By Steve Goddard
ESA’s Venus Express mission has been studying the planet and a basic atmospheric model is emerging.

Venus has long been the CO2 bogeyman of climate science. In my last piece about Venus I laid out arguments against the claim that it is a runaway greenhouse which makes Venus hot. This generated a lot of discussion. I’m not going to review that discussion, but instead will pose a few ideas which should make the concepts clear to almost everybody.
If there were no Sun (or other external energy source) atmospheric temperature would approach absolute zero. As a result there would be almost no atmospheric pressure on any planet -> PV = nRT.
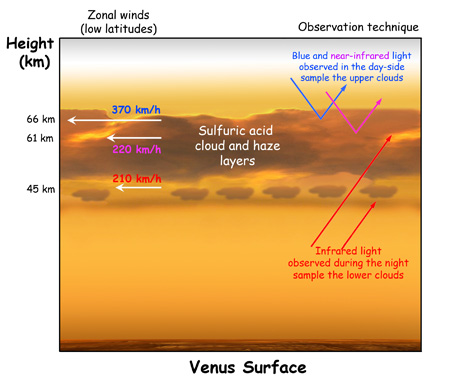
Because we have a sun providing energy to the periphery of the atmospheric system, the atmosphere circulates vertically and horizontally to maintain equilibrium. Falling air moves to regions of higher pressure, compresses and warms. The greater the pressure, the greater the warming. Rising air moves to regions of lower pressure, expands, and cools. The amount of warming (or cooling) per unit distance is described as the “lapse rate.” On Earth the dry lapse rate is 9.760 K/km. On Venus, the dry lapse rate is similar at 10.468 K/km. This means that with each km of elevation you gain on either Earth or Venus, the temperature drops by about 10C.
It is very important to note that despite radically different compositions, both atmospheres have approximately the same dry lapse rate. This tells us that the primary factor affecting the temperature is the thickness of the atmosphere, not the composition. Because Venus has a much thicker atmosphere than Earth, the temperature is much higher.
dT = -10 * dh where T is temperature and h is height.
With a constant lapse rate, an atmosphere twice as thick would be twice as warm. Three times as thick would be three times as warm. etc. Now let’s do some experiments using this information.
Experiment # 1 – Atmospheric pressure on Venus’ surface is 92 times larger than earth, because the atmosphere is much thicker and thus weighs more. Now suppose that we could instantly change the molecular composition of Venus atmosphere to match that of Earth. Because the lapse rate of Earth’s atmosphere is very similar to that of Venus, we would see little change in Venus temperature.
Experiment #2 – Now, lets keep the atmospheric composition of Venus constant, but instead remove almost 91/92 of it – to make the mass and thickness of Venus atmosphere similar to earth. Because lapse rates are similar between the two planets, temperatures would become similar to those on earth.
Experiment #3 – Let’s take Earth’s atmosphere and replace the composition with that of Venus. Because the lapse rates are similar, the temperature on Earth would not change very much.
Experiment #4 – Let’s keep the composition of Earth’s atmosphere fixed, but increase the amount of gas in the atmosphere by 92X. Because the lapse rates are similar, the temperature on Earth would become very hot, like Venus.
Now let’s look at measured data :

http://www.astro.wisc.edu/~townsend/resource/teaching/diploma/venus-t.gif

http://www.astro.wisc.edu/~townsend/resource/teaching/diploma/venus-p.gif
Note that at one Earth atmospheric pressure on Venus (altitude 50km) temperatures are only about 50 degrees warmer than earth temperatures. This is another indication that atmospheric composition is less important than thickness.
Conclusions : It isn’t the large amount of CO2 which makes Venus hot, rather it is the thick atmosphere being continuously heated by external sources. It isn’t the lack of CO2 on Earth which keeps Earth relatively cool, rather it is the thin atmosphere. Mars is even colder than earth despite having a 95% CO2 atmosphere, because it’s atmosphere is very thin. If greenhouse gases were responsible for the high temperatures on Venus (rather than atmospheric thickness) we would mathematically have to see a much higher lapse rate than on Earth – but we don’t.
WUWT commentor Julian Braggins provided a very useful link which adds a lot of important information.
“The much ballyhooed greenhouse effect of Venus’s carbon dioxide atmosphere can account for only part of the heating and evidence for other heating mechanisms is now in a turmoil,” confirmed Richard Kerr in Science magazine in 1980.
