Rate May Be Ten Times Faster, According to New Data

From Lamont-Doherty Earth Observatory: Some 56 million years ago, a massive pulse of carbon dioxide into the atmosphere sent global temperatures soaring. In the oceans, carbonate sediments dissolved, some organisms went extinct and others evolved.
Scientists have long suspected that ocean acidification caused the crisis—similar to today, as manmade CO2 combines with seawater to change its chemistry. Now, for the first time, scientists have quantified the extent of surface acidification from those ancient days, and the news is not good: the oceans are on track to acidify at least as much as they did then, only at a much faster rate.
In a study published in the latest issue of Paleoceanography, the scientists estimate that ocean acidity increased by about 100 percent in a few thousand years or more, and stayed that way for the next 70,000 years. In this radically changed environment, some creatures died out while others adapted and evolved. The study is the first to use the chemical composition of fossils to reconstruct surface ocean acidity at the Paleocene-Eocene Thermal Maximum (PETM), a period of intense warming on land and throughout the oceans due to high CO2.
“This could be the closest geological analog to modern ocean acidification,” said study coauthor Bärbel Hönisch, a paleoceanographer at Columbia University’s Lamont-Doherty Earth Observatory. “As massive as it was, it still happened about 10 times more slowly than what we are doing today.”
The oceans have absorbed about a third of the carbon humans have pumped into the air since industrialization, helping to keep earth’s thermostat lower than it would be otherwise. But that uptake of carbon has come at a price. Chemical reactions caused by that excess CO2 have made seawater grow more acidic, depleting it of the carbonate ions that corals, mollusks and calcifying plankton need to build their shells and skeletons.

In the last 150 years or so, the pH of the oceans has dropped substantially, from 8.2 to 8.1–equivalent to a 25 percent increase in acidity. By the end of the century, ocean pH is projected to fall another 0.3 pH units, to 7.8. While the researchers found a comparable pH drop during the PETM–0.3 units–the shift happened over a few thousand years.
“We are dumping carbon in the atmosphere and ocean at a much higher rate today—within centuries,” said study coauthor Richard Zeebe, a paleoceanographer at the University of Hawaii. “If we continue on the emissions path we are on right now, acidification of the surface ocean will be way more dramatic than during the PETM.”
The study confirms that the acidified conditions lasted for 70,000 years or more, consistent with previous model-based estimates. “It didn’t bounce back right away,” said Timothy Bralower, a researcher at Penn State who was not involved in the study. “It took tens of thousands of years to recover.”
From seafloor sediments drilled off Japan, the researchers analyzed the shells of plankton that lived at the surface of the ocean during the PETM. Two different methods for measuring ocean chemistry at the time—the ratio of boron isotopes in their shells, and the amount of boron –arrived at similar estimates of acidification. “It’s really showing us clear evidence of a change in pH for the first time,” said Bralower.
What caused the burst of carbon at the PETM is still unclear. One popular explanation is that an overall warming trend may have sent a pulse of methane from the seafloor into the air, setting off events that released more earth-warming gases into the air and oceans. Up to half of the tiny animals that live in mud on the seafloor—benthic foraminifera—died out during the PETM, possibly along with life further up the food chain.
Other species thrived in this changed environment and new ones evolved. In the oceans, dinoflagellates extended their range from the tropics to the Arctic, while on land, hoofed animals and primates appeared for the first time. Eventually, the oceans and atmosphere recovered as elements from eroded rocks washed into the sea and neutralized the acid.
Today, signs are already emerging that some marine life may be in trouble. In a recent study led by Nina Bednaršedk at the U.S. National Oceanic and Atmospheric Administration, more than half of the tiny planktic snails, or pteropods, that she and her team studied off the coast of Washington, Oregon and California showed badly dissolved shells. Ocean acidification has been linked to the widespread death of baby oysters off Washington and Oregon since 2005, and may also pose a threat to coral reefs, which are under additional pressure from pollution and warming ocean temperatures.
