Image Credits: NOAA – National Weather Service – Climate Prediction Center
By WUWT Regular “Just The Facts”
On the heels of Andrew Dessler’s Ozone Hole tweet, we have from the BBC:
“Researchers from the University of East Anglia have discovered evidence of four new gases that can destroy ozone and are getting into the atmosphere from as yet unidentified sources.”
“Scientists have identified four new man-made gases that are contributing to the depletion of the ozone layer.
Two of the gases are accumulating at a rate that is causing concern among researchers.”
“Other scientists acknowledged that while the current concentrations of these gases are small and they don’t present an immediate concern, work would have to be done to identify their origin.
“This paper highlights that ozone depletion is not yet yesterday’s story,” said Prof Piers Forster, from the University of Leeds.
“The concentrations found in this study are tiny. Nevertheless, this paper reminds us we need to be vigilant and continually monitor the atmosphere for even small amounts of these gases creeping up, either through accidental or unplanned emissions.
“Of the four species identified, CFC-113a seems the most worrying as there is a very small but growing emission source somewhere, maybe from agricultural insecticides. We should find it and take it out of production.”
The paper “Newly detected ozone-depleting substances in the atmosphere” Laube et al., paywalled, can be found here:
“Ozone-depleting substances emitted through human activities cause large-scale damage to the stratospheric ozone layer, and influence global climate. Consequently, the production of many of these substances has been phased out; prominent examples are the chlorofluorocarbons (CFCs), and their intermediate replacements, the hydrochlorofluorocarbons (HCFCs). So far, seven types of CFC and six types of HCFC have been shown to contribute to stratospheric ozone destruction1, 2. Here, we report the detection and quantification of a further three CFCs and one HCFC.”
“Our observations on air samples collected in remote regions of the atmosphere show the presence of four previously undetected ozone-depleting substances (ODSs). We have identified and quantified CFC-112 (CFCl2CFCl2), CFC-112a (CF2ClCCl3), CFC-113a (CF3CCl3) and HCFC-133a (CF3CH2Cl) in the atmosphere (Fig. 1). We have reconstructed their past abundances from air extracted from deep polar firn, which can provide a natural archive of atmospheric composition up to about a century back in time5. Our firn air measurements suggest that all four newly reported compounds are anthropogenic (see also Supplementary Information), with insignificant atmospheric abundances before the 1960s.”
For reference, the images the head of this article show the current Northern “Ozone Hole” within the Northern Polar Vortex, at 10 hPa/mb – Approximately 31,000 meters (101,700 feet). Draw your own conclusions…



Non Anglia, sed Angler – Just another fishy story from the super sleuths at Unbalievable Extroardinary Amaztatic.
Let me suggest the following “sources” of the new gaz:
1. Precisely where the latest spontaneous self immolating Tesla car is (was).
2. Look for a large, sweaty Stoat, pounding a keyboard and swearing viciously.
Was there really any need to read this after the words “East Anglia”?
CFC-112 and CFC-112a, CFC-113 and HCFC-133a are hardly NEW issues. They were banned under the Montreal Protocol. CFC-113a seems to be a by product of CCF-14 synthesis and/or decomposition of R-13.
see http://www.epa.gov/ozone/science/ods/index.html
“KNR says:
March 10, 2014 at 12:48 am
Researchers from the University of East Anglia – oxymoron?”
Can we just shorten it all to UEA=morons?
fhhaynie says:
March 9, 2014 at 4:24 pm
Asked the same question on earlier thread. Got this response from John M. What I’d like is some estimate of production which could be used to correlate with it.
I remember an episode of Family Ties (1982-1989) in which the younger daughter went on
a wicked environmental crusade, getting rid of all the “evil” chemicals in the house. This
included aerosol cans. But the government banned CFC propellants in 1978!
The other major use of CFC’s is as a refrigerant, but all CFC’s removed from an A/C unit
or refrigerator is treated like spent nuclear fuel rods! The stuff is sucked out of the system
and pumped into a storage tank and eventually destroyed. Can someone please tell me
how this can still be a problem?
I read in the supplementary information that they use multiple models!!!!…..
A state of the art model of trace gas transport in firn has been used in this study 16; compared with other similar models in 8). Such models need as input diffusion coefficient ratios in air
of the target species with respect to CO2. The values used calculated from critical
temperature and volume data are 203.83 for CFC-112 and CFC-112a, 187.38 for
CFC-113a and 118.49 for HCFC-133a, as detailed in the supplement of 8. Forward
firn models such as those inter-compared in 8 allow calculating concentrations in firn
from a known atmospheric history. Reconstructing atmospheric concentration
histories from depth – concentration profiles in firn requires to use inverse modelling
techniques. This inverse problem has multiple solutions S3. A robustness oriented
method for choosing the optimal solution, adapted to the scarcity of firn data (16 to 19
depth levels in this study), has been recently developed 21. The scarcity of
measurements is handled based on the mathematical development for robust solving
of inverse problems from S4. The reconstructed scenarios, together with their match of
the firn data are shown on Figures S11 and S12.
JustTheFacts
CFCs do not appear offer a plausible explanation for Northern “Ozone Hole”, i.e.
per NASA:
http://ozonewatch.gsfc.nasa.gov/facts/miniholes_NH.html
Apart from scale, how is that any different to the southern maxi-hole?
Given that southern depletion also appears to be nothing more than a temporary, cyclical,
seasonal rearrangement of ozone concentration, why does the Antarctic phenomena require
CFCs/chlorine & stratospheric ice crystals?
via Gary Pearse (previous thread), NASA again (annotated):
With an isolated & poorly insolated polar vortex, you don’t need any chlorine.
Khwarizmi says: March 10, 2014 at 6:01 pm
“Apart from scale, how is that any different to the southern maxi-hole?
Given that southern depletion also appears to be nothing more than a temporary, cyclical”
The Northern Ozone “Hole” wasn’t part of the CFC narrative and doesn’t fit well within it, thus it needs a different explanation. Otherwise they seem the same to me.
“With an isolated & poorly insolated polar vortex, you don’t need any chlorine.”
Yep, if there are a physical processes that adequately explain the existence of Ozone “Holes”, there is no need for extraneous CFC based chemical processes
My question is, how big would the Ozone “Hole” be if CFCs didn’t exist? The evidence indicates that it would be the same size, as Ozone “Hole” size appears to depend on how large and strong the Polar Vortex is, and how deep it penetrates into the atmosphere.
The IPCC and its alarmist faithful followers have been crying wolf for over two decades over dangerous AGW. In the process, they have brought science into disrepute. Consequently, even if these new gases were indeed a serious concern, none of us will give a damn!
fhhaynie says:
March 9, 2014 at 4:24 pm
Has anybody ever measured the amount of CFCs or chlorine actually in the “ozone hole” over time?
http://undsci.berkeley.edu/images/ozone/chlorine_graph.gif
http://upload.wikimedia.org/wikipedia/commons/thumb/1/16/Ozone_cfc_trends.png/250px-Ozone_cfc_trends.png
justthefactswuwt says:
March 10, 2014 at 6:51 pm
My question is, how big would the Ozone “Hole” be if CFCs didn’t exist? The evidence indicates that it would be the same size, as Ozone “Hole” size appears to depend on how large and strong the Polar Vortex is, and how deep it penetrates into the atmosphere.
I disagree, the data indicates that the Dobson number would be about 325 under the hole in the spring (October) rather than less than 100 at the minimum.
http://www.atm.ch.cam.ac.uk/tour/tour_images/total_ozone.gif
phil,
please try to explain why the highest concentrations of ozone on Earth are always found around the perimeter of the depletion region.
Look at the accretion disc instead of the hole, and try to explain it for us all.
Good luck!
Phil. says: March 11, 2014 at 10:21 am NOAA – National Weather Service – Climate Prediction Center – Click the pic to view at source[/caption]
NOAA – National Weather Service – Climate Prediction Center – Click the pic to view at source[/caption] NOAA – National Weather Service – Climate Prediction Center – Click the pic to view at source[/caption]
NOAA – National Weather Service – Climate Prediction Center – Click the pic to view at source[/caption] ” NASA – Earth Observatory – Click the pic to view at source[/caption]
” NASA – Earth Observatory – Click the pic to view at source[/caption] “Center for Atmospheric and Space Sciences, Logan, Utah – Click the pic to view at source[/caption]
“Center for Atmospheric and Space Sciences, Logan, Utah – Click the pic to view at source[/caption]
I disagree, the data indicates that the Dobson number would be about 325 under the hole in the spring (October) rather than less than 100 at the minimum.
No, even if this data was accurate, this would only effect the concentration of ozone within the Ozone “Hole”, the Ozone Hole itself would be same size:
[caption id="" align="alignnone" width="578"]
It terms of the data you present;
[caption id="" align="alignnone" width="578"]
it is suspect. Firstly, there is no identification of the source of the data on the graph, or on the page where it can be found:
http://www.atm.ch.cam.ac.uk/tour/part1.html
It appears to misleading version of this graph, which at least shows the source and variability of the associated measurements:
[caption id="" align="alignnone" width="578"]
Secondly, in “1956, the British Antarctic Survey set up the Halley Bay Observatory on Antarctica in preparation for the International Geophysical Year (IGY) of 1957. In that year, ozone measurements using a Dobson Spectrophotometer began.”
