note that the NIMBUS 4 satellite data on the left must be for ALL sources of radiation as seen from space, in this case, at some point over the tropical Pacific. The total emissions, amount to the integrated area under the curve, which unfortunately is not given. However, for comparison purposes, a MODTRAN calculator, looking down from 100 Km gives some interesting information for the figure, which is further elaborated in the tables below. Unfortunately the calculator does not give global data or average cloud/sky conditions, so we have apples and pears to compare, not only with Nimbus, but also with Trenberth. However, they all seem to be of somewhat similar order, and see the additional tabulations.
| Compare MODTRAN & “Trenberth”, looking down from 2 altitudes, plus Surface Temperature | ||||
| Location | Kelvin | 10 metres | 100 Km. | (Centigrade) |
| Tropical Atmosphere | 300K | 419 W/m^2 | 288 W/m^2 | (27C) |
| Mid-latitude Summer | 294K | 391 W/m^2 | 280 W/m^2 | (21C) |
| Mid-latitude Winter | 272K | 291 W/m^2 | 228 W/m^2 | (-1C) |
| Sub-Arctic Winter | 257K | 235 W/m^2 | 196 W/m^2 | (-16C) |
| Trenberth Global | 288K ? | 396 W/m^2 | 239 W/m^2 | (15C ?) |
| Compare MODTRAN & “Trenberth”, looking UP from 4 altitudes: W/m^2 | ||||
| Location | From 10 m | From 2 Km | From 4Km | From 6Km |
| Tropical Atmosphere | 348 | 252 | 181 | 125 |
| Mid-latitude Summer | 310 | 232 | 168 | 118 |
| Mid-latitude Winter | 206 | 161 | 115 | 75 |
| Sub-Arctic Winter | 162 | 132 | 94 | 58 |
| Trenberth Global | 333 Shown as coming from high cloud area (= BS according to MODTRAN) | |||



Tim Folkerts,
I think you made some assumptions about the Youtube experiment you cited.
I’ve just watched that video again, and ’tis strange but the infrared camera monitors the visible light from the candle fine, until the CO2 is pumped in!
Bob Fernley-Jones says:
‘Coming back to that Youtube experiment, it was only 1 minute long so I watched it. One thing that puzzled me was that the candle, with a colour temperature of over 1,000 C, emits a lot of visible light, yet the CO2 was demonstrated to extinguish that light. Doesn’t the greenhouse theory depend on CO2 not absorbing visible light?’
Bob, you are quite correct a greenhouse lets in most of the sun’s spectrum but blocks all of the LWIR. The atmosphere of course doesn’t do that, it blocks only in certain wavelengths, CO2 for instance doesn’t absorb in the 5 to 12 micron band, and it certainly doesn’t absorb in the visible range 0.38 to 0.76 microns. In fact standing outside of a greenhouse you would still see the candle flame but not get any heat from it. Well spotted its more of a charlatan trick than I thought!
People, maybe I am misunderstanding the last few comments above, but did you read the link I provided to the page that explains details about that experiment? The only thing happening is that a camera initially shows the image it picks up in the 4 micro-m range. When you add the CO2, most of that reception disappears. There is no visible light issue anywhere. The experiment demonstrates the effect of CO2 absorbing in that 4 micro range.
The problem with the video (as clarified below) is arguably suggests that CO2 traps a very wide range of radiation (when it does not). This flaw is made explicit when the host broadly states at the end that the candle “warmth” was trapped rather than state that the experiment specifically only covered a very narrow slice of the full spectrum and that the atmosphere covers much more but still much less than the full spectrum of the candle or of any body.
