Guest post by Reed Coray
The following example illustrates the issues I have with reasoning often used to argue that increasing the amount of CO2 in the Earth’s atmosphere will increase both the Earth’s surface temperature and the Earth’s atmosphere temperature. Immediately following is a direct quote from URL
http://www.school-for-champions.com/science/heat_transfer_earth.htm
“The present situation is that there has been an increase in infrared-absorbing gases in the atmosphere, such as carbon dioxide (CO2) and methane (CH4). Energy that would normally escape into space is absorbed by these molecules, thus heating the atmosphere and spreading through convection currents. The average temperature of the atmosphere has increased 0.25 °C since 1980, mainly attributed to an increase in infrared-absorbing gases in the atmosphere.”
Although the above statement makes no direct reference to Earth surface temperature, I believe it carries the implication that greenhouse gases in the Earth’s atmosphere increase the Earth’s surface temperature.
I make two comments: the first is relevant only if the above implication is valid, the second is relevant independent of the validity of the implication. First, placing matter adjacent to a warm surface such that the matter is capable of absorbing/blocking radiation to space from the warm surface can lead to a decrease in the warm surface’s temperature. Second, increasing the amount of the absorbing/blocking matter can lower the temperature of the absorbing/blocking material.
Take for example an internal combustion engine whose metal surface is exposed to a vacuum. In addition to doing useful work, the engine produces thermal energy (heat). That thermal energy will produce a rise in the temperature of the engine’s surface such that in energy-rate equilibrium the rate energy is radiated to space from the engine’s surface is equal to the rate thermal energy is generated within the engine. By attaching radiating plates to the engine’s surface, some of the energy radiated to space from the engine’s original surface will be absorbed/blocked by the plates; but because thermal energy can be transferred from the engine to the plates via both radiation and conduction, the temperature of the engine’s original surface will be lowered. This is the principle of an air-cooled engine[1]: provide a means other than radiation of transferring heat from an engine to a large surface area from which heat can be removed via a combination of conduction, convection and radiation, and the engine’s surface temperature will be lowered.
If plates at a temperature lower than the original engine surface temperature are attached to the engine, it’s true that the temperature of the plates will increase to establish energy-rate equilibrium. Once energy-rate equilibrium is established, however, increasing the plate radiating area (adding additional matter that blocks more of the energy radiated from the original engine surface) will likely lower the plate temperature.
Thus, blocking the amount of surface radiation escaping to space does not necessarily increase the surface temperature; and increasing the amount of radiation blocking material does not necessarily increase the temperature of that material. In both cases (the Earth/Earth-atmosphere and the internal combustion engine in a vacuum), the heat eventually escapes to space–otherwise the temperature of the Earth’s surface and the engine would continue to rise indefinitely. The difference isn’t that the energy doesn’t eventually escape to space (it does in both cases), the difference is in the path the energy takes to reach space. The amount of generated thermal energy in conjunction with the path the thermal energy takes to get to space determines temperatures along the path; and adding more material may increase or decrease those temperatures. To say that “Energy that would normally escape into space is absorbed by these molecules, thus heating the atmosphere…” by itself is unwarranted; because an equivalent statement for the case of adding extra plate material to the engine would be “Energy that would normally escape to space from an engine with small attached plates is absorbed by additional plate material, thus heating the plates…” For air-cooled engines, this statement is not true—otherwise the plate surface area of air-cooled engines would be as small as possible.
It’s fairly easy to visualize why (a) adding thermally radiating plates to an air-cooled engine might decrease the engine’s surface temperature, and (b) increasing the area of the radiating plates might decrease the plate temperature. It’s not so easy to visualize, and may not be true, why (a) adding greenhouse gases to the Earth’s atmosphere decreases the Earth’s surface temperature; and (b) increasing the amount of atmospheric greenhouse gases lowers the temperature of the Earth’s atmosphere. I now present one possible argument. I do not claim that the argument is valid for greenhouse gases in the Earth’s atmosphere, but I do claim that the argument might be valid, and can only be refuted by an analysis more detailed than simply claiming “Energy that would normally escape into space is absorbed by these molecules, thus heating the atmosphere.”
If we assume that (a) matter cannot leave the Earth/Earth-atmosphere system, and (b) non-greenhouse gases radiate negligible energy to space, then for a non-greenhouse gas atmosphere the only way thermal energy can leave the Earth/Earth-atmosphere system to space is via radiation from the surface of the Earth. The rate radiation leaves the surface is in part a function of both the area and temperature of the surface. For a greenhouse gas atmosphere, energy can leave the Earth/Earth-atmosphere system to space both via radiation from the Earth’s surface and radiation from greenhouse gases in the atmosphere. Suppose it is true that the density of greenhouse gases near the Earth’s surface is such that radiation emitted from low-altitude greenhouse gases does not directly escape to space, but is in part directed towards the Earth’s surface and in part absorbed by other atmospheric greenhouse gases. As the atmospheric greenhouse gas density decreases with increasing altitude, radiation emitted from high-altitude greenhouse gases can directly escape to space.
