Guest post by Reed Coray
The following example illustrates the issues I have with reasoning often used to argue that increasing the amount of CO2 in the Earth’s atmosphere will increase both the Earth’s surface temperature and the Earth’s atmosphere temperature. Immediately following is a direct quote from URL
http://www.school-for-champions.com/science/heat_transfer_earth.htm
“The present situation is that there has been an increase in infrared-absorbing gases in the atmosphere, such as carbon dioxide (CO2) and methane (CH4). Energy that would normally escape into space is absorbed by these molecules, thus heating the atmosphere and spreading through convection currents. The average temperature of the atmosphere has increased 0.25 °C since 1980, mainly attributed to an increase in infrared-absorbing gases in the atmosphere.”
Although the above statement makes no direct reference to Earth surface temperature, I believe it carries the implication that greenhouse gases in the Earth’s atmosphere increase the Earth’s surface temperature.
I make two comments: the first is relevant only if the above implication is valid, the second is relevant independent of the validity of the implication. First, placing matter adjacent to a warm surface such that the matter is capable of absorbing/blocking radiation to space from the warm surface can lead to a decrease in the warm surface’s temperature. Second, increasing the amount of the absorbing/blocking matter can lower the temperature of the absorbing/blocking material.
Take for example an internal combustion engine whose metal surface is exposed to a vacuum. In addition to doing useful work, the engine produces thermal energy (heat). That thermal energy will produce a rise in the temperature of the engine’s surface such that in energy-rate equilibrium the rate energy is radiated to space from the engine’s surface is equal to the rate thermal energy is generated within the engine. By attaching radiating plates to the engine’s surface, some of the energy radiated to space from the engine’s original surface will be absorbed/blocked by the plates; but because thermal energy can be transferred from the engine to the plates via both radiation and conduction, the temperature of the engine’s original surface will be lowered. This is the principle of an air-cooled engine[1]: provide a means other than radiation of transferring heat from an engine to a large surface area from which heat can be removed via a combination of conduction, convection and radiation, and the engine’s surface temperature will be lowered.
If plates at a temperature lower than the original engine surface temperature are attached to the engine, it’s true that the temperature of the plates will increase to establish energy-rate equilibrium. Once energy-rate equilibrium is established, however, increasing the plate radiating area (adding additional matter that blocks more of the energy radiated from the original engine surface) will likely lower the plate temperature.
Thus, blocking the amount of surface radiation escaping to space does not necessarily increase the surface temperature; and increasing the amount of radiation blocking material does not necessarily increase the temperature of that material. In both cases (the Earth/Earth-atmosphere and the internal combustion engine in a vacuum), the heat eventually escapes to space–otherwise the temperature of the Earth’s surface and the engine would continue to rise indefinitely. The difference isn’t that the energy doesn’t eventually escape to space (it does in both cases), the difference is in the path the energy takes to reach space. The amount of generated thermal energy in conjunction with the path the thermal energy takes to get to space determines temperatures along the path; and adding more material may increase or decrease those temperatures. To say that “Energy that would normally escape into space is absorbed by these molecules, thus heating the atmosphere…” by itself is unwarranted; because an equivalent statement for the case of adding extra plate material to the engine would be “Energy that would normally escape to space from an engine with small attached plates is absorbed by additional plate material, thus heating the plates…” For air-cooled engines, this statement is not true—otherwise the plate surface area of air-cooled engines would be as small as possible.
It’s fairly easy to visualize why (a) adding thermally radiating plates to an air-cooled engine might decrease the engine’s surface temperature, and (b) increasing the area of the radiating plates might decrease the plate temperature. It’s not so easy to visualize, and may not be true, why (a) adding greenhouse gases to the Earth’s atmosphere decreases the Earth’s surface temperature; and (b) increasing the amount of atmospheric greenhouse gases lowers the temperature of the Earth’s atmosphere. I now present one possible argument. I do not claim that the argument is valid for greenhouse gases in the Earth’s atmosphere, but I do claim that the argument might be valid, and can only be refuted by an analysis more detailed than simply claiming “Energy that would normally escape into space is absorbed by these molecules, thus heating the atmosphere.”