Halley Bay, is, unsurprisingly, on the coast of Antarctica;
[caption id="" align="alignnone" width="578"]
and thus is not well situated to measure a phenomenon that is usually centered over the South Pole. Halley Bay represented a single measurement location on a continent that is 5.405 million sq miles (14 million km²), and Ozone “Hole” that exceeds 18 million km² at its maximum. Furthermore the quality of the data is suspect, i.e.:
“The network included a station in Halley Bay, Antarctica, which is famous for the discovery of the ozone hole in the 1980s (Farman et al. 1985). At the same time observation techniques and calibration were improved and the procedures were standardised. This allows re-evaluating many total ozone series back to 1957 based on meta-information and calibration information.”
http://meteo.edu.vn/DATA/Books/Climate%20Variability%20and%20Extremes%20during%20the%20Past%20100%20Years/8.%20Total%20Ozone%20Observations%20During%20the%20Past%2080%20Years.pdf
Third, the TOMS Instrument satellite ozone data is also suspect, i.e.:
“Nimbus-7 TOMS Instrument and Satellite Information
The TOMS program began with the launch of TOMS Flight Model #1 on the Nimbus-7 spacecraft on October 24, 1978. Valid measurements started in November of that same year and the instrument continued to return data long after all other on-board experiments had failed. The TOMS instrument fell silent in May 1993. The software to derive useful information from the data returned by Nimbus 7 TOMS is the basis for the algorithm used to analyze all TOMS data and has gone through a lengthy evolutionary process bring it to the current version. The Version 7 processed data include a revised instrument calibration based on analysis of the entire 14.5 year data record (including a correction for a 0.2 nm wavelength error which caused a 3% absolute offset relative to Dobson) as well as an improved algorithm.
Algorithmic Improvements include:
use of wavelength “triplets” that correct for errors linear in wavelength
improved ISCCP cloud height climatology, higher resolution terrain height maps
use of improved profile shape selection to improve total ozone at very large solar zenith angles
use of a more accurate model for partially-clouded scenes improved radiative transfer calculations for table generation”
http://toms.gsfc.nasa.gov/n7toms/n7sat.html
NASA Ozone Data Source:
The data for 1979–1993 are from the TOMS instrument on the NASA/NOAA Nimbus-7 satellite.
The data for this 1993–1994 are from the TOMS instrument on the Soviet-built Meteor-3 satellite.
The data for 1996–October 2004 are from the NASA Earth Probe TOMS satellite.
The data for November 2004–2011 are from the OMI instrument (KNMI / NASA) onboard the Aura satellite. They are the OMTO3 that have been processed in a manner similar to the TOMS data from earlier years.
The ozone minimum is determined only from data actually contained in the processed satellite data. To calculate the ozone hole area and mass deficit, we fill in missing areas (bad orbits and polar night) from an atmospheric model. MERRA is a NASA reanalysis for the satellite era using a major new version of the Goddard Earth Observing System Data Assimilation System Version 5 (GEOS-5). The Project focuses on historical analyses of the hydrological cycle on a broad range of weather and climate time scales and places the NASA EOS suite of observations in a climate context. Since these data are from a reanalysis, they are not up-to-date. So, we supplement with the GEOS-5 FP data that are also produced by the GEOS-5 model in near real time.”
http://ozonewatch.gsfc.nasa.gov/meteorology/ozone_1990_MERRA_SH.html
“08-15-2007
Corrected Earth Probe Data
correction basis: NOAA-16 SBUV/2 ozone
time period: August 1996 – December 13, 2005
data products corrected: ozone, reflectivity
By mid-2000, the Earth Probe (EP) TOMS instrument degradation became so large that standard correction procedures could no longer produce accurate ozone. The problem is believed to be inhomogeneous degradation of the scanner mirror on TOMS that results in a calibration error that is different at different latitudes. We have warned users that the production EP ozone data should NOT be used for trend analysis.
We have now applied a correction to the Earth Probe data that stabilizes the EP ozone record. This empirical correction is based on the NOAA-16 SBUV/2 ozone record, with a solar zenith angle dependence that accounts for much of the spurious latitude dependence observed in the current data. Only the ozone and reflectivity records have been corrected. The aerosol index data and SO2 records are more complex and have not been corrected by this empirical correction.
Comparison with the ground network shows that the resulting ozone is stable within ± 1% over the 1996-2005 period. In the period 2002-2005 in the northern hemisphere, there is a residual seasonally-dependent error of ± 1.5% magnitude. These data should still NOT be used as a source for trend analysis since they are no longer independent.”
http://ozoneaq.gsfc.nasa.gov/news.md
So “long after all other on-board experiments had failed” it produced suspect data, which was then put through “a lengthly evolutionary process” that includes “revised instrument calibration”, “including a correction for a 0.2 nm wavelength error”, “as well as an improved algorithm” that included “a more accurate model for partially-clouded scenes” and “to calculate ozone hole area and mass deficit” they “fill in missing areas (bad orbits and polar night) from an atmospheric model.”
It seems that the TOMS instrument has a long history of issues and required “corrections”, thus the resultant Ozone “hole” data should be considered suspect.
Lastly, here is an interesting paper from 1986 that offers a sober assessment of the source of the Ozone “Hole”:
“Data from the Total Ozone Mapping Spectrometer (TOMS) on the Nimbus 7 satellite are compared with Dobson spectrophotometer measurements from the Antarctic, where a large decrease in October total ozone has been observed over the last decade. It has been hypothesized that the decrease is caused by local photochemical effects of man-made chlorofluorocarbons. The TOMS data, which start in 1978, agree well with the Dobson data, confirming the low values of recent years. However, during the southern hemisphere spring total ozone in the Antarctic is strongly affected by transport associated with the breakdown of the stratospheric vortex. The strength and timing of the vortex breakdown are highly variable and can have a large effect on monthly-mean total ozone values. The TOMS data suggest that variations in the stratosphefic circulation have contributed to the long-term decrease. Large variations in total ozone are not confined to the Antarctic. Zonal-mean ozone values have also decreased in the northern hemisphere spring during the TOMS observing period.”
“The TOMS measurements largely corroborate the exceptionally low October Dobson measurements made at HB for the years 1979-1982. The observed decrease in total ozone in the Antarctic during the last decade may have been caused by either photochemical or dynamical processes. Farman et al. [1985] and others have hypothesized that the changes are the result only of local photochemical effects. However, it is clear from the TOMS data that ozone transport and especially the timing of the vortex breakdown can have a large influence on the monthly mean total ozone, especially for October and November, when the vortex breakdown usually takes place. When the vortex breakdown occurs early (late) the October mean ozone is high (low). Figure 1 in Farman et al. also suggests that extreme high ozone values are associated with early breakdowns, while extreme low values are found in years with weak and late breakdowns. The vortex breakdowns of 1980-1982 were weaker and later than for 1978 and 1979. Thus, it is possible that the observed long-term ozone changes are in part the result of changes in planetary wave activity and the stratospheric circulation. Ozone fluctuations from interannual variability of the vortex breakdown may have been superimposed on trends due to chemical processes. A third possibility is that chemical- induced ozone changes have changed radiative heating rates sufficiently to alter the zonal-mean circulation and the propagation of planetary waves into the lower stratosphere, delaying the vortex breakdown. Continued monitoring from the ground and from satellites will be necessary to determine whether the low total ozone values in the Antarctic persist. A return of ozone values to early levels would contradict the chemical hypothesis.
Continued low ozone values, especially if they persist in both hemispheres, will support chemical depletion mechanisms and make dynamical explanations less likely. While the TOMS instrument continues to operate, it will provide valuable ozone trend data and information on the dynamics of the vortex breakdown.”
http://geotest.tamu.edu/userfiles/213/GL013i012p01193.pdf
No persistent Ozone “Hole” has formed in the Northern Hemisphere, rather the current Northern Ozone “Hole” appears to be the result of dynamical forces of the Northern Polar Vortex, thus the Southern Hemisphere Ozone “Hole” likely caused by the same dynamical forces.
Khwarizmi says: March 11, 2014 at 1:40 pm NOAA – National Weather Service – Climate Prediction Center [/caption]
NOAA – National Weather Service – Climate Prediction Center [/caption]
please try to explain why the highest concentrations of ozone on Earth are always found around the perimeter of the depletion region.
Look at the accretion disc instead of the hole, and try to explain it for us all.
Good luck!
Sure, the dynamical forces of the Polar Vortex displace the ozone, resulting in an Ozone “Surplus” around the Polar Vortex and associated Ozone “Hole”:
[caption id="" align="alignnone" width="400"]
justthefactswuwt says:
March 11, 2014 at 7:06 pm
Phil. says: March 11, 2014 at 10:21 am
“I disagree, the data indicates that the Dobson number would be about 325 under the hole in the spring (October) rather than less than 100 at the minimum.”
