Guest post by Reed Coray
The following example illustrates the issues I have with reasoning often used to argue that increasing the amount of CO2 in the Earth’s atmosphere will increase both the Earth’s surface temperature and the Earth’s atmosphere temperature. Immediately following is a direct quote from URL
http://www.school-for-champions.com/science/heat_transfer_earth.htm
“The present situation is that there has been an increase in infrared-absorbing gases in the atmosphere, such as carbon dioxide (CO2) and methane (CH4). Energy that would normally escape into space is absorbed by these molecules, thus heating the atmosphere and spreading through convection currents. The average temperature of the atmosphere has increased 0.25 °C since 1980, mainly attributed to an increase in infrared-absorbing gases in the atmosphere.”
Although the above statement makes no direct reference to Earth surface temperature, I believe it carries the implication that greenhouse gases in the Earth’s atmosphere increase the Earth’s surface temperature.
I make two comments: the first is relevant only if the above implication is valid, the second is relevant independent of the validity of the implication. First, placing matter adjacent to a warm surface such that the matter is capable of absorbing/blocking radiation to space from the warm surface can lead to a decrease in the warm surface’s temperature. Second, increasing the amount of the absorbing/blocking matter can lower the temperature of the absorbing/blocking material.
Take for example an internal combustion engine whose metal surface is exposed to a vacuum. In addition to doing useful work, the engine produces thermal energy (heat). That thermal energy will produce a rise in the temperature of the engine’s surface such that in energy-rate equilibrium the rate energy is radiated to space from the engine’s surface is equal to the rate thermal energy is generated within the engine. By attaching radiating plates to the engine’s surface, some of the energy radiated to space from the engine’s original surface will be absorbed/blocked by the plates; but because thermal energy can be transferred from the engine to the plates via both radiation and conduction, the temperature of the engine’s original surface will be lowered. This is the principle of an air-cooled engine[1]: provide a means other than radiation of transferring heat from an engine to a large surface area from which heat can be removed via a combination of conduction, convection and radiation, and the engine’s surface temperature will be lowered.
If plates at a temperature lower than the original engine surface temperature are attached to the engine, it’s true that the temperature of the plates will increase to establish energy-rate equilibrium. Once energy-rate equilibrium is established, however, increasing the plate radiating area (adding additional matter that blocks more of the energy radiated from the original engine surface) will likely lower the plate temperature.
Thus, blocking the amount of surface radiation escaping to space does not necessarily increase the surface temperature; and increasing the amount of radiation blocking material does not necessarily increase the temperature of that material. In both cases (the Earth/Earth-atmosphere and the internal combustion engine in a vacuum), the heat eventually escapes to space–otherwise the temperature of the Earth’s surface and the engine would continue to rise indefinitely. The difference isn’t that the energy doesn’t eventually escape to space (it does in both cases), the difference is in the path the energy takes to reach space. The amount of generated thermal energy in conjunction with the path the thermal energy takes to get to space determines temperatures along the path; and adding more material may increase or decrease those temperatures. To say that “Energy that would normally escape into space is absorbed by these molecules, thus heating the atmosphere…” by itself is unwarranted; because an equivalent statement for the case of adding extra plate material to the engine would be “Energy that would normally escape to space from an engine with small attached plates is absorbed by additional plate material, thus heating the plates…” For air-cooled engines, this statement is not true—otherwise the plate surface area of air-cooled engines would be as small as possible.
It’s fairly easy to visualize why (a) adding thermally radiating plates to an air-cooled engine might decrease the engine’s surface temperature, and (b) increasing the area of the radiating plates might decrease the plate temperature. It’s not so easy to visualize, and may not be true, why (a) adding greenhouse gases to the Earth’s atmosphere decreases the Earth’s surface temperature; and (b) increasing the amount of atmospheric greenhouse gases lowers the temperature of the Earth’s atmosphere. I now present one possible argument. I do not claim that the argument is valid for greenhouse gases in the Earth’s atmosphere, but I do claim that the argument might be valid, and can only be refuted by an analysis more detailed than simply claiming “Energy that would normally escape into space is absorbed by these molecules, thus heating the atmosphere.”
