By Steve Goddard
ESA’s Venus Express mission has been studying the planet and a basic atmospheric model is emerging.

Venus has long been the CO2 bogeyman of climate science. In my last piece about Venus I laid out arguments against the claim that it is a runaway greenhouse which makes Venus hot. This generated a lot of discussion. I’m not going to review that discussion, but instead will pose a few ideas which should make the concepts clear to almost everybody.
If there were no Sun (or other external energy source) atmospheric temperature would approach absolute zero. As a result there would be almost no atmospheric pressure on any planet -> PV = nRT.
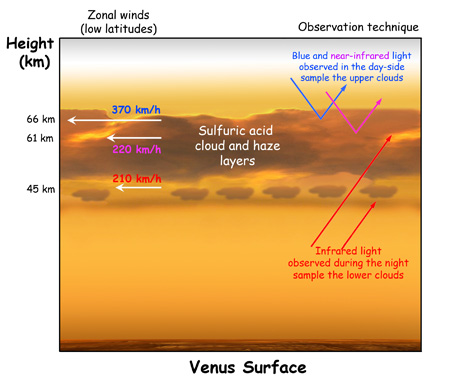
Because we have a sun providing energy to the periphery of the atmospheric system, the atmosphere circulates vertically and horizontally to maintain equilibrium. Falling air moves to regions of higher pressure, compresses and warms. The greater the pressure, the greater the warming. Rising air moves to regions of lower pressure, expands, and cools. The amount of warming (or cooling) per unit distance is described as the “lapse rate.” On Earth the dry lapse rate is 9.760 K/km. On Venus, the dry lapse rate is similar at 10.468 K/km. This means that with each km of elevation you gain on either Earth or Venus, the temperature drops by about 10C.
It is very important to note that despite radically different compositions, both atmospheres have approximately the same dry lapse rate. This tells us that the primary factor affecting the temperature is the thickness of the atmosphere, not the composition. Because Venus has a much thicker atmosphere than Earth, the temperature is much higher.
dT = -10 * dh where T is temperature and h is height.
With a constant lapse rate, an atmosphere twice as thick would be twice as warm. Three times as thick would be three times as warm. etc. Now let’s do some experiments using this information.
Experiment # 1 – Atmospheric pressure on Venus’ surface is 92 times larger than earth, because the atmosphere is much thicker and thus weighs more. Now suppose that we could instantly change the molecular composition of Venus atmosphere to match that of Earth. Because the lapse rate of Earth’s atmosphere is very similar to that of Venus, we would see little change in Venus temperature.
Experiment #2 – Now, lets keep the atmospheric composition of Venus constant, but instead remove almost 91/92 of it – to make the mass and thickness of Venus atmosphere similar to earth. Because lapse rates are similar between the two planets, temperatures would become similar to those on earth.
Experiment #3 – Let’s take Earth’s atmosphere and replace the composition with that of Venus. Because the lapse rates are similar, the temperature on Earth would not change very much.
Experiment #4 – Let’s keep the composition of Earth’s atmosphere fixed, but increase the amount of gas in the atmosphere by 92X. Because the lapse rates are similar, the temperature on Earth would become very hot, like Venus.
Now let’s look at measured data :

http://www.astro.wisc.edu/~townsend/resource/teaching/diploma/venus-t.gif

http://www.astro.wisc.edu/~townsend/resource/teaching/diploma/venus-p.gif
Note that at one Earth atmospheric pressure on Venus (altitude 50km) temperatures are only about 50 degrees warmer than earth temperatures. This is another indication that atmospheric composition is less important than thickness.
Conclusions : It isn’t the large amount of CO2 which makes Venus hot, rather it is the thick atmosphere being continuously heated by external sources. It isn’t the lack of CO2 on Earth which keeps Earth relatively cool, rather it is the thin atmosphere. Mars is even colder than earth despite having a 95% CO2 atmosphere, because it’s atmosphere is very thin. If greenhouse gases were responsible for the high temperatures on Venus (rather than atmospheric thickness) we would mathematically have to see a much higher lapse rate than on Earth – but we don’t.
WUWT commentor Julian Braggins provided a very useful link which adds a lot of important information.
“The much ballyhooed greenhouse effect of Venus’s carbon dioxide atmosphere can account for only part of the heating and evidence for other heating mechanisms is now in a turmoil,” confirmed Richard Kerr in Science magazine in 1980.