The greenhouse theory does not explain the even surface temperatures from the equator to the poles: “atmospheric temperature and pressure in most of the atmosphere (99 percent of it) are almost identical everywhere on Venus – at the equator, at high latitudes, and in both the planet’s day and night hemispheres. This, in turn, means the Venus weather machine is very efficient in distributing heat evenly,” suggested NASA News in April 1979. Firsoff pointed out the fallacy of the last statement: “To say that the vigorous circulation (of the atmosphere) smooths out the temperature differences will not do, for, firstly, if these differences were smoothed out the flow would stop and, secondly, an effect cannot be its own cause. We are thus left with an unresolved contradiction.”
======================================================
An update for those interested in what Venus looks like at the surface.
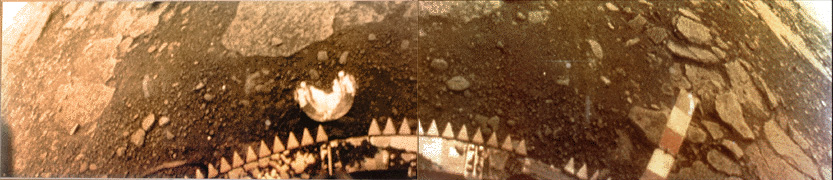
On March 1, 1982, the Soviet Venera 13 lander survived for 127 minutes (the planned design life was 32 minutes) in an environment with a temperature of 457 °C (855 °F) and a pressure of 89 Earth atmospheres (9.0 MPa). The photo composite above shows the soil and rocks near the lander.
Here’s another Venera image that shows a hint of yellow atmosphere. – Anthony

GEOTHERMAL activity; Here is an interesting article:
The Baffling Geology of Venus (TWO PAGES)
http://geology.about.com/od/venus/a/aa_venus.htm
It links to some essential reading stuff from the ESA where there is speculation that she is active.
(BTW, reportedly, one of the Soviet probes may have passed through a volcano plume, according to its “sniffer“)
Bob_FJ: May 11, 2010 at 3:45 pm
Hi Bob,
I’ll do things in reverse order:
– I don’t know of any reference that explicitly deals with the radiation phenomena of objects immersed in fluids. I’ll hunt around and see if I can find something, but I’m not all that hopeful about it.
– I wasn’t saying that the (ice + melted water) emits twice as much radiation, just that each one emits some, and that the result would be the total of the two. I ignored the complication that the fluid might not be transparent to the radiation from the internal solid. If that is important, then the emission, absorption, and re-emission process needs to be included. On Earth, and treating the atmosphere as your fluid, that does need to be taken into account because the atmosphere initially absorbs some of the outgoing radiation from the solid surface.
– In the case of the painted concrete, the photons don’t enter into the picture until you get to the paint layer. Before that, the energy transfers are mainly by conduction, and since phonons are a way to describe group vibrations of the atoms in the solid, you could use that approach to deal with things inside the concrete, but photons don’t get emitted “through” the paint. They are, in essence, created “by” the paint. The difference between paint on the one hand, and atmosphere or fluid on the other, is the matter of transparency. If you had a thick layer of see-through paint, you’d be back to something like the solid plus fluid situation. If not, then only the surface temperature of the paint is important for the radiation distribution pattern.
– Regarding your item (b), keep in mind that conduction requires physical contact, and convection requires “large-scale” movement. If we’re still talking about things like planets, neither process can result in energy loss to space. Only radiation can do that. The conduction and convection processes move energy around inside of the system, mostly conduction in solids, and convection in gases. Liquids are somewhere in between.
– When “thermal photons” are emitted, some energy of motion is lost by the molecules, thus reducing their Kinetic Energy. I think the glitch in your thinking is not noticing that these are not two different processes. One is simply the result of the other, and I believe that you might be “double-counting”.
– One more point, and this is perhaps just me being picky, is your use of the word HEAT. Strictly speaking, there is no such thing as heat, even though it is perfectly fine for everyday use, and generally causes no confusion. In Physics, however, using this term causes lots of unintended errors. Properly speaking, heating and cooling are processes, verbs, not nouns, that involve conduction, convection and radiation as means of transferring energy from one place or thing to another. The word heat (as a noun) is something left over from our earlier misinterpretations of energy and temperature and related things. (Look up ‘caloric’, ‘frigoric’, ‘phlogiston’ and ‘anti-phlogiston’.)