“Seawater carbonate chemistry is complex but the mechanism underlying ocean acidification is very simple,” said study lead author Donald Penman, a graduate student at University of California at Santa Cruz. “We can make accurate predictions about how carbonate chemistry will respond to increasing carbon dioxide levels. The real unknown is how individual organisms will respond and how that cascades through ecosystems.”
Other authors of the study, which was funded by the U.S. National Science Foundation: Ellen Thomas, Yale University; and James Zachos, UC Santa Cruz.
Phil
16K increase? where has that happened? Must have been sleeping and missed it
It is evident that the authors of this study have not come to grips with cause and effect relationships quite yet.
In a Limnology course I took in college we found out how difficult it was to determine the actual pH of seawater. Starting with DI-H2O we measured the pH and got a value. After bubbling N2 through the water the pH changed slightly (removal of residual CO2?). Adding NaCl to the concentration of sea water we got a different result. Measure actual sea water and get a different result, again. Filter the sea water with a 0.2 um membrane and get another pH. Change the temperature and get yet another pH result. Seawater that was kept in the dark vs. kept in the light? Yep…different pH results. Probably relates to seawater that was taken at 25 feet below the surface to seawater taken at the surface…different pH’s, again. Seawater taken from a ‘calm’ sea surface vs. an ‘active’ sea surface? Yep different pH’s.
Not to say that with extreme sampling control you can’t get reliable figures, but it takes some doing and to get equivalence to anyone else measurements to be comparable.
Phil
Thanks for that info. Now tell me what that means in regard to pH. F@ck all
Fresh water dissolves very little CO2, because it has no buffer capacity at all and therefore is slightly acidic.
===
Ferd, he was talking about fresh water ‘systems’…..
ex… Lake Malawi ph range from 7.7 to 8.6, and for Lake Tanganyika pH from 7.3 to 8.8, etc
JJ, too. says:
June 4, 2014 at 8:14 am
I couldn’t agree more.
I worked in labs or with labs for over 20 years. Unlike some of the ‘experts’ on this site
A few years ago a study appeared in PNAS, in which the effect of seawater pH on intracellular pH of coral was investigated. When the investigators decreased the pH of seawater from 8.0 to 7.4, the coral maintained its intracellular pH at 7.3 (note: also at pH 8.0, the intracellular pH was 7.3). When the seawater pH was brought to 7.2, the intracellular pH dropped from 7.3 to 7.2. They also investigated the effect on the pH of the mineralization zone and found that a decrease in pH from 8.0 to 7.4 caused a pH-drop in the mineralization zone from 8.3 to approx 8.0.
And the effect on mineralization? Well, not very large: in their study the authors found NO difference in extent of calcification between corals in seawater of pH 8.0 and 7.4!
kadaka (KD Knoebel) says:
June 4, 2014 at 7:08 am
From John Eggert on June 4, 2014 at 5:58 am:
kadaka: The estimation of pH using DIC and TA at a particular temperature, is a simple exercise in equilibrium chemistry. The estimation of DIC and TA is a complicated exercise in sampling with potential for substantial errors. (…)
I know, and I noticed there are nice equations for calculating them using pH. Which is easily read off a meter.
Not with the required precision and accuracy it isn’t, which is why research facilities use either the method referred to by Nick, using DIC and TA, or spectrophotometric methods.
Which was the issue. There was a simple question about measuring a quantity with accuracy, and Nicky wanted to show off by whipping out his big shiny throbbing intellect instead, with a complicated response about easily (for a chem major) deducing it.
For best accuracy and precision you use spectrophotometric methods, (±0.001 or better) not electrodes.
Why he keeps harping on some obsolete method kept around as an academic exercise when the modern faster way is long established, well, maybe he just likes telling passerby about his favored elaborate routine for keeping his intellect stimulated.