This is the rough transcript:
“I can show you how carbon dioxide affects us(?) climate using this heat sensitive of(?) infrared camera which is pouring away here, a candle, this glass tube which is hooked up to this (otherlouse?) canister of carbon dioxide gas. Now, if I light the candle you’ll see that on the monitor the camera picks up the flame perfectly. Look at that — the hot spots of glowing white. Now watch what happens when I turn on the carbon dioxide. Just keep your eye on the flame. The gas is invisible so you don’t see it filling the tube, but, as it comes in, you should see the candle itself disappear. There it goes. Look at that! What’s happening is that the carbon dioxide in the tube is effectively trapping the heat. The candle’s warmth no longer reaches the camera. ..instead it is absorbed by the carbon dioxide inside the tube.”
The first sentence says the experiment shows us how carbon dioxide affects the climate. This is not true 100%. That is, the experiment does not replicate the climate in every way and to proper degrees; however, it does show that CO2 can absorb heat, and current accepted science (by almost all experts in the field) states that this absorption has an effect in warming the climate.
That sentence also specifically states we are talking about an infrared camera.
The next sentence is basically correct although we can argue that no camera “picks up” anything “perfectly”.
“The hot spots of glowing white” is a descriptive phrase perhaps accurately describing the white on the monitor which refers to hotter parts of the flame.
“The gas is invisible” is essentially correct (to the naked human eye).
“You should see the candle itself disappear” likely refers to the image of the candle being shown to the audience, and it does largely disappear presumably as the CO2 fills the tube.
“What’s happening is that the carbon dioxide in the tube is effectively trapping the heat” and “instead it is absorbed by the carbon dioxide inside the tube”: “Effectively trapping the heat” sounds reasonably accurate to me, as the amount of radiation being picked up by the camera has apparently gone way down.
“The candle’s warmth no longer reaches the camera.” This is only true when referring to the 4 micro-m section. That statement and the introduction appear to refer to a much broader range of infrared being in play. That part is clearly misleading.
Yes, as you guys have said, there is the additional point that the particular band chosen to show off the effect is not the relevant one for the greenhouse effect.
However, the effect is already hacked to a narrow band, and picking one slice over another is not a major issue (at this point). It may call into question if in fact absorption works the same when you change bands, but science agrees, I think, that the essential physics doesn’t change when you change frequencies. I don’t know why the right peek wasn’t chosen. I assume the equipment was chosen to keep costs down since I think this is expected to be a demo for teachers to use in their science classes.
Bob & Robert,
You both seem to be mistaken about the camera being used. What difference does it make that the candle emits visible light in addition to IR??? The hot flame will emit both visible light and IR from roughly the same hot areas, so it would be about the same size & shape whether viewed with IR or visible light.
The camera is clearly an IR camera, and in fact the link Jose gave earlier discussed that the camera was indeed tuned to ~ 4 um IR so that the CO2 effect would be strongly noticeable. The fact that the candle also creates light of 0.5 um or 10 um photons is immaterial to this experiment. I consider constructing an experiment to show a specific feature of nature while eliminating extraneous details to be a standard, time-honored approach in science. Robert apparently thinks only charlatans would do such a thing. My only objection to the video is that, to be a little more accurate, a few times they should have said “some”. (eg “some of the IR is absorbed by the CO2”) to be a little more accurate. OTOH, I don’t expect a 2 minute TV demo to provide all the details.
If Robert wants to be that picky, then I could get picky about what he said.
>A greenhouse blocks all of the IR
No. Only some. Both CO2 and glass block SOME IR. Both Robert & the video gloss over this; Robert is just more emphatic about being inaccurate.
>a greenhouse lets in most of the sun’s spectrum but blocks all of the LWIR
Much better. NIR & SWIR get thru very well. MWIR gets thru somewhat. LWIR (8-15 um according to standard definitions) is almost entirely blocked by glass as claimed.
>you would still see the candle flame but not get any heat from it
Myrrh’s objections not withstanding, any wavelength of light transfers energy and hence “heat”. So the visible light would give you “heat”. The NIR & SWIR & MWIR would also certainly give you “heat” through the glass and could be picked up by cameras sensitive to these wavelengths.
