Guest post by Reed Coray
The following example illustrates the issues I have with reasoning often used to argue that increasing the amount of CO2 in the Earth’s atmosphere will increase both the Earth’s surface temperature and the Earth’s atmosphere temperature. Immediately following is a direct quote from URL
http://www.school-for-champions.com/science/heat_transfer_earth.htm
“The present situation is that there has been an increase in infrared-absorbing gases in the atmosphere, such as carbon dioxide (CO2) and methane (CH4). Energy that would normally escape into space is absorbed by these molecules, thus heating the atmosphere and spreading through convection currents. The average temperature of the atmosphere has increased 0.25 °C since 1980, mainly attributed to an increase in infrared-absorbing gases in the atmosphere.”
Although the above statement makes no direct reference to Earth surface temperature, I believe it carries the implication that greenhouse gases in the Earth’s atmosphere increase the Earth’s surface temperature.
I make two comments: the first is relevant only if the above implication is valid, the second is relevant independent of the validity of the implication. First, placing matter adjacent to a warm surface such that the matter is capable of absorbing/blocking radiation to space from the warm surface can lead to a decrease in the warm surface’s temperature. Second, increasing the amount of the absorbing/blocking matter can lower the temperature of the absorbing/blocking material.
Take for example an internal combustion engine whose metal surface is exposed to a vacuum. In addition to doing useful work, the engine produces thermal energy (heat). That thermal energy will produce a rise in the temperature of the engine’s surface such that in energy-rate equilibrium the rate energy is radiated to space from the engine’s surface is equal to the rate thermal energy is generated within the engine. By attaching radiating plates to the engine’s surface, some of the energy radiated to space from the engine’s original surface will be absorbed/blocked by the plates; but because thermal energy can be transferred from the engine to the plates via both radiation and conduction, the temperature of the engine’s original surface will be lowered. This is the principle of an air-cooled engine[1]: provide a means other than radiation of transferring heat from an engine to a large surface area from which heat can be removed via a combination of conduction, convection and radiation, and the engine’s surface temperature will be lowered.
If plates at a temperature lower than the original engine surface temperature are attached to the engine, it’s true that the temperature of the plates will increase to establish energy-rate equilibrium. Once energy-rate equilibrium is established, however, increasing the plate radiating area (adding additional matter that blocks more of the energy radiated from the original engine surface) will likely lower the plate temperature.
Thus, blocking the amount of surface radiation escaping to space does not necessarily increase the surface temperature; and increasing the amount of radiation blocking material does not necessarily increase the temperature of that material. In both cases (the Earth/Earth-atmosphere and the internal combustion engine in a vacuum), the heat eventually escapes to space–otherwise the temperature of the Earth’s surface and the engine would continue to rise indefinitely. The difference isn’t that the energy doesn’t eventually escape to space (it does in both cases), the difference is in the path the energy takes to reach space. The amount of generated thermal energy in conjunction with the path the thermal energy takes to get to space determines temperatures along the path; and adding more material may increase or decrease those temperatures. To say that “Energy that would normally escape into space is absorbed by these molecules, thus heating the atmosphere…” by itself is unwarranted; because an equivalent statement for the case of adding extra plate material to the engine would be “Energy that would normally escape to space from an engine with small attached plates is absorbed by additional plate material, thus heating the plates…” For air-cooled engines, this statement is not true—otherwise the plate surface area of air-cooled engines would be as small as possible.
It’s fairly easy to visualize why (a) adding thermally radiating plates to an air-cooled engine might decrease the engine’s surface temperature, and (b) increasing the area of the radiating plates might decrease the plate temperature. It’s not so easy to visualize, and may not be true, why (a) adding greenhouse gases to the Earth’s atmosphere decreases the Earth’s surface temperature; and (b) increasing the amount of atmospheric greenhouse gases lowers the temperature of the Earth’s atmosphere. I now present one possible argument. I do not claim that the argument is valid for greenhouse gases in the Earth’s atmosphere, but I do claim that the argument might be valid, and can only be refuted by an analysis more detailed than simply claiming “Energy that would normally escape into space is absorbed by these molecules, thus heating the atmosphere.”
If we assume that (a) matter cannot leave the Earth/Earth-atmosphere system, and (b) non-greenhouse gases radiate negligible energy to space, then for a non-greenhouse gas atmosphere the only way thermal energy can leave the Earth/Earth-atmosphere system to space is via radiation from the surface of the Earth. The rate radiation leaves the surface is in part a function of both the area and temperature of the surface. For a greenhouse gas atmosphere, energy can leave the Earth/Earth-atmosphere system to space both via radiation from the Earth’s surface and radiation from greenhouse gases in the atmosphere. Suppose it is true that the density of greenhouse gases near the Earth’s surface is such that radiation emitted from low-altitude greenhouse gases does not directly escape to space, but is in part directed towards the Earth’s surface and in part absorbed by other atmospheric greenhouse gases. As the atmospheric greenhouse gas density decreases with increasing altitude, radiation emitted from high-altitude greenhouse gases can directly escape to space.
Now it’s not impossible that since (a) in addition to radiation, heat is transferred from the Earth’s surface to greenhouse gases via conduction, and (b) convection currents (i) circulate the heated greenhouse gases to higher altitudes where energy transfer to space can take place and (ii) return cooler greenhouse gases to the Earth’s surface, that the process of heat transfer away from the Earth’s surface via greenhouse gases is more efficient than simple radiation from the Earth’s surface. Many engines are cooled using this concept. Specifically, a coolant is brought into contact with a heated surface which raises the coolant’s temperature via conduction and radiation, and the coolant is moved to a location where thermal energy transfer away from the coolant to a heat sink is more efficient than direct thermal energy transfer from the heated surface to the heat sink.