It’s you who’s ‘harping on some obsolete method’, incidentally pH is not measured by any method, it is calculated from measurements of other quantities, the ion selective probe that you are talking about measures a voltage which depends on [H+] via the Nernst equation and is calibrated in terms of the pH.
Sorry Phil
pH is measured by sticking a probe into a solution. We actually used a meter and not some esoteric calculation in ‘production control’. It would have cost thousands of dollars, otherwise.
This has developed into a really stupid discussion. I have managed to insult some people that I really didn’t mean to insult. Good night all
“suspected…” “…could be…” “CO2 ha[s] made seawater grow more acidic…”
No.
According to real world data from the Monterey Bay Aquarium’s intake pipe, located far out in the ocean, there has been no measurable change in pH.
Granted, this data stops several years ago, but it covers a time when CO2 was strongly ramping up.
The article also omits any mention of buffering — a major factor in any discussion of ocean pH; probably the major factor. Why no mention of buffering?
This is just the next in a long series of wild-eyed scares from the same people who have gotten every alarmist prediction wrong. No exceptions. They have been wrong about everything, from Polar bears, to accelerating sea level rise, to increased extreme weather events, to global warming, to vanishing Arctic ice, etc. They have been wrong about everything.
When one group makes constant predictions, and every one of them turns out to be flat wrong, rational people will at least say, “Hold your horses! We need to study this more — a lot more — before tilting at this latest grant-fed windmill.”
Alex says:
June 4, 2014 at 8:48 am
Sorry Phil
pH is measured by sticking a probe into a solution. We actually used a meter and not some esoteric calculation in ‘production control’. It would have cost thousands of dollars, otherwise.
I’m sure you do and I’m sure it’s sufficiently accurate for your purposes, however for measuring changing pH of ocean water that is inadequate, which is why the methods Nick and I described are necessary. By the way just because you use a meter doesn’t mean that there’s not an esoteric calculation embedded within the calibration.
The spectrophotometric method does cost thousands of dollars which is one reason why there is a prize awarded for the creation of “pH sensor technology that will affordably, accurately and efficiently measure ocean chemistry”. Perhaps you should enter your method, should be an easy way to pick up a couple of million$.
http://oceanhealth.xprize.org/competition-details/overview
“JJ, too. says:
June 4, 2014 at 8:14 am ”
What you are saying is that there is no “normal” value with which we can derive an anomaly (A diviation from “normal”. Thusmeaning, IMO, we simply do not know what is the “normal” pH of the oceans is).
Some 56 million years ago, a massive pulse of carbon dioxide into the atmosphere sent global temperatures soaring. In the oceans, carbonate sediments dissolved, some organisms went extinct and others evolved.
Might be the old case of wrong cause / wronge case. A quick search on lava flows 56 million years ago pops up.
The North Atlantic Igneous Province (NAIP) is one of the largest such on earth and extends from Baffin Island and Greenland northwards into the Arctic, east across to Norway and southwards down to Denmark, Scotland and Northern Ireland.
Outpourings of its magma created the Scottish islands of Skye, Rhum, Eigg, Canna and the basalt columns of the Giant’s Causeway and Fingle’s Cave.
Flood basalts from this time are still widely exposed on the Faroe Islands, Greenland and Baffin Island whilst Iceland remains a volcanically active ‘hot spot’ to this day.
It is known that the NAIP was particularly active at two periods in the deep past; the first phase was between 62 – 58 million years ago, with a second phase at the time of the PETM, between 56 – 54 million years ago when the area began to be uplifted; the continental plate split apart and emitted large volumes of magma.
https://sites.google.com/site/thepaleoceneeocenethermalmaxim/5-molten-magma-the-north-atlantic-igneous-province
Or
During the birth of Mount McKinley 56 million years ago when molten magma solidified deep beneath central Alaska, volcanic activity (eruptions at the surface) was also occurring in the park, and produced red, yellow and brown basalts, rhyolites, and other volcanic rocks.
http://vulcan.wr.usgs.gov/LivingWith/VolcanicPast/Places/volcanic_past_alaska.html
How many tons of lava at 1500 F does it take to cover the north Atlantic and would this tend to heat things up to a greater or lesser extent than CO2.
rogerknights says:
The problem with the “25% more acidic” statement is that it implies that acidification has progressed 25% of the way to neutrality, which isn’t so.