If we assume that (a) matter cannot leave the Earth/Earth-atmosphere system, and (b) non-greenhouse gases radiate negligible energy to space, then for a non-greenhouse gas atmosphere the only way thermal energy can leave the Earth/Earth-atmosphere system to space is via radiation from the surface of the Earth. The rate radiation leaves the surface is in part a function of both the area and temperature of the surface. For a greenhouse gas atmosphere, energy can leave the Earth/Earth-atmosphere system to space both via radiation from the Earth’s surface and radiation from greenhouse gases in the atmosphere. Suppose it is true that the density of greenhouse gases near the Earth’s surface is such that radiation emitted from low-altitude greenhouse gases does not directly escape to space, but is in part directed towards the Earth’s surface and in part absorbed by other atmospheric greenhouse gases. As the atmospheric greenhouse gas density decreases with increasing altitude, radiation emitted from high-altitude greenhouse gases can directly escape to space.
Now it’s not impossible that since (a) in addition to radiation, heat is transferred from the Earth’s surface to greenhouse gases via conduction, and (b) convection currents (i) circulate the heated greenhouse gases to higher altitudes where energy transfer to space can take place and (ii) return cooler greenhouse gases to the Earth’s surface, that the process of heat transfer away from the Earth’s surface via greenhouse gases is more efficient than simple radiation from the Earth’s surface. Many engines are cooled using this concept. Specifically, a coolant is brought into contact with a heated surface which raises the coolant’s temperature via conduction and radiation, and the coolant is moved to a location where thermal energy transfer away from the coolant to a heat sink is more efficient than direct thermal energy transfer from the heated surface to the heat sink.
One way to realize increased thermal transfer efficiency would be to use a coolant, such as greenhouse gases, that efficiently radiates energy in the IR band (i.e., radiates energy at temperatures around 500 K). Another way would be to spread the heated coolant over a large surface area. Since surface area increases with increasing altitude, thereby providing expanded “area” (in the case of a gas, expanded volume) from which radiation to space can occur, it’s not clear to me (one way or the other) that greenhouse gases won’t act as a “coolant” reducing both the temperatures of the Earth’s atmosphere and the Earth surface.
[1] It’s true that for most air-cooled engines the main transfer of heat from the engine plates is via a combination of (a) conduction of heat to the air near the plates, and (b) convection that replaces the warm air near the plates with cooler air. To aid this process, a fan is often employed, or the engine is located on a moving vehicle and the vehicle’s motion through an atmosphere provides the flow of air across the plates. Although conduction/convection may be the primary means of heat dissipation from the plates, radiative cooling also dissipates heat.
Ian W;
This would be an extremely simple undergraduate experiment. Set up a chamber of IR transparent material with a heated base and with say 10 liters of a mixture of N2 80% and O2 20%. >>>>
I would refer you to a similar actual experiment by Heinz Hug:
http://www.john-daly.com/artifact.htm
Note the link to the zip file with criticisms of the experiment which is well worth reading and shows that while the experiment is of value, it doesn’t allow us to draw firm conclusions about order of magnitude effects in the atmosphere.
Eli Rabett says: July 21, 2012 at 5:39 am
You wrote:
“Increasing concentrations of GHGs raises the altitude that GHGs can radiate to space in the blocked regions of the spectrum
Because of the lapse rate, the higher you go in the troposphere, the lower the temperature
This slows down the rate at which the Earth emits to space because it is now radiating at higher, colder altitudes”
Eli, I believe you are wrong.
First, any “slow down” in the rate the Earth emits energy to space must be transient–i.e., it can’t last forever. Otherwise, assuming an unchanged input rate of energy, a “slowed-down” energy rate implies an accumulation of thermal energy with time. At some point the outgoing rate must equal the incoming rate or all hell breaks loose. Thus, at some point the “higher/colder atmosphere” must radiate energy at the same rate as the “lower/warmer atmosphere.”