Now it’s not impossible that since (a) in addition to radiation, heat is transferred from the Earth’s surface to greenhouse gases via conduction, and (b) convection currents (i) circulate the heated greenhouse gases to higher altitudes where energy transfer to space can take place and (ii) return cooler greenhouse gases to the Earth’s surface, that the process of heat transfer away from the Earth’s surface via greenhouse gases is more efficient than simple radiation from the Earth’s surface. Many engines are cooled using this concept. Specifically, a coolant is brought into contact with a heated surface which raises the coolant’s temperature via conduction and radiation, and the coolant is moved to a location where thermal energy transfer away from the coolant to a heat sink is more efficient than direct thermal energy transfer from the heated surface to the heat sink.
One way to realize increased thermal transfer efficiency would be to use a coolant, such as greenhouse gases, that efficiently radiates energy in the IR band (i.e., radiates energy at temperatures around 500 K). Another way would be to spread the heated coolant over a large surface area. Since surface area increases with increasing altitude, thereby providing expanded “area” (in the case of a gas, expanded volume) from which radiation to space can occur, it’s not clear to me (one way or the other) that greenhouse gases won’t act as a “coolant” reducing both the temperatures of the Earth’s atmosphere and the Earth surface.
[1] It’s true that for most air-cooled engines the main transfer of heat from the engine plates is via a combination of (a) conduction of heat to the air near the plates, and (b) convection that replaces the warm air near the plates with cooler air. To aid this process, a fan is often employed, or the engine is located on a moving vehicle and the vehicle’s motion through an atmosphere provides the flow of air across the plates. Although conduction/convection may be the primary means of heat dissipation from the plates, radiative cooling also dissipates heat.
Eli Rabbit writes “This slows down the rate at which the Earth emits to space because it is now radiating at higher, colder altitudes”
Easy to say but harder to prove. At that increased altitude it is still the CO2 molecules that radiate according to a probability they have sufficient energy to do so. If you have more of them (as is the assumption that the atmosphere is well mixed) then you have more of them radiating.
Eli writes at his blog “Decreasing temperature slows down the rate at which each molecule can emit while decreasing density means there are fewer greenhouse gas molecules available to absorb or radiate the energy.”
Fewer than down lower thats true, but still more than there were previously at that altitude. so whilst they may be radiating less often, you cant escape the fact there are more of them doing so and its not at all intuitive what the net result is.
Dear RGB, please excuse my poorly articulated (Layman terminolgy) questions in the last paragraph of this post. Please follow the few posts to understand the questions in the last post, and, if you please, provide an answer, or answers if possible.
Can GHGs in the atmosphere receive conducted energy from non GHGs, (I think this is how they form a LTE; local thermal equilibrium) and then radiate that energy away?
If the answer to this question is yes, then are not those CO2 molecules (the ones which receive conducted energy from non GHGs) accelerating the loss of energy to space, which, in the absence of GHGs, would not be able to leave the atmosphere?
====================================================
Eli responds: Thee is a relatively rapid transfer of vibrational to kinetic energy in ghg molecules that absorb photons so essentially all greenhouse gas molecules that absorb photons do not re-radiate the energy (the radiation rate is five to six orders of magnitude slower, so only one in a million will reradiate promptly. OTOH there will be some ghg molecules that are excited by other collisions, the proportion being controlled by the local temperature.
=================================
Robert Austin says:
So the bulk non GHG’s which do not radiate appreciably at normal temperatures are the medium of storage and transport of the heat energy apprehended by the GHG’s,
About six percent of all CO2 at STP is vibrationally excited by the local thermodynamic equilibrium (2 fold degenerate, ~700 cm-1 vibrational energy, and 300K is ~200 cm-1), so essentially 6% is always ready to radiate, of course which molecules are excited is a constantly changing dance, but the amount of emission measured at various altitudes is in accord with this BOE,
===============================
Eli Rabett says:
July 21, 2012 at 8:23 pm
Konrad says:
July 21, 2012 at 6:11 pm
My very simple question for Joeldshore and Eli Rabett, can non condensing radiative gasses such as CO2 radiate as IR energy they have acquired conductively?
If you mean by conduction, T-V (translational to vibrational) energy transfer, yes
======================================
So, RGB the answer appears to be yes, but poorly quantified….”About six percent of all CO2 at STP is vibrationally excited by the local thermodynamic equilibrium (2 fold degenerate, ~700 cm-1 vibrational energy, and 300K is ~200 cm-1), so essentially 6% is always ready to radiate…”, OR…”so only one in a million will reradiate promptly. OTOH there will be some ghg molecules that are excited by other collisions, the proportion being controlled by the local temperature.”
So my questions are as follows. How much of the non GHG energy is radiated to space via collision with GHG molecues? If the GHG molecues were not present, how much longer would this energy stay within the atmosphere if it could only be conducted and convected about, but not radiated to space. And, as additional GHG molecues speed the escape of conducted Non GHG energy, would not this reduced residence time of conducted non GHG energy have to be subtracted from the increased residence time of IR energy raqdiating from the surface, and backradiating from the GHG molecues? TSI incoming is a consistent flow, so the energy gained or lost by either radiating conducted non ghg energy out, or keeping surface energy within the atmosphere is porportional to the residence time of the energies affected.