~~~~~~~~~~~~~~~
These details can be important if you are trying to do calculations, but for a general discussion it is good enough to know that both glass and CO2 block some IR while letting thru essentially all visible light, and that this fact helps explain the observed warming. (PS Of course, in a real greenhouse, the blocking of air flow is typically more important than the blocking of IR)
Well it looked a bit fishy to me. My wife looked it and said obviously the CO2 had extinguished the flame.
correction…… looked at it…..
Re: Candle flame.
I agree with Robert; Well it looked a bit fishy to me.
The image of the flame monitored by the infrared camera looked rather similar to what I’ve just checked indoors in both bright and dim daylight with my own candle and a long matchstick. I always thought that infrared photography gave different colours and contrasts to what can be seen in natural light. (As is employed in many galleries of such art photography).
Bob says: “I always thought that infrared photography gave different colours and contrasts to what can be seen in natural light. (As is employed in many galleries of such art photography).”
There are two very different sorts of IR imaging. The “traditional” version used “near IR” just beyond red (~ 0.7 – 1 um). This sort of IR is typically reflected IR from the sun that passes thru most glass easily (and is sometimes referred to as “reflected IR”). So a standard camera with film sensitive to near IR can pick it up, creating an image that looks sort of like black and white, but with odd contrasts. Furthermore, many digital cameras can pick this up (the CCD is sensitive to wavelengths beyond 0.7 um) so they can “see” remote controls that operate in this range.
The the other sort of imaging uses longer wavelengths of “thermal IR” that are produced by the heat of objects. This requires a more specialized camera (like the one in the video) that does not respond to visible light.
Google images of “infrared images” and you will get both types of images and you can generally tell the two apart quite easily.
Jose,
OK, this has apparently become an open thread after over 600 comments. (phew!)
You seem to have declared that exaggeration in AGW stuff is OK, providing that it is taken in context for appropriate observers, whoever they may be. However, I feel that when such crap is aimed at gullible people, especially including the young, that it is inexcusable.
AGW exaggeration often involves the deliberate deletion or omission of “inconvenient data”, such as in “hide the dendro-decline”, (= the divergence problem), but let me give another example:
The young glaciologist; Jason Box, was a co-author for IPCC AR4 WG1 Chapter 4.6.2.2.1 concerning the popular alleged disastrous melting of the Greenland icesheet:
http://www.ipcc.ch/publications_and_data/ar4/wg1/en/ch4s4-6-2-2.html
Yet, his earlier joint authorship of a paper suggested that Greenland air temperatures around 1930 – 1940, were on average higher, especially in summer, versus at the time of AR4. (IPCC 2007 report). I think he attributed the ~60-year cycle of the PDO as a significant correlation. However such inconvenient data had NO MENTION in that chapter.
OK, maybe rather than following many other IPCC contributing authors, perhaps he did not want to spruke his own paper. However, there were some later papers that were more definitive and confirmative, that could well have been employed into the report had the IPCC wanted them. (That is going by their acceptance of other post-cut-off, post-final-draft inclusions, when it suited the IPCC ideology and their UN charter to confirm CAGW)
Plugging 1000 C into my Planck/Wien’s Law spreadsheet I do indeed observe that the bulk of the emitted IR is in the 2.3 to 3.0 micron and 4.0 to 4.8 micron ranges with wery little in 15 micron range as Jose pointed out vis a vis the U tube experiment. The graphical printout confirms this and gives the peak intensity at 2.3 microns.
So tuning your IR camera to these wavelegths would give you the disappearing conjuring trick candle flame. However integrating the areas under Planck’s curve gives a me a relatively small percentage of the total emissive power that is absorbed by CO2.
I would re-do the experiment to reflect this fact. Clearly this would make people more sceptical
concerning the catastrophic etc as I have found when I explain this simple fact in discussions.
Jose_X says:
December 4, 2011 at 9:04 pm
What evidence can you offer that only radiation of a certain wavelength can warm things, in general?