Second, If you surround an active sphere (i.e., a sphere that internally generates thermal energy at a constant rate) with a co-centered, non-touching, spherical annulus where a vacuum exists everywhere else, (a) the temperature of the active sphere will rise above what its temperature would be in isolation, (b) the altitude at which heat radiates away from the sphere/annulus system will be increased and will increase as the outer radius of the spherical annulus increases, (c) the temperature through the spherical annulus will decrease with distance from the common center (i.e., the lapse rate through the spherical annulus will have the same sign as the Earth’s atmospheric lapse rate), and (d) the increased altitude will result in a lower heat-producing-sphere temperature. As with the Earth/Earth atmosphere, Thus, it’s true that heat radiated to space from the sphere/annulus system is radiated from a higher and colder surface, but it’s also true that the surface is also larger. Depending on the thermal conduction properties of the annulus, it can be shown that if the outer radius of the annulus is larger than a threshold radius, increasing the radius of the outer annulus results in a decreasing active sphere surface temperature. The surface temperature of the active sphere won’t drop below the active sphere’s surface temperature in isolation, but as the outer radius of the spherical annulus approaches infinity, the temperature of the active sphere’s surface in the sphere/annulus system will approach the active sphere’s surface temperature in isolation.
If for the above system, you connect the active sphere and the spherical annulus with highly thermally conduction rods, not only will all of the above be true, but depending on the thermal conduction properties of the rods (and the spherical annulus), the temperature of the active sphere’s surface can be made to be lower than the active sphere’s surface temperature in isolation.
I have written a paper that I believe proves the above assertions. That paper is, however, too long (approximately 22 pages) and too mathematical for posting as a guest post. However, I’d be happy to send that paper in PDF form to Anthony (or anyone else) capable of putting the paper on the net and provide a “link” to the paper.
O H Dahlsveen;
A thermos-flask cannot work if heat could radiate through a vacuum.
>>>>>>>>>>>>>
Good to know. That being the case, the earth ought to reach absolute zero in short order as there is no way for the sun to heat the earth due to all that vacuum between the two.
@ur momisugly Baa Humbug says:
July 21, 2012 at 5:57 am
Have found this page which may be the precursor to the missing pdf.
http://jinancaoblog.blogspot.com.au/2012/01/blog-post.html
Why the scientific basis of greenhouse gas warming is incorrect
“First, placing matter adjacent to a warm surface such that the matter is capable of absorbing/blocking radiation to space from the warm surface can lead to a decrease in the warm surface’s temperature.” It will always lead to an INCREASE in the surface’s temperatue..
In the case of a car radiator, and a Dimetrodon’s sail, one is in effect increasing the radiating surface area- leading to an increase in the rate of cooling. Adding greenhouse gases does NOT increase the radiating surface area of the atmosphere. The atmosphere won’t work like a radiator
gymnosperm:
That is actually not hard to estimate. The idea is to compare the fractional decrease in W/m^2 of the emission as one goes up in the atmosphere (due to the lower temperature) to the fractional increase in surface area due to the larger radius. In mathematical terms, one should compare (1/P)*(dP/dh) to (1/A)(dA/dh) where P is the intensity of emission in W/m^2, A is the surface area of a sphere, and h is the height of the emitting “surface”.
Working through the math [and using the Stefan-Boltzmann Law], one gets that (1/P)*(dP/dh) = (dT/dh)*4/T where T is the absolute temperature. (1/A)(dA/dh) is approximately 2/R_earth where R_earth is the radius of the Earth. A typical lapse rate in the atmosphere is dT/dh = -6.5 K per km and T = 255 K at the emitting level. Putting in numbers, I get that that
(1/A)(dA/dh) is about 0.00031 per km and (1/P)*(dP/dh) is about -0.10 per km, i.e., the emission per unit area decreases about 10% if the emitting level increases by 1 km while the area goes up by 0.03%.
In other words, the effect of lower temperature is about 300X as important as the effect of increasing surface area.
Bill Yarber says:
Time delays are not the correct way to think about situations that involve the continuous emission or absorption of energy at some rate. If someone turned the sun on for a few seconds and then turned it off again, you would be correct that the effect of adding GHGs would be to delay the cooling down but not the final temperature in the absence of a sun.
However, what is relevant to the case of a sun that emits energy to the Earth at a certain rate is that the Earth then has to emit energy back into space at that same rate. For a given surface temperature, GHGs reduce the rate at which energy is emitted to space (because the emission that successfully escapes to space comes from higher levels of the atmosphere where, because of the lapse rate, the temperature is colder). Thus, if you increase the levels of GHGs, the Earth will now be emitting energy at a slower rate than it is absorbing energy. This causes the Earth to warm…In fact, it warms until the radiative balance between emission and absorption is re-established.