Konrad writes “CO2 almost instantly re-radiates the outgoing IR radiation it intercepts, with around 50% of this radiated back towards the Earth’s surface.”
Not so. At sea level, the CO2 almost instantly gives up its absorbed energy to the rest of the atmosphere due to a collision. Collisions happen on the order of every 10^-7 seconds whereas it takes on average 10^-3 seconds to radiate. CO2 at sea level is a warming agent for the atmosphere and it occurrs right at ground level within tens of meters.
Ref : Pierrehumbert’s “Infrared radiation and planetary temperature”
http://geosci.uchicago.edu/~rtp1/papers/PhysTodayRT2011.pdf
Much further up in the atmosphere where collisions are less frequent this changes and is a fundamental property of the atmosphere that is not mentioned when it comes to the average altitude of radiation to space. It should be though.
Eli Rabett says:
July 21, 2012 at 5:39 am
“To maintain radiative balance (sun in, IR out) the entire Earth system warms until the temperature rises enough in the mid troposphere to restore the balance.”
But since Mainstream Climate Science’s predicted Tropical Tropospheric “Hot Spot” has not eventuated, then according to “the physics” of Mainstream Climate Science’s CO2 = GW hypothesis: there has been no “entire earth system” GW and in particular, no Global Mean Temperature increase.
David says
Now imagine a photon going from bottom of the jar to the top in a straight line. The rule that the photon has to follow is that if it hits a yellow bb, it goes straight through….
Henry says
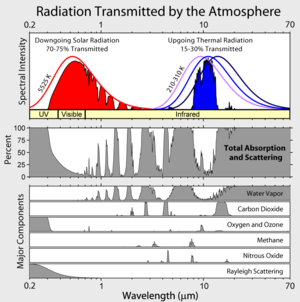
Well, this where you all go wrong. Carefully look at the picture that this post starts with. Ozone re-radiates more sunlight then earthshine. Water vapor re-radiates strongly in the IR coming from the sun, as does CO2.
Greg is right. You have to make a balancesheet and show us how much warming and how much cooling is caused by each GHG.
Look at water vapor and CO2 around 2 um and see how that makes a dent in the incoming solar radiation. Notice that the ozone shields us from ca. 15-20% of all sunlight by absorbing and re-radiating in the UV region. In fact, if you really grasp what you are seeing in this graph/ representation (from a cloudless day), you would realize that without the ozone and CO2 and H2O and other GHG’s you will get a lot more radiation on your head. In fact, you would probably fry.
For comprehensive proof that CO2 is (also) cooling the atmosphere by re-radiating sunshine, see here:
http://www.iop.org/EJ/article/0004-637X/644/1/551/64090.web.pdf?request-id=76e1a830-4451-4c80-aa58-4728c1d646ec
They measured this re-radiation from CO2 as it bounced back to earth from the moon. So the direction was sun-earth (day)-moon(unlit by sun) -earth (night). Follow the green line in fig. 6, bottom. Note that it already starts at 1.2 um, then one peak at 1.4 um, then various peaks at 1.6 um and 3 big peaks at 2 um. You can see that it all comes back to us via the moon in fig. 6 top & fig. 7. Note that even methane cools the atmosphere by re-radiating in the 2.2 to 2.4 um range.
This paper here shows that there is absorption of CO2 at between 0.21 and 0.19 um (close to 202 nm):
http://www.nat.vu.nl/en/sec/atom/Publications/pdf/DUV-CO2.pdf
There are other papers that I can look for again that will show that there are also absorptions of CO2 at between 0.18 and 0.135 um and between 0.125 and 0.12 um.
We already know from the normal IR spectra that CO2 has big absorption between 4 and 5 um.
So, to sum it up, we know that CO2 has absorption in the 14-16 um range causing some warming (by re-radiating earthshine) but as shown and proved above it also has a number of absorptions in the 0-5 um range causing cooling (by re-radiating sunshine). This cooling happens at all levels where the sunshine hits on the carbon dioxide same as the earthshine. The way from the bottom to the top is the same as from top to the bottom. So, my question is: how much cooling and how much warming is caused by the CO2? How was the experiment done to determine this and where are the test results? (I am afraid that simple heat retention testing might not work here, we have to use real sunshine and real earthshine to determine the effect in W/m3 / [0.03%- 0.06%]CO2/m2/24hours).
I am doubtful of the analysis of the spectral data. I have not seen any work that convinces me. In the case of CO2, I think the actual heat caused by the sun’s IR at 4-5 could be underestimated, i.e. the radiation of the sun between 4 and 5 may be only 1% of its total energy output, but how many Watts per m2 does it cause on earth? Here in Africa you cannot stand in the sun for longer than 10 minutes, just because of the heat (infra-red) of the sun on your skin.
In all of this we are still looking at pure gases. The discussion on clouds and the deflection of incoming radiation by clouds is still a completely different subject.
