Guest post by Reed Coray
The following example illustrates the issues I have with reasoning often used to argue that increasing the amount of CO2 in the Earth’s atmosphere will increase both the Earth’s surface temperature and the Earth’s atmosphere temperature. Immediately following is a direct quote from URL
http://www.school-for-champions.com/science/heat_transfer_earth.htm
“The present situation is that there has been an increase in infrared-absorbing gases in the atmosphere, such as carbon dioxide (CO2) and methane (CH4). Energy that would normally escape into space is absorbed by these molecules, thus heating the atmosphere and spreading through convection currents. The average temperature of the atmosphere has increased 0.25 °C since 1980, mainly attributed to an increase in infrared-absorbing gases in the atmosphere.”
Although the above statement makes no direct reference to Earth surface temperature, I believe it carries the implication that greenhouse gases in the Earth’s atmosphere increase the Earth’s surface temperature.
I make two comments: the first is relevant only if the above implication is valid, the second is relevant independent of the validity of the implication. First, placing matter adjacent to a warm surface such that the matter is capable of absorbing/blocking radiation to space from the warm surface can lead to a decrease in the warm surface’s temperature. Second, increasing the amount of the absorbing/blocking matter can lower the temperature of the absorbing/blocking material.
Take for example an internal combustion engine whose metal surface is exposed to a vacuum. In addition to doing useful work, the engine produces thermal energy (heat). That thermal energy will produce a rise in the temperature of the engine’s surface such that in energy-rate equilibrium the rate energy is radiated to space from the engine’s surface is equal to the rate thermal energy is generated within the engine. By attaching radiating plates to the engine’s surface, some of the energy radiated to space from the engine’s original surface will be absorbed/blocked by the plates; but because thermal energy can be transferred from the engine to the plates via both radiation and conduction, the temperature of the engine’s original surface will be lowered. This is the principle of an air-cooled engine[1]: provide a means other than radiation of transferring heat from an engine to a large surface area from which heat can be removed via a combination of conduction, convection and radiation, and the engine’s surface temperature will be lowered.
If plates at a temperature lower than the original engine surface temperature are attached to the engine, it’s true that the temperature of the plates will increase to establish energy-rate equilibrium. Once energy-rate equilibrium is established, however, increasing the plate radiating area (adding additional matter that blocks more of the energy radiated from the original engine surface) will likely lower the plate temperature.
Thus, blocking the amount of surface radiation escaping to space does not necessarily increase the surface temperature; and increasing the amount of radiation blocking material does not necessarily increase the temperature of that material. In both cases (the Earth/Earth-atmosphere and the internal combustion engine in a vacuum), the heat eventually escapes to space–otherwise the temperature of the Earth’s surface and the engine would continue to rise indefinitely. The difference isn’t that the energy doesn’t eventually escape to space (it does in both cases), the difference is in the path the energy takes to reach space. The amount of generated thermal energy in conjunction with the path the thermal energy takes to get to space determines temperatures along the path; and adding more material may increase or decrease those temperatures. To say that “Energy that would normally escape into space is absorbed by these molecules, thus heating the atmosphere…” by itself is unwarranted; because an equivalent statement for the case of adding extra plate material to the engine would be “Energy that would normally escape to space from an engine with small attached plates is absorbed by additional plate material, thus heating the plates…” For air-cooled engines, this statement is not true—otherwise the plate surface area of air-cooled engines would be as small as possible.
It’s fairly easy to visualize why (a) adding thermally radiating plates to an air-cooled engine might decrease the engine’s surface temperature, and (b) increasing the area of the radiating plates might decrease the plate temperature. It’s not so easy to visualize, and may not be true, why (a) adding greenhouse gases to the Earth’s atmosphere decreases the Earth’s surface temperature; and (b) increasing the amount of atmospheric greenhouse gases lowers the temperature of the Earth’s atmosphere. I now present one possible argument. I do not claim that the argument is valid for greenhouse gases in the Earth’s atmosphere, but I do claim that the argument might be valid, and can only be refuted by an analysis more detailed than simply claiming “Energy that would normally escape into space is absorbed by these molecules, thus heating the atmosphere.”