If the theory of reradiated heat were to be true then warm liquids placed into a vacuum flask, with its mirrored internal surfaces, would raise the liquid’s temperature by a considerable amount.
This is so, so, irrelevant. The Earth isn’t a vat of passive warm liquids. It is a vat of liquids being actively heated. I know that you know this because I have told you personally over and over.
Now take your flask. Fill it with a liquid and add a heater that turns on for 12 hours a day. Consider it with and without the mirrored surface. Which one on avergage is warmer? Uh-huh. I thought so.
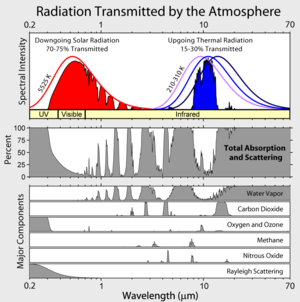
The point of the two figures showing insolation peaked around the visible part of the spectrum, where the atmosphere is nearly transparent, and radiative loss down in the IR is that the Earth is precisely a vacuum flask receiving additional heat every day. Surrounding it with a mirrored “surface” in the form of GHGs that don’t block the incoming heat but do block a fraction of the outgoing het absolutely causes differential warming just exactly the same way that your vacuum flask example would without any question whatsoever be warmer with the mirrored internal surface if there is heat production inside.
rgb
I don’t like analogies since they are hardly ever close enough to reality. They may tell you where to look but they don’t prove anything.
In this case the physics is very clear.
The earth ( by which I mean the earth and atmosphere combined) can only lose heat by radiation.
For the earth’s temperature to remain constant the energy from the sun absorbed by the earth must equal the energy radiated to space.
The energy radiated to space will depend on the temperature of the radiating element and it’s emissivity.
The energy radiated to space is spread across a range of wavelengths from about 3 micron to 70 microns.
If there were no atmosphere all wavelengths would be radiated from the earths surface.
Because of greenhouse gases, energy at wavelengths other than those within the atmospheric window (around 10 microns) are radiated from various levels in the atmosphere.
Wavelengths characteristic of water are radiated from all levels with an average temperature of around 250K whilst wavelengths between 14 and 18 micron (where CO2 absorbs) are radiated from the tropopause at a temperature of about 200K
The energy from the sun is in the UV/optical and very short IR range (less than 3 micron) so the presence of greenhouse gases does not significantly affect the energy absorbed so they do not alter the TOTAL amount of energy radiated either. The energy still has to balance.
Since an earth without greenhouse gases would radiate everything from the surface, the surface temperature would need to be such that it radiated the same as in the current scenario where the various wavelengths are emitted from molecules at temperatures ranging from about 288K (surface) to 200K (tropopause).
An estimate around 260K seems very plausible.
The global warming argument is that if you increase the level of CO2 further the altitude at which radiation in the 14 – 18 micron range takes place increases due to the reduced mean free path of the photon. Higher means colder so the radiation is less. Therefore the temperature at the surface has to increase to compensate.
However at the tropopause the temperature does not increase with altitude so this simple concept does not work. For AGW to be proven they will need to show a movement of the tropopause to greater altitude to allow for the temperature of the tropopause to reduce. I am not aware of any measurement demonstrating this.
So please stop trying to argue against the simple physics of greenhouse gases which is very sound and instead focus on the real issue of what the very very complex system of gases which makes up our atmosphere actually does in practice.
We need less discussion of simplistic analogies and more measurements.
And more Willis’s to do the analysis for us!
KR says:
July 21, 2012 at 9:29 am
See Santer et al 2003 (http://www.sciencemag.org/content/301/5632/479.short) among others:
Observations indicate that the height of the tropopause—the boundary between the stratosphere and troposphere—has increased by several hundred meters since 1979… This positive detection result allows us to attribute overall tropopause height changes to a combination of anthropogenic and natural external forcings, with the anthropogenic component predominating.
—————
Sorry, not very plausible. One has to monitor tropopause globally.
“Observations indicate…”
Tropopause fluctuates strongly and is at different height at different places. This paper is too weak.