CO2 also causes cooling by taking part in the life cycle. Plants and trees need warmth and CO2 to grow – which is why you don’t see trees at high latitudes and – altitudes. It appears no one has any figures on how much this cooling effect might be. There is clear evidence that there has been a big increase in greenery on earth in the past 4 decades. Therefore, there is a good chance that the total net effect of more carbon dioxide in the atmosphere could be close to zero. But unless we come up with the right test methods and measurements, we will never know for sure. For more on why it is considered highly unlikely that CO2 is a contributory cause to global warming, see here:
http://www.letterdash.com/henryp/global-cooling-is-here.
Hint: plot the development of the speed of warming (maxima).
which now stands at
+ 0.036 K per annum from 1974 (38 years)
+ 0.029 K per annum from 1980 (32 years)
+0.014 K per annum from 1990 (22 years)
-0.016 K per annum from 2000 (12 years)
Henry, your post appears correct. Any solar spectrum chart shows that about 98% of that energy lies between about 250 nm in the UV and 4.0 microns; with the remaining as 1% left over at each end. Such graphs often have superimposed on them the actual ground level (air Mass once) spectrum; that shows the amounts of that energy taken out by primarily O2, O3, and H2O, in the case of H2O which absorbs in the visible and near IR perhaps 20% of the total solar energy is capture by water VAPOR (clear sky) clouds are an additional loss over and above that. So as WV increases there is a corresponding reduction in TSI reaching the surface, and therefore a reduction in LWIR leaving the surface.
However, certainly all this is quantified in the climate models? I know that Steve McIntyre has been requesting an engineer style describtion of the GHE for some time. regarding my question here, (David says: July 21, 2012 at 11:15 pm) do you have any thoughts on how much non GHG energy is conducted to GHGs and leaves (radiates to space) the earth atmosphere system faster then it would if their was less GHG?
TimTheToolMan says: July 21, 2012 at 11:22 pm
———————————————————————
I have seen two basic theories of the greenhouse effect;
A- CO2 scatters 50% of outgoing LWIR it intercepts back to the surface slowing its rate of cooling.
B- CO2 directly heats the air molecules around it on intercepting outgoing LWIR.
I have found through empirical experiment that incident LWIR can slow the cooling rate of some materials, but not liquid water that is free to evaporatively cool. This rules out option A as a mechanism for CAGW.
You have chosen option B, stating “Not so. At sea level, the CO2 almost instantly gives up its absorbed energy to the rest of the atmosphere due to a collision. Collisions happen on the order of every 10^-7 seconds whereas it takes on average 10^-3 seconds to radiate”
If this rate of molecular collision directly equated to the speed of equalisation of energy states between molecules in air this would make air highly conductive and entirely unsuitable for use in double glazing.
Of course either option A or B as the primary CAGW mechanism matters little to the central question raised by Reed Coray’s post. No matter how CO2 is supposed to cause global warming, its ability to warm will be an inverse logarithmic function of its concentration in the atmosphere. However its ability to radiate to space energy it has acquired from conductive contact with Earth’s surface or atmosphere is a linear function of its concentration in the atmosphere.
The real problem lies within the physics themselves, it is the application of the Stefan Boltzmann planetary temperature to Earth (AKA -18C incident to 240W/m^2 insolation avged/spread globally), you’re pretending the Earth has no thermal capacity and behaves in accordance to a blackbody surface recieving 240W/m^2 insolation, or something close to the above behavior. In reality you have a ROTATING SPHERE, recieving 480W/m^2 insolation over 1/2 the spherical area when applying Holder’s inequality…the energy recieved via the Sun is mostly retained at ‘night’ by the Ocean/Atmosphere system..the oceans feature NO Diurnal Cycle and are for all intents and purposes a greenhouse fluid retaining heat with an enormous capacity.. and conduct heavily to an atmosphere composed of 99.8% non-emitting Oxygen/Nitrogen molecules (Though O2 absorbs heavily in the UV Spectrum).
Most of the 33C warming over the planetary Stefan Boltzmann temperature is via retention..in fact it is H2O-laden convection (AKA cloud albedo)that reduces the retention-driven planetary warming..it should be clear that retention is the issue as the high-temp of the “day” never occurs at noon rather you’ll see it occur around 3PM…same goes for the seasons at the poles, only the lag is 8 weeks.
The concept of equilibrium between the surface and atmosphere above the surface is also nonsense. GHGes which compose an avg of 0.2% of the atmosphere have no discernable impact on temperature..if anything the effect is negative given the enhancing of the water cycle/general convection.
Third and most important..if the Greybody temperature is 255K (and this mythical emission height exists at 14kft), then in that case the stratopause should not avg -55C or the conservation of energy is violated..this is, however, for another time.
Konrad writes “If this rate of molecular collision directly equated to the speed of equalisation of energy states between molecules in air this would make air highly conductive and entirely unsuitable for use in double glazing. ”
I’m not quite sure what you’re getting at there. The energy states of molecules in the atmosphere varies as per the Maxwell-Boltzmann distribution. There will always be some molecules at the energy state that CO2 needs to radiate but they wont all be CO2 molecules(!) and individually they wont stay there for long either.