If we assume that (a) matter cannot leave the Earth/Earth-atmosphere system, and (b) non-greenhouse gases radiate negligible energy to space, then for a non-greenhouse gas atmosphere the only way thermal energy can leave the Earth/Earth-atmosphere system to space is via radiation from the surface of the Earth. The rate radiation leaves the surface is in part a function of both the area and temperature of the surface. For a greenhouse gas atmosphere, energy can leave the Earth/Earth-atmosphere system to space both via radiation from the Earth’s surface and radiation from greenhouse gases in the atmosphere. Suppose it is true that the density of greenhouse gases near the Earth’s surface is such that radiation emitted from low-altitude greenhouse gases does not directly escape to space, but is in part directed towards the Earth’s surface and in part absorbed by other atmospheric greenhouse gases. As the atmospheric greenhouse gas density decreases with increasing altitude, radiation emitted from high-altitude greenhouse gases can directly escape to space.
Now it’s not impossible that since (a) in addition to radiation, heat is transferred from the Earth’s surface to greenhouse gases via conduction, and (b) convection currents (i) circulate the heated greenhouse gases to higher altitudes where energy transfer to space can take place and (ii) return cooler greenhouse gases to the Earth’s surface, that the process of heat transfer away from the Earth’s surface via greenhouse gases is more efficient than simple radiation from the Earth’s surface. Many engines are cooled using this concept. Specifically, a coolant is brought into contact with a heated surface which raises the coolant’s temperature via conduction and radiation, and the coolant is moved to a location where thermal energy transfer away from the coolant to a heat sink is more efficient than direct thermal energy transfer from the heated surface to the heat sink.
One way to realize increased thermal transfer efficiency would be to use a coolant, such as greenhouse gases, that efficiently radiates energy in the IR band (i.e., radiates energy at temperatures around 500 K). Another way would be to spread the heated coolant over a large surface area. Since surface area increases with increasing altitude, thereby providing expanded “area” (in the case of a gas, expanded volume) from which radiation to space can occur, it’s not clear to me (one way or the other) that greenhouse gases won’t act as a “coolant” reducing both the temperatures of the Earth’s atmosphere and the Earth surface.
[1] It’s true that for most air-cooled engines the main transfer of heat from the engine plates is via a combination of (a) conduction of heat to the air near the plates, and (b) convection that replaces the warm air near the plates with cooler air. To aid this process, a fan is often employed, or the engine is located on a moving vehicle and the vehicle’s motion through an atmosphere provides the flow of air across the plates. Although conduction/convection may be the primary means of heat dissipation from the plates, radiative cooling also dissipates heat.
Spector says:
July 23, 2012 at 10:28 am
The colder body just does not radiate as much. The standard IPCC thermal budget diagram
=================================================
There is no need to care about the “IPCC thermal budget diagram” because the warmism is stuck with its key assertion (colder body reduces cooling of warmer body by radiation) apparently not being proven experimentally.
It is just a narrative, rephrasing, repeating and so on.
RE; RACookPE1978:(July 23, 2012 at 10:35 am)
“Assume 1370 watts are present at the top of atmosphere at the equator.”
I believe that is the value at noon. Most of these calculations of this type are averaged over a full daily cycle. When albedo is also taken into account, (depending on what assumptions are made) I think the result can be close to 293 W/m² for the tropics.
Spector says:
That is not quite true . . . The colder body just does not radiate as much.
>>>>>>>>>>
I think this is the fundamental point that confuses a lot of people. In the absence of the colder body, what is there? People confine their thinking to the exchange of energy between the warm surface and the cold surface. We need to take one additional step and ask a simple question. If the cold surface was not there, what would be?
Answer: NOTHING!
Question: What is the temperature of “nothing”?
Answer: 2.7 degrees K, roughly -270 degrees C.
So, if we have a warm object at say 100 C exposed directly to space, it cools rather rapidly. If we insert a “cold” object at say -20 C in between the warm object and space, the warm object will obviously cool more slowly. The cooler object is only cooler relative to the warm object. But compared to NOT being there, the cool object is blazing hot. The cool object at -20 C is still 250 degrees C warmer than “nothing”.
Spector says:
July 23, 2012 at 10:58 am
RE; RACookPE1978:(July 23, 2012 at 10:35 am)
“Assume 1370 watts are present at the top of atmosphere at the equator.”
I believe that is the value at noon. Most of these calculations of this type are averaged over a full daily cycle. When albedo is also taken into account, (depending on what assumptions are made) I think the result can be close to 293 W/m² for the tropics.