/dr.bill
George Turner,
Are you reading that Virtis link right? I couldn’t see a reference to that window range. From the spectrum (Fig 1), that seems to be the range of CO2 absorption (confirming the greenhouse effect). I think the window they refer to is the narrow peak actually at 2.35 micron.
I think I’m reading it right. The window peaks at 2.3 microns but looks to average about 50% of that across the 2.2 to 2.5 micron range. This was apparently unexpected and discovered by accident during Earth-based observations that showed structures at 2.2 microns.
However, its normalized units.
*Digs deeper*
More on VIRTIS including a spectrum with actual units, but I’m inclined to think those are the units for the detector (receiving about 13 microwatts on a 1 cm^2 cell.
If CO2 was active (and broad spectrum) in this region there’s no way they could detect the trace gases.
I also ran across a VIRTIS link on vulcanism that indicated the conditions on Venus’ surface produces rocks with very low IR emissivity, which is another interesting avenue to investigate.
Hi dr.bill (Steven, you too if you can help answer this):
Didn’t want to put you on the spot, I know you have no idea who or what age you are conversing with initially. (I too have had troubles with a couple of redheads in the far past! ;))
Back on Venusian atmosphere, I don’t know how to convey this thought to you but to ask the question and have you ponder on it for a while.
Take a quiet and stable atmosphere on any planet with any one type of molecule as its gas, no convection, no rotation, no winds, purely hypothetically quiet but with proper physics. Looking at the atmosphere as concentric shells of thickness dz, can the molecular velocities vary from shell to shell to shell, and if they can vary, what force is maintaining the velocity differential. The gravitational pressure gradient is going to be there no matter what. Also, it’s quiet, there is no ‘work’ being done on the gas.
That was all I was trying to get to above. One part of me says no, they (the velocities) would equalize over time and stay in a equilibrium. That would mean it would always be hotter the deeper you go, the lapse rate, getting just enough radiation input to maintain the temperature. The other part of me says yes, by TD gas equations, the velocities must be somewhat different to satisfy the temperature and pressure differentials seen on Venus but I cannot seem to find why, what force would be doing that. That all ties into why there is a lapse rate in the first place. Get my gist now? It’s a rather deep question. To properly answer it I think you have to go a bit underneath to the basic properties supporting the statistical nature of thermodynamics (½mv²) which underlie temperature and pressure themselves.
George Turner says:
May 11, 2010 at 7:21 pm
I think I’m reading it right. The window peaks at 2.3 microns but looks to average about 50% of that across the 2.2 to 2.5 micron range. This was apparently unexpected and discovered by accident during Earth-based observations that showed structures at 2.2 microns.
However, its normalized units.
*Digs deeper*
More on VIRTIS including a spectrum with actual units, but I’m inclined to think those are the units for the detector (receiving about 13 microwatts on a 1 cm^2 cell.
If CO2 was active (and broad spectrum) in this region there’s no way they could detect the trace gases.
I ran CO2 spectra at ~90 atm and there’s a transmission window from 3840^cm-1 to 4600^cm-1, so yes there is a narrow window.
Wayne, that is the crux of the whole discussion and this has puzzled me even before this thread started. The question pondered is, when temperatures are being measured, what exactly are we measuring?. In space, in view of the sun, radiative photons on the sensor, in shadow, photons from background cosmic radiation. In water, an intimate contact between the sensor and the fluid. But what about air ? the ability of the sensor to measure temperature accurately is a function of the efficiency of kinetic energy to be transferred to the sensor, so, the more dense the air the more efficient this transfer is, and the temperature reading is higher. There is a gradient of efficiency as the sensor goes higher, less molecules hitting the sensor. Standardize the pressure, then read temp. at different altitudes and see what the difference is. My guess is that the gradient is nowhere as steep as the standard wet/dry lapse rate. The lapse rate increase in temperature with decreasing altitude is a artefact of increasing air density.
Until your question is answered “why is there a lapse rate in the first place” this argument is circular.
My proposal is not in any textbook that I know, and I welcome feedback if it is, but it is testable.