One way to realize increased thermal transfer efficiency would be to use a coolant, such as greenhouse gases, that efficiently radiates energy in the IR band (i.e., radiates energy at temperatures around 500 K). Another way would be to spread the heated coolant over a large surface area. Since surface area increases with increasing altitude, thereby providing expanded “area” (in the case of a gas, expanded volume) from which radiation to space can occur, it’s not clear to me (one way or the other) that greenhouse gases won’t act as a “coolant” reducing both the temperatures of the Earth’s atmosphere and the Earth surface.
[1] It’s true that for most air-cooled engines the main transfer of heat from the engine plates is via a combination of (a) conduction of heat to the air near the plates, and (b) convection that replaces the warm air near the plates with cooler air. To aid this process, a fan is often employed, or the engine is located on a moving vehicle and the vehicle’s motion through an atmosphere provides the flow of air across the plates. Although conduction/convection may be the primary means of heat dissipation from the plates, radiative cooling also dissipates heat.
I’ll play one more round anyway … “An alternate Earth 3 is the same as Earth 2, except that it has double the amount of O2 in the atmosphere. “
C: At Earth 3 TOA, is the amount of outgoing 15 um photons is LESS THAN Earth #1 or #2. The CO2 will remain reasonably well mixed, so the “N” moles of CO2 that make up the “top layer” and do most of the radiating will be be higher than before. Assuming the lapse rate is such that this new higher altitude is colder than the old, lower altitude, then the new, cooler “top layer” of CO2 will radiate less.
Presumably the other wavelengths will get more intense to make up the difference (ie the ground will get a little warmer.
(Now we get into potential secondary but important issues. For example, the inversion due to Ozone in the stratosphere. I am ignoring such other issues.)
Tim Folkerts, at post before your latest, you said:”The CO2 will remain reasonably well mixed, so the “N” moles of CO2 that make up the “top layer” and do most of the radiating will be be higher than before. Assuming the lapse rate is such that this new higher altitude is colder than the old, lower altitude, then the new, cooler “top layer” of CO2 will radiate less.”
Here’s some data on lapse rates from Griggs & Harries JoC 2007, Appendix Tables.
hPa T’ Ko 1970T Ko 1997T Ko 2003
1000 299.234 299.463 299.078
925 293.872 294.585 293.833
850 290.151 290.967 289.44
700 283.428 283.511 283.67
600 276.122 275.883 276.554
500 268.464 268.284 268.411
400 257.439 256.92 258.319
300 242.044 241.594 242.514
250 232.201 232.005 232.594
200 220.713 220.895 221.39
150 207.153 207.425 207.834
100 192.001 192.001 195.882
56.866 204.475 204.756 203.165
29.5643 217.385 219.476 215.503
14.792 228.791 228.063 227.923
4.60588 246.354 239.277 239.19
The Temps at tropsheric altitudes at and above 300 seem slightly warmer in 2003 than in 1970 and 1997, but cooling at hPa less than 100. Within each series the trends per hPa are negative at a faster rate in 2003 than in the earlier years:
1970: y = -6.126x + 299.56
R² = 0.7052
1997: y = -6.2822x + 300.59
R² = 0.7391
2003: y = -6.296x + 300.72
R² = 0.7538
What’s your take?
George says: “So we have one of these experimental near black body radiators Tim; you recommended I look up cavity radiation (I didn’t bother),”
But “cavity radiation” IS the way Planck derived the equations for BB radiation. I can’t decide if you feel you already know this topic (in which case you should know that the frequencies ARE quantized by the cavity) or that you feel it is not worth your time to understand the topic you are trying to explain.
“and it is so close to a perfect black body that we can’t measure the discrepancy; I think you said something along those lines.”
Yep .. i said that.
“So we have Planck’s exquisitely perfect formula for the black body radiation spectrum, containing nothing but fundamental constants of classical Physics…”
What? Planck’s constant is the EPITOME of non-classical physics! It is the key parameter that universally shows up in modern quantum physics. Planck was expecting that “h” would be a mathematical trick that he could eventually eliminate by letting it go to zero. It turned out that the constant named after him had to have one particular value, demolishing classical physics and ushering in quantum mechanics.
“and an actual real faux black body made of actual quantum mechanical materials, which we haven’t even specified; apparently don’t need to know it’s so close to perfect. ”
The whole point of cavity radiation is that the materials DON’T matter! Here are some pictures of “cavity radiators”:
* a beverage can: http://www.hoax-slayer.com/images/soda-can-top.jpg
* sewer grate: http://fc09.deviantart.net/images/i/2003/43/f/6/Sewer_Grate_01.jpg
* a hole in a pipe: http://www.mcgee-flutes.com/images/ClintHole.JPG
* an electric outlet: http://www.greenbang.com/wp-content/uploads/2008/11/electric-outlet.jpg
Note that in every case, the opening looks perfectly matte black, independent of the material on the interior of the cavity. Light can’t reflect from the “surface of the opening” Since there is nothing physically there to reflect light, so all incoming light is “absorbed” into the cavity –> emissivity = 1. Even with fairly reflective aluminum, the hole looks black –> emissivity close to 1. For large holes (compared to the cavity) and reflective materials, the hole won’t look truly black; for a large cavity, a small opening, and a rough/black surface, you can easily get the emissivity arbitrarily close to 1. This has been the “gold standard” for thermal radiation for over a century.
“How many possible ways, and material lists are there for building a near perfect black body radiator, all of which seem to behave the same regardless of their quite different quantum mechanical properties. Well it’s a rhetorical question Tim; no need to answer. “
There should be no need to answer, since the answer obviously is “infinite”. A small hole into a cavity constructed out of any material will approximate a blackbody.