If we assume that (a) matter cannot leave the Earth/Earth-atmosphere system, and (b) non-greenhouse gases radiate negligible energy to space, then for a non-greenhouse gas atmosphere the only way thermal energy can leave the Earth/Earth-atmosphere system to space is via radiation from the surface of the Earth. The rate radiation leaves the surface is in part a function of both the area and temperature of the surface. For a greenhouse gas atmosphere, energy can leave the Earth/Earth-atmosphere system to space both via radiation from the Earth’s surface and radiation from greenhouse gases in the atmosphere. Suppose it is true that the density of greenhouse gases near the Earth’s surface is such that radiation emitted from low-altitude greenhouse gases does not directly escape to space, but is in part directed towards the Earth’s surface and in part absorbed by other atmospheric greenhouse gases. As the atmospheric greenhouse gas density decreases with increasing altitude, radiation emitted from high-altitude greenhouse gases can directly escape to space.
Now it’s not impossible that since (a) in addition to radiation, heat is transferred from the Earth’s surface to greenhouse gases via conduction, and (b) convection currents (i) circulate the heated greenhouse gases to higher altitudes where energy transfer to space can take place and (ii) return cooler greenhouse gases to the Earth’s surface, that the process of heat transfer away from the Earth’s surface via greenhouse gases is more efficient than simple radiation from the Earth’s surface. Many engines are cooled using this concept. Specifically, a coolant is brought into contact with a heated surface which raises the coolant’s temperature via conduction and radiation, and the coolant is moved to a location where thermal energy transfer away from the coolant to a heat sink is more efficient than direct thermal energy transfer from the heated surface to the heat sink.
One way to realize increased thermal transfer efficiency would be to use a coolant, such as greenhouse gases, that efficiently radiates energy in the IR band (i.e., radiates energy at temperatures around 500 K). Another way would be to spread the heated coolant over a large surface area. Since surface area increases with increasing altitude, thereby providing expanded “area” (in the case of a gas, expanded volume) from which radiation to space can occur, it’s not clear to me (one way or the other) that greenhouse gases won’t act as a “coolant” reducing both the temperatures of the Earth’s atmosphere and the Earth surface.
[1] It’s true that for most air-cooled engines the main transfer of heat from the engine plates is via a combination of (a) conduction of heat to the air near the plates, and (b) convection that replaces the warm air near the plates with cooler air. To aid this process, a fan is often employed, or the engine is located on a moving vehicle and the vehicle’s motion through an atmosphere provides the flow of air across the plates. Although conduction/convection may be the primary means of heat dissipation from the plates, radiative cooling also dissipates heat.
OK, to answer some of my own questions, I looked at an atmospheric rad-spectrum, and the large CO2 “chunk” out of it centered at 667 cm-1 is at around -50C (~-60F). The altitude where the majority of the CO2 GHE is therefore where the atmos temp is -50C. Looking at various atmos temp profiles, -50C is close to, but not quite at the tropopause. OK, if the radiation height of the CO2 increases, there’s still alittle room for it to get colder, but not much. And the zero lapse-rate height would be a “limit” to how much it could rise to increase the insulation effect by that means.
But my question remains: Why would the altitude increase?
Beng says: “So far, I’ve increased my understanding from some of the replies in this thread (however incrementally). So it certainly isn’t “embarrassing” to me in any way, and I’d wager most others, too. Jeesh….”
But why should you have to wade thru a poor top post and hundreds of poor (and/or off-topic) replies in order to find the few replies that actually produced “incremental” increases in understanding. A well-written, on-topic top post would have provided more learning for more people with less effort and less possibility for continued misunderstanding of basic physics.
It’s cool that so many people are so passionate about science, but it is disappointing that so many of them are rather clueless (yet still feel they are expert enough to teach others). I’m sure a few people learn well by being told lots of wrong things and a few right things, but not most people.
beng – “…my question remains: Why would the [tropopause] altitude increase?”