No, even if this data was accurate, this would only effect the concentration of ozone within the Ozone “Hole”, the Ozone Hole itself would be same size:
No the hole is the result of the concentration distribution.
It terms of the data you present;
NOAA – National Weather Service – Climate Prediction Center – Click the pic to view at source
it is suspect. Firstly, there is no identification of the source of the data on the graph, or on the page where it can be found:
http://www.atm.ch.cam.ac.uk/tour/part1.html
It is the Hally Bay data shown on the NOAA site as indicated on the graph legend. You can read more about it here:
http://www.atm.ch.cam.ac.uk/tour/part2.html
It appears to misleading version of this graph, which at least shows the source and variability of the associated measurements:
As opposed to the British Antarctic Survey data which I showed which shows the confidence intervals for the annual october data. And you have it backwards the Hally Bay data came first!
Halley Bay, is, unsurprisingly, on the coast of Antarctica;
and thus is not well situated to measure a phenomenon that is usually centered over the South Pole. Halley Bay represented a single measurement location on a continent that is 5.405 million sq miles (14 million km²), and Ozone “Hole” that exceeds 18 million km² at its maximum.
Actually it’s perfectly situated and was the location from where the existence of the ‘hole’ was first detected by Farmar et al.!
Note the position of Hally Bay wrt the ‘hole’ in this image.
http://upload.wikimedia.org/wikipedia/commons/e/ea/NASA_and_NOAA_Announce_Ozone_Hole_is_a_Double_Record_Breaker.png
Furthermore the quality of the data is suspect, i.e.:
Not suspect at all, except for someone with a bias such as yourself.
No persistent Ozone “Hole” has formed in the Northern Hemisphere, rather the current Northern Ozone “Hole” appears to be the result of dynamical forces of the Northern Polar Vortex, thus the Southern Hemisphere Ozone “Hole” likely caused by the same dynamical forces.
That’s because the destruction of ozone is enhanced by some heterogeneous kinetics involving ice crystals (with nitric acid) in polar stratospheric clouds which need low temperatures for their formation (below -78ºC), these temperatures (and clouds) are encountered much more commonly over the Antarctic than the Arctic which is why such holes are infrequently encountered in the NH. These crystals accumulate chlorine in the form of ClONO2 during the winter, during the spring the sunlight melts the crystals thereby releasing a considerable amount of ClO and Cl at the same time and place as the UV arrives causing considerable O3 depletion.
Khwarizmi says:
March 11, 2014 at 1:40 pm
phil,
please try to explain why the highest concentrations of ozone on Earth are always found around the perimeter of the depletion region.
Look at the accretion disc instead of the hole, and try to explain it for us all.
Good luck!
No luck needed, it’s the result of Brewer-Dobson circulation. The air from the tropics rises and travels poleward and as it rises through the lower stratosphere ozone accumulates due to photolysis. Once it enters the region of high Cl and ClO then depletion begins.
Phil. says: March 12, 2014 at 10:57 am NOAA – National Weather Service – Climate Prediction Center [/caption]
NOAA – National Weather Service – Climate Prediction Center [/caption]
No the hole is the result of the concentration distribution.
No, the “hole” is the result of the dynamical forces of the polar vortex, including the naturally occurring low pressure area within the Polar Vortex;
[caption id="" align="alignnone" width="400"]
and because;
Air towards the top of the stratosphere and bottom of the mesosphere has lower concentrations of ozone;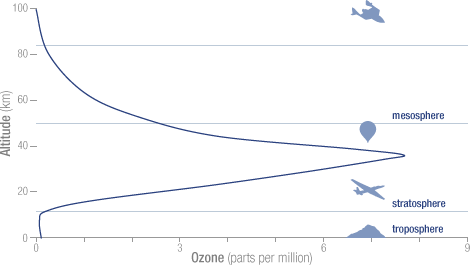 NOAA – National Aeronautics and Space Administration (NASA) Goddard Space Flight Center (GSFC) – Click the pic to view at source[/caption]
NOAA – National Aeronautics and Space Administration (NASA) Goddard Space Flight Center (GSFC) – Click the pic to view at source[/caption] NASA – Goddard Space Flight Center – Arctic Ozone Watch – Click the pic to view at source[/caption]
NASA – Goddard Space Flight Center – Arctic Ozone Watch – Click the pic to view at source[/caption] NASA – Goddard Space Flight Center – Arctic Ozone Watch – Click the pic to view at source[/caption]
NASA – Goddard Space Flight Center – Arctic Ozone Watch – Click the pic to view at source[/caption]
[caption id="" align="alignnone" width="578"]
As such, when this “air from very high altitudes descends vertically through the center of the vortex” it displaces the air below it, decreasing the concentration of ozone within the Polar Vortex. The combination of the low pressure area formed by the centrifugal force of the Polar Vortex and the air from very high altitudes with lower concentrations of ozone that descends through the center of the vortex, creates the “Ozone Hole”:
[caption id="" align="alignnone" width="600"]
[caption id="" align="alignnone" width="537"]
As opposed to the British Antarctic Survey data which I showed which shows the confidence intervals for the annual october data. And you have it backwards the Hally Bay data came first!
That’s funny, they have the same confidence intervals for the Dobson Spectrophotometer data in 1956 as for the TOMS data in 1995. Perhaps if they increase those intervals by an order of magnitude that graph would be more accurate….
Actually it’s perfectly situated and was the location from where the existence of the ‘hole’ was first detected by Farmar et al.!
Perfectly situated would be the center of Antarctica, i.e.:
“The ozone hole is in the center of a spiraling mass of air over the Antarctic that is called the polar vortex. The vortex is not stationary and sometimes moves as far north as the southern half of South America, taking the ozone hole with it.”
http://www.nasa.gov/centers/langley/news/factsheets/HALOE-Ozone.html
If Polar Vortex area is smaller, or breakup occurs earlier, there is no Ozone “Hole” above Halley to measure in October.
Not suspect at all, except for someone with a bias such as yourself.
Yes, a bias towards the facts. Apparently foreign to the climate science community these days…
That’s because the destruction of ozone is enhanced by some heterogeneous kinetics involving ice crystals (with nitric acid) in polar stratospheric clouds which need low temperatures for their formation (below -78ºC), these temperatures (and clouds) are encountered much more commonly over the Antarctic than the Arctic which is why such holes are infrequently encountered in the NH. These crystals accumulate chlorine in the form of ClONO2 during the winter, during the spring the sunlight melts the crystals thereby releasing a considerable amount of ClO and Cl at the same time and place as the UV arrives causing considerable O3 depletion.
Or because:
“The vortex is most powerful in the hemisphere’s winter, when the temperature gradient is steepest, and diminishes or can disappear in the summer. The Antarctic polar vortex is more pronounced and persistent than the Arctic one; this is because the distribution of land masses at high latitudes in the northern hemisphere gives rise to Rossby waves which contribute to the breakdown of the vortex, whereas in the southern hemisphere the vortex remains less disturbed.”
http://en.wikipedia.org/wiki/Polar_vortex
We’ll let Occam and the readers decide between those two options…
justthefactswuwt says:
March 12, 2014 at 10:04 pm
Phil. says: March 12, 2014 at 10:57 am
“No the hole is the result of the concentration distribution.”
No, the “hole” is the result of the dynamical forces of the polar vortex, including the naturally occurring low pressure area within the Polar Vortex;
The ‘hole’ is the result of ozone concentration regardless of the dynamical situation without the chemical depletion there would be no ‘hole’!
and because;
“in the center of the Antarctic vortex. Air from very high altitudes descends vertically through the center of the vortex, moving air to lower altitudes over several months.”
http://www.nasa.gov/centers/langley/news/factsheets/HALOE-Ozone.html
Air towards the top of the stratosphere and bottom of the mesosphere has lower concentrations of ozone;
The graph you showed applies to the atmosphere in general where the stratosphere is ‘stratified’, but does not apply over the Antarctic when the hole exists. Here’s some real data from that location:
http://www.cpc.ncep.noaa.gov/products/stratosphere/polar/gif_files/sp_profile.gif
As such, when this “air from very high altitudes descends vertically through the center of the vortex”
This quote is correct, however your assertion which follow is wrong!
it displaces the air below it, decreasing the concentration of ozone within the Polar Vortex.
Clearly the actual data shows that the ozone concentration above the ‘hole’ is actually higher than the ozone concentration at the lower altitudes. As that relatively ozone rich air falls below ~20km it encounters the Cl and ClO and the chemical kinetics takes over and that ozone is destroyed.
The combination of the low pressure area formed by the centrifugal force of the Polar Vortex and the air from very high altitudes with lower concentrations of ozone that descends through the center of the vortex, creates the “Ozone Hole”:
Clearly not!
“As opposed to the British Antarctic Survey data which I showed which shows the confidence intervals for the annual october data. And you have it backwards the Hally Bay data came first”!
That’s funny, they have the same confidence intervals for the Dobson Spectrophotometer data in 1956 as for the TOMS data in 1995. Perhaps if they increase those intervals by an order of magnitude that graph would more accurate….,
Total rubbish, you have no basis for that assertion, the BAS data was made using an instrument on the ground which is calibrated in situ and which still exists, what the TOMS instruments report has no bearing on the original BAS data.
