Guest post by Reed Coray
The following example illustrates the issues I have with reasoning often used to argue that increasing the amount of CO2 in the Earth’s atmosphere will increase both the Earth’s surface temperature and the Earth’s atmosphere temperature. Immediately following is a direct quote from URL
http://www.school-for-champions.com/science/heat_transfer_earth.htm
“The present situation is that there has been an increase in infrared-absorbing gases in the atmosphere, such as carbon dioxide (CO2) and methane (CH4). Energy that would normally escape into space is absorbed by these molecules, thus heating the atmosphere and spreading through convection currents. The average temperature of the atmosphere has increased 0.25 °C since 1980, mainly attributed to an increase in infrared-absorbing gases in the atmosphere.”
Although the above statement makes no direct reference to Earth surface temperature, I believe it carries the implication that greenhouse gases in the Earth’s atmosphere increase the Earth’s surface temperature.
I make two comments: the first is relevant only if the above implication is valid, the second is relevant independent of the validity of the implication. First, placing matter adjacent to a warm surface such that the matter is capable of absorbing/blocking radiation to space from the warm surface can lead to a decrease in the warm surface’s temperature. Second, increasing the amount of the absorbing/blocking matter can lower the temperature of the absorbing/blocking material.
Take for example an internal combustion engine whose metal surface is exposed to a vacuum. In addition to doing useful work, the engine produces thermal energy (heat). That thermal energy will produce a rise in the temperature of the engine’s surface such that in energy-rate equilibrium the rate energy is radiated to space from the engine’s surface is equal to the rate thermal energy is generated within the engine. By attaching radiating plates to the engine’s surface, some of the energy radiated to space from the engine’s original surface will be absorbed/blocked by the plates; but because thermal energy can be transferred from the engine to the plates via both radiation and conduction, the temperature of the engine’s original surface will be lowered. This is the principle of an air-cooled engine[1]: provide a means other than radiation of transferring heat from an engine to a large surface area from which heat can be removed via a combination of conduction, convection and radiation, and the engine’s surface temperature will be lowered.
If plates at a temperature lower than the original engine surface temperature are attached to the engine, it’s true that the temperature of the plates will increase to establish energy-rate equilibrium. Once energy-rate equilibrium is established, however, increasing the plate radiating area (adding additional matter that blocks more of the energy radiated from the original engine surface) will likely lower the plate temperature.
Thus, blocking the amount of surface radiation escaping to space does not necessarily increase the surface temperature; and increasing the amount of radiation blocking material does not necessarily increase the temperature of that material. In both cases (the Earth/Earth-atmosphere and the internal combustion engine in a vacuum), the heat eventually escapes to space–otherwise the temperature of the Earth’s surface and the engine would continue to rise indefinitely. The difference isn’t that the energy doesn’t eventually escape to space (it does in both cases), the difference is in the path the energy takes to reach space. The amount of generated thermal energy in conjunction with the path the thermal energy takes to get to space determines temperatures along the path; and adding more material may increase or decrease those temperatures. To say that “Energy that would normally escape into space is absorbed by these molecules, thus heating the atmosphere…” by itself is unwarranted; because an equivalent statement for the case of adding extra plate material to the engine would be “Energy that would normally escape to space from an engine with small attached plates is absorbed by additional plate material, thus heating the plates…” For air-cooled engines, this statement is not true—otherwise the plate surface area of air-cooled engines would be as small as possible.
It’s fairly easy to visualize why (a) adding thermally radiating plates to an air-cooled engine might decrease the engine’s surface temperature, and (b) increasing the area of the radiating plates might decrease the plate temperature. It’s not so easy to visualize, and may not be true, why (a) adding greenhouse gases to the Earth’s atmosphere decreases the Earth’s surface temperature; and (b) increasing the amount of atmospheric greenhouse gases lowers the temperature of the Earth’s atmosphere. I now present one possible argument. I do not claim that the argument is valid for greenhouse gases in the Earth’s atmosphere, but I do claim that the argument might be valid, and can only be refuted by an analysis more detailed than simply claiming “Energy that would normally escape into space is absorbed by these molecules, thus heating the atmosphere.”