Understanding the Tropopause
The tropopause is that altitude where convective heat transfer stops and the lapse-rate changes from a typical adiabatic decreasing temperature slope, typical of the troposphere, to the increasing temperature slope, typical of the stratosphere.
One thing, I think, that appears to be missing from our thermal radiation knowledge base is a good understanding or theory for radiation from a transparent medium that contains a sprinkling of internal radiation sources. I would assume the sources were in general thermal equilibrium with their containing medium. I note that if the active radiation layer of the Earth’s tropopause were 3 km thick, it would only take an average volume radiation power of 78.33 milliwatts per cubic meter to achieve the nominal 235 W/m2 radiated power level that some sources say is required balance the Earth’s radiation budget.
From one source I see that the typical temperature of the tropopause may be on the order of minus 55 degrees C. That is almost like a negative greenhouse effect [or an ice-locker effect] of 37 degrees relative to the minus 18 degrees C required to produce a surface flux of 235 W/m2 for the Earth.
Perhaps with a better understanding of how the tropopause unloads convected heat from below, we could more readily determine if the surface temperatures on the Earth and Venus are, as I suspect, controlled or regulated by the tropopause temperature (plus the lapse-rate span) or if the tropopause temperature is controlled by the surface temperature (minus the lapse-rate span) or if, perhaps, it were some intermediate average of these two alternatives.
I note that the Venus tropopause temperature bottoms out around -112 degrees C at an altitude of 100 km and on Earth the tropopause ranges between 11 km at the poles to 17 km at the equator where it might get as cold as minus 80 degrees C. As temperatures of these two planets are more similar at their tropopause altitudes than at their surface levels, I am more inclined to believe that surface temperatures are controlled or at least limited by tropopause conditions. Perhaps, in the case of the Earth, the higher tropopause temperatures are a result of more direct radiation from the surface.
I really don’t see why Mr Goddard’s ideas should be so controversial except, of course, because they question the doctrines of the AGW true believers.
Anyone who has experienced the sudden arrival of the warm Chinook wind on an otherwise frosty day east of the Rockies can attest to the great power of adiabatic warming.
They have the same kind of wind north of the Alps called the Föhn which is also the German word for hairdryer. As I explained to my grandchildren just the other day, hairdryers, like Chinooks, work on adiabatic principles, with the fan compressing and thereby heating the air, rather like the atmosphere of Venus.
Actually hairdryers don’t compress the air, they just accelerate it like a propellor or fan. To compress the air they’d need stator vanes, and to compress it significantly they’d need lots of paired rotors and stators. Hair dryers generate hot air with electrical heating coils in the direct flow of a high speed fan, which both cools the coils and blows the heated air toward a scalp.
🙂 <— understands many common household appliances.
Spector
“That is almost like a negative greenhouse effect [or an ice-locker effect] of 37 degrees relative to the minus 18 degrees C required to produce a surface flux of 235 W/m2 for the Earth.”
The arithmetic goes like this. About 40 W/m2 outward IR comes from ground through the atmospheric window (unabsorbed frequencies), at about 288K. Another 60W/m2 approx goes out from cloud tops, also fairly warm. The rest comes from varying altitudes, with about half from the tropopause at about 225 K. That all adds up to an average 255K.
You can see this reflected in the integral under the OLR spectrum.
@John A. Marr
“Anyone who has experienced the sudden arrival of the warm Chinook wind on an otherwise frosty day east of the Rockies can attest to the great power of adiabatic warming.”
but, that is another effect. Moist air is going up the mountains, cooling on its way, but water is condensing, thus, the lapse rate is a wet lapse rate, around 6.5K/km, now, on the other side of the mountains the now cool, dry air is sinking with dry lapse rate, which is around 10K/km. Thus, you convert latent heat into sensible heat, IMHO. Of course, there are also other explanations, but this is the common one.
Example: you have west wind. Moist air from Pacific or so is going up the Rockies, water is condensing, snow, rain, etc. The now cool,dry air reaches the top of the mountains, and is going down to Calgary. Chinook.