Because the height of the troposphere, the altitude of the tropopause, is set by the top layer of convection. Maximum tropopause altitude (west equatorial Pacific for example, ~17.5km) is where there are high surface temps and lots of convection, low tropopause where there is less convection (Antarctica for example, ~8km, where tropospheric mixing is mostly due to frontal system uplift).
And as the atmosphere heats up, there’s more convection, and the tropopause gains in altitude. There’s no hard “limit” to tropospheric altitude.
beng;
But my question remains: Why would the altitude increase?
>>>>>>>>>>>>>
part of the reason is simply the math regarding the % chance that any given photon radiated in an upward direction will escape directly to space. The denser the CO2 the higher up that photon has to have even a chance of seeing a free path to space.
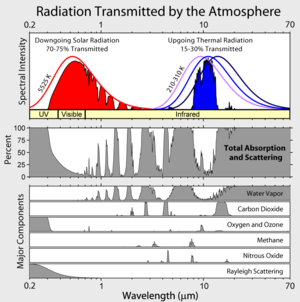
the other part of the reason has to do with water vapour which has absorption bands overlapping co2 (see chart in article above). At sea level in the tropics, water vapour is at 40,000 ppm, and completely overwhelms the effects of CO2. Going from 400 ppm to 800 ppm at sea level is as a consequence nearly meaningless. But at higher altitudes, colder temperatures reduce the amount of water vapour to very low amounts, so suddenly CO2 effects are much more pronounced compared to what would have happened otherwise.
Michael Tremblay;
Create any experiment which will take advantage of GHG’s to create an engine which will run on radiative energy alone.
>>>>>>>>>>>>
Given the number of times in this thread alone that it has been specified that the physics being explained is reliant upon an external energy source, one can only wonder if people like you even bother to read the explanations.
Shawnhet says:
Are you saying that they are somehow radiating more heat than one would expect given their temperature? Furthermore, the thermal structure of the tropical atmosphere is dominated by convection. The radiative transfer is already such as to maintain a larger lapse rate than exists but convection occurs and reduces the lapse rate approximately to the moist adiabatic lapse rate.
Sure, if the laws of radiative physics break down and they somehow emit more than allowed for by the laws of radiative physics, I suppose that is possible but seems rather unlikely (and calling it the “lapse rate feedback” would be a misnomer…probably want to call it the “magical emission feedback”). However, if we assume, as per the laws of radiative physics, that there is the expected dependence of emission on temperature then if you don’t have higher temperature at altitude then you won’t have the necessary emission to have the lapse rate feedback. [What the lapse rate feedback say, essentially, for those who don’t know is that if the atmosphere at altitude warms faster than at the surface, the surface doesn’t have to warm up as much in response to a radiative imbalance (due to, say, increasing greenhouse gases) as would otherwise be the case in order for the emission to increase enough to re-establish radiative balance.
cba says:
You are just picking nits. We know that the concept of a single emitting layer is an approximation. But doing the calculation more precisely is not going to change the factor of 300 down to anything remotely close to 1.
gymnosperm says:
No…That is not how it works with radiative transfer in gases. It is complicated but a simplified way to think about it is to imagine that you have a solid surface at some altitude that emits according to the Planck function. In the real system, density will have effects in terms of the exact distribution of emission with altitude but you are not going to magically get more emission than you would by imagining a spherical surface of emissivity 1.
eyesonu:
And, here we have the requisite comment expressing the logical fallacy that because one can find people willing to argue anything on the web, that means the science isn’t settled. In fact, as I noted, what this thread really demonstrates (at least to those who actually know the greenhouse effect is real) is how there are some people who will refuse to accept the scientific evidence no matter how strong when it leads to conclusions that conflict with what they want to believe for other reasons, a fact we already really knew from the evolution – creation debates.