That doesn’t make air a good conductor…
David says
However, certainly all this is quantified in the climate models?
Henry says
You are joking?
The so-called climate experts haven’t even figured out yet that there is a natural 50 year warming cycle followed by a 50 year cooling period –
Israel apparently knew about it (7×7 + 1 jubilee year) and I suspect Moses picked it up from the Egyptians, the pyramid builders, who were experts on everything that happened on the sun.
Did you do the plot and did you find the roots of the binominal (parabolic ) plot?
I am reasonably convinced that this cycle is caused by the sun-UV-O2-O3 cycle. The scare about the ozone falling was the greatest in the nineties when ozone was at its lowest, and it picked up since 1995, as can be expected by my theory….
Lester Via says:
July 21, 2012 at 6:13 pm
Bucky Cochrane says:
July 21, 2012 at 4:12 pm
The whole idea of GHG “absorbing heat” is erroneous. CO2 absorbs a photon, goes into the “bending” mode of molecular vibration and almost immediately radiates the photon which it absorbed. It cannot give up any fraction of this energy; there is no state between this 667 wavenumber excited state and its vibrational ground state. It cannot “warm the air”
Sorry Bucky but it does warm the air. The CO2 molecule is always vibrating from collisions with other air molecules. Due to these collisions, translational motion and vibratory motion are freely and very quickly exchanged at the gas pressures and temperatures typical of the lower atmosphere
Sorry, Lester, it does not warm the air. Explain yourself specifically without quoting specific heat ratios. etc. and vigorously waving your hands. The 010 state is a radiative decay state; look at CO2 laser diagrams. It would give over 1000C kinetic energy to a molecule if the 010 state were entirely converted to translational KE. How can all of the vibrational energy of CO2 molecules with atoms vibrating in opposite directions be converted to unidirectional KE? How is momentum conserved in such an interaction? PLUS, my model yields correct calculation of earth surface temperature. Remember, this ~.08 ev must be released in ONE interaction; there are no intermediate states. (Rotational states are in the micro wave region and will warm the gas, but we are not talking about microwave radiation) I am interested in your explanation if it is not a bunch of PY 101 platitudes.
JPeden says:
Your statement is very confused. Even if we assume you are correct about the data not showing the “hot spot”, that does not mean that things have not warmed. It just means that the places at altitude in the tropics that were expected to warm more rapidly than the surface have not warmed more rapidly than the surface. The most direct consequence of such a fact would be that the lapse rate feedback, a negative feedback in the climate models, shouldn’t be there and thus that the models may be UNDERESTIMATING the climate sensitivity a little bit.
However, the reality of the situation is that the data for the multidecadal trends in the tropics is not really good enough to conclude definitively whether the “hot spot” is missing or not. It is also noteworthy that the expected amplification of temperature variations with altitude does occur for temperature fluctuations over monthly to yearly time scales, severely constraining any explanations of how the models could be wrong in the basic prediction.
gymnosperm says (in reference to my comment here http://wattsupwiththat.com/2012/07/21/some-thoughts-on-radiative-transfer-and-ghgs/#comment-1039067 ):
Yes, CO2 gets into the stratosphere; I don’t think there is any argument about that. However, the mean emitting level of the Earth, i.e., the level at which most of the radiation emitted can successfully escape to space is still well within the troposphere. So, the vast majority of the emission to space occurs from the troposphere. Hence, one is justified in assuming the lapse rate that I assumed. Any corrections to that by doing the calculation more rigorously might change things a little bit…but even if my original calculation was off by a factor of 3 (which seems unlikely), we are still talking about the cooling with the decreasing emitting layer temperature effect being 100 times as important as the increasing emitting layer area effect.
”
Eli Rabett says:
July 21, 2012 at 4:35 pm
cba accuses Eli of being a character in a childrens novel and throws much detail against the wall which really does not shift the argument much. cb, if you want detail go read the science of doom articles on the greenhouse effect that KR provided. The mechanism remains what the Bunny pointed to.
http://scienceofdoom.com/roadmap/atmospheric-radiation-and-the-greenhouse-effect/
”
*************************
Perhaps you should try harder to make sense of it. The argument doesn’t have to shift much at all. A change of only from 3-5 deg C warming per co2 doubling down to around 1 deg C rise per co2 doubling changes things from CAGW to AGW or CGW to GW.
I didn’t have time to go completely over your reference – whose political affiliations are prominantly contained in the name of the website. I did notice that all the graphs were done using /cm instead of wavelength which tends to be more like speaking in ebonics rather than in english – quite understandable to a small fraction of the populace but almost meaningless to the majority. Since the graph shapes up differently with power per unit /cm versus per unit wavelength, it does help the graph look scarier and allows for much smaller bandwidths to be displayed, thus making the apparent effect of ghgs look more significant. I also didn’t see any mention of A. Eddington in the presentation of the radiative xfer approximations – but then maybe I was just scanning over it too rapidly.