Thus, if you increase the levels of GHGs, the Earth will now be emitting energy at a slower rate than it is absorbing energy. This causes the Earth to warm…In fact, it warms until the radiative balance between emission and absorption is re-established.
The problem is that it doesn’t “cause the Earth to warm”. The sun causes the Earth to warm, almost exclusively. The GHE causes the Earth to lose heat from the Sun more slowly and hence be warmer than it would have been without it.
Sorry to be picky, but half of the debate is people who do not understand that what you mean — and what a world of climate scientists mean — is not the literal meaning of the words you say. GHGs do not warm anything. They slow the cooling of something being actively warmed elsewhere, by other means, and just like the insulation in your walls makes you house warmer given a furnace inside than it otherwise would be, the Earth end up being warmer with them than it would be without them.
Clearly there are people that are confused by precisely this point. John Marshall, for example, with his “flask of warm” (but not actively warmed) liquid. Insulation doesn’t keep a house with no furnace warmer than the outside — at most it keeps at a uniform equilibrium temperature with the outside.
Yes, it gets very tedious explaining to them over and over again how the GHE works. But it isn’t made easier by claiming that it “warms” anything. It — as you do say — simply elevates the mean temperature until equilibrium is re-established between an internal source of heat delivered directly to the Earth’s surface by unblocked, non-reflected sunlight and heat loss via radiation out of an imaginary surface that contains the Earth and its atmosphere.
Space blankets “warm” humans exactly the same way. They don’t warm anything at all — no blanket does. But they do reflect back a fraction of the heat being radiated away from the human body. Since the human body continuously produces heat that has to be lost, this raises its equilibrium temperature. Insulation in the attic “warms” your house this way. It doesn’t actually warm anything — its a piece of inert spun fiberglass wool with a radiative shield — but it does slow the transmission of heat from the inside of your house to the outside, so that the inside is warmer when your furnace or other heat sources inside are turned on. It can be reduced all the way down to the heat equation itself where the mechanism of heat transmission is left completely ambiguous. If you actively heat a rod at one end, and hold the other end in a “bath” at a constant cooler temperature, then raising the thermal resistance of the pathway(s) in between by any means whatsoever — preventing any fraction of conduction, convection or radiation — will raise the temperature of the end being warmed relative to what it would have been with a better conductive pathway.
That, after all, is why we tend to cook in metal pans instead of asbestos ones. It is why a hundred watt light bulb works fine to heat an EZ-bake oven. It is why one is hotter lying around in the sun than one is in the shade, all things being equal. It isn’t rocket science, and it is utterly silly to claim that it doesn’t take place, that greenhouse gases overhead are incapable of raising the surface temperature relative to what they would have been without them, given active heating of the ground surface.
rgb
….has increased 0.25 °C since 1980, mainly attributed to an increase in infrared-absorbing gases in the atmosphere.”
Henry says
according to my sample it was +0.4 degrees C since 1980, and it was all due to natural causes….
http://www.letterdash.com/henryp/global-cooling-is-here
hint: it has to do with sun -UV-O2-O3 cycle
we still have about 33 years of cooling left….
“The average temperature of the atmosphere has increased 0.25 °C since 1980, mainly attributed to an increase in infrared-absorbing gases in the atmosphere.”
The average temperature between 1910 and 1945 increased by 0.7°C.
http://www.woodfortrees.org/plot/hadcrut3vgl/from:1910/to:1945
Until this, probably natural and twice as large increase has been reasonably explained by the models or orthodox climate scientists, please stop attributing the recent 0.3 °C increase to “greenhouse gases”.
Second, the average temperature decreased by 0.15°C since 2001
http://www.woodfortrees.org/plot/hadcrut3vgl/from:2001/to:2012
I ask where are the “greenhouse gases”?
davidmhoffer says on July 21, 2012 at 10:13 am:
“O H Dahlsveen;
A thermos-flask cannot work if heat could radiate through a vacuum.
>>>>>>>>>>>>>
Good to know. That being the case, the earth ought to reach absolute zero in short order as there is
no way for the sun to heat the earth due to all that vacuum between the two.”
=============
The Sun is radiating a form of energy that has the ability to penetrate the earth’s atmosphere. Upon hitting the top of the surface this energy interacts with the surface atom clusters or molecules. The electrons in the said atoms increase their speed resulting in increased molecular friction. A product of friction is always what we call “Heat”.