If we assume that (a) matter cannot leave the Earth/Earth-atmosphere system, and (b) non-greenhouse gases radiate negligible energy to space, then for a non-greenhouse gas atmosphere the only way thermal energy can leave the Earth/Earth-atmosphere system to space is via radiation from the surface of the Earth. The rate radiation leaves the surface is in part a function of both the area and temperature of the surface. For a greenhouse gas atmosphere, energy can leave the Earth/Earth-atmosphere system to space both via radiation from the Earth’s surface and radiation from greenhouse gases in the atmosphere. Suppose it is true that the density of greenhouse gases near the Earth’s surface is such that radiation emitted from low-altitude greenhouse gases does not directly escape to space, but is in part directed towards the Earth’s surface and in part absorbed by other atmospheric greenhouse gases. As the atmospheric greenhouse gas density decreases with increasing altitude, radiation emitted from high-altitude greenhouse gases can directly escape to space.
Now it’s not impossible that since (a) in addition to radiation, heat is transferred from the Earth’s surface to greenhouse gases via conduction, and (b) convection currents (i) circulate the heated greenhouse gases to higher altitudes where energy transfer to space can take place and (ii) return cooler greenhouse gases to the Earth’s surface, that the process of heat transfer away from the Earth’s surface via greenhouse gases is more efficient than simple radiation from the Earth’s surface. Many engines are cooled using this concept. Specifically, a coolant is brought into contact with a heated surface which raises the coolant’s temperature via conduction and radiation, and the coolant is moved to a location where thermal energy transfer away from the coolant to a heat sink is more efficient than direct thermal energy transfer from the heated surface to the heat sink.
One way to realize increased thermal transfer efficiency would be to use a coolant, such as greenhouse gases, that efficiently radiates energy in the IR band (i.e., radiates energy at temperatures around 500 K). Another way would be to spread the heated coolant over a large surface area. Since surface area increases with increasing altitude, thereby providing expanded “area” (in the case of a gas, expanded volume) from which radiation to space can occur, it’s not clear to me (one way or the other) that greenhouse gases won’t act as a “coolant” reducing both the temperatures of the Earth’s atmosphere and the Earth surface.
[1] It’s true that for most air-cooled engines the main transfer of heat from the engine plates is via a combination of (a) conduction of heat to the air near the plates, and (b) convection that replaces the warm air near the plates with cooler air. To aid this process, a fan is often employed, or the engine is located on a moving vehicle and the vehicle’s motion through an atmosphere provides the flow of air across the plates. Although conduction/convection may be the primary means of heat dissipation from the plates, radiative cooling also dissipates heat.
Hi Reed
I am never comfortable with statements such as “greenhouse gases slow down the rate of cooling”. It is not a description I would use because it is not clear what it means and leads to more confusion. I prefer to say – Greenhouse gases in the atmosphere make the SURFACE of the planet warmer than it would be without them.
As for your questions, I have some problem with wording of them and rather than bicker about that let’s get to the point. The amount of energy the earth receives from the sun is always going to be balanced by the amount that the earth radiates back to space. This is called the Radiation Balance. No matter how much greenhouse gas is put into the atmosphere this balance will always be restored. The amount of energy leaving the earth will equal the amount of energy coming in.
The important point is that the radiation balance applies at the top of the atmosphere (TOA). We do not live at the top of the atmosphere, we live on the surface. It is the temperature on the surface which is important to us, not the temperature or radiation balance at TOA. Greenhouse gases do not affect the rate at which radiation comes in or goes out, that is why I find statements like “greenhouse gases slow down the rate of cooling” can be confusing.
However, greenhouse gases DO affect the temperature at the surface. They help to keep the planet warm and habitable – and there are many descriptions of how they do that.
trcurtin says:
davidmhoffer says:
I just tried to find the post where this came up but couldn’t…There’s too many posts here on WUWT. And, as I think I tried to say from the wording, this is my understanding of what they said since the wording itself was a bit cryptic. However, they were clearly addressing the issue of why the temperature change one calculates naively from using the S-B Law with T=255 K and a forcing of about 3.7 W/m^2 or so gives a temperature that is a bit lower than what the models find for a doubling of CO2 (absent feedbacks).
The statement isn’t so much that it makes no difference but rather that it depends carefully on how one is defining the zero-feedback state. And, while one might prefer one definition to another, I don’t think there is any right or wrong one.