Tim Folkerts,
You answered C for question 3. In question 1 sphere 2 cooled faster due the presence of CO2. In question 2 the alternate Earth 2 atmosphere was able to cool more effectively due to the conductive transfer of energy to CO2 that then radiated it to space. Yet for Earth 3 you suggest that increased CO2 would reduce the atmospheres cooling ability.
The correct answer to question 3 is B. Given the imaginary absence of outgoing 15 um IR from the surface of Earth 3 the increased quantity of CO2 in the atmosphere increases the amount of outgoing photons at the 15 um energy level.
On Earth 1 however the surface emits 15 um IR. On Earth 1 CO2 can both cool and warm. Its initial effect should be warming. The original question posed by Reed Corays post heading this thread is when do the secondary cooling effects exceed these initial warming effects.
Konrad:”On Earth 1 however the surface emits 15 um IR. On Earth 1 CO2 can both cool and warm. Its initial effect should be warming. The original question posed by Reed Corays post heading this thread is when do the secondary cooling effects exceed these initial warming effects”
I haven’t really been following your discussion with Tim, nor the original questions in your discussion but I think it is clear the effect of CO2 is *always* warming under *our Earth’s* conditions when the surface is being heated *and* there is a gravitationally driven lapse rate. Cooling effects would be from the increased SB emission at the warmer temps plus any other negative feedbacks that are beyond the scope of this discussion.
It is fine to argue that CO2 has a cooling effect under certain (non-Earthlike) circumstances but doing so doesn’t establish that CO2 will have a cooling effect under Earthlike conditions.
Again, the GH effect is proposed to work because radiation from the Earth’s surface (after being warmed from the sun) cannot leave the atmosphere as quickly or easily as it would without the presence of GH gases because that energy on leaving the Earth’s surface is continually absorbed and re-emitted. This relative inefficient means of radiating energy to space *combined* with the fact that a gravitationally induced lapse rate pulls hot air towards the surface implies the existence of a GH effect (of some magnitude). It is all these properties of the Earth acting together that produce a GH effect.
Cheers, 🙂
AJB says
http://wattsupwiththat.com/2012/07/21/some-thoughts-on-radiative-transfer-and-ghgs/#comment-1043301
Henry says
sorry ,
very bad internet connection here (in South Africa)
I don’t yet have a clue as to what you are trying to say
trccurtin says
http://wattsupwiththat.com/2012/07/21/some-thoughts-on-radiative-transfer-and-ghgs/#comment-1043667
0.7 is not good enough
try getting it >0.95
http://www.letterdash.com/henryp/global-cooling-is-here
Konrad, you originally said “except that it has double the amount of O2 [not “CO2″] in the atmosphere. ”
But now you are saying “Yet for Earth 3 you suggest that increased CO2 would reduce the atmospheres cooling ability.”
So we are comparing apples and oranges. We have Question 3a -> Doubling O2; and Question 3b –> Doubling CO2. (Although the same answer still holds).
For Question 3a (O2) I stand by my conclusion stated previously.
For question 3b, there will STILL be warming. You incorrectly conclude The correct answer to question 3 is B. Given the imaginary absence of outgoing 15 um IR from the surface of Earth 3 the increased quantity of CO2 in the atmosphere increases the amount of outgoing photons at the 15 um energy level.”
The key point you overlooked is that a relatively short distance of CO2 will block all (>99.9%) of the 15 um photons.So the photons that leave the surface are all blocked by the first km of CO2.
* The photons that are heading up at 1 km altitude were created by CO2 ~ 1 km up, where the temperature will be ~ 10 C cooler than the surface.
*The photons that are heading up at 2 km altitude were created by CO2 ~ 2 km up, where the temperature will be ~ 20 C cooler than the surface.
…
There will be some top of atmosphere level (call it “TOA2” corresponding to Question 2) where there are no longer enough CO2 molecules higher up to block the outgoing 15 um photons. Looking down from above this altitude we will see the BB curve from the ground (somewhere around 300K), with “bites” missing where CO2 has blocked the ground’s IR. The “depth” of those bites will tell you the “TOA” temperature.
If you put in more CO2, you are not blocking any more of the ground’s 15 um photons. But you are putting more CO2 above the previous “TOA2” level, blocking some of those IR photons from that cold TOA2 level. The NEW TOA3 will be a bit higher up, and (due to the lapse rate) will be even COLDER than TOA2. Thus the 15 um photons emitted to space will be even weaker.
I know it sounds backwards, but MORE CO2 leads to LESS outgoing IR (because the IR is coming from colder places that radiate poorly)!
PS. There is a SECOND effect that will also reduce the outgoing IR. The “ragged edges” of the absorption band allow some ground IR photons near 15 um to escape. With more CO2, the “ragged edges” absorb more of the plentiful IR photons from the warm ground. These will be replaced by sparse IR photons from the cooler atmosphere..
PPS. At some point in a real atmosphere there could well be an inversion in the lapse rate. I am assuming the temperature continues to drop with altitude above TOA2. If not, then adding more CO2 would have little effect. (And given that the actual TOA on earth is not that far from the Tropopause, this is a significant concern in my opinion.)
PPPS. You can model all this yourself with MODTRAN. http://geoflop.uchicago.edu/forecast/docs/Projects/modtran.html I would suggest removing the water vapor to make the effects of CO2 more distinct. Try looking down from various altitudes from 0 to 20 km. You can see the “bites” getting deeper as the air gets colder. (You can also see the “ragged edge” and “tropopasue” effects if you pay attention.)
“””””…..Tim Folkerts says:
July 25, 2012 at 9:39 pm
George says: “So we have one of these experimental near black body radiators Tim; you recommended I look up cavity radiation (I didn’t bother),”
But “cavity radiation” IS the way Planck derived the equations for BB radiation. I can’t decide if you feel you already know this topic (in which case you should know that the frequencies ARE quantized by the cavity) or that you feel it is not worth your time to understand the topic you are trying to explain. …..”””””