Jose, I’m sorry, I don’t have the time to got through this further with you as you’re still getting distracted from the basics – I’ve given you the mechanism to your question above, the difference between electronic transitions and atomic/molecular vibrational:
I can see that you have a great facility for thinking about all this, from the range and volume of your questions, but until you have a thorough grasp of this basic difference you will continue to fall into the traps of terms distorting meaning, for example, “absorbed” means different things according to context. Visible light’s energy is not absorbed by the molecules of water, water is a tranparent medium for visible light, bullet point 3 on the list on that page, so that “absorbed” in ‘visible light is absorbed by the ocean because it disappears at different levels’ can’t be referring to the same meaning of “absorbed” in the difference between absorption and transmission of visible light in the fluid gaseous atmosphere above us, where it is briefly absorbed by the electrons of the molecules of oxygen and nitrogen before being reflected/scattered out, bullet point 2, and not being absorbed on electronic transition level by the water molecules, because it is transmitted through without being absorbed, bullet point 3. Neither of these are using the energy to heat up the molecules, which takes greater energy than they possess, to move whole atoms and molecules into vibration, which is heat, thermal energy.
Now, which bullet point on that list describes what happens to visible light which is neither reflected nor transmitted on encountering matter but its energy absorbed as chemical energy in photosynthesis? This too does not create heat, it creates sugars.
“Absorbed” does not automatically mean that energy creates heat.
That however is the claim of the AGWSF energy budget, KT97 and ilk, which is how they say visible light, the shortwaves, directly convert land and ocean to heat, directly heat up land and ocean, and, they say that the Sun’s actual thermal energy doesn’t play any part in this, they say it doesn’t even reach the surface. But we know in traditional physics that the heat we feel from the Sun, its thermal energy on the move to us via radiation (contrasting with heat transfer by conduction and convection) is the invisible thermal infrared.
So, we know, in traditional physics and not the science fiction physics of AGW promotion, that visible light’s energy works on electronic transition level, this energy utilised differently depending on the physical circumstances, as plants utilising it for creation of sugars or electrons of molecules scattering and reflecting it as in the atmosphere, and we know that it takes a bigger more powerful energy to move the whole molecule into vibration which is what heat, the invisible thermal infrared wavelength which is thermal energy, does. That is how water molecules are heated, by heat, when the whole molecule absorbs the invisible thermal infrared heat energy from the Sun the water in our ocean is heated. Visible light on electronic transition level doesn’t have the power to do this, its power is so miniscule that it doesn’t even get to be absorbed by the electrons of water molecules, but is transmitted through, transmitted being the correct technical term for this process.
Now, if you’d just concentrate on this, the very basic real physical differences between visible wavelengths, and the short of the AGWSF energy budget, and thermal infrared, you’d see why I keep asking for proof that “visible and shortwave solar directly heat land and oceans” – as in the claim in the cartoon above.
Until you deal with my question, the question I’m actually asking here, you will continue to get distracted by questions that you will be able to answer yourself when you fully grasp what this difference is.
Have a go. Prove the claim in the cartoon, prove that visible light heats land and oceans.
Unless you can do so, and you are actively defending that claim so it is up to you to prove it when I’ve asked, I see your agility of mind as merely avoiding the challenge.
I have shown you from traditional physics why it can’t.
Tom Folkerts says:
I am simply saying that “evaporative cooling” is synonymous with the 80 W/m^2 of energy leaving the surface. The oceans are the ‘wet t-shirt around the earth’.
The oceans also absorb/dissolve an awful lot of CO2 far more than one would expect from Henry’s Law constant.
CO2- Water Solubility
Henry’s-law constant for CO2-water solutions is 1.42×10^3 at 20 C . At equilibrium the oceans therefore should hold only 30% of the 2,900 giga tonnes of the atmospheric CO2 if simple Henry’s-law solubilities were applied not 50 times which is the actual figure.
Myrrh, you have not shown me “from traditional physics why it can’t”. Traditional physics contradicts your viewpoint.