Repeated for effect.
What caused the burst of carbon at the PETM is still unclear.
Probably the Kochasaurus.
In the last 150 years or so, the pH of the oceans has dropped substantially, from 8.2 to 8.1
What are the local highs and lows in pH? I find it hard to believe this matters beyond the margins. Also, what kind of measurement precision is that? The change is the same as the smallest increment, which suggests the error bound is 100% of the signal.
I did indeed, sorry. In a hurry to go teach. The answer to the second question is that many physical quantities are best understood in terms not of absolute values, but rather the log of the absolute values. Sound intensity, for example. Humans are exposed daily to sounds that range over at least ten to twelve orders of magnitude in intensity, with somewhat rarer exposures to another six or seven orders of magnitude (before the sounds become intense enough to not count as sounds any more, but as shock wave fronts that are likely to certain to be fatal). The human ear, amazingly, can render all of 16 or 17 orders of magnitude as perceptible sound, from the faintest sound we can hear to jet engines or guns being fired nearby. Somewhere between a 30-06 being fired near our heads and a nearby object generating a substantial sonic boom, sound stops being “sound” to our ears (and incidentally causes instant damage to our hearing) and become a blast wave from e.g. an explosion.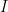 and one at intensity
and one at intensity 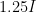 , you almost certainly could not differentiate the louder sound from the fainter one — that is an approximately 1 decibel increase, where sound intensity is rended into a log scale sound level relative to a reference intensity
, you almost certainly could not differentiate the louder sound from the fainter one — that is an approximately 1 decibel increase, where sound intensity is rended into a log scale sound level relative to a reference intensity 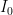 , the “threshold of hearing”, at
, the “threshold of hearing”, at  Watts/meter^2. In contrast, music at a rock concert 30 or 40 meters from the stage can easily be 1 Watt/meter^2! We can hear both easily as sound, the latter sound that is likely slowly damaging our hearing.
Watts/meter^2. In contrast, music at a rock concert 30 or 40 meters from the stage can easily be 1 Watt/meter^2! We can hear both easily as sound, the latter sound that is likely slowly damaging our hearing. etc, but that too involves a lot of nearly pointless writing. It is far easier to form $-\log_{10}(P_{H+})$, or more properly as $-\log_{10}(a_{H+})$ where
etc, but that too involves a lot of nearly pointless writing. It is far easier to form $-\log_{10}(P_{H+})$, or more properly as $-\log_{10}(a_{H+})$ where  is the activity of the hydrogen ion in a solution. The activity is defined relative to the chemical potential of a “standard” electrode, which can be directly mapped to a voltage generated on the electrode and measured by measuring the electrode voltage. This activity does directly involve
is the activity of the hydrogen ion in a solution. The activity is defined relative to the chemical potential of a “standard” electrode, which can be directly mapped to a voltage generated on the electrode and measured by measuring the electrode voltage. This activity does directly involve 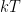 (or
(or  , if you are a chemist instead of a physicist) so one has to know the temperature at the electrodes to get anything like a precise measurement. I’m certain that there is other chemistry that can confound it, as well — the presence of other electrolytes or contaminants in the water, perhaps.
, if you are a chemist instead of a physicist) so one has to know the temperature at the electrodes to get anything like a precise measurement. I’m certain that there is other chemistry that can confound it, as well — the presence of other electrolytes or contaminants in the water, perhaps.