>>>>>>>>>>>>>>>
No, it is the value presented by the sun to the earth if earth was a flat plane directly facing the sun. Adjust for albedo, subract 30%. Adjust for curvature of earth and rotation of earth divide by four. That gives you a rough average which is useless except for the most basic of discussions. I suggest
http://eos.atmos.washington.edu/erbe/
For actual data on relative energy flux at different latitudes going both up and down. Note that the result in the tropics is WELL above 293 w per m2.
RACookPE1978 Asks:
“What absorbed that 360 watts that are NOT transmitted, and where does that energy “go” if O2 and N2 do not radiate or absorb IR energy?
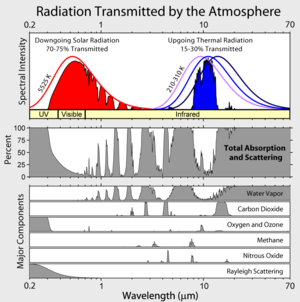
Look at the figure at the very top of this post: “Radiation Transmitted by the Atmosphere”.
* Ozone absorbs most of the UV.
* gaseous water absorbs a bunch of solar IR
* Rayleigh scattering sends some of it back to space
* Its not on the diagram, but aerosols absorb some, too.
All together, that would add up to about 360 W/m^2. Most of it would heat the atmosphere directly; some is lost back to space
<davidmhoffer says:
July 23, 2012 at 11:03 am
So, if we have a warm object at say 100 C exposed directly to space, it cools rather rapidly. If we insert a “cold” object at say -20 C in between the warm object and space, the warm object will obviously cool more slowly. The cooler object is only cooler relative to the warm object. But compared to NOT being there, the cool object is blazing hot. The cool object at -20 C is still 250 degrees C warmer than “nothing”.
davidmhoffer, I know you’re frustrated with the amount of time you’re having to spend on this issue, but I think it’s time well spent.
You’re getting better and better at explaining your point and probably getting more converts than you think.
Greg House – “The warmists problem is that they can not produce any real genuine falsifiable scientific experimental proof of that “reducing its rate of heat loss”. They just keep “explaining”, rephrasing and mostly suggesting I should prove their claim.”
Greg, I would suggest you look at the spectra linked/shown at the top of the page (http://upload.wikimedia.org/wikipedia/commons/thumb/7/7c/Atmospheric_Transmission.png/300px-Atmospheric_Transmission.png). The smooth curve on the right represents the spectra of a ‘blackbody’, the most efficient thermal radiator possible. The jagged blue spectra shows the actual emissions from our atmosphere to space, shaped by the various GHG’s in our atmosphere.
That emission spectra is lower than the blackbody spectra, meaning that the Earth must be at a higher temperature to radiate the same energy (the area under the spectral curve). As observed from space, the IR emissivity of the Earth is about 0.612, 61% as efficient as a blackbody. Feed that into the Stephan-Boltzmann equation (one of the best established relationships in physics):
Power = emissivity * SB Constant (5.670 373(21)×10^−8 W·m^−2·K^−4) * T^4
240 = 0.612 * SBc * T^4
T = 288.37 K = 15.2 C
And the shape of that spectra changes as we add more GHG’s, in a fashion clearly measurable from space, (Harries et al 2001, https://workspace.imperial.ac.uk/physics/Public/spat/John/Increase%20in%20greenhouse%20forcing%20inferred%20from%20the%20outgoing%20longwave%20radiation%20spectra%20of%20the%20Earth%20in%201970%20and%201997.pdf). We’ve observed the atmosphere emit IR less and less efficiently.
That’s the greenhouse effect in the simplest terms – increasing GHG’s decrease the amount of energy radiated at any particular temperature, creating an imbalance between incoming/outgoing energy, and hence the world warms in compensation – until that imbalance is cancelled out.
—
You have repeatedly demanded that folks show you the experiments to prove their case. They have. Thermal radiation, cooler objects (yet warmer than the background) adding to the temperature of warmer objects, and the 33 C higher temperature of the Earth than would be seen without GHG’s – those have all been proven. And the various experiments you have been pointed to (which all have been done in high schools across the world) are simply opportunities for you to observe the same thing yourself. Your repeated demands are, therefore, rather disingenuous.
Greg House:”Again: a colder body does radiate. But there is no law of thermodynamics nor a “well-established rule of physics” saying that a colder body can warm (or reduce cooling of) a WARMER body, in our case by means of radiation.”