“Actually it’s perfectly situated and was the location from where the existence of the ‘hole’ was first detected by Farmar et al.!”
Perfectly situated would be the center of Antarctica, i.e.:
As shown by the images Hally Bay is close to the center of the ‘hole’, in the image you show above it’s approximately equidistant with the S Pole.
The HALOE site which you quoted mostly concentrates on the destruction of ozone by chemical means, for example:
“Ozone Destruction
Ozone in the stratosphere is destroyed when it combines with chlorine, forming oxygen and chlorine monoxide. A single chlorine molecule can destroy 100,000 ozone molecules in its lifetime.”
And: “HALOE’s measurements have settled a crucial scientific issue by confirming that CFCs are responsible for the elevated chlorine levels in the stratosphere that lead to ozone loss.”
“HALOE is the first instrument that has been able to confirm the influence of human activities on the amount of ozone-destroying chlorine in the stratosphere.”
Apparently you ignored the whole site except the last paragraph, did you hope that no-one would read it?
Phil. says: March 13, 2014 at 8:11 am NOAA – National Aeronautics and Space Administration (NASA) Goddard Space Flight Center (GSFC) – Click the pic to view at source[/caption]
NOAA – National Aeronautics and Space Administration (NASA) Goddard Space Flight Center (GSFC) – Click the pic to view at source[/caption] NASA GSFC, USA; image prepared by CSIRO Atmospheric Research – Click the pic to view at source[/caption]
NASA GSFC, USA; image prepared by CSIRO Atmospheric Research – Click the pic to view at source[/caption]
The ‘hole’ is the result of ozone concentration regardless of the dynamical situation without the chemical depletion there would be no ‘hole’!
No, I have clearly demonstrated that there would be a “hole” without “chemical depletion”, hence why there is also “a water vapor hole, a nitrogen oxide hole”;
http://books.google.com/books?id=B93SSQrcAh4C&lpg=PA283&ots=d0-uBRjmyI&dq=%22water%20vapor%20hole%22%20polar%20vortex&pg=PA283#v=onepage&q=%22water%20vapor%20hole%22%20polar%20vortex&f=false
and “measurements of low methane concentrations in the vortex”.
http://onlinelibrary.wiley.com/doi/10.1029/93GL01104/abstract
What do you think causes the water vapor, nitrogen oxide and methane “holes”?
The graph you showed applies to the atmosphere in general where the stratosphere is ‘stratified’, but does not apply over the Antarctic when the hole exists. Here’s some real data from that location:
http://www.cpc.ncep.noaa.gov/products/stratosphere/polar/gif_files/sp_profile.gif
[caption id="" align="alignnone" width="400"]
“The polar vortex extends from the tropopause (the dividing line between the stratosphere and troposphere) through the stratosphere and into the mesosphere (above 50 km). Low values of ozone and cold temperatures are associated with the air inside the vortex.” NASA
Your graph demonstrates my point quite well, thank you.
Clearly the actual data shows that the ozone concentration above the ‘hole’ is actually higher than the ozone concentration at the lower altitudes. As that relatively ozone rich air falls below ~20km it encounters the Cl and ClO and the chemical kinetics takes over and that ozone is destroyed.
What are you looking at? Both of the graphs show that there is clearly less Ozone up at 30, 40 and 50 km, which is the “air from very high altitudes” that “descends vertically through the center of the vortex, moving air to lower altitudes over several months.”
http://www.nasa.gov/centers/langley/news/factsheets/HALOE-Ozone.html
Total rubbish, you have no basis for that assertion, the BAS data was made using an instrument on the ground which is calibrated in situ and which still exists, what the TOMS instruments report has no bearing on the original BAS data.
I cited 5 sources supporting my assertion that the quality of the historical Ozone data is suspect, here;
http://wattsupwiththat.com/2014/03/09/mysterious-new-man-made-gases-pose-threat-to-ozone-layer/#comment-1588123
and you cited “rubbish”. I think I won that one…
As shown by the images Hally Bay is close to the center of the ‘hole’, in the image you show above it’s approximately equidistant with the S Pole.
Yes, in that image, but in other images it’s not:
[caption id="" align="alignnone" width="578"]
The reason for the increased prevalence and persistence of the Southern Ozone “Hole” in October is well documented, i.e. “several studies (including Waugh and Randel 1999; Waugh et al. 1999; Karpetchko et al. 2005; Black and McDaniel 2007) have indicated a trend over the 1980s and 1990s toward a later vortex breakdown.”
http://www.columbia.edu/~lmp/paps/waugh+polvani-PlumbFestVolume-2010.pdf
The HALOE site which you quoted mostly concentrates on the destruction of ozone by chemical means, for example:
Yes, sad that NASA was already that offtrack back in 1996. Now they’re pushing silly Warmist propaganda, e.g.:
http://climatekids.nasa.gov/
Apparently you ignored the whole site except the last paragraph
No, that paragraph is in the middle. I read it all and determined some of it to be factually inaccurate.
did you hope that no-one would read it?
Yes, you got me, that’s why I posted a link to it, to trick people into not reading it…
justthefactswuwt says:
March 13, 2014 at 9:23 pm
Phil. says: March 13, 2014 at 8:11 am
Your graph demonstrates my point quite well, thank you.
Only if you’re completely unable to read a graph!
“Clearly the actual data shows that the ozone concentration above the ‘hole’ is actually higher than the ozone concentration at the lower altitudes. As that relatively ozone rich air falls below ~20km it encounters the Cl and ClO and the chemical kinetics takes over and that ozone is destroyed.”
What are you looking at? Both of the graphs show that there is clearly less Ozone up at 30, 40 and 50 km, which is the “air from very high altitudes” that “descends vertically through the center of the vortex, moving air to lower altitudes over several months.”
Clearly I have to explain the graph you’re looking at since you’re either incapable of reading it or are so consumed by your bias that you don’t want to see it!
At about 31km the pO3 is ~3mPa, as the air descends to ~23km the pO3 increases to ~6mPa due to the photolysis reactions. As it descends over the next couple of km the pO3 rapidly drops to less than 1mPa, so clearly there is more O3 at 30km than at 20km not less as you incorrectly assert above. Your mechanism is contradicted by the data and the chemical kinetic mechanism is supported by the data as shown. Note that this decrease occurs in October but not in July, when the pO3 continues to increase, peaking at ~16mPa where the temperature and pressure has increased and the chemical mechanism (Chapman etc) is expected to deplete O3. Below ~13km and above ~23km there’s no difference between Oct and July values which your mechanism is unable to explain!
I cited 5 sources supporting my assertion that the quality of the historical Ozone data is suspect, here;
You cited no such data regarding the BAS data which I cited earlier.
“As shown by the images Hally Bay is close to the center of the ‘hole’, in the image you show above it’s approximately equidistant with the S Pole.”
Yes, in that image, but in other images it’s not:
It’s under the ‘hole’ in all those images you showed since 1984 (when the ‘hole’ was still small) the S Pole was at the edge in Oct 84 by the way.
Phil. says: March 14, 2014 at 7:28 am NOAA – National Weather Service – Climate Prediction Center[/caption]
NOAA – National Weather Service – Climate Prediction Center[/caption] NOAA – National Weather Service – Climate Prediction Center [/caption]
NOAA – National Weather Service – Climate Prediction Center [/caption] NOAA – National Weather Service – Climate Prediction Center [/caption]
NOAA – National Weather Service – Climate Prediction Center [/caption] NOAA – National Weather Service – Climate Prediction Center [/caption]
NOAA – National Weather Service – Climate Prediction Center [/caption] NOAA – National Weather Service – Climate Prediction Center [/caption]
NOAA – National Weather Service – Climate Prediction Center [/caption] NOAA – National Weather Service – Climate Prediction Center – Click the pic to view at source[/caption]
NOAA – National Weather Service – Climate Prediction Center – Click the pic to view at source[/caption] NOAA – National Weather Service – Climate Prediction Center – Click the pic to view at source[/caption]
NOAA – National Weather Service – Climate Prediction Center – Click the pic to view at source[/caption] NOAA – National Weather Service – Climate Prediction Center[/caption]
NOAA – National Weather Service – Climate Prediction Center[/caption] NOAA – National Weather Service – Climate Prediction Center[/caption]
NOAA – National Weather Service – Climate Prediction Center[/caption] NOAA – National Weather Service – Climate Prediction Center[/caption]
NOAA – National Weather Service – Climate Prediction Center[/caption] NOAA – National Weather Service – Climate Prediction Center [/caption]
NOAA – National Weather Service – Climate Prediction Center [/caption] NOAA – National Weather Service – Climate Prediction Center [/caption]
NOAA – National Weather Service – Climate Prediction Center [/caption] NOAA – National Weather Service – Climate Prediction Center [/caption]
NOAA – National Weather Service – Climate Prediction Center [/caption] NOAA – National Weather Service – Climate Prediction Center [/caption]
NOAA – National Weather Service – Climate Prediction Center [/caption] NOAA – National Weather Service – Climate Prediction Center [/caption]
NOAA – National Weather Service – Climate Prediction Center [/caption] NOAA – National Weather Service – Climate Prediction Center [/caption]
NOAA – National Weather Service – Climate Prediction Center [/caption] NOAA – National Weather Service – Climate Prediction Center [/caption]
NOAA – National Weather Service – Climate Prediction Center [/caption]
I asked you “What do you think causes the water vapor, nitrogen oxide and methane “holes”?” and you did not respond. Can we take this as an admission that you cannot answer this question without contradicting your claim that “regardless of the dynamical situation without the chemical depletion there would be no ‘hole’!”?