Tim, rather than us slinging around a lot of chaff, I would like to simply present some assorted facts and notions for the consideration of anyone who wants to think openly about this question.
First MY position is that Blackbody Radiation is classical Physics, and is NOT quantum Mechanics.
You assert the exact opposite; that Blackbody radiation is the epitome of Quantum Mechanics.
So some demonstrable facts. I recently purchased a very complete Textbook Treatise on Quantum Mechanics, since ALL of my own personal high school and University Textbooks, were lost in shipping about 51 years ago; sad occurrence.
This modern very complete text on Quantum Mechanics Contains NOWHERE in its 400 or so pages, the word BLACKBODY, nor the combination BLACKBODY RADIATION. Odd that a highly regarded Reference Textbook, would contain not a word about something that is the epitome of its subject matter.
You mention cavity radiation, and assert that there are an “infinite” number of ways of building a very reasonable facsimile (I concur). Odd, that we both agree that a very good approximation to ideal blackbody radiation, is obtainable from an arbitrary shaped and dimensioned cavity made out of quite arbitrary real physical world materials, each of which has an entirely different set of electronic energy levels, and associated quantum numbers associated with it; none of which seem to have ANY influence on the absorbed or emitted radiation or its spectrum; yet those quantum number lists and tables are an essential part of the calculable molecular or atomic absorption/emission spectrum of those real materials, with programs like HITRAN for example.
Speaking of HITRAN, where in that program does one find the knob for TEMPERATURE, or PRESSURE, or DENSITY ? Can I use HITRAN to calculate the black/grey/blue/pink emission spectrum of say a solid piece of Cobalt, which is a metal solid, that Mr Kelly asserts does not contain atoms/molecules that can collide with each other.
I can recommend to anyone the two volume set of books called “The Constitution of Binary Alloys.” Which provides complete phase diagrams of every possible binary composition of the known stable elements; specially including, the Fe/C phase diagram, sometimes known as the “Steel” diagram, which is a well known sold metal, and contains multitudes of crystalline lattice types, all of which are actually chemical compounds of various composition; which normally would indicate that the atoms are already in permanent contact with each other so their electron clouds, are already mingled; to the point where many of those electrons simply take leave of their atom, and drift freely anywhere in those crystalline lattices.
Yes as a result of the intimate proximity of those atoms in a solid, their individual electron structures and energy levels interract to form bands of energy levels, such as the Valence Band or the Conduction Band in which the free electrons can roam. And yes, those band structures in both metals, and semi-conductors do result in energy and wavelength specific absorptions and emssions of DISCRETE SPECTRAL LINES, whose frequencies are first order quite Temperature independent. The do vary with Temperature to some extent, simply because the Temperature does affect the lattice constant, so the crystal can grow or shrink (thermal expansion) and that results in detail alteration of the energy band levels. There is NO WAY that such Temperature variable band structure shifts can result in very broad band black body or thermal radiation emission.
Sorry \, but solids DO emit line spectra (more like bands)that are only slightly second order Temperature dependent, but they also emit a complete continuum of quite non-quantized Thermal (black body like) radiation, as do all other materials that have a non zero Temperature.
Plasma Physicists, are well aware that plasmas (like the sun) emit continuum spectra, that result from the fact that free electrons of quite arbitrary and non-quantized energy (in the lab co-ordinate frame) can drop into an ionised atom, and end up at one of its defined electron quantized energy levels, and when you add (x) a continuous value to (n) an integer value, you still end up with (x+n) being a continuous value; NOT a quantized value.
It must seem odd to readers, that blackbody radiation; the epitome of quantum mechanics is quite independent of the specific quantum numbers of ANY real material, including that used to form a “Cavity Radiator”, and depends entirely on the Temperature of that source or cavity; while at the same time, the resonance line/band spectra of real materials, including both the GHGs, as well as the ordinary neutral gases, are intrinsically independent of the Temperature of the source material, and vary from the intrinsic line frequencies, simply as a result of ordinary classical Physics phenomena, such as the Doppler effect, of the lab space velocity of the emitting or absorbing molecules (Temperature broadening) and the mean time between collisions of the molecules (Pressure/Density Broadening).
It is important for the lay readers to understand the fundamental difference between the “GHG mode” of energy absorption or emission, that IS a direct consequence of the specific molecular/atomic electron structure; AND IS QUANTUM MECHANICS, and the quite different absorption and emission of a continuum of frequencies, that are not related in ANY way to the specific quantized energy level structures of the source materials, and ARE absorbed/emitted by ALL physical materials that have a Temperature, by means of processes that depend only on that Temperature, and not on the material or its quantum energy level properties.
Yes Planck DID propose that the radiated energies of “radiation” at ANY specific frequency (f) of radiation should be in integral multiples of hf, h being a constant he introduced, to be determined experimentally, BUT he did NOT specify that those FREQUENCIES themselves be quantized; they can be of ANY value, and hence so can the emitted energy packet energy, it is still a continuum spectrum, and in particujlar it is NOT a picket fence of closely spaced spectral lines, as are the real high resolution molecular band spectra of GHG absorptions/emissions.
So Tim, you assert that cavity radiation is quantized by the cavity. What specific physical properties of the cavity determine what those quantized energy levels are, since we apparently are in agreement that NOTHING about the specific nature of the cavity, either size or shape or material properties, seems to have ANY influence on the spectrum of the BB radiation, or its amount; the Temperature alone dtermines the entire spectrum.