As RGB has pointed out, CAGW extremists have been known to actually use legitimate physics in preparation of their hysterical claims. Of course it doesn’t take a long time to find that some, like hansen, sometimes quickly depart from that and invent new things like ‘characteristic’ radiating altitude – where in reality nothing of any significance is actually radiating into space from that altitude because its either radiating through there from the ground at wavelengths unaffected by any ghg molecules or its being absorbed by higher layers except for the slight decrease in line width due to a reduction in pressure – which also increases the peak’s ability to absorb/emit.
Radiative transfer can get a rather good estimate of how much power is absorbed or transferred through clear skies – but not through clouds. It cannot tell you the sensitivity of Earth’s temperature to a change in the amount of power transferred / absorbed. The vast majority of measurements intended to determine this have ignored albedo variation which can be several times that of the change in co2 power absorption. Simple averages, mostly of real numbers can give you a real average sensitivity and that is a far cry from the usual estimates, being less than than straight stefan’s law estimates which means there’s net negative feedback present. It also shows just how far off you people are when it comes to the dellusions of massive amounts of positive feedback that have some sort of tiny relative stability. Also, your big h2o vapor feedback bugaboo is simply BS. Even a 5 deg C shift in T for surface and atmospheric column at constant RH would net you scarcely 30% and h2o is every bit a log function just like co2 and is roughly linear over almost a dozen halvings just like co2. Hint, even a 30% increase in absolute humidity for h2o provides less additional power absorption than a co2 doubling at the tropopause.
Thanks to all that have seriously participated in the discussions on this thread. I has been very interesting. There is certainly no consensus here. To some it may appear that no progress has been made. To me it appears that a lot has been made as ideas are debated, yet far from any settled consensus. This has been real peer review.
Some have argued for a specific point (possibly correctly in that right) against another supporting the same point but with the inclusion of a related concept which led to arguments of minute details where they were basically in agreement. Seemed a little chaotic at times. But the entire discussion of climate is actually a chaotic discussion of a complex and chaotic system. There are just too many variables effecting every little piece of the many interrelated variables involved to ever come close to any settled conclusions with regards to the overall concept of understanding climate. Simply put, chaos can’t be fully explained. It is just that, chaos. But it is a real intellectual challenge to try to learn as much about the chaos as possible. That’s why most of us are here. Then there is always the token trolls and those with an agenda.
Again, thanks to all.
Joel says
your statement is very confused
Henry says
it is you who is confused
a) you have no proof that the net effect of more CO2 is warming rather than cooling, as I requested you to give to me in previous postings
b) you have no tests or measurements that you have collected yourself, so you rely on others
c) JPeden asked about the temperature drop that we note since the beginning of the new milennium, to which you obviously have no reasonable explanation – seeing that CO2 is still rising.
You are most certainly not the (truthful) prophet Joel from the bible to whom you were named after.
http://www.letterdash.com/HenryP/more-carbon-dioxide-is-ok-ok
”
joeldshore says:
July 22, 2012 at 6:43 am
gymnosperm says (in reference to my comment here http://wattsupwiththat.com/2012/07/21/some-thoughts-on-radiative-transfer-and-ghgs/#comment-1039067 ):
Carbon dioxide seems to be getting into the stratosphere. I doubt anyone knows how. Ozone holes? The tops of those thunderheads that dome in? As you undoubtedly know at the tropopause the lapse rate is inverted and temperature increases with altitude, eventually reaching levels nearly as warm as the surface.
I’m going to leave the math to you, but the trouble with, “It’s not difficult to calculate”, and, “It’s simple physics”, is that the reality seems not to be easy or simple.
Yes, CO2 gets into the stratosphere; I don’t think there is any argument about that. However, the mean emitting level of the Earth, i.e., the level at which most of the radiation emitted can successfully escape to space is still well within the troposphere. So, the vast majority of the emission to space occurs from the troposphere. Hence, one is justified in assuming the lapse rate that I assumed. Any corrections to that by doing the calculation more rigorously might change things a little bit…but even if my original calculation was off by a factor of 3 (which seems unlikely), we are still talking about the cooling with the decreasing emitting layer temperature effect being 100 times as important as the increasing emitting layer area effect.
”
*********************************
It’s nonphysical. There is no level where most of the radiation emitted can successfully escape. 70% of the radiation from the surface under clear skies makes it through the atmosphere, some of it after being absorbed and reradiated numerous times. No layer absorbs or emits significant amounts of power. You are dealing with hansen’s dellusions based on the amount of power radiated from a blackbody or greybody at a given temperature and that doesn’t exist above the surface under clear sky conditions.
The actual result is that your emitting altitude factor is no more important than your area increase effect. One is very small but meaningful, the other is meaningless because it doesn’t exist. Each layer has contributions to emission and absorption but it takes a 1 dimensional model to determine it as it is a function of pressure as well as temperature that affects the line shape.
****
Eli Rabett says:
July 21, 2012 at 5:39 am
Increasing concentrations of GHGs raises the altitude that GHGs can radiate to space in the blocked regions of the spectrum
Because of the lapse rate, the higher you go in the troposphere, the lower the temperature
This slows down the rate at which the Earth emits to space because it is now radiating at higher, colder altitudes.