The greenhouse theory does not explain the even surface temperatures from the equator to the poles: “atmospheric temperature and pressure in most of the atmosphere (99 percent of it) are almost identical everywhere on Venus – at the equator, at high latitudes, and in both the planet’s day and night hemispheres. This, in turn, means the Venus weather machine is very efficient in distributing heat evenly,” suggested NASA News in April 1979. Firsoff pointed out the fallacy of the last statement: “To say that the vigorous circulation (of the atmosphere) smooths out the temperature differences will not do, for, firstly, if these differences were smoothed out the flow would stop and, secondly, an effect cannot be its own cause. We are thus left with an unresolved contradiction.”
======================================================
An update for those interested in what Venus looks like at the surface.
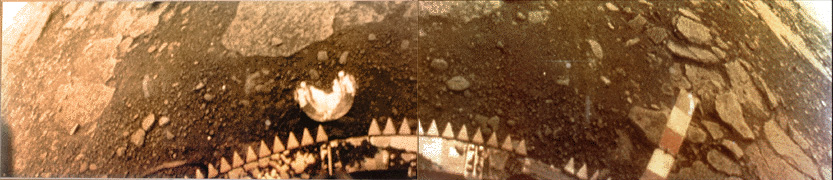
On March 1, 1982, the Soviet Venera 13 lander survived for 127 minutes (the planned design life was 32 minutes) in an environment with a temperature of 457 °C (855 °F) and a pressure of 89 Earth atmospheres (9.0 MPa). The photo composite above shows the soil and rocks near the lander.
Here’s another Venera image that shows a hint of yellow atmosphere. – Anthony

Nick Stokes
Again PV = nRT
The North Pole has equal pressure (as you said.) n is fixed and the temperature is lower, so the volume also has to be lower. Lower volume means with fixed n means higher density (as you said.)
What is confusing to you about this? The ideal gas law works just fine, like it always has.
Nick Stokes
Perhaps you are being confused by my use of the word “thinner?” I am using “thinner” to mean exactly that – the height of the atmosphere is smaller at the poles, because the atmosphere is more dense at the lower temperatures.
The whole point of these articles is to demonstrate that Earth could not become like Venus, unless all the limestones dissociated.
Ummmm? Whats Up With That?
I think you have taken a fairly common statement “the atmosphere of both Earth and Venus demonstrate Greenhouse heating” and constructed a straw man “AGW will cause a runaway Venusian Greenhouse.” There may be some idjits bouncing around claiming that – but it ain’t mainstream AGW. Step away from the straw man, Steve.
Steve Goddard:
Yes, and there’s a fundamental problem this observation presents to conventional physics. There’s no reason that the lower layers shouldn’t reach thermal equilibrium with the upper layers and warm them to the same temperature regardless of this pressure difference.
Take geothermal heat. About 200km down all rock is molten at at least 1000C. That heat can not ultimately go inward; it must all come outward. There should be an inverse square relation to the heat dissipation. The surface only being a fraction of an Earth radius higher than 200km down should be:
Earth radius = 6371km.
1/(Earth radius)^2 divided by 1/(Earth radius – 200km)^2 times 1000C = 938C.
The Earth’s surface should be 938C. It appears heat is disappearing. Meanwhile, the ocean below the thermocline is refrigerated at 2 to 4C when it should be boiling from the geothermal heat. The mid/low ocean can’t be cooling from evaporation on the surface because the upper ocean layer is warmer than the mid to lower levels. Heat can’t be extracted from a colder place through a hotter place. It appears to be a case where energy is either created or destroyed.
My own belief is that cooling might be caused by a thermoelectric effect related to the vertical voltage gradient.
Steve, there is apparently some sort of relation between temp and pressure in the air but it’s not one of cause and effect. We just don’t know what it is yet.
Steve Goddard
Well, Paul Clark said:
“If Steve was right you would have to take an oxygen tank with you to the Arctic because low temp would mean low pressure. Low pressure systems would have lower temperature than high ones.”
to which you responded:
“Try again. Low temperature means low volume in Antarctica. The atmosphere is thinner at the poles.
PV = nRT
I really suggest that everyone pass a high school chemistry class before trying to tackle this problem.”
Nothing Paul said related to the height of the atmosphere.
Ron Broberg
The idea that Earth could become like Venus was a central point of Sagan’s TV show – Cosmos. A whole generation was corrupted by this idea.
Nick Stokes:
“Thinner” I believe refers here to height not density. See this.
The reasons for this are a lot more complex that just e.g. the ideal gas law, though. (Ref.