I’ll probably have at best only sporadic web access over the next several days, so I may be slow to respond to future discussions.
Konrad says:
Unfortunately, what it illustrates is only that you still don’t understand the science of radiative transfer in the atmosphere. Your linear function cooling effect is totally fictional. The NET effect of the fact that CO2 molecules absorb and emit radiation is an approximately logarithmic radiative forcing (in the current concentration regime) with concentration.
”
Phil. says: July 23, 2012 at 1:48 pm
Outgoing flux=Incoming flux
∴ Surface radiation – absorption by GHGs = Incoming flux (constant)
So k*T^4 – f*ghg([CO2]) = constant
so if ghg([CO2]) goes up then T^4 must go up by the same amount. So if T^4 goes up by 4% then T goes up by ~1%..
Thanks again for your response.
I agree with your math–i.e., I agree that in an expression x – y = constant, if x goes up then y goes up by the same amount. I want to make sure, however, that we’re not talking at cross purposes regarding what you mean by “flux”. In the context of our discussion, the Stefan-Boltzmann equation, the k*t^4 term in your equation is “energy rate” per unit area. [If you want to include the surface energy in the constant “k”, then the term in your equation corresponds to energy rate. However, in neither case does it correspond to energy itself.] That is, (a) “surface radiation” is the rate (not the amount of) energy leaves a surface (either per unit area or for the total surface, which for his discussion I’ll take to mean “for the total surface”), (b) “absorption by GHGs” is the rate GHGs absorb energy, and “Incoming flux” is the rate energy enters the system comprised of the surface and the GHGs. Furthermore, I assume your equation applies to a state of “energy-rate” equilibrium–i.e., sufficient time as elapsed that the individual terms in your equation do not change with time.
If this interpretation is correct, then according to your equation as greenhouse gas levels increase, the temperature of the surface will increase to keep the equation in balance. So far so good. However, your equation implies that the rate energy is absorbed by greenhouse gases is never zero–in fact is a non-zero constant. So if your equation represents something physical, then isn’t it legitimate to ask: where does the energy that over time keeps being absorbed by (i.e., keeps accumulating) in the GHGs go? For energy rate equilibrium, if energy is absorbed by GHGs at a non-zero constant rate, doesn’t that energy have to leave the greenhouse gases at the same rate? If this is the case, shouldn’t your equation have in addition to a non-zero term for the “rate of energy aborption by GHGs” also have a non-zero term for the “rate of loss of energy by GHGs?”
”
**********************************
WRONG!
incoming flux is not a constant! It contains variables with variational effects greater than that of a co2 doubling from variables that are greater in magnitude than all co2 contributions. While the sun is rather constant, Earth’s albedo is not and the incoming flux that stays is the difference between this relative solar constant and that power returned to space as albedo reflection. Also, the actual solar incomming power varies by several times the effect of a co2 doubling as Earth travels through its orbit – as I recall about 90 W/m^2 peak to peak difference between perihelion and aphelion. Note these are two items are different factors. The net result of the latter is that the TSI reaching the southern hemisphere TOA is significantly greater than that reaching the northern hemisphere compared with the equivalent power effect of a co2 doubling, Combine that with the realization that most of Earth’s land mass is in the northern hemisphere and surface albedo of water tends to be under 0.04 as compared to Earth’s average of around 0.30 and voila, simple explanations would demand that the southern hemisphere be much hotter than the northern hemisphere – which it isn’t.
One of the ‘tells’ (giveaways where gamblers inadvertently show their position to their competition) I saw early on concerning the CAGW literature was the treatment of albedo as essentially a constant along with the lip service about albedo change being due to human induced land use changes being another threat to earth (mother goddess).
While one can do rather well using averages and stefan’s law to understand things, to assume albedo has little to no variation is to utterly fail to understand the more important factors involved in the whole problem.
It’s actually quite easy and rather accurate to determine Earth’s sensitivity to changes in W/m^2 incoming power being absorbed and the need for temperature variations to make up for the changes to achieve a balance. It’s also quite easy to see the ongoing presence of net negative feedback where the Earth’s change in temperature to achieve balance is less than that of a simple black body. All can be done by using a few known values and running stefan’s law forward and backwards a few times.