****
Why does the altitude that radiation escapes get higher? I don’t see it. As GHGs increase in concentration, their absorption/emission-bands widen, and so more of the earth’s total IR emission comes from the tropopause relative to warmer, lower altitudes. So the surface IR “window” (that gets past GHGs) must increase its emission (surface gets warmer) to get back to ~equilibrium. Fine. But I still don’t see why the emission height must change. At the tropopause the lapse-rate has gone to zero due to increasing absorption of UV by ozone. Raising the emission height there won’t have any effect from altitude change. If the emission height got even higher, it would get into warmer, stratospheric air, which would be an anti-greenhouse effect.
joeldshore says:
July 22, 2012 at 6:30 am
==========================
Curious post. Joel Shore appears to be saying that the missing hot spot is either “A” an indication that the lack of overall atmospheric heat is an indication that it is : worse the we think”, or “B” the measurements are wrong. When this is your only two possible answers I think the potential for confirmation bias is greatly enhanced.
Joel, perhaps you could put the models numbers on these questions…
Any solar spectrum chart shows that about 98% of that energy lies between about 250 nm in the UV and 4.0 microns; with the remaining as 1% left over at each end. Such graphs often have superimposed on them the actual ground level (air Mass once) spectrum; that shows the amounts of that energy taken out by primarily O2, O3, and H2O, in the case of H2O which absorbs in the visible and near IR perhaps 20% of the total solar energy is capture by water VAPOR (clear sky) clouds are an additional loss over and above that. So as WV increases there is a corresponding reduction in TSI reaching the surface, and therefore a reduction in LWIR leaving the surface.
However, certainly all this is quantified in the climate models? I know that Steve McIntyre has been requesting an engineer style describtion of the GHE for some time. Regarding my questionS here, (David says: July 21, 2012 at 11:15 pm) do you have any thoughts on how much non GHG energy is conducted to GHGs and leaves (radiates to space) the earth atmosphere system faster then it would if their was less GHG?
This is complicated. Tim the tool man states …At sea level, the CO2 almost instantly gives up its absorbed energy to the rest of the atmosphere due to a collision. Collisions happen on the order of every 10^-7 seconds whereas it takes on average 10^-3 seconds to radiate. CO2 at sea level is a warming agent for the atmosphere and it occurrs right at ground level within tens of meters. so several question come to mind. How much of the energy in CO2 which it is “giving up” via conduction to non GHGs, came from conducting cooling, non GHG molecues? In which case it is neither warming or cooling, just acting as if it was another non GHG molecue. However, it does still radiate at times? How often? If it radiates towards space is it not potentially radiating conducted “non GHG energy” away from the planet faster then that energy would leave if it only encountered non radiating non GHGs.? How much more energy is moved via convection if the intial affect is primarily warming? How much energy is absorbed via an acceleration of the hydrological cycle? How do these ratios change at diaparate elevations, as the higher up in the atmosphere one goes, the more time is required between conduction via collisions, and a higher percentage of energy is radiated?, Numbers numbers numbers.?????
David says:
July 22, 2012 at 7:55 am
==========
😉
This is really dopey:
“So, radiation from a colder body directed to the warmer body is radiation, but no warmist has been able to present a real falsifiable scientific experiment proving that this sort of radiation can warm the warmer body (or slow down it’s cooling, whatever).”
Does this clown think that the warmer body is transparent to the radiation from the colder body? Does it pass through without any effect? Or it detours around the warmer body? Bounces off?
Suspend a heating element in a vacuum chamber and heat it to a constant temperature of 500 F. Suspend another element near the first and heat it to 300 F. Work on that.
Henry@David
Just to clarify: I am watching with some amusement a lot of scholar discussions on the green house effect as I realised again that the people that I encounter on most scientific blogs don’t understand the chemistry principle of absorption and subsequent re-radiation. In fact very few people do understand it because if they did they would have raised the alarm bells ringing long time ago. But they all got stuck at Tyndall and Svante Arrhenius. …
They know that CO2 (carbon dioxide) “absorbs” in the 14-16 um region. Most people think that what it means is that the molecules absorbs photons here which then subsequently get transferred as heat to neighbouring molecules. Then it absorbs again, and so on, and so on…and all the absorbed light is continuously transferred to heat…
Although this may happen up to a certain saturation point as soon as the light or radiation hits on the gas, that is in fact not what is causing the heat entrapment.
I happen to be familiar with spectrophotometry. You have to understand what actually happens when we put a beam of light of certain wavelength on a sample of liquid or gas.
We have various spectrophotometers that can measure the various ranges of UV-visible -IR etc. Usually you have the option to vary the wavelength of the beam of light, either manually or automatically.