The ear is almost completely insensitive to much less than a doubling of sound intensity. If I presented you with two sounds one at intensity
pH is precisely such a quantity. The concentration of hydrogen ions in a water-based solution can vary by 14 orders of magnitude (although it is moderately difficult to reach the extreme ends of this range chemically). Sure, one can write it out with all of those zeros as you did above, but that is silly. One can use scientific notation, and write it as
Lots of other things vary by many orders of magnitude and are best described on a log scale in e.g. decibels rather than in absolute terms. Enough so that engineers (and physicists, and I’m sure chemists) have some simple rules of thumb. Doubling a quantity is an increase of 3 dB (decibels). Increasing it by an order of magnitude adds 10 dB. Halving a quantity subtracts 3 dB. The true beauty of log scales is that multiplication maps into addition — we don’t care what the original base is, doubling e.g. any sound intensity will increase its sound level in decibels by 3. Doubling the hydrogen ion activity in any solution will decrease the pH (from whatever original value it had) by 0.3.
rgb
The problem with the “25% more acidic” statement is that it implies that acidification has progressed 25% of the way to neutrality, which isn’t so.
Repeated for effect.
And I actually agree. This is very misleading. Acidity is generally measured, given, discussed in pH, and it makes this sound like a big change, rather than a tiny one (with various possible sources of error, which may or may not be significant).
rgb
Acidity is the quantitative measure of how much hydroxide ion can be titrated. Those who claim that the acidity of sea water has changed by 25% are incorrectly ignoring the presence of bicarbonate and other “acidic” ions that can neutralize OH-. Assuming that the models are correct, acidity has increased by a few percent.
Robert Brown says: June 4, 2014 at 10:58 am
“Lots of other things vary by many orders of magnitude and are best described on a log scale in e.g. decibels rather than in absolute terms.”
Actually, there is a more cogent reason here. The law of mass action, which provides equilibrium relations, is multiplicative. The conservation laws are additive, which often ends up meaning that when one concentration changes, equilibrium is maintained by changing just one other, and that changes in ratio. So here (in sea), for example, it’s a rough rule that a 25% rise in H+ requires a 25% rise in [H2CO3], even tho the latter is in much higher concentration. That’s how buffering works.
There is a typo in the first sentence.
“Some 56 million years ago, a massive pulse of carbon dioxide into the atmosphere sent global temperatures soaring.”
should read
“Some 56 million years ago, a massive pulse of global temperature sent carbon dioxide soaring.”
Ancient Koine greek has four “ifs”: if and it is, if and it isn’t, if and it might be or might not be, and if – I wish it was but it is not.
IF (Koine type 4) the scientific community really wanted to know whether CO2 in the atmosphere was “acidifying” the ocean or not, there is a straightforward way to find out.
Deep ocean water has been separate from the surface for years to centuries while surface water is in contact with air. Compare CO2 levels in ocean bottom water to surface water. What’s the problem?
How do universities get away with making press releases with such sensationalized garbage, half-truths, and flat-out lies? Papers I’ve read studying the PETM found that temperatures began to rise 60 m.a., 4 million years before the CO2 spike, and during this increase in temperature CO2 levels actually were falling. This early temperature rise was also about 3 times higher than the temperature rise following the CO2 spike.
richardscourtney says: June 4, 2014 at 6:48 am
“Really? Why then did the many not object when e.g. Al Gore did make that attribution in his sci-fi movie?”
The objection was made. It was item 4 on Judge Burton’s list of nine. The New Scientist blog commented:
“Historically, global warming events at the end of ice ages have not been triggered by rises in atmospheric CO2 concentrations. However, as explained in “Climate myths: Ice cores show CO2 increases lag behind temperature rises, disproving the link to global warming”, this does not disprove that CO2 warms the atmosphere and that rising CO2 emissions have cause warming since the 20th century.”
That’s the view of the non-few.
“And if CO2 did not cause the temperature rises in the PETM and at the other times then what was the cause of those temperature rises?
Importantly, if CO2 did not cause those temperature rises in the past then how and why is “burning fossil fuel” doing it “in modern times”?”
The cause of PETM etc warming is a research topic. I don’t know the answers. But for CO2 to cause warming, something has to be forcing CO2 into the atmosphere. In past times, there is mostly nothing obvious to do that. Now there is. It’s us.