Incoming power average = 341 w/m^2, Earth’s average T = 288.2k , Earth’s albedo as we’ve measured it = 0.30. Stefan’s law gives us 391 w/m^2 for a bb at 288.2k. Incoming absorbed power is (1-albedo)*341w/m^2 = 239w/m^2. For balance, what escapes from the surface and atmosphere to space must equal what is absorbed = 239w/m^2 which gives us 255k which is what a bb with 0.30 albedo located at Earth’s orbit would be without an atmosphere blocking the escape of some of the IR. The difference is 33k or 33 deg C warming for all of the absorbed outgoing power that doesn’t escape to space. Take the difference of what leaves the surface and what escapes = 391-239 = 152 w/m^2 is captured in the atmosphere and doesn’t escape. Note that only about 2/3 of this is ghgs and clouds and aerosols etc. make up the other 1/3 and also that only 0.61 or 61% (239/391) of what leaves the surface escapes to space.
We have atmospheric effects blocking 152W/m^2 and providing warming of 33 deg C. 33/152 = ~0.22 deg C / W/m^2 change. This is actually the real average for Earth with all the feedbacks present. Note it is not the politically defined co2 doubling sensitivity that contains supposed additional feedbacks and assume co2 has a specific amount of w/m^2 for a doubling.
The straight calculation for a bb blanketed by the atmosphere would be to assume that for a 1 W decrease in outgoing power, 239 to 238 W/m^2, the Earth’s surface would have to heat up by enough for stefan’s law to return that 238 to 239 – but remember – only 61% of what is radiating from the surface escapes so we’d need to increase the T by enough so that 1/0.61 = 1.6 W/m^2 or 391 + 1.6 = 392.6W/m^2 which gives us (reversing stefan’s law equation) 288.47 or 0.27 deg C over the original T of 288.2k. Comparing this to the 0.22deg C rise, you can see it takes less warming to regain balance than that of the simple black body – which indicates that there is actually net negative feedback present instead of positive feedback.
Since the accepted increased power absorption for a co2 doubling is 3.7W/m^2, our sensitivity for a co2 doubling without additional feedbacks due to the rise in temperature would be 3.7 * 0.22 = 0.8 deg C. Positive feedback is extremely limited in values for there to be a stability and considering that the Earth has quite a variation in parameters that affect our balance, it is nuts to talk about high values. Let’s put a number or two to this though.
Atmospheric ghg absorption depends on the actual number of molecules of a gas in the air column. Absolute humidity fits this bill but relative humidity, RH, does not. If we increase T by 5 deg C in the entire atmospheric column where water vapor is present and if we hold RH constant – a common assumption for climatologists, we can consult the table that shows we get a 30% increase in absolute humidity. Like co2, h2o vapor is in the log region and has been for many doublings. It has roughly a little over twice the effect of co2 in the atmosphere and is roughly contributing a little over twice the doubling effect of co2. Since a 30% increase is a far cry from a full doubling, one finds there to be about 3.1 W/m^2 contribution for a full 5 degree increase in T which gives us 3.7w/m^2 (co2) +3.1w/m^2(h2o) = 7.8w/m^2 increase in our assumed 5 deg C rise due to a co2 doubling. Using our sensitivity of 0.22 deg C/W/m^2, we find we have enough power absorption to raise T by 0.22 x 7.8 = 1.7 deg C using our co2 forcing and our h2o ipcc proclaimed “primary” feedback. This leaves only 5-1.7 = 3.3 deg C of feedback missing from our consideration and it means that h2o vapor cannot be primary. Also, it means that this other feedback is far greater than stability limits allow. One can go back and assume 2 deg C rise from a co2 doubling + feedback but that substantial contribution from h2o vapor was due to the assumption of a full 5 deg C rise in the entire atmospheric column. If you drop that to 2 deg C, you lose most of that 3.1w/m^2 and it becomes a small fraction of the co2 forcing value
A final question or comparison shows the ridiculous nature of things when it comes to high sensitivity/effects. Co2 has been essentially a log function for almost a dozen doublings to get to our present value. Put another way, a co2 doubling now has the effect of about 10% of the total effect of co2 . A 5 deg C rise in temperature would require 5/0.22 = 22.7 W/m^2 of added forcing and co2 directly only can contribute 3.7w/m^2 and h2o could only contribute around 3.1 w/m^2. Considering that ghgs contribute to only around 100 w/m^2 total at present time, to claim that a co2 doubling could affect the total contribution by 20% is utterly and totally ridiculous.