If the gas or liquid is completely transparent, we will measure 100% of the light that we put through the sample coming through on the other side. If there is “absorption” of light at that specific wavelength that we put through the sample, we only measure a certain % on the other side. The term “extinction” was originally used but later “absorption” was used to describe this phenomenon, meaning the light that we put on was somehow “absorbed”. I think this was a rather unfortunate description as it has caused a lot of confusion since. Many people think that what it means is that the light of that wavelength is continually “absorbed” by the molecules in the sample and converted to heat. If that were true, you would not be able to stop the meter at a certain wavelength without over-heating the sample, and eventually it should explode, if the sample is contained in a sealed container. Of the many measurements that I performed, this has never ever happened. Note that in the case of CO2, when measuring concentrations, we leave the wavelength always at 4.26 um. Because the “absorption” is so strong here, we can use it to compare and evaluate concentrations of CO2.
The best way to experience re-radiation for yourself is to stand in a dark forest just before dawn on a cloudless night. Humidity must be high. Note that water vapour also absorbs in the visible region of the spectrum. So as the first light of sun hits on the water vapour you can see the light coming from every direction. Left, right, bottom up, top down. You can see this for yourself until of course the sun’s light becomes too bright in the darkness for you to observe the re-radiated light from the water vapour. This is also the reason why you will quickly grab for your sun glasses when humidity is high, because even with the sun shining for you from your back and driving in your car, you can feel on your eyes that the light from the sun is re-radiated by the water vapor in the atmosphere.
A third way to experience how re-radiation works is to measure the humidity in the air and the temperature on a certain exposed plate, again on a cloudless day, at a certain time of day for a certain amount of time. Note that as the humidity goes up, and all else is being kept equal, the temperature effected by the sun on the plate is lower. This is because, like carbon dioxide, water vapour has absorption in the infra red part of the spectrum.
We can conclude from all these experiments that what actually happens is this:
in the wavelength areas where absorption takes place, the molecule starts acting like a little spherical mirror, the strength of which depends on the amount of absorption taking place inside the molecule. We may assume that at least 50% of a certain amount of radiation is sent back in a radius of 180 degrees in the direction where it came from. (However, because the molecule is very small and therefore might behave more or less like a sphere, it could be up to ca. 62,5% ). This re-radiation in the sun’s spectrum and in the earth’s spectrum is the cooling effect, or warming effect, respectively, of a gas that is hit by radiation. An effect that is very similar to this, is also observed when car lights are put on bright in humid, moist and misty conditions: your light is returned to you!!
Unfortunately, in their time, Tyndall and Arrhenius could not see the whole picture of the spectrum of a gas which is why they got stuck on seeing only the warming properties of a gas (i.e. the closed box experiments).
If people would understand this principle, they would not singularly identify green house gases (GHG’s) by pointing at the areas in the 5-20 um region (where earth emits pre-dominantly) but they would also look in the area 0-5 um (where the sun emits pre-dominantly) for possible cooling effects. If you really want to understand what happens in the atmosphere, this rough graph / representation (on a cloudless day) is very important:
http://albums.24.com/DisplayImage.aspx?id=cb274da9-f8a1-44cf-bb0e-4ae906f3fd9d&t=o
….now carry on at to my previous post to davidmhoffer……sorry about the confusionswith the two Davids…
Joel,
What of density? If your construct of “mean radiating altitude” has any physical basis it would be the density of radiating molecules, yet ultimately more radiating molecules means more photons escaping to space.
Joel Shore:”Your statement is very confused. Even if we assume you are correct about the data not showing the “hot spot”, that does not mean that things have not warmed. It just means that the places at altitude in the tropics that were expected to warm more rapidly than the surface have not warmed more rapidly than the surface. The most direct consequence of such a fact would be that the lapse rate feedback, a negative feedback in the climate models, shouldn’t be there and thus that the models may be UNDERESTIMATING the climate sensitivity a little bit.
However, the reality of the situation is that the data for the multidecadal trends in the tropics is not really good enough to conclude definitively whether the “hot spot” is missing or not. It is also noteworthy that the expected amplification of temperature variations with altitude does occur for temperature fluctuations over monthly to yearly time scales, severely constraining any explanations of how the models could be wrong in the basic prediction.”
At the risk of pulling this thread off-topic, I have heard you make this statement before and I can’t see how it could be true. You seem to be arguing that the only way that the hotspot to be missing is for there to be no more condensation of WV on average than when the air was drier. It seems to me much more likely that the models are wrong about how efficiently those levels of the atmosphere radiate heat to space(for whatever reasons). If those levels are much more efficient in this regard than the models assume, then it is possible for the hotspot to be missing and the lapse rate feedback to be present. Further, since it seems to be highly unlikely that there is no lapse rate feedback, I would say that this second alternative is much more likely to be taking place (assuming there is no hotspot).
Cheers, 🙂
****
Sam Yates says:
July 21, 2012 at 1:42 pm
…Mr. Watts, a question; do you look over these guest posts before throwing them up here? Because, honestly, it does not do much to improve your credibility when you gladly host things like this, which betray a massive lack of comprehension of very, very basic physics. It’s your site, of course, and yours to run as you see fit, but…If I were you, I might be a bit more picky about things like this post. Quite frankly, this is embarrassing.
****
So far, I’ve increased my understanding from some of the replies in this thread (however incrementally). So it certainly isn’t “embarrassing” to me in any way, and I’d wager most others, too. Jeesh….