Only if you’re completely unable to read a graph!
“Clearly the actual data shows that the ozone concentration above the ‘hole’ is actually higher than the ozone concentration at the lower altitudes.
We seem to be at an impasse here. When there is not a Polar Vortex in the Southern Hemisphere, does ozone concentration increase or decrease between 25 km and 50 km?
As that relatively ozone rich air falls below ~20km it encounters the Cl and ClO and the chemical kinetics takes over and that ozone is destroyed.
Really?… Then why in the Northern Hemisphere is there currently an Ozone “Hole” at 2 hPa/mb – ~42 km;
[caption id="" align="alignnone" width="450"]
and a bigger one at 5 hPa/mb ~ 35 km;
[caption id="" align="alignnone" width="450"]
and an even bigger one at 10 hPa/mb ~ 31 km;
[caption id="" align="alignnone" width="450"]
a pronounced one, surrounded by a large Ozone “Surplus”, at 30 hPa/mb ~ 23 km;
[caption id="" align="alignnone" width="449"]
and then it shrinks significantly at 50-hPa/mb ~ 20 km;
[caption id="" align="alignnone" width="452"]
before it disappears at 70-hPa/mb ~ 18 km;
[caption id="" align="alignnone" width="451"]
and there’s nothing at 100-hPa/mb ~ 15 km:
[caption id="" align="alignnone" width="450"]
And lest you think at this is only the case in Northern Hemisphere, and in the Southern Hemisphere magically, “ozone rich air falls below ~20km it encounters the Cl and ClO and the chemical kinetics takes over and that ozone is destroyed”, let us also look at the Southern Ozone “Hole”:
On October 15, 2013, in the Southern Hemisphere, the Ozone “Hole” started even higher at .5 hPa/mb – ~55 km;
[caption id="" align="alignnone" width="450"]
grews at 1 hPa/mb – ~50 km;
[caption id="" align="alignnone" width="450"]
and was quite pronounced at 2 hPa/mb – ~42 km;
[caption id="" align="alignnone" width="450"]
weakened at 5 hPa/mb ~ 35 km;
[caption id="" align="alignnone" width="450"]
split into two lobes at 10 hPa/mb ~ 31 km;
[caption id="" align="alignnone" width="450"]
and reinvigorated, with a large Ozone “Surplus” at 30 hPa/mb ~ 23 km;:
[caption id="" align="alignnone" width="449"]
Unfortunately, NOAA’s Ozone Mixing Archives;
http://www.cpc.ncep.noaa.gov/products/stratosphere/sbuv2to/archive/sh/
don’t go below 30 hPa/mb ~ 23 km, so we can’t see the magical “encounters” “below ~20km” where the “Cl and ClO and the chemical kinetics takes over and that ozone is destroyed”. However, it is apparent that the air within the Polar Vortex “above ~20km” has low concentrations of ozone, thus your assertion “that relatively ozone rich air falls below ~20km” is falsified. Can you admit that you are wrong?
Clearly I have to explain the graph you’re looking at since you’re either incapable of reading it or are so consumed by your bias that you don’t want to see it!
At about 31km the pO3 is ~3mPa, as the air descends to ~23km the pO3 increases to ~6mPa due to the photolysis reactions. As it descends over the next couple of km the pO3 rapidly drops to less than 1mPa, so clearly there is more O3 at 30km than at 20km not less as you incorrectly assert above.
Now you are just playing games because you know you’re losing. There is clearly less O2 with altitude. I never asserted that “is more O3 at 30km than at 20km”, I wrote that “that there is clearly less Ozone up at 30, 40 and 50 km”. You can play with the starting point, ~23 km, 20 km or ~ 18 km depending on day, season, vortex size, strength and penetration, but that doesn’t change the fact that there is ” is clearly less Ozone in the air up at 30, 40 and 50 km which “descends vertically through the center of the vortex, moving air to lower altitudes over several months.”
http://www.nasa.gov/centers/langley/news/factsheets/HALOE-Ozone.html“.
Furthermore, Ozone-Sonde don’t measure absolute Ozone concentration rather;
“ozone-sonde observations often use the ‘partial pressure’ of ozone as their unit. This is the pressure that the ozone molecules would exert if all the other molecules in the air were removed. If you look at Figure 5 the O3 is presented as a pressure in milli-Pascals (mPa). You can convert these numbers to a mixing ratio by dividing by the pressure of the air at that point. So for example at the surface the O3 partial pressure is 5 mPa. The pressure at that point is ~1013 hPa so the mixing ratio of O3 is 5×10-3 /1013×102 = 5×10-8 molecules of O3 per molecule of air or 50 parts per billion.
Sometimes we are interested in the total amount of O3 above our heads. In this case we use Dobson units. If you image you took all the O3 from above your head and brought it down to the surface at 0ºC and a pressure of 1013 hPa how thick would it be? For historical reasons this thickness is given in units of 0.01 mm. So if you brought all the O3 above your head down to the ground at 0ºC and it formed a column 3 mm thick the O3 column would be 300 DU”
http://www.met.reading.ac.uk/~sws05ajc/teaching/ozonesonde.pdf
Since atmospheric pressure decreases rapidly with height;
[caption id="" align="alignnone" width="449"]
ozone-sonde observations using “‘partial pressure’ of ozone as their unit” can be misleading to the uninformed.
For those interested, this site offers an archive of Ozone South Pole, Antarctica Vertical Profiles;
http://www.esrl.noaa.gov/gmd/dv/iadv/graph.php?code=SPO&program=ozwv&type=vp
including the one Phil cited.
Your mechanism is contradicted by the data and the chemical kinetic mechanism is supported by the data as shown. Note that this decrease occurs in October but not in July, when the pO3 continues to increase, peaking at ~16mPa where the temperature and pressure has increased and the chemical mechanism (Chapman etc) is expected to deplete O3. Below ~13km and above ~23km there’s no difference between Oct and July values which your mechanism is unable to explain!
No it’s not, there is a simple dynamical mechanism versus the convoluted chemical mechanism that you assert, i.e. the Polar Vortex is still developing and penetrating through the atmosphere in July;
[caption id="" align="alignnone" width="449"]
on August 1st, 2013, their first reading, the Polar Vortex has barely reached 30 hPa/mb ~ 23 km;
[caption id="" align="alignnone" width="449"]
whereas by October 15th, 2013 it has penetrated much deeper, thus the lower Ozone Mixing ratio at 30 hPa/mb ~ 23 km:
[caption id="" align="alignnone" width="449"]
A simple dynamical mechanism explains the observations, whereas the convoluted chemical mechanism you assert is falsified by the observations that the Ozone Hole extends well above the supposed “~20km” where you claim “relatively ozone rich air falls below” and “encounters the Cl and ClO and the chemical kinetics takes over and that ozone is destroyed.”
You cited no such data regarding the BAS data which I cited earlier.
I cited that it is a single location on the coast and that:
“in the 1980s” “observation techniques and calibration were improved and the procedures were standardised. This allows re-evaluating many total ozone series back to 1957 based on meta-information and calibration information.”
http://meteo.edu.vn/DATA/Books/Climate%20Variability%20and%20Extremes%20during%20the%20Past%20100%20Years/8.%20Total%20Ozone%20Observations%20During%20the%20Past%2080%20Years.pdf
“Re-evaluating many total ozone series back to 1957 based on meta-information and calibration information” is the very definition of suspect data…
It’s under the ‘hole’ in all those images you showed since 1984 (when the ‘hole’ was still small) the S Pole was at the edge in Oct 84 by the way.
It is still measure the outer edge of a swirling polar vortex. Clearly the South Pole location is preferred, hence why they now do ozone-sondes from there. And the reason the increased prevalence and persistence of the Southern Ozone “Hole” in October since 1984 is well documented, i.e. “several studies (including Waugh and Randel 1999; Waugh et al. 1999; Karpetchko et al. 2005; Black and McDaniel 2007) have indicated a trend over the 1980s and 1990s toward a later vortex breakdown.”
http://www.columbia.edu/~lmp/paps/waugh+polvani-PlumbFestVolume-2010.pdf
justthefactswuwt says:
March 14, 2014 at 8:54 pm
Phil. says: March 14, 2014 at 7:28 am
I asked you “What do you think causes the water vapor, nitrogen oxide and methane “holes”?” and you did not respond. Can we take this as an admission that you cannot answer this question without contradicting your claim that “regardless of the dynamical situation without the chemical depletion there would be no ‘hole’!”?