For starters, you keep ignoring what nasa and other respectable websites have said directly, that sunlight can be turned into heat naturally. You have not explained the “blackbody” effect as observed non-ideally in all sorts of objects and as taught by virtually any university. Here, I’ll add another quote from a harvard webpage, https://www.cfa.harvard.edu/~jbattat/a35/blackbody_color.html , “[i]f we had infrared goggles, we would be able to see objects at room temperature by their emitted light.”
You tried to use a thermo website, but it never said anything about visible light not being turned into IR or “heat”. In fact, thermodynamics supports the view that many and/or most natural processes generate heat.
From http://en.wikipedia.org/wiki/Irreversible_process
> All complex natural processes are irreversible… A certain amount of “transformation energy” will be used as the molecules of the “working body” do work on each other when they change from one state to another. ..During this transformation, there will be a certain amount of heat energy loss or dissipation due to intermolecular friction and collisions; energy that will not be recoverable if the process is reversed.
The quotes you took from various wikipedia pages also fail to claim or iirc even suggest that visible light cannot be turned into IR.
Q1 — Let me ask a question, what do you theorize happens to the vast quantity of the sunlight absorbed by objects on earth?
[This is an extension of the sunglasses example you haven’t addressed yet.]
According to the very Trasparency and Translucency wikipedia page you used for quotes in your recent comment most sunlight appears to be absorbed:
> When light strikes an object, it usually has not just a single frequency (or wavelength) but many. Objects have a tendency to selectively absorb, reflect or transmit light of certain frequencies. That is, one object might reflect green light while absorbing all other frequencies of visible light. Another object might selectively transmit blue light while absorbing all other frequencies of visible light.
Q2 — I have another minor question, were you suggesting that plants can absorb sunlight and store that energy without generating “heat”? Wouldn’t that violate the second law of thermodynamics?
From the same http://en.wikipedia.org/wiki/Irreversible_process
> Many biological processes that were once thought to be reversible have been found to actually be a pairing of two irreversible processes. Whereas a single enzyme was once believed to catalyze both the forward and reverse chemical changes, research has found that two separate enzymes of similar structure are typically needed to perform what results in a pair of thermodynamically irreversible processes.
Q3 — ..and I’ll repeat, why do you keep ignoring what most universities are teaching (eg, blackbody)?
These are all well established multi-disciplinary physics theories, not “climate science” theories.
Bob, didn’t Tim just show that you are not infinitely precise either? Has it ever been in doubt that I (as likely most people) believe that being imprecise and not knowing everything is a fact of life? I said that I think the demo presentation should be adjusted and that the disclosure page should be clearly marked (a part of doing experiments/demos in connection with science is that you reveal a fair amount of your “secrets”).
Also, it is you that is going against established science in denying man has a non-negligible effect on the environment a la “global warming”. Pointing to a few potential mistakes here or there in a ton of information doesn’t prove you right or me negligent for accepting the science. I clearly don’t accept unconditionally, but the volume of the evidence is not on your side. To take a point I conditionally accepted, that one of Mann’s paper should not have received the value that it got, that doesn’t prove anything against climate science except that a few data points and one study are under question.
It seems you are criticizing aspects of climate science. That is good and healthy.
Bob>> Chapter 4.6.2.2.1 concerning the popular alleged disastrous melting of the Greenland icesheet
The conclusion from that section on Greenland:
> Assessment of the data and techniques suggests a mass balance for the Greenland Ice Sheet ranging between growth of 25 Gt yr–1 and shrinkage of 60 Gt yr–1 for 1961 to 2003, shrinkage of 50 to 100 Gt yr–1 for 1993 to 2003 and shrinkage at even higher rates between 2003 and 2005. Lack of agreement between techniques and the small number of estimates preclude assignment of statistically rigorous error bounds. Interannual variability is very large, driven mainly by variability in summer melting, but also by sudden glacier accelerations (Rignot and Kanagaratnam, 2006). Consequently, the short time interval covered by instrumental data is of concern in separating fluctuations from trends.
Basically, it says the jury is still out but enough evidence apparently supports that the rate of ice melting in certain parts (Greenland Ice Sheet) has been growing.