Therefore heat is a product of energy-use, it can no more be emitted as radiation than speed can.
Remember radiation cannot be seen by the human eye. Nor can it be “seen” by any modern derivative of the “Thermopile”. All that can be “seen” is the source of radiation. The heat-source can be seen as light. Radiation, at certain wave-lengths, from the Sun only turns into “Light” upon interaction with the atmosphere. – Once again read Tyndall and Fourier.
By the way, sarcasm becomes no-one.
Convection does not occur without radiation or conduction first but it is mostly conduction. Air in direct contact with a hot surface is warmed by conduction. Once it is warm, it begins to convect which pulls cool unheated air in to replace it, which is itself warmed by conduction, convects away, pulls in more unheated air, etc. Any heat loss by radiation by the surface is tiny compared to the heat lost to conduction. Radiation does not become the primary heat loss mechanism until one gets to the top of the troposphere and the air can no longer convect upwards (because the air above is warmer). BUT, what this does is in effect increase the surface area from which radiation is occurring. Consider the surface area of a sphere the size of the surface of the Earth. Now consider the surface area of a sphere the size of the tropopause. That heat is spread over much more area from which to radiate.
Henry@rgb
we had this same argument before
show me, EXACTLY, how much cooling and how much warming each GHG causes?
http://www.letterdash.com/HenryP/the-greenhouse-effect-and-the-principle-of-re-radiation-11-Aug-2011
including the amount of cooling CO2 causes by taking part in photo synthesis
which I guess has been increasing explosively since the last estimate was taken in 1974
Anyone who has done any work with flight qualified electronics, for example, knows that the rule of thumb is that you lose about 10% efficiency for every thousand feet of altitude for a heatsink. Over 10,000 feet, convection cooling is just about useless and the dominant heat loss becomes radiative. Air cooled engines don’t work well at altitude because the air becomes thin. But at the surface, heat lost due to radiation is much less than the heat loss due to conduction to the air and the air then transporting the heat away by convecting.
“Eli Rabett says:
July 21, 2012 at 5:39 am
Sorry Reed, you miss the point, but you are not alone. Eli remembers eminent analytical chemists who missed the same point in print many years ago.
What happens is that effectively GHG block radiation from reaching space across most of the IR.
This includes IR emitted from the GHGs low in most of the troposphere.
Increasing concentrations of GHGs raises the altitude that GHGs can radiate to space in the blocked regions of the spectrum
Because of the lapse rate, the higher you go in the troposphere, the lower the temperature
”
You’ve evidently spent too much time as a character in children’s stories.
first off, ghg blocking is not most of the IR or most of the energy and that’s water vapor. Increasing concentrations of ghgs increase the absorption but also increase the emission (to a slightly lesser extent). As one goes up in the atmosphere, the pressure, and hence the pressure broadening, decreases along with temperature. That affects the bandwidth of emission/absorption line so higher up there is always more absorption very close to the line center but further down there is a significant smearing of the energy over a broader bandwidth which is not going to be affected at the higher altitudes. The lapse rate is nothing more than conservation of energy so it will adapt based upon the power flow in and out of any altitude. That power flow is the total in and out for radiation, conduction, and convection.
What is absorbed in the clear sky is only a fraction of the surface emission and since line width (and peak intensity) depends upon pressure, there are contributions from all heights involved in an emission/absorption line. The net result for a 288.2k surface is about 279 W/m^2 of power leaving the Earth through clear skies (including emissions from the atmosphere). A doubling of co2 would reduce that by about 3.7W/m^2 down to 275 W/m^2. To refresh your memory, the average incoming solar power is 239 W/m^2 after accounting for about 0.3 average albedo which leaves a 40 W / m^2 deficit that must be dealt with by clouds, aerosols, and atmospheric scattering. Otherwise, our average T would have to be reduced to around 280 K for energy balance, reducing the average T to around 45 deg F from around 56 deg F.