Ok, so let’s see if we can unpack this a bit. If a colder body does radiate, and we can hopefully agree that radiation *can* warm(or reduce the cooling of) something what is it about the *radiation* that the colder body emits that stops it from doing so. Are you arguing that there are two types of radiation: radiation from cooler bodies and radiation from warmer ones or what?
It seems to me that one of the obvious consequences of your line of thought is that a warm object in a vacuum would cool *less* slowly than an object in the atmosphere. The warm object will radiate at the same rate in the atmosphere as in a vacuum but in an atmosphere it can also cool due to convection and conduction. OTOH, if we allow that cool objects can slow the cooling of warming ones, we can easily see how objects in an atmosphere will stay warmer than the same object in a vacuum, even though an object in a vacuum cannot cool through conduction and convection(and one in the atmosphere can).
Cheers, 🙂
yes davidmhoffer:
“So, if we have a warm object at say 100 C exposed directly to space, it cools rather rapidly. If we insert a “cold” object at say -20 C in between the warm object and space, the warm object will obviously cool more slowly. The cooler object is only cooler relative to the warm object. But compared to NOT being there, the cool object is blazing hot. The cool object at -20 C is still 250 degrees C warmer than “nothing”.”
The problem that the intransigent dopes have is that they have heard that heat always travels from a warm object to a cooler object. Actually, there is an interchange of heat/radiation. The colder object receives more radiation from the warmer object than it gives. Net effect for the colder object is that it gets warmed up. The warmer object cools less rapidly, because it is receiving some radiation from the colder object. At lest that is the crazy idea that physicists and engineers dreamed up, long ago.
davidmhoffer says:
beng says:
Fine…I will address it. It is based on a simple misconception: The ~1 degC temp change at the surface is obtained by taking the ~1 degC temperature change at the TOA and extrapolating down to the surface under the assumption that the lapse rate remains constant as the atmosphere warms.
Is that assumption a good one?
(1) It is not perfect, but it is not too bad. In fact, the lapse rate is expected to decrease a little bit with increasing temperature because the moist adiabatic lapse rate decreases with increasing temperature.
(2) It doesn’t matter.
Now, (2) might seem a bit strange of a claim, but here is the reason: The models all correct for the fact that the lapse rate decreases with temperature by including a feedback called the “lapse rate feedback”. It is a negative feedback that accounts for this.
So, what David Hoffer is arguing about comes down to just a matter of definition: The climate scientists talk about the zeroth-order temperature change being what you get if you assume a constant lapse rate and then to the extent that the lapse rate is not constant, it gets corrected, lowering the temperature change at the surface. By contrast, David Hoffer wants to take this correction and put it into the zeroth-order temperature change that gets talked about.
Either way you do it, you get the same answer. I think the way climate scientists do it makes more sense to them (and to me) because the climate modelers understand that the correct way to look at things is to consider the TOA radiation budget and then think about the lapse rate to figure out what happens at the surface. David’s way is wedded to thinking about the radiation budget at the surface which is usually not very helpful because for the surface energy budget, convection is very important. Trying to reason things out just on the basis of the radiative part of the equation tends to be very incomplete.
TimTheToolMan says:
The models and theories of radiative transfer in the atmosphere are backed up by a wealth of experiments. Basically, the entire field of remote sensing would cease to exist if it were wrong. If you are going to be consistent in your skepticism, you would also have to disbelieve Spencer and Christy’s entire body of work on the temperature record of the troposphere. Heck, you probably couldn’t even really believe the IR weather satellite photos that you see on TV.
Greg House says:
July 23, 2012 at 10:07 am
Phil. says:
July 23, 2012 at 9:21 am
…but there are products based on this, …Light bulbs with an IR reflective coating which causes the filament to heat up and emit more visible.
http://hirheadlights.com/USPTO%20HIR%20bulbs.pdf
http://www.pegasuslighting.com/par38-halogen-ir-light-bulbs-48w-flood-25-degree.html
=================================================
Look, just multiplying the claim is no proof of the claim being correct.
The manufacturer CLAIMS his bulb is more efficient, the same goes for the patent. Now, WHERE IS THE SCIENTIFIC PROOF FOR THAT? Nowhere, apparently.
We are talking about science here, not about marketing.
No some of us are talking about science you are not!
What did your last slave die of? Don’t be so lazy and get off your ass and google the items I referred you to and learn something about physics and engineering. I’m not going to do all the work for you, there’s enough information there for to find data about the increase of output/watt.