Personally, I have a VERY limited knowledge of QUANTUM MECHANICS. I am fortunate to have the counsel of some folks, who are steeped in QM. In deference to those individuals, some known to almost everybody by sight (although unknowingly) , but not by name; I am not going to drop any names; but I did ask one such person with whom I am acquainted, just what QM could add to my simple classical Newtonian/Hertzian model of how BB radiation is emitted during atomic/molecular collisions due to Temperature; and his response was almost one of shock and horror. QM could add nothing, and only muddy the waters further. You see QM simply replaced causal deterministic classical Physics, with a set of possible outcomes, of which only the statisitical probabilities of each could be found, and that simply trying to observe the existence of any one of those happenstances, immediately caused all of the alternative possibilities to simply evaporate; no matter which of the possibilities one chose to test for.
Frankly, I have not found giggle/wiki/yoohoo/ faceback/whatever, nor their lookalikes to be very rigorous or useful, in studying any of these subject matters. I defy anyone to find on the web ANY fundamental Physical explanation for the actual originating mechanism for the emission of thermal radiation; excluding of course simple one liners like “acceleration of charge”; and no don’t read back to me, my own description of how it happens, that has been already posted here at WUWT.
But I’m not going to waste any more of Anthony’s valuable bandwidth, on this subject. I could run over to some Physics specific blog, and chat with others there (mostly academics); but I’d much rather try to help even one other person, to try and grasp some of this stuff; it’s all interesting, and mostly all understandable by anyone. As I have said; Ignorance is NOT a disease; we are all born with it. But stupidity has to be taught; and sadly the web is full of those willing and able to teach it. Fortunately there is still a good supply of those trying to help others understand; and Anthony has managed to attract many of those folks, who come here, and try to educate the rest of us.
George E. Smith; says on July 25, 2012 at 4:28 pm:
“I might come back to see who wants to join you.”
============
George E, will you come back to join us even if I am not going to join Tim?
I am the idiot or “super idiot” even – who does not believe radiation can be seen by the “human eye” or indeed by Mellony’s thermopile, or any modern derivative thereof. –
I have performed many experiments – in order to prove, or disprove – this “pesky” chameleon that goes under the name of “AGW”. My “experiments”, I must admit, are not always performed under “Ideal Laboratory Condition” (ILC). – However, I have for many years, proved – to myself at least. – That “cooling by radiation” – ( that is what we shall have to call it,) is “quite insignificant”
I do not know much about IR radiation and the “wave-bands” that locks it in, or makes it convenient for us to measure it, but by now I do understand that “Thermodynamics” and “Thermoradiationics” is/are not interchangeable sciences.
George E. Smith,
Just FYI, Wiki’s page for Planck’s law: “Classical physics provides an account of some aspects of the Planck distribution, such as the Stefan–Boltzmann law, and the Wien displacement law. Other aspects of the Planck distribution cannot be accounted for in classical physics, and require quantum theory for their understanding.”
http://en.wikipedia.org/wiki/Planck%27s_law
“So Tim, you assert that cavity radiation is quantized by the cavity. What specific physical properties of the cavity determine what those quantized energy levels are, since we apparently are in agreement that NOTHING about the specific nature of the cavity, either size or shape or material properties, seems to have ANY influence on the spectrum of the BB radiation, or its amount; the Temperature alone dtermines the entire spectrum.”
I’m not an expert here by any means but I believe it is its *shape* that accounts for the ability of a cavity to approximate a blackbody. Again from the wiki page: “Though perfectly black materials do not exist, in practice a black surface can be accurately approximated.[4] As to its material interior, a body is completely black to a certain wavelength if it is completely opaque to that wavelength; that means that it absorbs all of the wavelength that penetrates the interface to enter the body; this is not too difficult to achieve in practice. On the other hand, a perfectly black interface is not found in nature. The best practical way to make an effectively black interface is to simulate an ‘interface’ by use of a small hole in the wall of a large cavity in a completely opaque body, with a controlled temperature. Radiation entering the hole has almost no possibility of escaping the cavity without being absorbed by multiple impacts with its walls.[13]”
Cheers, 🙂
“””””…..Shawnhet says:
July 26, 2012 at 2:12 pm
George E. Smith,
Just FYI, Wiki’s page for Planck’s law: “Classical physics provides an account of some aspects of the Planck distribution, such as the Stefan–Boltzmann law, and the Wien displacement law. Other aspects of the Planck distribution cannot be accounted for in classical physics, and require quantum theory for their understanding.”
http://en.wikipedia.org/wiki/Planck%27s_law …..”””””
Total and utter balderdash. The Stefan-Boltzmann “law” is nothing more than the complete integral over all wavelengths, of the Planck black body spectrum equation.
How can one obtain SB from classical Physics, if one cannot obtain the spectrum except with quantum mechanics; and you can tell wiki I said that.
Tim Folkerts says:
July 26, 2012 at 11:05 am
Konrad, you originally said “except that it has double the amount of O2 [not “CO2”] in the atmosphere. ”
But now you are saying “Yet for Earth 3 you suggest that increased CO2 would reduce the atmospheres cooling ability.”
——————————————————————————————–
Tim,
My apologies, your original answer to question 3 as posted was correct. I had typed that comment from my “smart” phone and it had “corrected” CO2 to O2. Question 3 should have read –
“Question 3 references Question 2. An alternate Earth 3 is the same as Earth 2, except that it has double the amount of CO2 in the atmosphere.”
However you state “For question 3b, there will STILL be warming.”
There could be less cooling than Earth 2, but it is inconceivable that there could be warming on Earth 3 when 15 um photons are still escaping to space.