It is a cautionary tale from which much can be learned.
joeldshore says:
July 22, 2012 at 6:30 am
JPeden says:
But since Mainstream Climate Science’s predicted Tropical Tropospheric “Hot Spot” has not eventuated, then according to “the physics” of Mainstream Climate Science’s CO2 = GW hypothesis: there has been no “entire earth system” GW and in particular, no Global Mean Temperature increase.
Joel replies:
“Your statement is very confused. Even if we assume you are correct about the data not showing the “hot spot”, that does not mean that things have not warmed.”
Exactly: according to “Mainstream Climate Science’s” own predictions, the subsequent empirical data effectively reduces to absurdity “the physics” contained within the GCMs – involving the specific hypothesis that [increasing] CO2 = GW, and likewise predicting the empirical existence of a tropical tropospheric “hot spot”.
To begin with, Mainstream Climate Science’s own GCM “physics” did not manage to produce a distinct measurable empirical effect as advertised from increasing CO2 concentrations, while GMT did in fact increase over the period of ~1975-1998.
Likewise, there has been no empirical effect produced or detected from “the physics” of CO2 = GW during the subsequent ~15 yr., as manifestd by the failure of GMT to increase over this period as per “the physics” of continued increases in CO2 concentrations, a failure to warm which was specifically predicted by GCMs to not occur under the CO2 = GW hypothesis – unless of course CO2 concentrations did not increase.
“It just means that the places at altitude in the tropics that were expected to warm more rapidly than the surface have not warmed more rapidly than the surface.”
Yes and finally, the empirical data shows that the CO2 = GW hypothesis failed according to its own [lapse rate + water vapor abetted] predictions from its own “physics” – according to which there also shouldn’t have even been any GW from any cause, because the now claimed to be ‘nonspecific’ sign of any GW “hot spot” simply did not occur as predicted by the mainstream GCM “physics”!
Bottom line in these two cases, Joel: Mainstream Climate Science “physics” hasn’t been able to provide or detect any empirical warming from “the physics” of its CO2 = GW hypothesis.
Henry at Tim
I did not encounter too many experts here, but I am glad you are here! Perhaps you can help me. In the graphic that this post starts off with, we see the spectrum of CO2, but obviously the presentation of the graph looks as if the CO2 causes almost all the absorption at 14-16 in the atmosphere. In reality this cannot be true. The concentration of water vapor alone is ca. 15x greater than CO2, so at the very most, the CO2 cuts off only a little corner of that area of radiation from earth not covered by the absorption by water vapor.. Apart from that, if you look very closely, you can see that there is also absorption of oxygen and ozone in the 14-16. Now I have been posting that it is the variation in ozone that seems to be the cause of global warming and its increase is now the cause of global cooling. The observed global warming and subsequent global cooling all follows on nice parabolic curves, as proven from my results, as reported in my earlier posts here. The question I have is this: is the (weak) absorption by oxygen/ozone at 14-16 caused by the ozone or by the oxygen?
Konrad says:
July 21, 2012 at 6:11 am
JeffC says: July 21, 2012 at 5:15 am
“GHG does not block radiation, it absorbs and then re-transmits … a better term than block would be slows …”
I believe JeffC to be correct. CO2 almost instantly re-radiates the outgoing IR radiation it intercepts, with around 50% of this radiated back towards the Earth’s surface….
_______________________________
That is one of the fallacies. 50% is not radiated back towards earth. Think in 3 dimensions not in 2 dimensions because the CO2 molecule and the earth are three dimensional. Also energy can be transferred via collision to other molecules so the energy headed in the direction of the earth is less than 50%.
The second point brought up by Sleepalot @ur momisugly July 21, 2012 at 4:21 am, is the day/night cycle of off-again/on-again solar insolation and the rapid response of the air and land temperatures to the “switching -off” of the sun’s energy. Sleepalot’s link
Sleepalot @ur momisugly July 21, 2012 at 4:53 am then points out the actual effects of the GHG water vapor on the temperature by comparing high vs low humidity.
Take a good hard look at those two pieces of real world data and ask yourself what it is telling you.