You want a verifiable experiment, place a heated filament at the focus of an uncoated, transparent spherical mirror (i.e. one that doesn’t reflect), measure the output at a certain power setting. Coat the mirror with a dichroic coating which reflects 95% of the IR above 1 micron and passes 95% of the light below 1 micron, now measure the output at the same power setting. You’ll find the temperature of the filament will go up with the corresponding change in light output.
Or if that’s too hard for you, immerse a small, bare Pt/Rh thermocouple in the flame of a Meker burner and measure the output of the Th/C. Then place a quartz tube around the Th/C to act as a radiation shield, measure the output again. You should find a increase in voltage consistent with a temperature increase of ~100ºC.
joeldshore;
So, what David Hoffer is arguing about comes down to just a matter of definition:
>>>>>>>>>
Indeed it does.
The definition in AR3 is that CO2 doubling = 3.7 w/m2 = +1 degree AT THE EFFECTIVE BLACK BODY TEMPERATURE OF EARTH which is -20C and considerably colder than average earth surface temps. This definition is carried over into AR4. If you are arguing that this definition tanslates 1:1 at earth surface, then I challenge you to explain why the definition was worded in the context of effective black body temperature in the first place. I challenge you also to explain how 3.7 w/m2 modeled at TOA can punch through the atmospheric column and result in not just getting through unimpeded, which would be a challenge unto itself, but to arrive at earth surface at 5.5 w/2. Where exactly did it pick up the extra 1.8 w/m2? Why was none of absorbed by water vapour and CO2 on the way down?
You can’t explain this away with lapse rate. The definition relies on the temperature at -20 C which is effective black body temperature of earth for a reason. If the value scaled 1:1 at earth surface there never would have been a reason to define it that way in the first place, and it is the only definition that provides for an energy balance as per SB Law.
So sorry, your lapse rate arm waving doesn’t cut it. The definition of CO2 doubling = +1 degree was made in reference to the effective black body temperature of earth for a reason. If it scaled 1:1 to earth surface then the definition would have been at earth surface, there would have been no reason to complicate it further.
The only reason for using effective black body temperature of earth that I can see as the reference point is because it does result in a higher number to present to the public.
Tim Folkerts says:
July 23, 2012 at 8:13 am
This is simply semantics, not science. ……
>>>>>>>>>>>>>>>>>>
Thanks for replying Tim.
You’re right that it is only semantics but you have to remember who the intended audience is. You and I could talk all day about the Greenhouse Effect and various Atmospheric Effects and understand the differences perfectly well. People in authority often use semantics to deliberately mislead people who do not understand. Use of the word greenhouse to describe this effect lends itself to misinterpretation. Proponents of CAGW love to use the word because most laypeople link greenhouses to warming, even though they may not know how a greenhouse actually works. On the other side, opponents of CAGW should avoid using the word precisely because their intended audience (laypeople) automatically link it to warming. – Just my own little rant 😉
Maybe I am taking too cynical a view of the level of understanding of climatologists. My viewpoint is skewed by reading about the unscientific and unethical misdeeds of a few who are creating a stereotypical view of their profession. This also accounts for the growing public distrust of scientists as evidenced by public opinion polls.
—-
” ‘If a CO2 molecule (or any molecule for that matter) is absorbing a photon of a specific frequency and re-emitting a molecule of the same frequency, where is the energy transfer?’
That is subtle, but let me try an explanation.
* A CO2 molecule can gain vibrational energy by absorbing a photon
* A CO2 molecule can gain vibrational energy by colliding with nearby molecules
* A CO2 molecule can lose vibrational energy by emitting a photon
* A CO2 molecule can lose vibrational energy by colliding with nearby molecules
It is important to note that a CO2 molecule could gain energy from a photon and lose that energy via a collision, or vice versa.”
I see I made a little mistake substituting the word molecule where I intended to say photon. Nevertheless, your explanation ignores my mistake and answers my question. Unfortunately I should have clarified what I am actually trying to understand.
Most of your explanation involves the conductance of energy away from the molecule, not the radiation of energy away from the molecule. If you were to take a single CO2 molecule in isolation and subject it to a stream of photons of the correct frequency for it to absorb, what happens? Does the vibrational energy of the molecule continue to increase, does the radiative energy convert into another form of energy, does the rate of photons emitting increase, or does something else happen?
RACookPE1978 says:
July 23, 2012 at 10:35 am
….A question not addressed yet.