Hi Wayne,
I think we had better knock off with the redhead stuff before we get clobbered by Pamela Grey or someone! Yeah, I know, I’m a wuss… 🙂
Anyway, I’ve got your model for you (Wayne’s World ?? – sorry!), but you have to stop working me so hard! On the other hand, I might get a bonus from the exercise. I’m giving a TD test on Friday, and I’m thinking of putting this on as a question in the atmospherics part. Hope none of them are reading this thread. 🙂
So then, if you don’t mind, along with your no-convection stipulation, I simplified things a bit further by irradiating your planet uniformly from all directions, and I gave it a transparent atmosphere. That just leaves conduction processes acting, and you can’t avoid those. What you get is essentially an atmosphere that is one big troposphere, right up to the “top” (which isn’t, of course, precisely defined – it just fades away into space).
Under these conditions, the TD processes are pretty much the same as keeping one end of an iron bar at a high temperature and the other end cold, except that the bar doesn’t have a finite length, and the low temperature is the background temperature of space, or pretty much 0 kelvins for our purposes. Because there’s a hot end and a cold end, however, you do get a temperature gradient, just like with an iron bar, so the question is how to get a fix on what that should be. We could do it by working with the thermal conductivity of air or other gases, but I decided to “cheat” a little bit for the sake of expediency.
In regular analysis of the troposphere, we use a linear function T = To – Ly, where L is the lapse rate (which is just a regular thermal gradient, regardless of what it’s caused by, or what its value might be). An alternative is the Isothermal Approximation. This leads to what people call the Barometric Equation (for pressure), which is a decreasing exponential. It works surprisingly well, despite the rather bad assumption that the surface temperature persists right up to the tropopause. The lapse rate version is much better, but we can’t use either of them for your model because they give “stupid results” once you get a bit above the tropopause.
What has to happen is that the gradient will be largest when close to the hot end, and will stay pretty much constant for some distance, just like the iron bar and our own troposphere, but will eventually diminish because of the “infinite length”. It turns out that there’s an easy way to get something just like that, by re-writing the temperature equation given above, and then treating it as the first two terms in the expansion of a decreasing exponential. In other words: T/To = (1 – Ly/To) becomes T/To = exp(-Ly/To). The quantity L/To is quite small (about 1/30 or 1/40 km or so), which makes this a good mathematical approximation, and it guarantees that we will also match “normal behaviour” for things like iron bars and tropospheres.
All you need after that are the Ideal Gas Law and the Hydrostatic Equation: dP/dy = -ρg, and the rest is just a matter of solving one differential equation. Here are the results, which you can stick into a spreadsheet and do some plotting. I’ve normalized everything, so all the values either start at 1 and end at 0, or the other way around in the case of the cumulative mass function (the total mass M below a given altitude).
T/To = exp(-Ly/To)
P/Po = exp(-(mg/RL)(exp(Ly/To) – 1))
ρ/ρo = (P/Po)exp(Ly/To)
M/Mtot = 1 – P/Po
For “play purposes”, I’d advise putting To, L, and m (the molar mass) in separate cells somewhere, and use them as parameters in your cell formulas instead of hard-coding them. Then you just change any one of them and all the values and the graphs will adjust by themselves.
Just for reference, here some of the values you should get if y is measured in km:
L = whatever you like (5 to 10 K/km is typical in our atmosphere)
To = whatever you like (but 288K is “average” for Earth)
mg/R = 34.157 K/km for air, 51.91 for CO2, etc…
By the way, those results above give a dead match to the International Standard Atmosphere profile for the troposphere if you use 6.5 for the lapse rate and 28.96 for the molar mass, so in that sense, you can use them for “semi real world” stuff as well as just playing with them. Let me know how it works out, but you may run the risk of getting an overtime slip if you keep slave-driving me like this. 🙂
/dr.bill
I gotta hand it to ya, Steve, you sure do generate lots of comments.
What’s the duration record for a thread on WUWT. How long do we have to keep going to beat it?
Reply: You’ve got another 1228 comments to go to beat this post. ~ctm
[Actually, the this article is within less than 400 posts of the Climategate article. -Moderator]
Hi Again Wayne,
I forgot one formula in my previous post, and actually it was the one that you seemed most interested in, namely the molecular speeds. My bad! So here’s the addendum:
v/vo = exp(-Ly/2To), where vo = sqrt(3RTo/m)
vo is the speed at ground level, and as you can see, because the temperature decreases with altitude, then so does the speed of the molecules, but not nearly as quickly because of the square root.