>> However, there were some later papers that were more definitive and confirmative, that could well have been employed into the report had the IPCC wanted them. (That is going by their acceptance of other post-cut-off, post-final-draft inclusions, when it suited the IPCC ideology and their UN charter to confirm CAGW)
You might be right that the Greenland section may have been a section where the draw of the papers submitted and accepted on time could have affected the conclusions. [I would entertain links if you have them.] I do note that a number of quoted studies were rather recent (post 2005 range).
As concerns global warming, the main claims are about global averages. Locally, any number of atmosphere/land/water variations (plus overall global cycling and plain old statistical anomalies) might lead to some parts cooling over that time. It’s easier to get the little details wrong over 100 year “forecasts” than the main trends.
>> Greenland air temperatures around 1930 – 1940, were on average higher.. I think he attributed the ~60-year cycle of the PDO as a significant correlation. However such inconvenient data had NO MENTION in that chapter.
What do you mean? If there is correlation with a cycle, that would support the view of not over-emphasizing the air temps then. The ice melts over a prolonged period is much more significant especially given how water absorbs much more heat than air.
Of course, did you realize the title of that section?
“4.6.2.2 Measured Balance of the Ice Sheets and Ice Shelves” is not about air temperatures. It is more than reasonable to neglect discussion of air temperatures.
Myrrh, sorry I have misspelt your name (it seems some of the time I spell it myrhh).
Q: How do you justify how we use electrical energy to heat up an oven very hot? The heat comes from a glorified electrical battery.
Transducers are all around us. Energy is always changing forms. [We eat apple created via sunlight and use that converted energy to chop down the tree for extra warmth.]
If energy can so readily change forms, why is it so hard to accept that excited electron states in molecules (because of visible light and other non-IR radiation) can be turned into IR radiation?
Maybe I’ll research the quantum mechanical description of just this process — widely used physics, not merely “climate science”.
Jose_X,
Without commenting on the accuracy of Myrrh’s statements, let me just say that a few others have already tried reasoning with Myrrh in other threads, presenting many of the same sort of arguments you have given. So far, no one has been able to convince him that he has made any significant errors in logic or science.
Jose_X @ur momisugly December 8, at 9:24 am
Sorry Jose, I don’t have the links, but if you look around for Box, Chylek, Polyakov, or Lesins, + Greenland, you should find something.
I take exception to this:
Here is one of Jason Box’s co-authored papers, where in the extract, they seem to think that it’s all about air temperatures being a consideration, and pointing to the big warming in 1920, which apparently Box and other IPCC authors somehow omitted:
Global Warming and the Greenland Ice Sheet by Chylek P.1; Box J.E.2; Lesins G.3 2004
http://www.ingentaconnect.com/content/klu/clim/2004/00000063/F0020001/05140445
Jose_X says:
December 8, 2011 at 9:09 am
Myrrh, you have not shown me “from traditional physics why it can’t”. Traditional physics contradicts your viewpoint.
No it doesn’t. I’ve given you traditional physics, the difference between Heat and Light.
For starters, you keep ignoring what nasa and other respectable websites have said directly, that sunlight can be turned into heat naturally.
And I have warned of the infiltration of this junk science into the education system. What NASA used to teach was that the heat we feel from the Sun was the invisible thermal infrared, now it teaches that thermal infrared doesn’t even reach the surface of the Earth.. Do you see the disjunct here? Which is why I keep trying to get you to concentrate on the very basics, the difference between Heat and Light.
You have not explained the “blackbody” effect as observed non-ideally in all sorts of objects and as taught by virtually any university. Here, I’ll add another quote from a harvard webpage, https://www.cfa.harvard.edu/~jbattat/a35/blackbody_color.html , “[i]f we had infrared goggles, we would be able to see objects at room temperature by their emitted light.”
If you keep being distracted before you have a firm grasp of the basics then you’ll continue to be confused.
You tried to use a thermo website, but it never said anything about visible light not being turned into IR or “heat”.