Water vapor decreases in the stratosphere because of condensation, after all that’s how Brewer discovered the circulation that’s named after him. As it descends over the pole the extremely low temperature (-78ºC) causes even more condensation (PSCs), NO reacts to form nitric acid which also condenses in the PSCs it is also sequestered in the form of ClONO2. Again these processes are chemical in nature something you choose to ignore. Methane is depleted in the stratosphere by oxidation (with OH) and is a source of about a third of stratospheric water vapor.
“Clearly the actual data shows that the ozone concentration above the ‘hole’ is actually higher than the ozone concentration at the lower altitudes.”
We seem to be at an impasse here. When there is not a Polar Vortex in the Southern Hemisphere, does ozone concentration increase or decrease between 25 km and 50 km?
The data I provided from sondes shows that O3 increases between 50km and 25km then below 25km dramatically drops how does your mechanism explain that? The graph shows that above 25km the O3 profile is the same in July and Oct i.e. it is not effected by the presence of the vortex, it is below 25km where the changes occur!
And lest you think at this is only the case in Northern Hemisphere, and in the Southern Hemisphere magically, “ozone rich air falls below ~20km it encounters the Cl and ClO and the chemical kinetics takes over and that ozone is destroyed”, let us also look at the Southern Ozone “Hole”:
A bit disingenuous of you to omit the qualifier ‘relatively’ from the above quote! It might seem like ‘magic’ to someone like you who doesn’t understand chemical kinetics but to a chemical kineticist it’s elementary.
I looked at the data you provided, on that date the ‘hole’ is centered over the peninsula, at the various altitudes the data is as follows:
Altitude min O3
55km 2.5ppm
50 4.0
42 5.0
35 8.0
31 8.0
23 2.0!
So your data bears out the sonde data I cited, O3 increases as the air descends then dramatically drops below about 25km, which is due to chemical reactions.
Unfortunately, NOAA’s Ozone Mixing Archives;
http://www.cpc.ncep.noaa.gov/products/stratosphere/sbuv2to/archive/sh/
don’t go below 30 hPa/mb ~ 23 km, so we can’t see the magical “encounters” “below ~20km” where the “Cl and ClO and the chemical kinetics takes over and that ozone is destroyed”.
As shown above we see the start of that with a 4-fold drop in O3 concentration!
However, it is apparent that the air within the Polar Vortex “above ~20km” has low concentrations of ozone, thus your assertion “that relatively ozone rich air falls below ~20km” is falsified. Can you admit that you are wrong?
Actually your data confirms the data that I cited that above 20km the air has higher concentrations of O3 than below during the times of the spring ‘hole’. So will you now admit that you are wrong as shown by your own data?
“At about 31km the pO3 is ~3mPa, as the air descends to ~23km the pO3 increases to ~6mPa due to the photolysis reactions. As it descends over the next couple of km the pO3 rapidly drops to less than 1mPa, so clearly there is more O3 at 30km than at 20km not less as you incorrectly assert above.”
Now you are just playing games because you know you’re losing.
No, that is an accurate description of the sonde data and as shown above of the data you produced!
There is clearly less O2 with altitude. I never asserted that “is more O3 at 30km than at 20km”,
No I said that and was correct, I quoted you correctly, unfortunately the data shows you to be wrong, as the air descends the concentration of O3 increases until kinetics takes over below ~25km in the spring and it dramatically decreases.
I wrote that “that there is clearly less Ozone up at 30, 40 and 50 km”. You can play with the starting point, ~23 km, 20 km or ~ 18 km depending on day, season, vortex size, strength and penetration, but that doesn’t change the fact that there is ” is clearly less Ozone in the air up at 30, 40 and 50 km which “descends vertically through the center of the vortex, moving air to lower altitudes over several months.”
Clearly both sets of data show that this ‘fact’ is not true!
Furthermore, Ozone-Sonde don’t measure absolute Ozone concentration rather;
“ozone-sonde observations often use the ‘partial pressure’ of ozone as their unit.
Which is an ‘absolute’ measure, ppm is a ‘mixing ratio’ or ‘relative’ measure.
ozone-sonde observations using “‘partial pressure’ of ozone as their unit” can be misleading to the uninformed.
Perhaps, I work with them on a regular basis and don’t find them to be misleading, in any case the mixing ratio data you cite tells the same story so there should be no confusion.
A simple dynamical mechanism explains the observations, whereas the convoluted chemical mechanism you assert is falsified by the observations that the Ozone Hole extends well above the supposed “~20km” where you claim “relatively ozone rich air falls below” and “encounters the Cl and ClO and the chemical kinetics takes over and that ozone is destroyed.”
The observations indicate that as the air descends the concentration of O3 inside the vortex increases until it reaches ~25km whereupon it rapidly decreases, you have not explained that with your dynamical mechanism.
“Re-evaluating many total ozone series back to 1957 based on meta-information and calibration information” is the very definition of suspect data…
You claimed that you cited 5 references, now it’s just one which doesn’t demonstrate what you claim. It’s not ‘suspect’, it’s what good scientists do, you constantly check and recalibrate your apparatus and keep good records of all your readings and the calibration, and constantly re-evaluate procedures and data. That’s why I kept sources of analytical standard gas mixtures in my laboratory at significant expense.
Phil. says: March 15, 2014 at 7:26 am Figure 2.14: Ozone Concentration vs. Height (Ajavon, et al., 2007)- Click the pic to view at source[/caption]
Figure 2.14: Ozone Concentration vs. Height (Ajavon, et al., 2007)- Click the pic to view at source[/caption] Figure 2.14: Ozone Concentration vs. Height (Ajavon, et al., 2007)- Click the pic to view at source[/caption]
Figure 2.14: Ozone Concentration vs. Height (Ajavon, et al., 2007)- Click the pic to view at source[/caption] NOAA – National Weather Service – Climate Prediction Center[/caption]
NOAA – National Weather Service – Climate Prediction Center[/caption]
Water vapor decreases in the stratosphere because of condensation, after all that’s how Brewer discovered the circulation that’s named after him. As it descends over the pole the extremely low temperature (-78ºC) causes even more condensation (PSCs), NO reacts to form nitric acid which also condenses in the PSCs it is also sequestered in the form of ClONO2. Again these processes are chemical in nature something you choose to ignore. Methane is depleted in the stratosphere by oxidation (with OH) and is a source of about a third of stratospheric water vapor.
So you think that the Ozone “Hole”, Water Vapor “Hole”, Nitrogen Oxide “Hole” and Methane “Hole” all occur within the Polar Vortex solely due to condensation and chemical mechanisms? Excluding condensation and chemical mechanisms, do you think there would be any “Holes” within the low pressure area and descending cold air in the Polar Vortex?
The data I provided from sondes shows that O3 increases between 50km and 25km then below 25km dramatically drops how does your mechanism explain that? The graph shows that above 25km the O3 profile is the same in July and Oct i.e. it is not effected by the presence of the vortex, it is below 25km where the changes occur!
That’s when the Polar Vortex/Polar Night Jet moves below 25 km on its decent, i.e.:
“Throughout the winter, air from the upper stratosphere moves poleward and descends into the middle stratosphere. In the SH lower to middle stratosphere, strongest descent occurs near the edge of the polar vortex. The NH shows a similar pattern in late winter, but in early winter strongest descent is near the center of the vortex. Strong barriers to latitudinal mixing exist above =420 K throughout the winter. Below this, the polar night jet is weak in early winter, so air that descends below that level mixes between polar and middle latitudes. In late winter, parcels descend less, and the polar night jet moves downward, so there is less latitudinal mixing. The degree of mixing in the lower stratosphere thus depends strongly on the position and evolution of the polar night jet.”
“Diagnostics of mixing show a strong barrier to mixing at the edge of the polar vortex in the lower stratosphere, even into December in the SH, In the middle stratosphere, stratospheric warmings cause mixing to increase, and the vortex edge, as identified by a minimum in mixing, to move to higher PV values. This is consistent with previous studies showing the shrinking of the vortex and weakening of PV gradients during stratospheric warmings. As expected, considerably more mixing is seen in the NH than in the SH, In the lower stratosphere, in early winter the ,po]ar night jet usually does not extend significantly below =400 K, and parcels that descend below this level experience little barrier to mixing into mid-latitudes. As the winter progresses, the polar night jet moves downwards, and the parcels descend more slowly. Consistent with this, our simulations show considerably less mixing of polar air into mid-latitudes in late winter than in early winter in both hemispheres. In the presence of this polar night jet structure in the lower stratosphere and the sub-tropical jet in the upper troposphere, the degree of mixing of between polar and mid-latitude air in the lower stratosphere is strongly dependent on the amount of descent.”
http://trs-new.jpl.nasa.gov/dspace/bitstream/2014/33809/1/94-0540.pdf
Here is a simple view of the Ozone Layer;
[caption id="" align="alignnone" width="451"]
here is what happens to Column Ozone when the Polar Vortex descends into and through the Ozone Layer:
[caption id="" align="alignnone" width="451"]
and here is an animation of it occurring in 2013:
http://www.esrl.noaa.gov/gmd/dv/spo_oz/movies/index.html
The dynamical mechanism explains all of the observations and Occam takes over from there…
A bit disingenuous of you to omit the qualifier ‘relatively’ from the above quote!