Assume 1370 watts are present at the top of atmosphere at the equator.
Approximately 360 watts are absorbed by the atmosphere before reaching the ground. What absorbed that 360 watts that are NOT transmitted, and where does that energy “go” if O2 and N2 do not radiate or absorb IR energy?
_______________________________
Just looking at the diagram at the top of the page. Incoming solar insolation is absorbed by CO2, H2O and O3. It is also reflected by H2O in the form of clouds.
This also goes back to what Sleepalot and I were pointing out. (See my comment)
I would like to add that in looking again at Sleepalot’s data
He picks May which is midway between the vernal equinox and the summer solstice and therefore the sun would be midway between the equator and the Tropic of Cancer (the latitude line at 23.5° North) so the solar insolation at both locations would be roughly equal with a bit more expected in Barcelos, Brazil.
ALTITUDE:
Barcelos, Brazil elevation ~ 30 meters (100 ft)
Adrar, Algeria ~ Elevation: 280 metres (920 feet)
One would expect a drop in temperature of ~ 4C due to altitude for Adrar, Algeria so the difference between locations, taking into account altitude is ~ 8C higher in Adrar which is further north but with much lower humidity.
Albedo?
Photos Adrar, Algeria and Barcelos, Brazil
A quick search shows some work has been done on clouds, humidity and solar insolation but it is not something I (or my computer) could handle. I found this quite interesting since it is obvious from the chart at the top of this page that water vapor (not clouds) does effect the amount of surface insolation and I would expect to easily find information on it… info based on real life data collection and not models.
There is also the International Satellite Cloud Climatology Project (ISCCP)
And this interesting graph of top of the atmosphere vs surface vs air variation in net shortwave energy over time. link from NASA The link also contains a similar longwave radiation graph.
RE: davidmhoffer: (July 23, 2012 at 11:09 am)
“No, it is the value presented by the sun to the earth if earth was a flat plane directly facing the sun”
Thanks for pointing that out. You are correct, it was not a tropical forcing value. I see the example I cited seems to have been a rough estimate for a cloud-free surface.
From the Kiehl and Trenberth diagram above it looks like the ‘official’ forcing value is 235 W/m² which seems to imply a net albedo of about 31 percent and a 25 percent ratio of the Earth’s solar cross-section area to its spherical surface area.
Reed Coray says:
July 23, 2012 at 9:33 am
Phil. says: July 23, 2012 at 7:53 am
“Because the outgoing radiation increases as the fourth power of temperature whereas the effect of CO2 is logarithmic. Consequently the stationary point where incoming energy equals outgoing energy goes up but with bounds.”
Phil, thank you for your response.
However, I’m not sure I follow your answer. You say the outgoing radiation increases as the fourth power of the temperature whereas the effect of CO2 is logarithmic. It’s not clear what you mean by effect. I’ll assume you mean: “Temperature is proportional to the logarithm of the amount of CO2 in the atmosphere.” If my assumption is incorrect, please let me know; and clarify your meaning.
Outgoing flux=Incoming flux
∴ Surface radiation – absorption by GHGs = Incoming flux (constant)
So k*T^4 – f*ghg([CO2]) = constant
so if ghg([CO2]) goes up then T^4 must go up by the same amount. So if T^4 goes up by 4% then T goes up by ~1%..
davidmhoffer says:
I think you are confused here. Nothing is punching through the atmospheric column to get to the surface. The idea is that an increase in greenhouse gases lowers the Earth’s emission back out into space by ~3.7 W/m^2 and the temperature at the effective radiating level has to rise by ~1 C to compensate. If this were to occur in such a way that the lapse rate remained constant (which it doesn’t), the temperature at the surface would rise by the same amount and that means that the surface itself would emit an additional ~5.5 W/m^2.
Look, you can complain all you want about definitions because the point is that, at the end of the day, what matters is the total temperature rise that occurs at the surface, which includes all of the feedbacks. The theoretical surface temperature rise that occurs in the absence of feedbacks is a theoretical concept which depends on what you define as the zeroth-order change and what you define as the feedbacks. It is useless to argue what its value is in the absence of some arbitrary choice of definitions as to how you are going to define it.
However, what you are definitely not allowed to do is do what you did in your original post on the matter ( http://wattsupwiththat.com/2012/07/21/some-thoughts-on-radiative-transfer-and-ghgs/#comment-1040463 ), which was to argue that they got the zeroth order temperature rise wrong and thus that they got the temperature rise in the presence of feedbacks wrong (i.e., you claimed that they got 2.6 C but should have gotten 1.8 C).