It didn’t have to… 🙂 I began, by giving you traditional science about Heat. Until and unless you understand that how will you understand the difference between Heat and Light?
In fact, thermodynamics supports the view that many and/or most natural processes generate heat.
From http://en.wikipedia.org/wiki/Irreversible_process
> All complex natural processes are irreversible… A certain amount of “transformation energy” will be used as the molecules of the “working body” do work on each other when they change from one state to another. ..During this transformation, there will be a certain amount of heat energy loss or dissipation due to intermolecular friction and collisions; energy that will not be recoverable if the process is reversed.
The quotes you took from various wikipedia pages also fail to claim or iirc even suggest that visible light cannot be turned into IR.
It didn’t have to say that, it was describing Light… 🙂 What I gave was the basic mechanisms of what happens to visible, shortwave, on meeting matter, electronic transitions. If you had bothered to understand what was being said about Heat and that it takes vibrational energy to move molecules to this, you would have a grasp of the difference by now between that and electronic transitions, get some scale perspective here. The difference in size between near infrared, not hot, and thermal infrared is between microscopic and pin head size, visible light is even smaller than near infrared. You’re still not concentrating.., you’re not bothering to give this any thought as in ‘getting a feel’ for it, but instead without understanding what I’ve said already you continue to confuse yourself by bringing in irrelevancies or questions that you could answer yourself if you’d bothered to spend time digesting what traditional physics says of the differences.
Q1 — Let me ask a question, what do you theorize happens to the vast quantity of the sunlight absorbed by objects on earth?
[This is an extension of the sunglasses example you haven’t addressed yet.]
According to the very Trasparency and Translucency wikipedia page you used for quotes in your recent comment most sunlight appears to be absorbed:
> When light strikes an object, it usually has not just a single frequency (or wavelength) but many. Objects have a tendency to selectively absorb, reflect or transmit light of certain frequencies. That is, one object might reflect green light while absorbing all other frequencies of visible light. Another object might selectively transmit blue light while absorbing all other frequencies of visible light.
I don’t have to theorize what happens to the vast quantity of the sunlight absorbed by objects on earth, we know what happens, light enables us to see the world via reflection, pigmentation, and is utilised for photosynthesis. Those four bullet points show what is actually happening in the processes of Light contacting matter, and note, not all absorbed as the third bullet points out, transmission means that light is not absorbed, as in light travelling through a transparent medium, water. Bullet four covers photosynthesis, some of the light is absorbed for chemical changes and some reflected back out, which takes back to bullet 2.
Q2 — I have another minor question, were you suggesting that plants can absorb sunlight and store that energy without generating “heat”? Wouldn’t that violate the second law of thermodynamics?
Plants are not storing that energy, it is being utilised as chemical energy to produce sugars, not to produce heat. The laws of thermodynamics have nothing to do with this, light isn’t hot…
Q3 — ..and I’ll repeat, why do you keep ignoring what most universities are teaching (eg, blackbody)?
These are all well established multi-disciplinary physics theories, not “climate science” theories.
Irrelevant. I’m talking about the difference between real Light and real Heat..
Bringing this into the ‘explanations’ for the AGWSF energy budget is a distraction from real physics in these arguments, by eliminating the real differences between the different wavelengths, claiming that ‘all energy is the same and all create heat’, etc. Get back to the basic differences between wavelengths, their different properties relative to each other such as size, character, etc., the different processes, such as the difference between electronic transitions on an electron scale and thermal vibration on an atomic/molecular. Then, for example, you could move on to looking at the differences between these in the ionising and non-ionising categories, such as UV has both. Sets are useful tools here, as you’ve given above, http://hyperphysics.phy-astr.gsu.edu/hbase/mod3.html, different wavelengths do different things because they are different.
The category I’m covering is the difference between Heat and Light. Because, the claim from the AGWSF junk energy budget says that visible light directly heats land and oceans, and this is physically impossible because of visible light’s properties and processes.