John Eggert says: June 4, 2014 at 6:29 am
“Nick: The entire point of the paper is that pH has changed. If you don’t know how extensive the readings were 100 years ago, you too agree that there is no basis for saying the pH has changed.”
No, I’m just saying that I don’t know. I could find out. So could others here.
“And wikipedia for a source? “
Yes. It’s easily linked, and for matters like carbonate chemistry, it is fine. Did you find any errors?
Robert Brown says:
June 4, 2014 at 10:58 am
pH is precisely such a quantity. The concentration of hydrogen ions in a water-based solution can vary by 14 orders of magnitude (although it is moderately difficult to reach the extreme ends of this range chemically). Sure, one can write it out with all of those zeros as you did above, but that is silly. One can use scientific notation, and write it as 10^{-3}, 10^{-4}, 7.432 \times 10^{-5} … etc, but that too involves a lot of nearly pointless writing. It is far easier to form $-\log_{10}(P_{H+})$, or more properly as $-\log_{10}(a_{H+})$ where a_{H+} is the activity of the hydrogen ion in a solution. The activity is defined relative to the chemical potential of a “standard” electrode, which can be directly mapped to a voltage generated on the electrode and measured by measuring the electrode voltage. This activity does directly involve kT (or RT, if you are a chemist instead of a physicist) so one has to know the temperature at the electrodes to get anything like a precise measurement. I’m certain that there is other chemistry that can confound it, as well — the presence of other electrolytes or contaminants in the water, perhaps.
The pH notation is convenient for use with an electrode because the Nernst equation is linearly dependent on log[H+] so it’s very convenient to calibrate in terms of pH. Other methods such as titration are linearly dependent on [H+] so if you want to know how much acid it would take to neutralize a solution pH isn’t what you want. Back in the early 20th century when the pH scale was first devised, there were no computers or calculators so the log notation was a very convenient shorthand to describe [H+] and rendered the arithmetic simpler as Robert describes.
Lots of other things vary by many orders of magnitude and are best described on a log scale in e.g. decibels rather than in absolute terms. Enough so that engineers (and physicists, and I’m sure chemists) have some simple rules of thumb. Doubling a quantity is an increase of 3 dB (decibels). Increasing it by an order of magnitude adds 10 dB. Halving a quantity subtracts 3 dB. The true beauty of log scales is that multiplication maps into addition — we don’t care what the original base is, doubling e.g. any sound intensity will increase its sound level in decibels by 3. Doubling the hydrogen ion activity in any solution will decrease the pH (from whatever original value it had) by 0.3.
Yes so calculating pOH from pH in aqueous solution becomes pOH=14-pH for example.
rgbatduke says:
June 4, 2014 at 11:01 am
“The problem with the “25% more acidic” statement is that it implies that acidification has progressed 25% of the way to neutrality, which isn’t so.”
Repeated for effect.
And I actually agree. This is very misleading. Acidity is generally measured, given, discussed in pH, and it makes this sound like a big change, rather than a tiny one (with various possible sources of error, which may or may not be significant).
Not to anyone who understands chemistry it doesn’t. For example, blood pH is normally between 7.35 and 7.45, which makes it sound like a very narrow range whereas in fact [H+] varies by about 25%. A sustained drop in blood pH of 0.1 in pH doesn’t sound like much but is likely to prove fatal, saying that the Hydrogen ion concentration was 25% below the normal range would be more likely to convey the necessary degree of urgency!
If you want to evaluate the solubility product of aragonite for example:
it’s Ksp=[Ca++] *[CO3–]
but [CO3–] is given by DIC/(1+[H+]/K2 +[H+]^2/K1K2) (where logK1=-6.39 and logK2=-10.39
so the change in absolute [H+] is what’s relevant not the change in pH.
So in the context of dissolving shells it’s the relative change in [H+] that counts not the change in pH.