Reed Coray says:
July 23, 2012 at 9:40 pm
Fifth Question: If we’ve reached this question, we are in agreement that the “rate of cooling of the Earth/Earth-atmosphere system” is unaffected by the presence of greenhouse gases. Is it then logical to argue that the presence of greenhouse gases in the Earth’s atmosphere “slow down the rate of cooling” and it is the “slow down of the rate cooling” that causes the Earth/Earth-system temperature to be different in the presence/absence of atmospheric greenhouse gases?
Yes, because it is the rate of loss from the surface that is reduced by the presence of GHGs, however the loss from the TOA is unaffected once steady state is achieved.
Reed Coray says: July 23, 2012 at 9:40 pm …
Reed, I like what you said.
Perhaps the biggest misconception you address is how “fast” or “slow” energy enters and leaves. As you quite properly state, we are interested in the RATE that energy enters or leaves, dE/dt (in Watts), not the SPEED it enters or leaves, dv/dt (in m/s). To be more accurate, people should say the rate “increases” or “decreases”. While the the term “speed up energy loss” could be correctly interpretted to me “increase dE/dt”, others can easily interpret it to mean something about the “speed of the photons” or “the time it takes a give ‘dE’ of energy to get from the ground to the top of the atmosphere”. Too many people seem to be hung up on the idea that the speed matters, when in fact it is irrelevant.
Kudos.
~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~
In the series of questions you ask, I think you could clarify your ideas a bit by throwing in the terms “average” and “steady-state” and “net” occasionally, but overall I agree with what you said.
Basically, after the series of rhetorical questions, you have established that 341 W/m^2 arrive at the earth from the sun, and 341 W/m^2 leave again to space (on average after steady-state conditions have been achieved). You have the information from the top edge of Trenberth’s energy flow diagram: http://www.agci.org/classroom/images/trenberth_energy.png.
NOW comes the interesting part — the rest of the diagram!
1) What happens between the TOA (top of atmosphere) and the ground, and how does this affect surface temperature?
2) How does continually changing the CO2 level affect things and how far from steady-state does that make the earth?
3) How do OTHER factors (variable sunlight, variable orbit, variable albedo, variable humidity …) affect the average, steady-state temperature?
4) Does it even make sense to talk about “steady state” when there are changes constantly occurring on scales from minutes to millions of years?
I have been following the comments here, smiling and LOL, sometimes, and I have come to the same conclusion that I came after my initial investigations,
on whether my carbon footprint is harmful to earth,
http://www.letterdash.com/HenryP/more-carbon-dioxide-is-ok-ok
As a scientist who not only follows the truth, but also knows the Truth, what I notice is that the pro AGW camp increasingly wants to make it seem that atmospheric chemistry and physics is so difficult that they themselves hardly understand it – and indeed they often have to consult each other on questions being posed -their income depends on it!!!
Yet, what I have found, is, that all you need is a basic understanding of probability theory and sampling technigues, which is taught at most third year university level statistics.
You are being all deluded into looking at AVERAGE temps. which has an awful lot of weather NOISE when you should be looking at MAXIMA, which gives you a proper look at the energy input by the sun.
http://www.letterdash.com/henryp/global-cooling-is-here
cba:”Atmospheric ghg absorption depends on the actual number of molecules of a gas in the air column. Absolute humidity fits this bill but relative humidity, RH, does not. If we increase T by 5 deg C in the entire atmospheric column where water vapor is present and if we hold RH constant – a common assumption for climatologists, we can consult the table that shows we get a 30% increase in absolute humidity. Like co2, h2o vapor is in the log region and has been for many doublings. It has roughly a little over twice the effect of co2 in the atmosphere and is roughly contributing a little over twice the doubling effect of co2. Since a 30% increase is a far cry from a full doubling, one finds there to be about 3.1 W/m^2 contribution for a full 5 degree increase in T which gives us 3.7w/m^2 (co2) +3.1w/m^2(h2o) = 7.8w/m^2 increase in our assumed 5 deg C rise due to a co2 doubling.”