As for some some sort of characteristic radiating altitude, it doesn’t exist. Most comes from the surface and, where present, the cloud tops. The lines include components throughout the atmosphere, including some from the stratosphere and beyond. What is actually emitted at some sort of ‘characteristic’ radiating alitude is actually almost nothing. First off, no component from the continuum away from the ghg spectral lines exists for clear skies. Next, the atmosphere above some parcel of air at this ‘characteristic’ altitude will be almost the same pressure and will absorb virtually all of the emissions and will reradiate almost the same amount and so forth as the pressure broadening and temperatures decrease with altitude.
When one looks at the system more as a whole, things get far worse for CAGW. What becomes apparent is that there is a strong net negative feedback at work which precludes the possibility of a positive net feedback.
If one tries to push watervapor as a strong feedback, they come up very short. If one claims that some slightly increased temperature will reduce the cloud cover and hence cause an albedo related positive feedback, one is faced with the severe problem of claiming we are at a maxima where decreased T results in less clouds and increased T results in less clouds and so could never have more than 62% cloud cover, despite examples like Venus which provides an example of 100% cloud cover – even though it’s not h2o vapor clouds. It also means that increased energy going into a more active water cycle results in fewer clouds despite the fact that there is no change in physics to reduce the clouds. This comes from an earlier hansen & lacis paper which ASSUMES the effect rather than the more commonly accepted situation of constant relative humidity as T rises and that has been magically transformed into the gospel.
rgbatduke says:
I said, “This causes the Earth to warm” where the antecedent of “This” that I was referring to was the fact that “the Earth will now be emitting energy at a slower rate than it is absorbing energy”. Clearly, if the Sun isn’t there, the Earth will not be emitting energy at a slower rate than it is absorbing energy.
So, I don’t see anything wrong with my statement. Nonetheless, I do agree with you on the larger point of the sort of things that do seem to trip people like John Marshall up and thus I appreciate your re-emphasis of the fact that it is necessary for there to be input from an external (or internal) energy source like the sun in order for the addition of GHGs to lead to a situation where the Earth is warmer than before.
Richard111 says:
July 21, 2012 at 4:01 am
“As an ex-motorcyclist, first hand experience leads me to agree with much of what Reed Coray writes. As a layman in the sciences I must rely on educational web sites that explain heat transfere and IR radiation and such like. I am led to believe that gases in the atmosphere can absorb OR radiate specific radiation bands dependant on the local temperature. It cannot do both at the same time. Wein’s Law will give the peak temperature at any specific IR wavelength. Using Wein’s Law to look at CO2 I find that the 2.7 micron band peaks at ~800C, the 4.3 micron band at ~400C and the 25 micron band at about -80C!! I understand only limited areas of the Earth’s surface might radiate at up to 50C so the 2.7 and 4.3 micron bands will NEVER be exited enough to absorb any energy from the surface. They might absorb a very little from the sunlight but that is working as a coolant. The so called standard surface temperature of the Earth is said to be 15C, well above the the -80C temperature level of the 15 micron band for CO2. The problem now is most of the CO2 molecules in the atmosphere will be at a temperature comensurate with the adiabatic lapse rate starting at the surface. So assuming a drop of 10C per kilometre altitude air temperature should be down to -80C at about 9.5 kilometres altitude, almost the tropopause. Only then will the CO2 molecules be cool enough to absorb radiation at 15 microns.
BUT! There is indeed nothing to stop the CO2 radiating at 15 microns and some of that radiation reaching the surface. Now another BUT! The surface, except at possibly a small area at the south pole, is well above -80C!! Any element, black body or not, does not absorb radiative energy below its peak temperature.
A CO2 molecule IS a black body with rather specific characteristics. And so is any other gas molecule in the atmosphere.
Since I am completely unable to see any ‘greenhouse’ effect in the atmosphere I need more education. Please post links that will this layman”.
Well done Richard111. As one layman to another let me say that you have a better understanding of the basic physics involved, than most climatologists. The reason why you are unable to see any “greenhouse” effect in the atmosphere is that there simply is none. The “greenhouse” effect is junk science. There can be no such effect. A zillion words have been prattled about CO2 that is based on the unquestioning acceptance of a very simple but erroneous theory. The top physicists scorn the theory – the conscientious layman has to work his way through from ground zero doing all of the tedious maths along the way.
I can’t read all the comments to see if these points were already made:
1) The conc of H20 and CO2 vary in the atmosphere; they are not homogeneously distributed.