Imagine and alternate Earth. Earth 1.5 has no 15 um IR radiated from the surface and no CO2 in the atmosphere. You appear to agree that increasing CO2 concentration between Earth 1.5 and Earth 2 will initially allow CO2 to acquire energy conductively and radiate it to space, however you appear to be claiming further increases in CO2 toward the conditions of Earth 3 will reduce this ability. When adding CO2 to the atmosphere of an alternate earth such as earth 1.5 at how many ppm would CO2 reach its peak ability as a coolant and start to lose effectiveness?
All 15 um IR photons transport the same energy. If more escape to space then conductive to radiative cooling increases.
Uh, George, I think you misunderstood what the wiki page is saying. It is saying that some aspects of Planck’s law (and, hence, blackbodies) are explicable in classical terms and some are not.
For instance, see here:
http://en.wikipedia.org/wiki/Ultraviolet_catastrophe
IAC, there are ways to derive S-B that do not reference Planck’s law (which makes sense because SB came first).
http://en.wikipedia.org/wiki/Stefan%E2%80%93Boltzmann_law
Cheers, 🙂
“Total and utter balderdash. … How can one obtain SB from classical Physics if one cannot obtain the spectrum except with quantum mechanics?”
Its laid out here, if you want to try to understand. It is actually a rather elegant proof using thermo and E&M. http://en.wikipedia.org/wiki/Stefan%E2%80%93Boltzmann_law
It is not uncommon for a net result (eg the value of an integral) to be know when the exact nature of the the details (the function being integrated) are not know. It is also not uncommon for the classical limit of a problem is known without understanding the QM details.
In this case, a relationship dealing with radiation pressure is known without knowing the actual distribution of photon energies
“You see QM simply replaced causal deterministic classical Physics …”
That is ONE of the things QM did, but that is certainly not all.
“So Tim, you assert that cavity radiation is quantized by the cavity. What specific physical properties of the cavity determine what those quantized energy levels are?”
The dimensions of the cavity determine the possible standing waves. The math is simplest if you assume a rectangular box. Check your QM book for “particle in a box”. Apply the principles to massless bosons.
“It must seem odd to readers …”
QM and modern physics does indeed often seem odd. That doesn’t mean it is wrong.
“This modern very complete text on Quantum Mechanics Contains NOWHERE in its 400 or so pages, the word BLACKBODY, nor the combination BLACKBODY RADIATION. Odd that a highly regarded Reference Textbook, would contain not a word about something that is the epitome of its subject matter.”
It is your turn to quote me accurately. I said “Planck’s constant is the EPITOME of non-classical physics! It is the key parameter that universally shows up in modern quantum physics. ”
For your complaint to be valid, your QM text would have to have “not a word” about h or h-bar.
In any case, BB radiation is not the simplest nor the most fundamental application of QM. It is a starting point historically of QM, but very few text books take a historical approach to teaching science (thank goodness!). You can learn a LOT of QM before ever seeing BB radiation as an application of QM principles.
Konrad says “Imagine and alternate Earth. Earth 1.5 has no 15 um IR radiated from the surface and no CO2 in the atmosphere. You appear to agree that increasing CO2 concentration between Earth 1.5 and Earth 2 will initially allow CO2 to acquire energy conductively and radiate it to space, however you appear to be claiming further increases in CO2 toward the conditions of Earth 3 will reduce this ability. When adding CO2 to the atmosphere of an alternate earth such as earth 1.5 at how many ppm would CO2 reach its peak ability as a coolant and start to lose effectiveness?”
Interesting! The short answer is I don’t know the optimal value, but I am sure it exists. But now we have all sorts of details that would start to come into play to get a specific number.
* With NO CO2, then there will be no radiation in the missing band. Or put another way:
>>the radiation near 15 um is like a BB with a temperature of 0 K
* With a TINY bit of CO2, there is not enough CO2 higher up to really block any of the IR from the lower CO2. The outgoing 15 um IR will come from throughout the atmosphere (but mostly from the warm lower atmosphere, since there is more CO2 there). The “TOA” for 15 um IR might be only 200 m up.
>>The radiation near 15 um is like a BB with a temperature just slightly cooler than the surface.
* With LOTS of CO2, then the IR from the warm, lower layers of the atmosphere will be blocked, only the weak stream of 15 um photons from the cold upper atmosphere can escape.
>>The radiation near 15 um is like a BB with a temperature much cooler than the surface.
So as we add CO2, we will go smoothly from no 15 um IR to much 15 um IR and then back to little 15 um IR.
This would mean that
>a world with NO CO2 would be warmest,
>a world with A LITTLE CO2 would be coolest, and
>a world with LOTS OF CO2 would be almost as warm as a world with no CO2.
(A reminder to all: this is all predicated on the surface not radiating any 15 um photons, which is not how the earth behaves.)
The following quotation is from “Computational Blackbody Radiation” by Claes Johnson:
“A blackbody thus absorbs and emits frequencies below cut-off without getting warmer, while absorbed frequencies above cut-off are not emitted but are instead stored as heat energy increasing the temperature.
A blackbody is thus like a high-pass filter, which re-emits frequencies below a cut-off frequency while capturing frequencies above cut-off as heat.”
Antennas work by absorbing energy, converting radiation into electrical signals which have POWER. At room temperatures, radio frequencies are well below cut-off. So the first phrase of this quotation seems is in error, making the second paragraph untrue.
Tim Folkerts says on July 26, 2012 at 4:20 pm:
““Total and utter balderdash. … How can one obtain SB from classical Physics if one cannot obtain the spectrum except with quantum mechanics?”