This “relatively ozone rich air” that “falls below ~20km”, what is it relatively rich in relation to?
It might seem like ‘magic’ to someone like you who doesn’t understand chemical kinetics but to a chemical kineticist it’s elementary.
It’s magic, because there is simple dynamical mechanism to explain the observations and a complex, convoluted and unnecessary CFC based chemical mechanism that doesn’t appear to be borne out by the observations, as seen by the failure of the Montreal Protocol to put an end all of these “Holes”…
So your data bears out the sonde data I cited, O3 increases as the air descends then dramatically drops below about 25km, which is due to chemical reactions.
So now you’ve moved the “chemical reactions” goal posts up to “25km”? Is this because the observations don’t support your statement that “relatively ozone rich air falls below ~20km it encounters the Cl and ClO”? According to NOAA’s Earth System Research Laboratory, Global Monitoring Division:
“Antarctic ozone depletion occurs primarily between the altitudes of 12 and 20 kilometers, a region where polar stratospheric clouds, necessary for the chlorine-catalyzed chemical ozone destruction process, readily form.”
http://www.esrl.noaa.gov/gmd/dv/spo_oz/1220plot.html
So tell us, at what altitude does the “magic” occur?
Actually your data confirms the data that I cited that above 20km the air has higher concentrations of O3 than below during the times of the spring ‘hole’. So will you now admit that you are wrong as shown by your own data?
That doesn’t prove anything. This isn’t a simple linear process, it is a massive swirling Polar Vortex with anticyclones occurring around it, i.e.:
“Stratospheric anticyclones are present and interact with the polar vortex in a variety of situations. Stratospheric ‘‘surf zones,’’ for example, [McIntyre and Palmer, 1984] are generally not zonally symmetric in midlatitudes but result from the presence of one or more large-scale, quasi-stationary anticyclones [Fairlie and O’Neill, 1988]. Thus, in the discussion of planetary wave breaking (PWB) [McIntyre and Palmer, 1983], the presence and involvement of anti-cyclones needs to be emphasized [see also O’Neill and Pope, 1988; O’Neill et al., 1994]. This PWB process is efficient at irreversibly mixing air of different origins, however, the strongest mixing does not necessarily occur in the core of anticyclones. Regions of ‘‘chaotic advection’’ [Pierce and Fairlie, 1993] occur along the periphery of anticyclones. Stratospheric anticyclones also play a critical role in the following processes. They create high latitude nonlinear critical layers [Salby et al., 1990] along their central latitude. The ‘‘Kelvins cats eye’’ solution [Warn and Warn, 1978] is a closed anticyclonic circulation [O’Neill and Pope, 1988]. Cross-equatorial flow, visible in tracer fields [Randel et al., 1993; Chen et al., 1994], and the movement of material lines [Waugh, 1993], are associated with anticyclonic air with low isentropic potential vorticity (PV). Zonal harmonic waves 1 and 2 otherwise refer to 1 or 2 anticyclones present around a latitude circle. Anticyclones have long been regarded as important in the development of sudden stratospheric warmings (SSW) [e.g., Scherhag, 1952; Labitzke, 1977, 1981a, 1981b; McIntyre, 1982], a
phenomenon characterized by rapid warming and deceleration of the polar night jet (PNJ). In particular, ‘‘Canadian’’ type warmings are due to the intensification of the Aleutian High (hereafter AH) [Labitzke, 1977].
http://www.aos.wisc.edu/~mag/polarvortices.pdf
“A three-dimensional picture has been drawn of the final warming in the stratosphere. The breakup of the polar vortex is characterized by large fragments of polar air being stripped from the vortex and drawn out into increasingly narrow tongues in low latitudes. In the SH, this process is relatively slow, and these fragments may maintain their identity for weeks; in the NH, the final warming is dynamically induced, and is more rapid, with parcels from the mid-stratospheric polar vortex being spread throughout the hemisphere by early April in the year shown here.The large degree of interannual variability in the NH late winter suggests that many variations on this behavior might be observed during other years, For the period of UARS observations, the vortex in the lower stratosphere remains intact, and many of the air parcels confined within it, into December in the SH, and into April in the NH, well after the vortex has broken up in the mid-stratosphere.”
http://trs-new.jpl.nasa.gov/dspace/bitstream/2014/33809/1/94-0540.pdf
Thus ‘regions of ‘‘chaotic advection’’’ due to anti-cyclone interactions can occur and Polar Vortex strength and persistence can vary significantly from top to bottom, in fact “the vortex in the lower stratosphere” can remain “intact, and many of the air parcels confined within it” “well after the vortex has broken up in the mid-stratosphere.” The data you cite are well within the expected observations of the dynamical mechanism described.
No I said that and was correct, I quoted you correctly, unfortunately the data shows you to be wrong, as the air descends the concentration of O3 increases until kinetics takes over below ~25km in the spring and it dramatically decreases.
Firstly, you didn’t quote me, you wrote “so clearly there is more O3 at 30km than at 20km not less as you incorrectly assert above.”, which is not a “quote” but rather an intentional misstatement. I wrote that “that there is clearly less Ozone up at 30, 40 and 50 km”, and my statement is correct. You just want to get bogged down in the minutiae…
Perhaps, I work with them on a regular basis and don’t find them to be misleading, in any case the mixing ratio data you cite tells the same story so there should be no confusion.
Then you should appreciate why the observations support a dynamical mechanism.
The observations indicate that as the air descends the concentration of O3 inside the vortex increases until it reaches ~25km whereupon it rapidly decreases, you have not explained that with your dynamical mechanism.
Yes, I have, i.e. Vortex decent below ~25km causes O3 to rapidly decrease, as the low pressure and relatively low ozone in the air descending within the polar vortex create an area of low Ozone concentration within the Ozone layer. And thus an Ozone “Hole” occurs, with no need for a chemical mechanism.
You still have not explained why there is an Ozone “Hole” at 2 hPa/mb – ~42 km;
[caption id="" align="alignnone" width="450"]
above the “~20km” or “25km” where you claim “relatively ozone rich air falls below” and “encounters the Cl and ClO and the chemical kinetics takes over and that ozone is destroyed.”
If air is descending within the Polar Vortex, and “the chemical kinetics” due to “encounters the Cl and ClO” occur at “~20km” or now “25km”, why is there an Ozone “Hole” at ~42 km?
You claimed that you cited 5 references, now it’s just one which doesn’t demonstrate what you claim.
More of the minutiae, I cited 1 reference on the quality of the BAS data and 4 references on the quality of TOMS data. You are just trying to waste time, try focusing on the substance of the debate that you are losing.
It’s not ‘suspect’, it’s what good scientists do, you constantly check and recalibrate your apparatus and keep good records of all your readings and the calibration, and constantly re-evaluate procedures and data. That’s why I kept sources of analytical standard gas mixtures in my laboratory at significant expense.
I am all for checking and recalibrating your apparatus, keeping “good records of all your readings and the calibration”, and constantly re-evaluate procedures and data.” However, what they did was “Re-evaluating many total ozone series back to 1957.” Re-evaluating old measurements sounds a lot like the Bucket Model games that were played with HadSST3, HADISST and ERSST.v3b;
http://wattsupwiththat.com/2013/05/25/historical-sea-surface-temperature-adjustmentscorrections-aka-the-bucket-model/
and it is definitely suspect.
justthefactswuwt says:
March 16, 2014 at 1:11 am
Phil. says: March 15, 2014 at 7:26 am
So you think that the Ozone “Hole”, Water Vapor “Hole”, Nitrogen Oxide “Hole” and Methane “Hole” all occur within the Polar Vortex solely due to condensation and chemical mechanisms? Excluding condensation and chemical mechanisms, do you think there would be any “Holes” within the low pressure area and descending cold air in the Polar Vortex?
Correct, those ‘holes’ as you describe them all involve reactive and condensible species.
Firstly, you didn’t quote me, you wrote “so clearly there is more O3 at 30km than at 20km not less as you incorrectly assert above.”, which is not a “quote” but rather an intentional misstatement. I wrote that “that there is clearly less Ozone up at 30, 40 and 50 km”, and my statement is correct. You just want to get bogged down in the minutiae…
The ‘minutiae’ you refer to is the actual data which you chose to ignore! Below is the actual quote that I referred to.
Your graph demonstrates my point quite well, thank you.
“Clearly the actual data shows that the ozone concentration above the ‘hole’ is actually higher than the ozone concentration at the lower altitudes. As that relatively ozone rich air falls below ~20km it encounters the Cl and ClO and the chemical kinetics takes over and that ozone is destroyed.”
What are you looking at? Both of the graphs show that there is clearly less Ozone up at 30, 40 and 50 km, which is the “air from very high altitudes” that “descends vertically through the center of the vortex, moving air to lower altitudes over several months.”