2) The charts showing absorption clearly indicate the vast majority of the absorption is due to water vapor and not CO2.
3) In the peak of IR emission from the Earth’s surface, Water already absorbs about 2/3 of the energy where CO2 absorbes. The main CO2 band overlaps the shoulder of the main water band.
Bottom line: CO2 hardly matters. What does matter is water. And as we know, water isn’t just vapor, it’s also condensed droplets that reflect incoming radiation.
And of course, water vapor cools the surface as it evaporates. Then it rises to much higher altitude where it cools and condenses, carrying and releasing heat.
The analysis tends to be static, assumes homogeneity, is clearly incomplete, and has exceedingly poor predicitve value. Let’s just allow the Earth to do what it’s done for a few billion years without our ‘help’. You are welcome to go to the very warm place, Greenies.
If you take a planet without an atmosphere and add one to it, it will impede the outflow of radiation. The surface will necessarily heat until the point at which the incoming and outgoing energy fluxes reestablish equilibrium. That much is simple.
Now, what happens with the GHG effect as temperature increases? The radiation from the surface is sure to have the form of a blackbody radiation curve. As the peak of the curve reaches the region in which the gases absorb and re-radiate, the impedance maxes out. As the temperature moves higher, the GHG effect decreases. We reach the point of maximum warming potential for that particular gas, and the temperature stabilizes there.
A good analogy is floodgates in a dam. You place a dam across a river, and the water rises until it reaches the floodgates, water starts spilling through, and the rise is checked.
Where it gets interesting is, what happens if we add gases with lower warming potential? This is akin to opening up floodgates lower down in the dam. The water recedes. It may continue to flow out of the upper floodgates, but at a reduced rate. The more we open up the lower floodgates, the more the level of the water behind the dam recedes.
In an atmosphere with two major GHG emitters, the warming potential of the shorter wavelength emitter sets the level of the upper “floodgates”, and the warming potential of the longer wavelength emitter sets the level of the lower. If we are at a point where significant “water” (i.e., energy in the analogy) is spilling through the upper floodgates, then adding more lower floodgates will reduce the level of the water behind the dam (i.e., the surface temperature in the analogy).
In the Earth’s atmosphere, we have such a situation. The upper floodgates are set by the warming potential of CH4. Significant energy is spilling out of them. What, therefore, naturally happens when we add lower warming potential CO2 which, in effect, opens up floodgates lower in the “dam”? The retained energy, and necessarily the surface temperature, goes down.
O H Dahlsveen;
By the way, sarcasm becomes no-one
>>>>>>>>>>>>>>>>.
If Einstein was around to respond to your last diatribe he most likely would have used that classic quip “that’s not right, that’s not even wrong”. Your first statement was blatantly incorrect and got the treatment it deserved. Your subsequent response is so far divorced from reality that it hardly deserves even that.
Ken Harvey says:
I think you would be hard-pressed to find physicists who don’t accept the theory. Even among climate change “skeptics” who can be classified as physicists, like Roger Brown (commenting here as rgbatduke), Fred Singer, Will Happer, Freeman Dyson, etc., you don’t find any who reject the basic greenhouse effect theory. And, amongst the larger physics community, there is not only acceptance of the greenhouse effect but generally acceptance of the danger of AGW, as evidence by such things as the American Physical Society statement on climate change, the fact that the two textbooks that we use at RIT to teach introductory physics both discuss AGW, etc., etc.
As a physicist to a layman, let me say that both of you have very little understanding of the basic physics involved and that most climatologists, many of whom were trained in physics or closely allied fields, do.
Juray V says
Second, the average temperature decreased by 0.15°C since 2001
Henry@Juray
True. According to my sample it was about 0.2 since 2000, globally. Not a lot: I think most thermometers used at homes will not have picked up on it, as indeed most people did not. But some places are cooling down faster, like Anchorage. I have two weather stations there that are reporting cooling of about 1.5K (=1.5 degrees C) since 2000. Unfortunately, the worst is still to come.
I suppose if you live in Alaska the writing is on the wall. I would pack my bags….We still have 33 years of cooling lying ahead of us….
http://www.letterdash.com/henryp/global-cooling-is-here