Now, unless you or anyone else can prove that visible light direct from the Sun can actually heat land and oceans, you shouldn’t be using that cartoon as the basis for this real world’s energy budget.
Tim Folkerts says:
December 8, 2011 at 12:48 pm
Jose_X,
Without commenting on the accuracy of Myrrh’s statements, let me just say that a few others have already tried reasoning with Myrrh in other threads, presenting many of the same sort of arguments you have given. So far, no one has been able to convince him that he has made any significant errors in logic or science.
🙂 No more argument from you then..
Jose_X – here’s a recent reply from me to one such still trying to convince me that I’ve significantly erred in logic or science:
http://wattsupwiththat.com/2011/12/02/foia-is-not-enough-why-not-legally-mandate-transparency-in-climate-research-a-modest-proposal/#comment-822762
Bob >> “Here is one of Jason Box’s co-authored papers”
Abstract > This suggests that the Greenland ice sheet and coastal regions are not following the current global warming trend.
Interesting. I wonder how knowledgeable the author was on the Greenland ice sheet back in 2004. I hear that ice melting is one of those areas climatologists apparently have been underestimating. Maybe there are more ideas and measurements to shake out in the upcoming years in this area.
This one paper’s possible surmising doesn’t suggest to me that a section of the report on ice sheets should include a discussion on surface temperatures, especially if this issue were to have been unsettled (for example, perhaps there has been genuine confusion among researchers over surface temperatures possibly not correlating well with ice sheet melt). If unsettled, they might not mention it and keep the discussion to what they are most sure about. And, of course, this author could simply have been wrong in the paper on that point.
Personally, if that view were to have been representative (air temp being linked to ice melt), I would think they would want to mention it. To judge better, we would have to look at a representative selection of relevant papers and then armchair quarterback.
I can’t see foul play or suspect biased judgement on so little info, but it’s an interesting observation nevertheless.
Myrrh,
Here’s another one for you. It was a warm day yesterday ~30C, and I popped up to the shops without footwear. Big mistake: I had to negotiate some tarmac (blacktop), and it was painfully hot, yet strangely, paler pavements in the sun were OK. Now I wonder why that should be! No need to reply this time either, I’m just having fun.
Mods – I posted a reply to Jose_X before I posted to Tim, it hasn’t appeared.
[Reply: I checked the spam folder. Sorry, it’s not there. Please re-post. ~dbs, mod.]
Jose_X @ur momisugly December 8, 9:24 am
Actually, I feel that the biggest problem in so-called climate science is exaggeration by the alarmist “scientists” commonly by exclusion of “inconvenient” data, and more importantly the political roll-on from there.
The MBH 99 hockey-stick was the charging white stallion in the 3AR (2001) IPCC report with six (or seven?) prominent displays within the report and even larger wall versions behind subsequent rostra.
If I remember correctly, in the 4AR 2007 report, the IPCC wisely buried MBH 99 amongst I think it was eleven spaghetti graphs all purporting to verify MBH 99. There were a large number of “scientists” involved. This is the junk that is used to scare policymakers etc, that 20 th Century had unprecedented warming.
Incidentally, in addition to hiding the decline of tree-ring correlation with instrumental temperatures since around 1940 or 1960 there are a bunch of other naughties in all these spaghetti graphs. One of the worst is probably what caught the eye of Severinghaus, before he knew about hide the decline, that there is a strange flattening of the curves in about the last 15 or 20 years. In fact, there is another cheat here in that in a 30-year CPA smoothing as is required for time-series smoothing, the smoothed line should stop 15 years short of the end raw data for lack of 15 years of future data beyond the centre-point. No worries, these “scientists” simply add non-existent data/ fiddle, (that I could elaborate), to show what they want.
I don’t want to overload you with references, but perhaps after you have finished and digested the Casper and Jesus paper stuff, you could get some more context here:
http://wattsupwiththat.com/2011/12/05/tim-barnett-on-the-hockey-stick-statistics-were-suspectthe-rest-of-the-team-knew-of-problems-with-manns-reconstruction/