Your statement clearly is wrong, here is that data you were looking at since you appear to have difficulty with graphs:
Alt O3
40 no data
30 ~3 mPa
25 ~5
23 ~7
20 ~1
17 ~0 (in July ~15)
15 ~1 (~15)
12 ~4 (~4)
10 ~4
So the “air from very high altitudes” that “descends vertically through the center of the vortex, moving air to lower altitudes over several months”, shows a gradually increasing level of O3 due to photolysis and chemical reaction. Between 23km where it peaks and 20km the O3 concentration drops to about zero and stays that way until you get below 15km (the range where PSCs are formed). In what way is there clearly less Ozone up at 30km than in the ‘hole’?
The data you provided goes to higher altitudes:
Altitude min O3
55km 2.5ppm
50 4.0
42 5.0
35 8.0
31 8.0
23 2.0!
It shows the same effect, there is more O3 at 50, 40 and 30 km than there is at 23km.
“The observations indicate that as the air descends the concentration of O3 inside the vortex increases until it reaches ~25km whereupon it rapidly decreases, you have not explained that with your dynamical mechanism.”
Yes, I have, i.e. Vortex decent below ~25km causes O3 to rapidly decrease, as the low pressure and relatively low ozone in the air descending within the polar vortex create an area of low Ozone concentration within the Ozone layer. And thus an Ozone “Hole” occurs, with no need for a chemical mechanism.
How on earth does this describe what happens? At 31km this descending air has 8ppm and then it drops to 2.0 by 23km, how if there’s no chemistry, by your dynamical mechanism how does that happen? You are the one who is relying on magic, you’re saying that air with 8ppm of O3 descends a few km and then 3/4 of the O3 just disappears! Where does it go?
You still have not explained why there is an Ozone “Hole” at 2 hPa/mb – ~42 km;
above the “~20km” or “25km” where you claim “relatively ozone rich air falls below” and “encounters the Cl and ClO and the chemical kinetics takes over and that ozone is destroyed.”
Now I see your problem, you don’t know what the ozone hole is! At 42km you see the descending air per Brewer-Dobson, that is normal. In the summer and fall that descending air gradually increases to ~16ppm between ~20 and 15km, that is ‘normal’. In the spring that peak drops to about zero due to the photolysis reactions, that is the Ozone hole! In the 1999 data I cited the data above 23km is unchanged between July and October and July has a Dobson number of 255, in October the Dobson number drops to 111, entirely due to the changes between 23 and 10km, that is the ‘hole’, it always refers to the changes in the lower stratosphere not the upper stratosphere. The fact that it is defined by the Dobson number appears to have confused you, that defines the extent of the ‘hole’, but the ‘hole’ itself is the loss of ozone in the lower stratosphere.
Phil. says: March 16, 2014 at 6:43 am NOAA – National Weather Service – Climate Prediction Center[/caption]
NOAA – National Weather Service – Climate Prediction Center[/caption]
Correct, those ‘holes’ as you describe them all involve reactive and condensible species.
I don’t necessarily disagree that they “all involve reactive and condensible species.” The question you did not answer, is “excluding condensation and chemical mechanisms, do you think there would be any “Holes” within the low pressure area and descending cold air in the Polar Vortex?”
So the “air from very high altitudes” that “descends vertically through the center of the vortex, moving air to lower altitudes over several months”, shows a gradually increasing level of O3 due to photolysis and chemical reaction. Between 23km where it peaks and 20km the O3 concentration drops to about zero and stays that way until you get below 15km (the range where PSCs are formed). In what way is there clearly less Ozone up at 30km than in the ‘hole’?
You seem to be confused by the data from one day and at very specific altitudes, versus seeing the bigger picture. I will offer two very simple statements with supporting data:
Ozone concentrations tend to decrease with height starting at approximately ~23 km:
[caption id="" align="alignnone" width="450"]
The Polar Vortex transports air from higher in the atmosphere to lower in the atmosphere:
“In the NH vortex, air parcels which were initialized at 18 km on November 1, descended about 6 km by March 21, while air initially at 25 km descended 9 km in the same time period. This represents an average descent rate in the lower stratosphere of 1.3 to 2 km per month. Air initialized at 50 km descended 27 km between November 1 and March 21.
In the SH vortex, parcels initialized at 18 km on March 1, descended 3 km, while air at 25 km descended 5–7 km by the end of October. This is equivalent to an average descent in the lower stratosphere of 0.4 to 0.9 km per month during this 8-month period. Air initialized at 52 km descended 26–29 km between March 1 and October 31. In both the NH and the SH, computed descent rates increased markedly with height. The descent for the NH winter of 1992–1993 and the SH winter of 1992 computed with a three-dimensional trajectory model using the same radiation code was within 1 to 2 km of that calculated by the one-dimensional model, thus validating the vortex averaging procedure. The computed descent rates generally agree well with observations of long-lived tracers, thus validating the radiative transfer model.”
https://earthref.org/ERR/59278/
As a result, one would expect to see an Ozone “Hole” below ~23, because air parcels with lower concentrations of Ozone are descended below this point within the Polar Vortex. Do you agree or disagree?
The data you provided goes to higher altitudes:
Altitude min O3
55km 2.5ppm
50 4.0
42 5.0
35 8.0
31 8.0
23 2.0!
It shows the same effect, there is more O3 at 50, 40 and 30 km than there is at 23km.
Again, this is one day and is occurs above “the altitudes of 12 and 20 kilometers” where “Antarctic ozone depletion occurs primarily”.
http://www.esrl.noaa.gov/gmd/dv/spo_oz/1220plot.html
Do you think NOAA is wrong about the height where “Antarctic ozone depletion occurs”? Furthermore, using the data I provided above and you cited, Ozone at ~55 km 2.5ppm, “air initialized at 52 km descended 26–29 km between March 1 and October 31”, Ozone at ~ 23 km “2.0!”, magic how that happens…
How on earth does this describe what happens? At 31km this descending air has 8ppm and then it drops to 2.0 by 23km, how if there’s no chemistry, by your dynamical mechanism how does that happen? You are the one who is relying on magic, you’re saying that air with 8ppm of O3 descends a few km and then 3/4 of the O3 just disappears! Where does it go?
These are parcels of air within a swirling and sometimes displaced, perturbed and split polar vortex, descending over many months. It is a chaotic dynamical system that is constantly in motion. You seem to be looking a single day and assuming that presents the whole picture.
At 42km you see the descending air per Brewer-Dobson, that is normal. In the summer and fall that descending air gradually increases to ~16ppm between ~20 and 15km, that is ‘normal’.
Great, now we are making headway, you accept that there is a naturally occurring Ozone “Hole” within the Polar Vortex at ~42 km. Now tell us, at what altitude does this natural Ozone “Hole” become an unnatural Ozone “Hole”?
in October the Dobson number drops to 111, entirely due to the changes between 23 and 10km, that is the ‘hole’
Yes, this is explained by the Polar Vortex forming and air parcels descending 10s of kilometers within it. What part of the concept are you struggling with?
The fact that it is defined by the Dobson number appears to have confused you, that defines the extent of the ‘hole’
Not confused, more amused. The mainstream definition of the Ozone “Hole” is arbitrary and laughable i.e.:
“A ‘hole’ is arbitrarily defined as an area where the volume of ozone is less than 220 Dobson units, a decrease of about one-third of the normal value”:
http://books.google.com/books?id=I-Br1CEx8fcC&pg=PA181&lpg=PA181&dq=220+dobson+arbitrary+ozone&source=bl&ots=Q4I0b1Cr0D&sig=laqwocak0P_l2xyb6ZotyUMonZw&hl=en&sa=X&ei=y3EmU93WIaOf0AHekoGQCA&ved=0CDoQ6AEwAw
“We noted that the ozone hole is not actually a hole but a region of heavily depleted ozone in the atmosphere this is defined, slightly arbitrarily, as region where the total ozone column (TOC) is less than 220 DU”
http://books.google.com/books?id=YZzGFPnaEv0C&pg=PA406&lpg=PA406&dq=220+dobson+arbitrary+ozone&source=bl&ots=k2RAuouiqu&sig=Nr_-3cXmDDTvxNCAUiRdKWjUGb0&hl=en&sa=X&ei=y3EmU93WIaOf0AHekoGQCA&ved=0CFMQ6AEwBw#v=onepage&q=220%20dobson%20arbitrary%20ozone&f=false
“The value of 220 Dobson Units is chosen since total ozone values of less than 220 Dobson Units were not found in the historic observations over Antarctica prior to 1979.”
http://earth.rice.edu/earthupdate/atmosphere/topics/ozone/topic_01.html
As I pointed out above the “historic observations over Antarctica prior to 1979” were limited to a single station on the coast and the data has since been “re-evaluated”. What a joke…
but the ‘hole’ itself is the loss of ozone in the lower stratosphere.
You haven’t demonstrated any “loss”, whereas I have clearly demonstrated the dynamical mechanisms of decent and low pressure. Excluding chemical mechanisms, what do you think the value in Dobson Units of total column ozone would be due to the naturally occurring Ozone “Hole” within the Polar Vortex?