#1. The solar eclipse data tells you the earth & air temperature response (in low humidity) to a change in solar energy is FAST!
#2 The effect of the addition of water vapor (~ 4%) is not to raise the temperature but to even the temperature out. The monthly high is 10C lower and the monthly low is ~ 10C higher when the GHG H2O is added to the atmosphere in this example. The average temperature is about 4C lower in Brazil despite the fact that Algeria is further north above the tropic of Cancer. Some of the difference is from the effect of clouds/albedo but the dramatic effect on the temperature extremes is also from the humidity.
I took a rough look at the data from Brazil. Twelve days were sunny. I had to toss the data for two days because it was bogus. The average humidity was 80% for those ten days. The high was 32 with a range of 1.7C and the low was 22.7C with a range of 2.8C. Given the small range in values over the month the data is probably a pretty good estimate for the effects of humidity only. You still get the day-night variation of ~ 10C with a high humidity vs a day-night variation of 35C without and the average temp is STILL going to be lower when the humidity is high.
This data would indicate GHGs have two effects. One is to even out the temperature and the second is to act as a “coolant” at least if the GHG is H2O.
The latent heat of evaporation could be why the average is 4C lower when in Brazil vs Algeria. As one of the commenters here at WUWT mentioned using temperature without humidity to estimate the global heat content is bad physics.
davidmhoffer, I think this jar would absolutely show the silliness of the CO2 concern. Even 8 blue BBs in a jar of 9,592 yellow BBs will look incredibly sparse in comparison to the 400 red BBs.
Now, create four jars:
Jar #1: 4 blue BBs + (400 + σ) red BBs
Jar #2: 8 blue BBs + (400 – σ) red BBs
Explain that Jar #1 is displaying base case CO2 with slightly higher than average water vapor.
Explain that Jar #2 is displaying the supposed catastrophic case for CO2 with slightly lower than average water vapor.
Now, ask the observer if it makes sense that CO2 could have a measurable effect (even 1°C) on sustained global temperatures given the fact that it’s quite obvious that any conceivable amount of blue BBs is a rounding error compared to the red BBs.
Ask if it makes any sense to conclude that earth’s climate system could be that sensitive to the blue BBs despite the fact that the system demonstrates relative stability during periods that the red BBs constantly fluctuate on an enormous scale (in comparison).
But an environmentalist has convinced me that if I remove all of the insulation the average temperature inside will go down, so I do. What do you think will happen to the average temperature?
Do you have big, transparent, south-facing windows with a thin IR-blocking film in them so that sunlight streams in every day, heating the floors? Because if you do, I suspect the environmentalist might be right.
The Earth’s surface and atmosphere aren’t heated from the top down (outside in) — they are heated from the inside out because a significant fraction of sunlight directly heats the surface after passing through the atmosphere without being either absorbed or reflected. Which makes most of the metaphors that have been proposed in this thread completely wrong.
Treat sunlight delivered to the surface correctly as a heat source heating an object inside a vacuum flask and cooling only by radiation, and it becomes quite clear that absorbing from a high temperature black body (the Sun) in the visible band, but radiating from a much cooler black body (the Earth) in the IR band, creates a situation where adding insulation in the form of atmospheric CO_2 that blocks direct radiation from the surface in a chunk of the IR spectrum can only raise the temperature of the surface to compensate until equilibrium is maintained, energy in equals energy out.
The Earth is not a closed system. It receives a huge amount of energy in the form of sunlight every day. It loses all of this energy, every day! At least on average. If you reduce the rate of loss in one channel without altering the input, you MUST increase the rate of loss in all other channels until the two balance. The only thing that can increase the rate of radiative loss in the unblocked channels is higher temperature, since the outgoing radiation is at least mostly thermal in the first place.
rgb
wobble
you totaly and completely missed the point of the explanation
Here’s your answer: Less than what the world is spending now.
Try to remember that technological advances accelerate over time. If you believe that the equivalent of $30 trillion is a reasonable bet against probability weighted negative effects of warming and that such warming will start to negatively impact the world’s wealth within 30 years, then it’s irrational to start spending $1 trillion per year now.