The actual fact is that they did the calculation in a totally consistent way. (In fact, I don’t think they ever calculate a zeroth-order surface temperature rise. They just calculated the actual temperature rise that occurs including all the feedbacks.) You are doing something that is inconsistent (putting in your definition of the zeroth-order effect with their definition of the feedbacks) to arrive at a lower value for the predicted temperature rise that is just plain wrong. It’s as simple as that.
David: To summarize my point simply (and assuming the sensitivity number that you have claimed that AR5 will use is correct), what the climate scientists have essentially done is said, “Our models show on average that doubling CO2 produces a temperature rise of 2.6 C. This can be thought of as a ‘bare’ response of about 1 C multiplied by a feedback factor of about 2.6.”
What you now have come along and said is, “No…I am going to look at things in a way I think is more natural and that gives a bare response of 0.7. Then when I multiply by your feedback factor of 2.6, I get a 1.8 C rise.”
Unfortunately, it doesn’t work that way.
davidmhoffer says:
July 23, 2012 at 11:03 am
So, if we have a warm object at say 100 C exposed directly to space, it cools rather rapidly. If we insert a “cold” object at say -20 C in between the warm object and space, the warm object will obviously cool more slowly.
======================================================
That is the same apparently experimentally unproven narrative.
You really should make an effort and understand that telling people what you think is real and proving that it is real are 2 different things.
It looks very bad for you, guys. You key assertion does not appear to have any basis in real science.
Buy the way, that does not mean automatically that this key assertion is false, it only means that it is baseless. At the same time, given what an enormous amount of money the warmists have at their disposal, the apparent lack of the key proof is a very strong indication that their key assertion and thus their built on this assertion “greenhouse effect” are simply false.
Nevertheless, I am still open to any experimental scientific proof. Who knows, maybe the guys here on this thread are just not competent enough to present it.
[Moderator’s Note: This has gone on long enough. Greg, you have managed to pick a fight with half the people commenting on this blog and have gotten insulting in the process. You are suggesting that Dr. Shore, Dr. Brown and Dr. Felton are incompetent or worse. They have all answered, with great patience, your requests for evidence and proof and you’ve blown it off. You can submit a post explaining your position, citing Dr. Wood if you wish, and it will be published here. For most of us, the “Greenhouse Effect” is not controversial. If you think otherwise, defend it in a post and the comments you wil get or bug off. -REP]
Phil. says: July 23, 2012 at 1:48 pm
Outgoing flux=Incoming flux
∴ Surface radiation – absorption by GHGs = Incoming flux (constant)
So k*T^4 – f*ghg([CO2]) = constant
so if ghg([CO2]) goes up then T^4 must go up by the same amount. So if T^4 goes up by 4% then T goes up by ~1%..
Thanks again for your response.
I agree with your math–i.e., I agree that in an expression x – y = constant, if x goes up then y goes up by the same amount. I want to make sure, however, that we’re not talking at cross purposes regarding what you mean by “flux”. In the context of our discussion, the Stefan-Boltzmann equation, the k*t^4 term in your equation is “energy rate” per unit area. [If you want to include the surface energy in the constant “k”, then the term in your equation corresponds to energy rate. However, in neither case does it correspond to energy itself.] That is, (a) “surface radiation” is the rate (not the amount of) energy leaves a surface (either per unit area or for the total surface, which for his discussion I’ll take to mean “for the total surface”), (b) “absorption by GHGs” is the rate GHGs absorb energy, and “Incoming flux” is the rate energy enters the system comprised of the surface and the GHGs. Furthermore, I assume your equation applies to a state of “energy-rate” equilibrium–i.e., sufficient time as elapsed that the individual terms in your equation do not change with time.
If this interpretation is correct, then according to your equation as greenhouse gas levels increase, the temperature of the surface will increase to keep the equation in balance. So far so good. However, your equation implies that the rate energy is absorbed by greenhouse gases is never zero–in fact is a non-zero constant. So if your equation represents something physical, then isn’t it legitimate to ask: where does the energy that over time keeps being absorbed by (i.e., keeps accumulating) in the GHGs go? For energy rate equilibrium, if energy is absorbed by GHGs at a non-zero constant rate, doesn’t that energy have to leave the greenhouse gases at the same rate? If this is the case, shouldn’t your equation have in addition to a non-zero term for the “rate of energy aborption by GHGs” also have a non-zero term for the “rate of loss of energy by GHGs?”