Its laid out here, if you want to try to understand. It is actually a rather elegant proof using thermo and E&M. http://en.wikipedia.org/wiki/Stefan%E2%80%93Boltzmann_law
It is not uncommon for a net result (eg the value of an integral) to be know when the exact nature of the the details (the function being integrated) are not know. It is also not uncommon for the classical limit of a problem is known without understanding the QM details””
C.mon Tim, grow up “The Stefan–Boltzmann law, also known as Stefan’s law, states that the total energy radiated per unit surface area of a black body per unit time (also known as the black-body irradiance or emissive power), j*, is directly proportional to the fourth power of the black body’s thermodynamic temperature T (also called absolute temperature):”
The Earth, that we live on is not a “black body” – It can be seen from space. – OK, so North Korea may be a perfect black body but if so, – it is so, only in “jokes”
Please classify the Earth exactly where it belongs in the scale between a “Black” and a “White” body before you make your calculus.
I.E. The Earth can be seen from space, so we know it is not a “Black Body”
Therefore “Blackbody Calculations” have no place whatsoever in our world of “climate calculations”
I’m confused by your response, O H Dahlsveen. I am going to assume that you were confused by a missing close-quote. The first paragraph (“Total and utter balderdash. … “) was George’s opinion on classical derivations of SB. The second paragraph (“It is not uncommon for a net result …”) was my reply, stating that you can indeed derive S-B classically (but not the wavelength distribution – that requires QM). Unfortunately, I left off the close-quote ( and this time I also didn’t include italics) at the end of George’s words, so it would be easy to not notice which parts were mine and which were George’s. Hopefully that clarifies things a little.
I don’t disagree with the quote you included about S-B, but it doesn’t really address the issue you quoted about classical vs QM derivations of S-B.
I do, however, disagree with what you wrote later.
“I.E. The Earth can be seen from space, so we know it is not a “Black Body”
Therefore “Blackbody Calculations” have no place whatsoever in our world of “climate calculations”
1) Yes, we all know the earth is only approximately a black-body; it is not a “true blackbody”.
2) In this context, we are discussing the absorption/emission of IR for the earth, so reflection of visible light is not germane and tells us nothing about the earth as a blackbody for thermal IR. Many materials reflect visible light quite well, but can be considered “blackbodies” for thermal IR.
3) The fact is that BB calculations put an absolute upper limit on thermal radiation (at every individual wavelength) from a surface at a given temperature, which is quite useful.
4) Furthermore, ideal BB radiation is indeed a very good approximation for the radiation from the much of the ground, from the oceans, and from clouds, so BB do indeed have a major place in climate calculations. Of course, for accurate calculations for real surfaces, actual emissivity values should and would be included (possibly as a function of wavelength), but the theoretical BB equation is still needed as a starting point.
~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~
And now, I think it may be time to drop out of this discussion. I suspect we have all learned about as much as we can from this discussion and it is time to move on.
Just for Reference: I believe a black-body is assumed to have so many electromagnetic resonant states that radiation at any wavelength is uniformly possible and Planck’s Law is an accurate representation of the power spectrum that will be emitted on that assumption.
Ref: http://en.wikipedia.org/wiki/Planck%27s_law
The transparent gases of the atmosphere do not qualify as black-bodies, but their spectra are punctuated by a limited number of resonances.
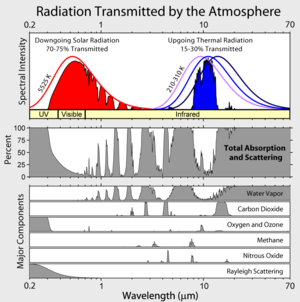
If anyone wants to see what the 15 micron absorption-emission spectrum of CO2 really looks like, there is a NASA Virtual Planetary Laboratory website based on the HITRAN 2008 database that will plot it out for you:
http://vplapps.astro.washington.edu/vplselectmicro
select:
wavelength_low:=13 µ m
wavelength_high=17 µ m
choose molecules=CO2 in the first box and click ‘findplots’
I believe the multiple lines are from various possible quantum states of the molecule. As stated, this is only for the most abundant isotope combination for CO2–that containing no abnormal isotopes of carbon or oxygen.
This has been a very interesting thread, thank you Reed.
Thanks also to Tim Folkerts and rgbatduke for battling against the tide.
Thanks to Spector for the superb VPL link.
George E. Smith; says: July 26, 2012 at 12:52 pm
“Personally, I have a VERY limited knowledge of QUANTUM MECHANICS”
I knew I would eventually agree with something you said George.
And, on the entertainment side, thanks for the amusement features provided by Mr. Dahlsveen. “The Earth can be seen from space, so we know it is not a Black Body”. Great. Was this part of an attempt to say the most stupidest things in a single blog?
So I decided to take a look at Tim’s Wiki reference in detail to see for myself their “rather elegant proof” ? I also spent some time looking into the Quantum Mechanics Textbook I referenced.
They did say this in their introduction to QM:- “”The important parameter in quantum mechanics is Planck’s constant h = 6.626 x 10^-34 Js …….Classical Physics treated electromagnetic radiation as waves. It is particles called photons whose quantum of energy is hbar.omega where omega is the classical angular frequency””.
They then launch into Schroedinger/s equation and so on, with nary a mention of Planck or black bodies or blackbody radiation.
No argument from me as to the importance of (h) to QM; as to being “The important parameter”, I dunno, since as I have said, I know very little of QM.
Notice how all this talk of BB radiation refers to frequencies, and wavelengths; which are quite clearly properties of WAVES, as used in classicl Physics. Anybody got a Planck spectrum formula using the language of quantum mechanics, and not mentioning the frequencies or wavelengths of classical physics; some photonic language perhaps ?
The constant that Planck introduced (h) is of course nothing more than the energy contained in any single cycle of any frequency of the waves of classical physics.
But off to wikiland, where they tell us Planck’s radiation law:-
L = 2hc^2.lambda^-5 / ( exp hc/lambda.kT – 1)
Well it’s got an (h) in it so it must be quantum mechanics.