Straw Man? LMAO – This is just one of thousands of similar articles:
http://www.is.wayne.edu/mnissani/a&s/GREENHOU.htm
Just to clarify my last comment: there is a PV = nRT relation for temps and pressure for changing situations in such as low pressure systems, Hadley cells, etc. But, it does not apply to the correlation between temp and pressure vertically as found on Earth and Venus.
And because of the rotation of the earth/atmosphere system, troposphere goes to 7km at the poles and to 17km at the equator.
In general, I believe that an adiabatic lapse rate in a planetary atmosphere is evidence of an active convection process. Without convection, such as in the stratosphere, higher parcels of air may be warmer than those below. Perhaps there must always be a greenhouse effect to force the tropopause above the ground level.
If Venus had an atmosphere as thick as it is now, but with a composition similar to the Earth, I suspect an active convection system would also exist and produce a lapse rate similar to that of the current Venusian atmosphere. Perhaps this is why Hungarian scientist Ferenc Miskolzi has said the magnitude of the greenhouse effect is limited by the thickness of the atmosphere.
>>>Ben Schumacher says: May 9, 2010 at 2:06 pm
>>>An atmosphere should approximately follow an adiabat — that is,
>>>pressure and temperature varying at the adiabatic lapse rate —
>>>provided the conditions are suitable for convection.
You don’t need convection for an adiabatic lapse rate – it is purely gas density/pressure that makes the gas hot. I have adiabatic warming in my bicycle pump, but no convection.
http://en.wikipedia.org/wiki/Lapse_rate
But adiabatic temperatures can reduce, as the heat is conducted or radiated away, as in my bicycle pump getting cooler – and the result on Venus would be a cooler but denser atmosphere (but roughly the same pressure, I presume, because of the same weight of gas above the surface).
So the current high temperatures on Venus must be augmented by solar input (or geothermic processes) to keep this high temperature. I would suggest that the atmosphere is thick enough and opaque enough to absorb a large amount of solar radiation.
.
.
>>>Paul Clark says: May 9, 2010 at 9:50 pm
>>> There’s no reason that the lower layers shouldn’t reach thermal
>>>equilibrium with the upper layers and warm them to the same
>>>temperature regardless of this pressure difference.
They may well do, in terms of molecular velocities (temperature), but the lower atmosphere has many more molecules/cm2 – so that same ‘temperature’ per molecule equals much more heat. That’s the whole point about compressing gasses and an adiabatic lapse rate.
But again that higher pressure does not preclude the lower atmosphere radiating and convecting away its heat, and becoming denser and cooler.
A HIGH PRESSURE GAS DOES NOT HAVE TO BE WARM. Touch the sides of a diver’s gas cylinder, and tell me if it is warm. Thus Venus must have a thermal input, to maintain its high temperature – otherwise it would end up like the diver’s very dense, very high pressure, but very cold gas cylinder.
.
Just one question about the Venera pics – why did they never ‘normalise’ the colours? They have a colour chart there for comparison, but I have never seen an image of the surface as it would look under Earth-like light conditions.
.
@Paul Clarke and others: The Pressure-Temperature link exists only in gasses that are vertically mixed. Water (being essentially incompressible) doesn’t do this even with vertical mixing. The earth’s crust doesn’t do this either. Not only is it almost incompressible, but it is also essentially static. So comments about a relationship between pressure and the temperatures of the sea and earth’s crust are way off topic.
A column of gas without vertical mixing would also be all the same temperature. Wherever there is some amount of vertical mixing however, the high pressure regions low down will be heated by this, and the upper low pressure regions will be cooled. A temperature gradient will then form.
Overall temperatures will be set by the part of the atmosphere which is in radiative equilibrium with space. On earth most radiation reaches the ground, and the surface itself is directly heated. Therefore the temperature at the surface mostly sets the overall temperature of the atmosphere. Since the rest of the atmosphere is at much lower pressure it will be much cooler than the radiative equilibrium temperature. On venus it is not the surface but the the upper cloud layers that are directly heated by the sun. The upper clouds therefore reach the temperature of radiative equilibrium with space, and the surface, which is at much higher pressure will end up being much hotter.
All this is approximate and ignores other effects, including the greenhouse effect which deals with radiative transfer within the atmosphere. But to first order this serves as an adequate explanation of why Venus is so much hotter than the Earth.
Steve.
If you are implying from this post that Venus should have a much higher laps rate, due to its 99% CO2 atmosphere being a so-called ‘greenhouse gas’, that is not immediately obvious in your post.
You need a graph of lapse rate with 0.1% CO2 (Earth) and a lapse rate with 99% CO2 (Venus) being much steeper (because the latter should, according to green logic, capture more solar energy, which is distributed through the Venusian atmosphere by convection).