I must say I don’t follow the derivation of your 3.1 W/m2 number above so let me show how I look at these numbers in the past. According to most of the numbers I have the seen, the total magnitude of the WV portion of the GH effect is supposed to be 75W/m2 and each additional degree of warming should put enough WV into the air to increase the WV-GHE by 2W/m-2. Assuming that the WV GH effect is basically logarithmic (like CO2) gives you something around 23 or 24W/m2 for a doubling of WV(GH of doubling/ln(2)=2Wm^-2/ln(1.06)).
However, this amount per this calculation, we can see that WV has only had the equivalent of a bit more than 3.2 doublings of WV (75w/m2/24W/m2). OTOH, CO2 has apparently had about 8.5 ~ 32W/m2 of CO2 GH effect/3.7 W/m2 of GH effect per doubling. Given that WV is supposed to be a better GH absorber and there is more WV for the vast majority of the atmosphere below the effective radiating level only logical way I can match these two numbers is to assume that WV has a negative feedback associated with it that reduces its net warming effect by at least 1/2.
Cheers, 🙂
Sorry about the last paragraph above – a couple of missing words may make it hard to understand. Here it is as I meant to write it.
However, given the 24W/m2 per the above calculation, we can see that WV has only had the equivalent of a bit more than 3.2 doublings of WV (75w/m^2/24W/m^2). OTOH, CO2 has apparently had about 8.5 ~ 32W/m2 of CO2 GH effect/3.7 W/m2 of GH effect per doubling. Given that WV is supposed to be a better GH absorber and there is more WV than CO2 for the vast majority of the atmosphere (below the effective radiating level) only logical way I can match these two numbers is to assume that WV has a negative feedback associated with it that reduces its net warming effect by at least 1/2. I believe that assuming WV has had the equivalent of about 6 doublings where each doubling has (on average) a 12W/m2 effect than arguing that it has had about three doublings where each doubling has(on average) a 24W/m2 effect.
Cheers, 🙂
Reed, I believe I have a more succint way to phrase the point I was trying to make above. IMO, the addition of GH gases slow down the rate of cooling while heating the surface speeds it up again. At equilibrium then, the rate *the Earth as a whole* cools with GH gases is the same rate at which it would cool without them. It is still accurate to say both that GH gases slow the rate of cooling and that the Earth as a whole cools at the same rate.
Cheers, 🙂
Michael Tremblay says:
July 24, 2012 at 1:05 am
Thanks Eli, I think that I found most of my answers under Wikipedia’s description for ‘Spontaneous Emission’. Your direction to IR multiphoton dissociation is interesting because it confirms a thought that I had that if you exceed the absorption and stimulated emission balance you can break the molecular bonds – unless I miss the mark, this can occur naturally but the chances of it happening are about the same as the odds of me winning the next lottery without buying a ticket 😉
You can’t beat the house, but if you provide other paths into or out of the system (e.g. feeding the upper level from reaction or a third state or providing an empty state you can emit to), you can get the same effect. This is basically what lasers do. The neatest one Eli knows is the CO2 laser in the upper atmosphere of Mars that Mike Mumma discovered.
joeldshore says:
July 24, 2012 at 5:08 am
“Unfortunately, what it illustrates is only that you still don’t understand the science of radiative transfer in the atmosphere. Your linear function cooling effect is totally fictional.”
—————————————————————————————————————-
Given your claim about my lack of understanding I believe you owe me an answer to the following multiple choice question –
I have two 1m spheres of thin LDPE plastic (transparent to LWIR) in a sunless vacuum in deep space. Both spheres contain gas at an initial temperature of 100C. Sphere 1 contains 100% O2. Sphere 2 contains 99% O2 and 1% CO2. Which sphere cools faster? Is the answer :
A. Sphere 1 cools faster.
B. Sphere 2 cools faster.
C. Sphere 1 and 2 cool at the same rate.
Or
D. None of the above.
A simple one letter answer is all that is required A, B, C or D. No hand waving, no “putting in context”, no reframing the question. Simply A, B, C or D.
Konrad says: July 24, 2012 at 2:24 pm
“I have two 1m spheres of thin LDPE plastic … “
I’m trying to decide if there is some “trick” that I am missing, since the answer seems glaringly obvious. The sphere with CO2 can radiate IR, the sphere without CO2 cannot (to any noticeable extent). Thus the sphere with CO2 can cool effectively, while the other can’t.