And with this, I agree! In fact, I make the same argument! That was easy, right?
My usual argument is that the best thing to do about CAGW even if it might be right is almost nothing! Now, at any rate. Continue to invest heavily in research that can bring alternative energy resources to maturity, because this sort of research will almost certainly have a positive ROI regardless of CAGW, and it a trivial fraction of your trillion a year. Invest modest amounts in pilot projects that help us work out the scaling and true costs of these technologies as they go along. Is a solar updraft tower a viable, competitive, feasible energy producer? It will be difficult to say without building not just one, but several of them, using what we learn from one to build the next one. Is a solar updraft tower built at the top of a mountainside collector a viable competitive energy producer? Maybe, but first one has to build a few regular towers without the mountainside to learn the engineering to find out.
These sorts of things cost at most a few billion a year, and many of them have trillion dollar payoffs if they hit. What I don’t like — and at least some climate scientists who believe in AGW and at least some negative sequellae aren’t thrilled with either — is carbon trading, large scale subsidies of immature technologies that don’t have a clear positive ROI, and so on.
Personally, I think that Solar PV technology is going to be a no-brainer within roughly 8 to 10 years, with or without further government funded research, faster with than without. I do argue with those that for some reason seem to oppose it (I can’t imagine why). Solar is damn close to break even as far as ROI is concerned even without a subsidy, and a factor of 2 improvement in cost per watt will put it solidly into the black for a large fraction of the United States without a subsidy. Or a breakthrough in energy storage would do it. Or both would do it fast, with a lot of money on the line. Even if I’m wrong what’s the cost? A teensy fraction of the money pissed away in Iraq, money that could have funded a HUGE amount of solar energy (even at a subsidized loss) for a far greater ROI. Solar should be implemented when implementing it is profitable; I just think it will be profitable soon.
If it is, then who cares about CAGW? Long before carbon trading has any useful effect (probably infinitely long given a latter date of “never”) the problem will be resolving itself simply because it is cheaper that way and makes people more money, or the evidence for CAGW will be unmistakable and we’ll be taking more directed (and more justified) action.
rgb
Try this: in a jar the height of the atmosphere put clear BBs and blue BBs. 8 blue BBs for every 10,000 clear. Look at the jar from the top. Kinda blue, ain’t it? Do you suppose there is any way a photon originating at the earth’s surface could reach space without colliding with a blue BB, or two?
The top physicists scorn the theory – the conscientious layman has to work his way through from ground zero doing all of the tedious maths along the way.
They do? Damn, why didn’t I know that. Who are these “top physicists” that scorn the reality of the greenhouse effect?
Not that there probably aren’t one or two — finding a dozen physicists that agree on anything complex can be challenging. But if you showed any of them the TOA spectrographs they’d change their minds. I mean, physicists aren’t usually stupid.
rgb
That’s an inaccurate assumption. I didn’t miss anything. I understood your point about a long jar being opaque looking from the top down, and I certainly didn’t say that you were wrong. I merely stated that your jar would absolutely show the silliness of the CO2 concern.
wobble;
I merely stated that your jar would absolutely show the silliness of the CO2 concern.
>>>>>>>
In that case, you totaly and completely missed the point.
Please Bucky Cochrane, QuantumPhysicistPhil, RGB, say more. This seems to play into the misinterpretation of Tyndall’s experiment that began this entire mess of misinterpretation. If any one will read just page 13 and 14, just two pages, and get a vivid mental image of Tyndall’s experiment you will see that he merely proved that carbon dioxide and other ‘greenhouse’ gases absorb and emit LW radiation. That is a fact I think anyone versed in physics agrees.
http://books.google.com/books?id=RT9-bP9p9zQC&pg=PA13&dq=Tyndall
But look deeper, if Tyndall would have used a rock salt 3 foot by 3 inch tube instead of the tube of IR absorbing glass, that same experiment would have reported entirely different results, the gas in the tube would have cooled, not warmed. Then only a portion of the LW from the source copper plate would have been reaching the far thermo-piles on either side of the far copper plate.