Spector;
From the Kiehl and Trenberth diagram above it looks like the ‘official’ forcing value is 235 W/m² which seems to imply a net albedo of about 31 percent and a 25 percent ratio of the Earth’s solar cross-section area to its spherical surface area.
“”””””””””””””
To be precise, take the post albedo number and multiply by 0.5 for curvature of the earth and then multiply by 0.5 yet again to accomodate day-night.
[Moderator’s Note: This has gone on long enough. Greg, you have managed to pick a fight with half the people commenting on this blog and have gotten insulting in the process. You are suggesting that Dr. Shore, Dr. Brown and Dr. Felton are incompetent or worse. They have all answered, with great patience, your requests for evidence and proof and you’ve blown it off. …For most of us, the “Greenhouse Effect” is not controversial.
==============================================
OK, yes they did answer my comments but just for the record: none of them did present what I asked for: a link to a real genuine falsifiable scientific experiment proving the key assertion the notion of “greenhouse effect” is based on, namely that a colder body can reduce cooling of a warmer body by means of radiation.
I do not quite understand, how after all that the “greenhouse effect” can still be considered “not controversial”.
REPLY: The moderators have called me in on this. Let me make this simple, the greenhouse effect is a well established property of radiative physics in our atmosphere, one that I have observed firsthand through experimentation. There is no controversy over its existence, for did it not exist, neither would we. There is controversy however, over the magnitude of forcings and induced feedbacks. Take a 48 hour time out and leave the issue alone – Moderating this is tiresome for all involved. – Anthony
I have a very simple multiple choice question for those still commenting on this thread.
I have two 1m spheres of thin LDPE plastic (transparent to LWIR) in a sunless vacuum in deep space. Both spheres contain gas at an initial temperature of 100C. Sphere 1 contains 100% O2. Sphere 2 contains 99% O2 and 1% CO2. Which sphere cools faster? Is the answer :
A. Sphere 1 cools faster.
B. Sphere 2 cools faster.
C. Sphere 1 and 2 cool at the same rate.
Or
D. The spherical chicken found in the same region of space has “Property of Pierrehumbert” tattooed on its butt.
joeldshore;
Look, you can complain all you want about definitions because the point is that, at the end of the day, what matters is the total temperature rise that occurs at the surface, which includes all of the feedbacks.
“””””””””””””””””
And once again Joel, my point remains that IPCC AR3 defined the temperature increase as being at the effective black body temperature of earth and NOT at the surface. AR4 uses the same values at AR3, but doesnèt specify the definition, so we are left to assume that it is the same. There is no reasoning nor evidence provided in AR3 or AR4 to support the notion that the temperature change at effective black body temperature of earth (-20 C) will be equal to the temperature change at the surface (+15 C). I find it doubtful from many perspective to assume that it would be! That would require a consistant 1 degree change from TOA to surface, and that is simply illogical on multiple fronts. Further, you say
Joel Shore;
The idea is that an increase in greenhouse gases lowers the Earth’s emission back out into space by ~3.7 W/m^2 and the temperature at the effective radiating level has to rise by ~1 C to compensate.
“”””””””””””””””
Sorry, but that is not the case, and since you and I have discussed this before, Im going to assume that you have misworded it. Unless doubling CO2 increases the absorption of SW in the first place, at equilibrium, nothing changes.
240 w/m2 comes in
240 w/m2 is radiated out.
CO2 doubles
240 w/m2 comes in
240 w/m2 is radiated out.
What changes is the temperature gradient from TOA to surface. How many watts going “up” and from WHERE. Will the lower reaches of the atmosphere and earth surface be warmer as a result. I believe so. Will it be linear from surface to TOA? I doubt it. Some parts more, some parts less. To suggest that there will be the same temperature rise at surface where the greatest number of w/m2 is required to generate it and due to preponderance of water vapour, CO2’s effects are minimized, as it does at 14,000 feet where (roughly) the effective black body temperature of earth occurs and where CO2 effects are more pronounced due to lack of water vapour just doesn’t make sense.
If there is one thing we know about the system as a whole, it is ugly complex. Assuming that the effects of CO2 at effective black body temperature of -20 and at surface at +15 are both 1 degree is nonsensical, and if it were true, then there would have been no need to define it that way in the first place.