But this is something that Venus does not display, with both Earth and Venus having the same lapse rate.
Ergo – CO2 is not the big driver of temperature on Venus, and nor on Earth.
.
OK, I’ll try this once more.
When using the Ideal Gas Law, or any other equation of state for a gas, it is important to consider ALL of the quantities involved. It is also important to consider the circumstances. A sealed rigid volume such as a SCUBA tank isn’t the same as an inflated rubber balloon, and neither of those is the same as an open column of air “standing up” in a gravitational field. Details matter, and people have been conflating many issues incorrectly, or forgetting certain constraints and complications.
The Gas Law can be written as P = (N/V)kT, and the quantity N/V is called the particle density. There is no fixed volume for our atmosphere. Its volume can increase or decrease as it expands and contracts. In this context, it is the product of particle density and temperature that must remain fixed if the pressure is not to change.
This happens all the time, all around the planet. The surface temperature changes RADICALLY with time and location. Nevertheless, the pressure at sea level NEVER changes in any SIGNIFICANT way from 1000mb, no matter where you are on Earth, or what time of the day or time of the year it happens to be. Yes, it does change, and that is why we use barometers, but the AMOUNT of change is MINISCULE in comparison with the corresponding changes in temperature. Sea level pressure in the Arctic is essentially the same as sea level pressure in the Caribbean, despite the large differences in temperatures.
When you start climbing above sea level, however, you can’t just use the Gas Law by itself. You have to consider the effects of gravity. One of the consequences is the fact that pressure and particle density do not change in the same way as we go to higher altitudes. Both temperature and particle density decrease as we move upward in the troposphere, and because pressure depends on the product of these things, the pressure decreases more quickly than either temperature or particle density. At the tropopause, the particle density is typically down to about 30% of its sea level value, but the air pressure has gone down faster, and will only be about 20% of its sea level value.
The devil is in the details, and many people on this thread, and particularly Steve, are ignoring them. Actually, I think the tide has reversed on this. Many other posters appear to “get it”, but Steve doesn’t seem to be doing so. Another case of “right answer, wrong logic”, I guess.
/dr.bill
Jacob says:
May 8, 2010 at 4:14 pm
Why is the atmosphere on Venus 92 times more dense than on Earth? That is the big question, that I don’t understand.
Now, if the atmosphere is denser – it means there are 92 times more molecules of gas in the same volume (or layer) of the atmosphere, therefore they would absorb 92 time more outgoing radiation (much more anyway). So, the heat on Venus’ surface is caused by the greenhouse effect after all, by the fact that the greenhouse effect is much stronger there, due to the density of the atmosphere, and not necessarily due to it’s composition (Co2).
If pressure alone could cause heating – why isn’t the bottom of the oceans very hot?
_________________________________________________________________________
J.Hansford says:
You are confusing gasses and liquids Jacob… Seems a few others are doing the same. Gasses are compressible, liquids are not…..
_________________________________________________________________________
PV=nRT is the ideal GAS law. How it applies to Venus and Earth is the topic under discussion.
“A law relating the pressure, temperature, and volume of an ideal gas. Many common gases exhibit behavior very close to that of an ideal gas at ambient temperature and pressure. The ideal gas law was originally derived from the experimentally measured Charles’ law and Boyle’s law. Let P be the pressure of a gas, V the volume it occupies, and T its temperature (which must be in absolute temperature units, i.e., in Kelvin). Then the ideal gas law states
PV=nRT
where n is the number of moles of gas present and R is the universal gas constant,….”
Source: http://scienceworld.wolfram.com/physics/IdealGasLaw.html
dr.bill
Remarkable that I have to explain this again. Please read my posts carefully before posting.
At temperatures where the atmosphere is a gas, the pressure is fixed by the weight of the column of air. The temperature controls the volume (i.e height of the atmosphere.) That is why the cold poles have a thinner atmosphere than the tropics.
P = T *c/V
This is the most fundamental physics. When the temperature decreases, the volume has to decrease. The volume is area * height. Area is fixed, so the only thing which can change at the cold poles is the height of the atmosphere.
Consider a cylinder containing a quantity of gas. The cylinder is closed at one end and has a piston at the other. Suppose now we force in the piston, compressing the gas. What happens to the temperature of the gas?
Applying the ideal gas law, using the equation pV = nRT, the volume V is reduced by the compression, and nR is unchanged, so, other things being equal, T must be reduced. Thus we reach the remarkable conclusion that compressing a gas reduces its temperature!