B. Sphere 2 with some CO2 will cool faster. Final answer. I am 99.9% sure Joel would agree (we tend to agree on basic physics like this).
I’m curious what the NEXT question is and why this question is important.
A number of commenters claim the so-called location of outgoing radiation is moved up due to GHGs
1. Where is the observational evidence of this?
2. Why do theoretical calculations on MODTRAN show the temperature profile of the atmosphere in the troposphere and stratosphere does not change at all between CO2 concentration inputs of 350 and 3500?
http://forecast.uchicago.edu/Projects/modtran.html
Konrad says: July 24, 2012 at 2:24 pm
“I have two 1m spheres of thin LDPE plastic … “
I believe the balloon surface of LDPE plastic would radiate to space at the same rate in both scenarios with equivalent starting temperatures, and therefore answer C
Hockey Schtick asks “Why do theoretical calculations on MODTRAN show the temperature profile of the atmosphere in the troposphere and stratosphere does not change at all between CO2 concentration inputs of 350 and 3500?”
As I understand it, MODTRAN is designed simply to calculate the radiation from given initial conditions, not to actually predict those initial conditions. So concentrations of gases, amount of cloud cover, and temperature profiles (among other things) are INPUTS to this program. If you want a different profile, you would program it into the computer.
Hockey Schtick says: “I believe the balloon surface of LDPE plastic would radiate to space …”
Since the plastic was postulated to be transparent, then I assumed it was 100% transparent. This means it can neither absorb nor emit IR => emissivity = 0. (In reality, the plastic might well emit IR better than such a small volume of CO2, but that is not what the question was focusing on, I think).
Tim,
The correct answer is of course B. The CO2 in the sphere acquires energy conductivly from the O2 and then radiates it to space. The point of the question was to get Joel to acknowledge that CO2 in our atmosphere can radiate energy to space that it has acquired conductivly from Earths surface and atmosphere. CO2s ability to cool in this manner should be a linear function of its concentration in the atmosphere.
Joel Shore: it is all too clear that you did not have time to read the Grigg Harries (GH, JoC, 2007) paper properly before rushing to judgment on me. What you quote from the careless Abstract to their paper is misleading as nearly the whole paper deals only with their data on differences in brightness temperatures by wave number in the spectra between 1970 and 2003. The brightness temperatures within the IR spectrum are NOT the same as the temperature at any given altitude, and while they claim to have shown that bT increased between 1970 and 2003, their own data in their Tables A1-5 show no correlation between actual temperatures by altitude in the atmosphere, which range from 192 K to 299 K and their brightness temperatures (by wave number) in their Fig.6, which range only between 288 and 296 K in Fig.6c and 240 to 278 K in Fig. 6f.
I should not have to tell you that brightness temperature is the temperature a black body in thermal equilibrium with its surroundings would have to have in order to duplicate the observed specific intensity of an object at a frequency v, and has little to do with the GH observed temperatures by altitude in the troposphere and stratosphere. Similarly the wave numbers in the GH Figures bear no relation to the altitudes in the atmosphere in Tables A1-6.
The GH discussion of the NCEP and ECMWF data in Tables A1-A6 in their Appendix does not mention the temperatures at each altitude (in hPa), so they failed to notice either that the temperatures in 1970, 1997, and 2003, are virtually identical in each year at each hPa, or that there are declining trends between 1970 and 1997, and between 1997-2003, so NO warming at all.
Unfortunately Griggs and Harries do not provide the raw data for the brightness temperatures shown in their Figures, but to judge from their Fig.6’s c and f panels, there is really no visible difference between their brightness temperatures for 1970, 1997 and 2003 in the wave numbers between 800 and 940 cm/1, or wave numbers 1280 to 1400 c/1.
Konrad, I am not sure which Tim you are referring to, but I would certainly also have answered (B). Has Joel rsponded?
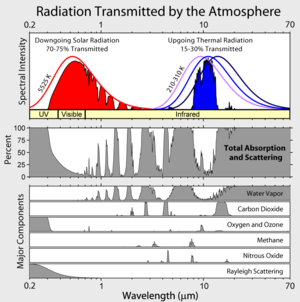
You are missing something.