Using O2, for example, with To = 288K, gives vo = 474m/s. At an altitude of 10km, the speed has decreased, but only to 423m/s (using L = 6.5K/km), which is a much smaller decrease than for T or P or ρ.
/dr.bill
Well i had some comments here earlier, have they been removed?
Venus atmosphere I guess absorb the short wave light from the Sun, like the Earths oceans do. And when Venus atmosphere is isolated/insulated 90 times more than Earths atmosphere, the long wave I.R. has a much much longer way to go and thus stays longer in Venus atmosphere. In addition there is no or little water in Venus Atmosphere to make clouds to give negative feedback on the climate so Venus atmosphere is therefore mostly extra crispy?
And this is what makes Venus atmosphere warmer?
dr.bill says:
May 12, 2010 at 4:44 am
I forgot one formula in my previous post, and actually it was the one that you seemed most interested in, namely the molecular speeds. ..
Thanks Bill for the info. That’s might help. I also found the proper equations which bring density into the temperature calculations, I just knew it was there somewhere. Take a look at my question at May 11, 2010 at 7:27 pm later and see if you come up with the same. I can only account for about half of the dT at molecular level. With equal velocities the temp should be much higher at surface, or conversely, lower at high altitudes. That is why it seems to be top driven, not from the surface.
My wild guess is that what is being shown is most of the incoming radiation energy is being absorbed by high levels in the atmosphere and re-radiated back out to space, never making it to the surface keeping velocities higher always greater. This would be in a smooth distribution vertically of coarse. Look closely at Venus’s lapse curve, its convex while Earth’s is concave. Is this because our atmosphere is much more transparent?
(And the numbers I posted near the top, not only was the formatting trashed but most of the calculations are trash also. Never focused on the central values before pressing submit, was only focusing on the √90 factor. But I caught and fixed them. Oops! Patience would help. That is why I said earlier that I am not totally versed in TD, I still make such mistakes.).
Later.
dr.bill, to be clearer you might change:
Is this because our atmosphere is much more transparent?
to read
Is this because our atmosphere is much more transparent and therefore mostly bottom (or surface) driven in contrast to Venus?
dr.bill,
Going call this an end for a while. You have given enough for me to digest for some time! Thank again.
Also, I will adjust the 3 factor in the equations depending on the atom or molecule being assumed as the gas as degrees of freedom vary, I was following that all along. Now just need to numerically integrate to see if I can get a little clarity if possible.
Still, I will always wonder on the avg. molecular velocities in a gradient density environment, as a gravity held atmosphere, how could they possibly be different in a mostly opaque, dense, and smoothly radiated atmosphere such as Venus, intriguing.
re: wayne: May 12, 2010 at 10:28 am
and wayne: May 12, 2010 at 10:34 am
Hi Wayne,
One thing you should note is that I (personally) don’t think Venus is “top-driven” – except in the sense that the energy which heats the surface of the planet likely comes in at the top in the same way that sunlight coming into the Earth’s atmosphere heats the surface of our planet. After you get a warm surface, however, no matter how you get it, and as long as it is sustained in its warm condition, there isn’t a whole lot of mystery about the generalities of what follows. Details, sure. Big picture, no.
For a column of gas in a gravitational field, more weight will be pressing down on the molecules at the bottom than on those higher up, so there will be a pressure gradient. This is adequately described in a normal atmosphere by the equation: dP/dy = -(mg/RT)P. In order to solve this equation, you need to know how T depends on y. In the tropospheres of Earth and Venus, temperature decreases with altitude. Once you know the details of that variation, the rest is just Math, and what you get are decreasing values for Temperature, Pressure, Particle Density, and Particle Velocity as you move upwards.
Those statements hold true for all the predominant normal conditions that prevail in the atmosphere, and I don’t want to get side-tracked onto ephermeral things like Chinooks and temperature inversions, ash clouds, or any other short-term and geographically-isolated phenomena. They occur. There are reasons for them, but they are not the normal day-to-day behaviour of the atmosphere, which is what we, or at least I, have been focused on. The force holding up the molecules isn’t mysterious either. It’s the same kind of buoyancy described by Archimedes Principle. It’s what holds up the Goodyear blimp.