Now I admit since the atmosphere is more or less homogenous horizontally, but radiation is line-specific unlike the glass that absorbs then re-emits in a gray body type spectrum redistributing that energy spherically in any direction so the factor of conversion to thermal would be greatly different in the two cases.
See anything there?
Don Monfort says:
July 22, 2012 at 8:49 am
Does this clown think that the warmer body is transparent to the radiation from the colder body? Does it pass through without any effect? Or it detours around the warmer body? Bounces off?
Suspend a heating element in a vacuum chamber and heat it to a constant temperature of 500 F. Suspend another element near the first and heat it to 300 F. Work on that.
=========================================================
Yeah, I know this tale: http://www.drroyspencer.com/2010/07/yes-virginia-cooler-objects-can-make-warmer-objects-even-warmer-still/ .
Inexperienced readers might get the impression that it is real, but it is not. It is just a so called “thought experiment” with no reference to a real genuine falsifiable scientific experiment. Which leads to the conclusion that that notion has never been proven. It was just an “explanation” of a fiction.
As for “Work on that”, it looks rather typical for warmists: when asked for experimental proofs of their claims, they either say it is not necessary because it is so in theory or suggest the opponents do the experiment. Or they give “explanations” of what they have yet to prove. Unbelievable.
So, radiation from a colder body directed to the warmer body is radiation, but no warmist has been able to present a real falsifiable scientific experiment proving that this sort of radiation can warm the warmer body (or slow down it’s cooling, whatever). in the energy of each and every photon, and those photons are indeed absorbed by pretty much anything capable of absorbing photons at those frequencies, even the cold liquid helium, by scattering light back onto an object, adds energy to it compared to the energy that would be there if there were no cold mirror, no cold helium.
in the energy of each and every photon, and those photons are indeed absorbed by pretty much anything capable of absorbing photons at those frequencies, even the cold liquid helium, by scattering light back onto an object, adds energy to it compared to the energy that would be there if there were no cold mirror, no cold helium.
Since I’m not a “warmist”, technically this is true. However, I’ve suggested two or three real falsifiable scientific experiments that you can do in the privacy of your own home that show that reflected radiation can lead to a higher equilibrium temperature of a body being warmed and in dynamic equilibrium with radiative heat loss.
But you don’t want to actually perform such an experiment, not even if it is simple enough for a high school student to perform.
Here’s an even simpler experiment. Visit this website:
http://en.wikipedia.org/wiki/File:2_Helium.png
The picture, you will note, is alleged to be a small container of liquid helium, at a presumed temperature of perhaps 4K. It is, you will note, really really cold.
Yet we can see it! Oh, my, gosh! Photons of visible light are somehow being scattered by this helium and making it to our eye, where they — gasp — warm the eye relative to the temperature it would have if the helium were replaced by nothing. In fact, we can see a fire and feel its heat even if the light we see, spectrally emitted from a hot system is reflected from a very cold mirror indeed. Since the light we see reflected in a cold mirror or scattered from cold liquid helium carries
Greg, sooner or later you’re going to have to realize that you can actually do experiments that falsify your bizarre belief that cold things can’t affect the rate that things cool (and hence, affect their equilibrium temperature when they are being warmed). Personally, I don’t understand how you could really end up thinking that. Spend a single windless night in a desert, sleeping under the stars, then come to North Carolina and spend a windless night in my back yard at the ocean, sleeping under the same stars with a relative humidity of maybe 60% or 80%. Look at the temperature differential almost anywhere at night with and without overhead clouds. I learned about the greenhouse effect in boy scouts at the age of 12, back in the 60s when there wasn’t the slightest bit of concern about global warming and if anything, there was talk of a return of glaciation. It was expressed as a simple rule — nights with cloud cover are almost always warmer (on the ground) than clear, dry nights.