Of course this is quite fallacious. In reality, compressing the gas increases its temperature because the kinetic energy of the moving piston is transferred to the molecules of the gas. To balance the equation, the pressure p is also increased. If the cylinder is perfectly insulated the increased T will be maintained indefinitely, so long as the piston is held in position. In practice, perfect insulation is impossible, and T will fall by conduction and radiation until it returns to equilibrium with the environment. As T falls, assuming the piston is held in position, pressure will also fall, though not back to its original level before the gas was compressed.
In this example the fallacy in the original argument was obvious, because we knew the conclusion was false, and it was easy to see the loophole in ‘other things being equal’. But in other cases the fallacies may not be obvious. The moral is that the equation of the ideal gas law cannot be applied blindly without considering the physical processes involved.
experiment #5 Imagine the venusian atmosphere would be still heavier and no sunlight would reach the surface; the temperature would still be greater and would you still speak about greenhouse effect to explain the great temperature at the venusian surface?
“Why? Because the greenhouse effect is logarithmic. After the first few percent, additional CO2 makes much less difference to the temperature.
Consider earth, where a doubling of CO2 only increases temperatures by <1 (Lindzen) to 3 (IPCC) degrees C."
Again, NOT TRUE. The forcing from CO2 is logarithmic only within certain a certain range about the current concentration. At very high concentrations it approaches linearity again as the 15 um band saturates and the weak bands become predominant. This has been pointed out to you multiple times, and yet you keep repeating it.
If you want a reference, see Figure 1 in:
http://journals.ametsoc.org/doi/pdf/10.1175/1520-0469%281977%29034%3C0448%3AARCMSO%3E2.0.CO%3B2
(I might also recommend http://www.agu.org/journals/ja/v085/iA13/JA085iA13p08223/, one of the earlier calculations of the full temperature profile of Venus' atmosphere)
DavidB
If you push a piston down into a cylinder, you are exerting a force which directly increases the pressure. That is why the temperature increases. Your claim that “everything else is held equal” is incorrect.
Exactly what I have been trying to get Steve to respond to. He refuses to acknowledge that because PV=nRT does not include any term for gravity, it does NOT mean that a gas is magically immune to the law of gravity. It is basic physics.
Steve, true or false, if there was no gravity there would be no atmospheric pressure?
You might escape earth’s gravity if you run fast enough but you won’t escape this question.
Another thing Steve, you dismissed my notion that water made all the difference between Venus and Earth per my reasoning. I did some further thinking that I hope you will at least consider and comment without going back in time to limestone.
I looked up the annual rainfall on earth and came up with the number ~5 x10^14 cubic meters. Every last drop of that rain represents an amount of latent heat energy to evaporate it and the huge majority of that heat went into liquid water
on the ground, (a small amount happened in the air, i.e. virga).
So I went further and astounded myself even doubting the numbers I came up with. Maybe I made a mistake or relied on wrong data or something but please follow it and you be the judge of that.
If you take that number and divide by seconds per year it comes to 16 million m^3/sec. At 1000 kg per m^3 water, that’s 16 billion kg of water evaporating on mostly the surface per second.
<a href="http://www.thermexcel.com/english/tables/vap_eau.htm"Evaporating one kg of water requires 2,257,920 joules of heat. That works out to 3.4×10^16 watts of evaporation – something Venus, without water, does NOT have going for it.
So I divided that number by earth’s surface and get about 67 watts/m^2.
If that is correct then it most definitely is NOT something to snort at!
NASA puts incoming solar irradiance at 340 watts/m^2 so that comes to 20% of earth’s ERB being manipulated by water enthalpy as I think another poster above correctly termed it – ‘a heat engine’.
Recognize that although the heat is collected at the surface it is NOT released at the surface … it is released at cloud formation altitude. The heat was carried up totally unaffected by very significant amount of ‘green house’ gases below cloud formation altitude. Pick the average altitude AGL – I don’t know. 5000 feet? 10000 feet?
As I see it, whatever the best average altitude value, it represents a chunk of atmosphere that ~20% of solar heating did NOT have to pass through on its journey re-radiating out to space because it was carried up there mechanically. (And remember, just because the water vapor cooled off as it rose to that altitude it did NOT lose any heat! “adiabatic”.)
So I stand on my assertion that having 2/3 of our planet covered in water is what drives our climate and the reason we are so much cooler than Venus. PV=nRT has nothing to do with it.
Dave
The weak bands are linear, but with a small slope. That is why they call them “weak bands.”