They also give us the Stefan Boltzmann law, which is just classical physics.
E = pi^2.k^4 / (60h^3c^2) T^4
I see it also has an (h) in it. Must be the energy per cycle version of (h), since it was derived years before the Planck formula.
But I diligently read the entire wiki post on S-B and some of the linked subjects as well.
But I never did find the part where it says that only certain frequencies (spectral lines) and energies are allowed, because they are quantized; you know the discrete modes that are quantized by the cavity; you know, the ones that you get our of a soda pop can or drainage grate, or even an electric power socket.
If you;re a ham, good luck at getting the signal from your 2 metre rig into the pop can or electric power socket; might get some of it into the drain grate black body cavity radiator.
“””””…..George E. Smith; says: July 26, 2012 at 12:52 pm
“Personally, I have a VERY limited knowledge of QUANTUM MECHANICS”
I knew I would eventually agree with something you said George……”””””
Well MikeB; don’t be so bashful; come to think of it, this is about the very first time YOU have EVER made any reference to ANYTHING, I have ever posted; so I must presume that most of it simply went over your head. Really it is not all that hard to understand.
And I’m still waiting for someone to point out where the Temperature knob is in HITRAN or MODTRAN, so we can use it to do Blackbody Radiation calculations;, but I also noticed on wiki, that they do actually have a reference to “Thermal Radiation”, and what it is, so all you folks in Myrrh’s club, can stop pretending, that you have never heard of it.
And if anybody has a good table of all the discrete spectral line frequencies, for the black body cavity radiator (all those quantized modes), it would be nice to get a copy of them.
Bottom line Earth is not a black body and not even a real gray body. By the time all the corrections are made we are not even approximating at all. I support George E. Smith’s statement and I DO know alot of QM and not just ‘about’. All you need is classical physics for temperature which is AVKE of molecules, no need to even go to atoms in most cases, unless you want to know more underpinnings in a layered abstraction model.
“The constant that Planck introduced (h) is of course nothing more than the energy contained in any single cycle of any frequency of the waves of classical physics.”
Not even close. Classically “any single cycle ” of EM waves could have any conceivable energy by having arbitrary large or small E&B fields.
“But I never did find the part where it says that only certain frequencies (spectral lines) and energies are allowed”
You must not have recognized that part, since any derivation of Planck’s Law will list these. For example, you could look in Wikipedia. http://en.wikipedia.org/wiki/Planck%27s_law. Look in the section labeled “Derivation”. The allowed wavelengths will also certainly be in your QM textbook in a section about a “particle in a box” but you might need to generalize to 3D if the book only does the 1D case. (The photon is the particle; the cavity is the box.)
“They also give us the Stefan Boltzmann law, which is just classical physics.
E = pi^2.k^4 / (60h^3c^2) T^4
I see it also has an (h) in it. Must be the energy per cycle version of (h), since it was derived years before the Planck formula.”
1) The FORM of S-B can be derived classically, ie P ∝ T^4. Deriving the constant in the equation requires QM.
2) As already noted, the “energy per cycle” interpretation is wrong.
3) That is the equation for P/A = E/(t*A) = the power per unit area, not the equation for energy. If anything, the equation would have to be for “energy per unit time per unit area per cycle”, whatever that might mean.
4) Minor point: that should be h-bar^3, not h^3. That is an easy mistake to make.
“But I diligently read the entire wiki post on S-B and some of the linked subjects as well.”
Wikis and webpages are not bad ways to review advanced physics topics, but clearly they are not effective ways to learn it for the first time.
“… so I must presume that most of it simply went over your head. Really it is not all that hard to understand.”
Can you truly not see the irony here? You admit you don’t understand QM and have made repeated fundamental mistakes, but you feel qualified to judge the understanding of others. Two Nobel prize winning physicists said “Nobody understands quantum mechanics” (Feynman) and “Anyone not shocked by quantum mechanics has not yet understood it.” (Bohr) yet you feel it not hard to understand.
Wikipedia is pure garbage, and this is especially true for physics and chemistry. Wikipedia is only good for things like what color hat did Paris Hilton wear while on vacation in Europe.
The problem with understanding of physics:
http://staff.science.uva.nl/~eberg/Antwerpen/Diagnostische%20toetsen/Heat%20and%20TemperatureTPT%20YeoZadnik.pdf
Power is energy flow. That is why these words are sometimes used interchangeably. One watt per square meter is the same as one joule (the meter-kilogram-second (MKS) unit of energy) per second per square meter.
If one were in a closed room where all surfaces were the same temperature, each surface would emit the same number of photons at that temperature as it would in outer space. In the fixed temperature room, however, each surface would also be receiving photons that would replace the energy being emitted. The photons emitted all these surfaces are real and detectable even though there is no net loss of energy from any surface.
The real reason that CO2 cannot be the serious global warming hazard that many fear is that its absorption band stands like a one-foot diameter tree in the middle of a ten foot wide stream. Almost all of the effect of additional CO2 is to absorb photons already being totally absorbed at a slightly lower altitude.
Here, again, is a curve showing almost no difference between the calculated energy (or power) escaping the Earth’s troposphere (‘Radiative Forcing’) for CO2 concentrations of 300 PPM and 600 PPM.
http://en.wikipedia.org/wiki/File:ModtranRadiativeForcingDoubleCO2.png
Note that most of this radiation originates in the troposphere as a result of thermal energy convected up from the surface. That is why the surface absorption plot shown in the main article can be very misleading as only (390-324)=66 of the 235 joules per second per square meter shown escaping the troposphere on the classic Kiehl-Trenberth diagram actually is radiated directly from the surface.
http://wattsupwiththat.files.wordpress.com/2009/11/trenberth-color-best.jpg