In both cases the air overhead is cooler than the ground, Greg. The clouds are cooler. The air is cooler. You have to go up to the thermosphere before you start to find air that is warmer, where the “warmer” air is so thin that nobody cares. Yet we have a readily observable, systematic, reproducible temperature difference on the ground, one that transcends wind, conduction, convection, evaporation — all things being measurably equal on the ground, a cloudy night will be warmer than a cloudness night. I don’t believe you could have lived long enough to learn to write without observing this yourself countless times.
This one systematically falsifiable observation is all it takes to refute you: So, radiation from a colder body directed to the warmer body is radiation, but no warmist has been able to present a real falsifiable scientific experiment proving that this sort of radiation can warm the warmer body (or slow down it’s cooling, whatever).
Except that colder clouds overhead seem to warm the ground or slow down its cooling or whatever. They also backscatter even visible light. Mere humidity — it doesn’t even require actual clouds — warms the ground or slows its cooling or whatever. Water is a powerful greenhouse gas — much more important than CO_2 at least once the CO_2 warming (which isn’t very great) is saturated.
It is an undeniable fact that radiation trapped, reflected, or scattered by a colder body carries energy. It is a fact that energy, steadily being delivered to an object, will cause it to be warmer than it would without the delivery of energy, all things being equal. There is nothing in the second law of thermodynamics applied to open, non-equilibrium systems that prevents heat from flowing from a warm object to a cold one more slowly if the cold object warms up (but remains colder than the warm object). Indeed, even straight up conduction will proceed more slowly and the warm object will be warmer at all times starting from identical initial conditions even without an active heat source there.
I fail to see your argument. What you are asserting is falsified by simple, everyday experience and common sense. Of course a colder body can affect the cooling rate of a warmer body, a statement that is logically identical to the things that you claim no real experiment can show. They show little else!
How they affect it and by how much they affect it requires analysis of the physics involved. But you can’t even start to participate in the grown-up arguments until you get over your childish assertion that the heat equation itself is somehow wrong, that modulating the temperature of a cold reservoir in contact with a warm reservoir doesn’t affect the rate that the warm reservoir cools.
Once you accept the fact that a cold gas can most certainly affect the rate of radiative cooling of a warmer surface the same as any other physically coupled thermal reservoir, we can move on. And that will happen when, in 2050? When hell freezes over? When you are fiddling with your car battery and it delivers a powerful shock across your frontal lobes that shocks you momentarily into sanity a la Stanley Stupid? When you find yourself stranded in your car on a -20F night and your space blanket — in spite of being a thin sheet of aluminized mylar more or less in equilibrium with the -20 F outside air, in spite of the fact that the car is closed and there is basically no difference in convective cooling with or without the blanket — saves your life by reflecting your own body heat back at your body, thereby slowing your rate of energy loss?
Who knows?
So Dr, Spencer is a warmist too. You are thick, Greg. Get yourself two radiant heaters from Costco. Turn one on high and the other on low. Stand between them. They both warm you. Right Greg? Remove yourself from between the heaters. Now they are warming each other. Oh, but that can’t be. That would dispel your willful ignorance. Thick types like you are the best friends the warmists have.
rgbatduke says:
July 22, 2012 at 4:22 pm
Greg, sooner or later you’re going to have to realize that you can actually do experiments that falsify your bizarre belief that cold things can’t affect the rate that things cool …
=======================================================
Yeah, first of all you omitted the word “warmer”: “warmer things”. Of course, it is just a coincidence that you have distorted the central point you apparently can not prove.
As for my alleged belief, I am completely open to any conclusion. All you need is to prove your point. Until now you have failed to provide any link to a real genuine falsifiable scientific experiment. Maybe you can not for whatever reason, but let us hope (other) warmists will come to help. Or you can keep trying to sell your narratives as science, no problem, people are not stupid and will understand what is going on and what “climate science” is based on. Please, do not go away, we need you.
michael hammer says:
July 21, 2012 at 3:52 am
The 8th comment in the thread. Should have stopped reading, after that one.