The model that I worked out for you is the simplest one that I could think of, since the only active ingredients are gravity and a warm surface, with only contact conduction for energy transfer, and everything else removed. Even under such simplified and minimialistic conditions, however, all of the general variations that I described above hold true. Plot the graphs. Look at them. The details would change if a slightly different variation of T with y were used, but not the big picture.
In short then, I have a feeling that you might be looking for the answer to a couple on “non-questions” in the matter of particle velocities and forces. I’m not saying that these aren’t involved in the processes, but I also don’t think there’s anything odd about them. When temperature decreases, particles slow down (the other way around, actually, but I won’t get picky). When particles sit on top of each other, buoyancy supports the higher ones. I don’t see any mysteries in this.
/dr.bill
re: wayne: May 12, 2010 at 1:35 pm
Hi Wayne,
Our last posts seem to have passed in transit.
One final item regarding the “model”: in addition to letting the lapse rate, surface temperature, and molar mass change, you can also change gravity, so it would be convenient to leave those as “settable” parameters. I hope you sort things out in your contemplations, and if I have been of any small help, it has been my pleasure.
All the best.
/dr.bill
dr.bill, Reur May 11, 2010 at 5:37 pm
Concerning S-B emission from a body immersed in a fluid, (maybe such as the Venus atmosphere); thanks for your comments Bill.
In the analogy of painted concrete, you say that there would be emission ONLY from the paint, assuming the paint to be opaque. On the other hand, if the paint were hypothetically transparent, the concrete would then emit through the paint unhindered. It is thus apparent that if immersed in a fluid, the amount of radiation from a body is dependent on the level of opacity in the fluid. (considering also your earlier comments on wet versus dry water ice)
On Venus, at 92 bars, and with mostly CO2 in intimate contact with the basaltic rocks, that fluid is obviously very opaque to infrared, apart from some small windows in the shorter wavelengths.
Thus, whilst S-B may be “near enough” on planet Earth, with only its trace levels of GHG’s, it would seem to me that its application on Venus’s surface is badly compromised. (without addition of some other theory)
Furthermore, I would think that most of the surface HEAT loss is via conduction/ convection/ advection. Whereupon, at the higher altitudes, that HEAT is more able to convert to EMR and escape to space.
~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~
There are other points in your comments to discuss, but right now, I don’t want to distract from the important considerations above
re Phil: May 11, 8:25
I ran CO2 spectra at ~90 atm and there’s a transmission window from 3840^cm-1 to 4600^cm-1, so yes there is a narrow window.
I calculated that 799 W/m^2 could be emitted into that window (blackbody), and since parts of the window are used to image the surface from orbit, some of the radiation is making it directly into space. Other parts of the window are being used to look at constituents of the lower atmosphere, so CO2 emissions (and thus absorption) aren’t occuring there.
Even is the surface emissivity is 0.25, that would still be 200 W/m^2 radiating up from Venus surface, which is more radiation than the surface receives from the sun. If the surface has this negative radiative flux then its temperature is being maintained by atmospheric convection/conduction (or it requires that as supplemental energy).
If this is the case then the surface radiation exchange (ingoing and outgoing) is cooling the atmosphere, not warming it, and I would think the surface temperature is being maintained by the rest of the atmospheric dynamics.
However, I don’t have hard numbers on the fluxes.
As a thought experiment I could imagine that the sunlight shifted totally to the blue end of the spectrum (which doesn’t reach Venus surface) but maintained the same overall radiative flux, being totally absorbed or reflected by the cloud layers. Then there would be no downward flux in the visible spectrum at the surface, ensuring a net negative radiative flux at the surface, but I’d bet the overall atmospheric temperatures would stay largely unchanged.
So many comments it’s difficult to follow the scientific discussion in just a few minutes, so I’ve probably missed something, but I’m struggling with the basic theory here regarding pressure and temperature. I probably covered some of the relevant physics at school/uni, but that was a long time ago now and I’ve not had to apply it to this sort of scenario so presumably I’m missing something or got my logic wrong?
I’m imagining trying to model a hypothetical simple atmosphere where all we need for the model is PV=nRT. So, if air is rising, the pressure (P) drops, but I don’t understand why in this simple model temperature (T) drops. Would not P1xV1 = P2xV2? i.e. the gas is free to expand as it rises and the pressure drops; i.e. the volume (V) increases? Isn’t PV a constant; as P drops, V increases, therefore P1V2 = P2V2 therefore no change in T? If V did not change, then I presume T would drop, but presumably V is increasing, that’s why the air gets thinner with altitude?