Please compare the following:
1. The radiation spectrum of a body with a temperature of roughly 5,500 K
2. The radiation spectrum of a body with a temperature of roughly 280 K
3. The absorption spectrum of CO2
Konrad says: “The point of the question was to get Joel to acknowledge that CO2 in our atmosphere can radiate energy to space that it has acquired conductivly from Earths surface and atmosphere. CO2s ability to cool in this manner should be a linear function of its concentration in the atmosphere.”
If that was your point, then you haven’t succeeded.
We agree that …
* Yes, CO2 can receive energy from O2.
* Yes, CO2 can radiate energy to space.
* Yes, Sphere B will cool faster.
But …
1) There is no reason to expect this to be linear. For small enough concentrations and short enough distances, the effect would be approximately linear. But as the length and/or concentration increased you get a “saturation” effect (and you should (at least approximately) exponentially approach some maximum emission of IR). At some point, the CO2 can’t radiate to a greater degree because CO2 molecules near the outside block IR that was emitted by the inner CO2 molecules. For something as small as a 1 m sphere, this saturation effect would (I strongly suspect) be minimal, but for the earth’s atmosphere, the IR that gets emitted to space by CO2 comes from near the top of the troposphere. Adding more CO2 will not make significantly more IR come out.
2) This is more subtle, but you can only say that CO2 in the atmosphere has an “ability to cool” the earth in a very restrictive sense. It’s like having a bank of bright lights and setting some dimmer lights in front of that panel. Yes, the dim lights are still providing some light, but they block the light that would have been coming from the brighter lights behind them. The net effect is of these “extra lights” to make things darker!
The warm earth is the “bank of bright (IR) lights” (which is missing in your analogy with the spheres, and hence invalidates many of the conclusions you might try to draw). The cool CO2 is the “dimmer lights in the way”. Yes, the CO2 emits IR which “cools” the earth, but the net effect is to make the earth “darker” for outgoing IR radiation (as you can clearly see in satellite IR measurements). In the case of the earth, the “total IR emitted” must eventually equal the total sunlight absorbed. So on the earth, the “bright panel of lights” have to get turned up (ie get warmer) until the outgoing light is the same as it would have been with out the “dim bulbs” blocking the light.
Or put another way, the CO2 in the atmosphere cools the earth, but it does that cooling so poorly (compared to the warmer surface behind it), that the net effect is to warm the earth!
”
Hockey Schtick says:
July 24, 2012 at 7:38 pm
A number of commenters claim the so-called location of outgoing radiation is moved up due to GHGs
1. Where is the observational evidence of this?
2. Why do theoretical calculations on MODTRAN show the temperature profile of the atmosphere in the troposphere and stratosphere does not change at all between CO2 concentration inputs of 350 and 3500?
http://forecast.uchicago.edu/Projects/modtran.html
”
************************
at the simplest level, one would expect it to. In reality, under clear skies most of it originates from the surface and the rest is smeared across the altitudes. Under cloudy skies, most originates from the cloud surface and the rest from the altitudes above that.
I believe the project giving observational information is ARM. It provides rather interesting graphs that show blackbody curves of various temperatures and shows where the absorption occurs is really just emission at lower temperatures.
MODTRAN calculator is a simple program that provides limited abilities to alter the inputs. I doubt they do a conservation of energy calculation for the air column nor would it be accurate without the inclusion of convection. for what it is, the MODTRAN calculator is a nice program that can provide a nice bit of information. However, it does not convey any serious information one way or the other when it comes to GW, much less prove AGW or CAGW.
just remember, all charletons that peddle their electric or magnetic cures for dropsy, cancer, and the common cold have some sort of actual scientific trappings or demo equipment to generate electricity and magnetism to convince the rubes that it’s not just talk. The first digital computer I ever saw as a kid was at a carnival side show – telling fortunes. Mine was that I’d grow up tall rich and famous. It was only 25% accurate. It got the part right about growing up. LOL
Hockey Schtick says:
July 24, 2012 at 7:38 pm
2. Why do theoretical calculations on MODTRAN show the temperature profile of the atmosphere in the troposphere and stratosphere does not change at all between CO2 concentration inputs of 350 and 3500?
Because you omitted to adjust your initial conditions. Try again but this time adjust the ‘ground T offset’ until you get the same ‘Iout’, otherwise you’re forcing it to have the same profile.