I haven’t studied any of this properly, so probably wrong, but (and as a frequent visitor to mountains I’ve often pondered climate and temperature differences at different locations and altitudes) I was thinking the reason why temperature drops with altitude is not due to pressure, but (assuming absorption of radiation is relatively small in comparison) imagined that the main heating effect came from the sun heating the planet’s surface (rather than the atmosphere directly), and this in turn heating the atmosphere by conduction and convection, so the further away (higher up) from the surface (i.e. distance not pressure), the less heat the atmosphere has picked up. Thus explaining why when I go to Norway in June I can camp at 3000ft on one of the large plateaus and sit outside my tent in shorts anytime it’s sunny (with temperatures subjectively comparable to or warmer than the valleys 3000ft below), but if I camp at 3000ft on a Scottish Munro it’s far too cold!! ie. when on a plateau the breeze I’m getting passed close to lots of ground heated by the Sun, even though I’m at 3000ft, but on a Scottish peak, surrounded by land and valleys 3000ft lower the air passing me has not been close to much sun warmed ground.
re: dr.bill: May 12, 2010 at 2:41 pm
Hi Bill, one last post.
We did pass in the posts. I looked later and said, that wasn’t here a while ago. Well, that happens in blogs.
I did notice on one of your first post you mentioned you understanding that radiation was heating the surface and the heat moves upward in the atmosphere. That is where I diverged; because of a new thought that had entered my mind.
Due to a lapse rate, the lower one km is warmer ‘by a thermometer’ than the km layer above it. I asked myself (and I’m not speaking of radiation, that is another separate topic), does the heat energy in the lower and hotter layer tend to warm the layer above by conduction. After much thought I think not. Why? It’s hotter by a thermometer. TD says the heat will move but it doesn’t. Why? The molecules must have the same mean velocity. That is where I started speaking of the molecular velocity and the density differences. You can’t even get that to happen in a lab here on Earth. Only a gravity held atmosphere has this aspect. In a lab gases at different densities will instantly equalize, in an atmosphere, they won’t, vertically that is.
Whoa was my response to myself. I had never thought of temperature, pressure, density and thermometers in that manner. If that heat will not and cannot migrate upward by conduction, then the molecules, on an average (actually a Boltzmann distribution I think, maybe Maxwell), must have the same mean velocity, that is why the ‘heat’ does not move by conduction. Whoa, that IS a lapse rate. But I try to doubt myself a first, and second, and a third time. But that one aspect seemed correct even though that basically goes against normal thermodynamic principles when loosely passed in conversations about normal substances here on the ground. It must have to do with the density gradient by gravity.
The big thought is that if the above is possible and actually true then in an atmosphere, due to the density gradient of the gases, energy can pass from a cooler to warmer area by conduction as long as the warmer area is also at a greater density and only to a certain degree. Solving for v using (½ ρ • equal_vol • v_rms2 = 3RT / 2, I think 5 not 3 for co2) on each area will tell you if heat can move by conduction alone. The molecules of the cooler layer must actually have greater avg. velocity than the molecules in the warmer layer, density differential does the counter-intuitive magic. That is why I said you would have to go underneath normal TD to the equations underlying. New thought, right? So Venus type of atmosphere could be totally or partially top driven, heat moving down even though that is initially thought impossible by common TD jargon.
I could be totally wrong on this and I have before. After while with the relationships you have supplied above I’ll probably prove myself wrong, but that is science, right, exploring new thoughts, new ideas, new ways to look at the same thing from a different aspect.
If I try a model, I am well versed in gravitational aspects and will leave it variable, in fact, integrated as the height in the atmosphere varies. Have programmed for decades and one area was ephemeris software for near-exact positions of the planets and asteroids. (solar system simulation). I have always had a deep interest in astronomy and later physics.
There is another aspect in Venus’s atmosphere that has a big factor in internal re-radiation and scattering since it is so very thick at >60km. The dip of the horizon. At 50 km a much greater portion of the radiation, randomly directed, is re-radiated back to space than downward to the surface, but that is separate factor for me to handle.
If you should ever see my view on the density gradients and temperature, either way, let me know here. Once again, thanks for the help and an open ear!